Abstract
The aim of the study was to verify the production of PAF and the activity of PAF acetyl-hydrolase (PAF-AH), the enzyme involved in the catabolism of this phospholipid mediator, in migraine attacks. Their levels were determined during migraine crises in serial samples of internal jugular venous blood taken from five migraine patients without aura, who were admitted to the hospital during the crises. Internal jugular venous blood samples were taken immediately after catheter insertion at 1, 2, and 4 h after attack onset, and within 2 h from its cessation. PAF was purified by high-performance liquid chromatography (HPLC) and determined by radioimmunoassay method. The enzymatic activity of PAF-AH was measured by reverse-phase HPLC, based on the derivatization with 7-diethylaminocoumarin-3-carbonylazide. In the internal jugular venous blood of migraine patients without aura (MO), an increase was observed in PAF levels, which was already evident at the time of catheter insertion (885.6 ± 82.8) and at the first hour (868.4 ± 65.24) (
Keywords
Introduction
Platelet-activating factor (PAF) (1-O-alkyl-2-acetyl-sn-glycero-3-phosphorylcholine) is a phospholipid belonging to the family of biologically active acetylated phosphoglycerides, exerting a wide range of biological activities, of which one of the most relevant is the induction of platelet activation and aggregation (1). The involvement of PAF has been hypothesized during migraine attacks, but no studies of this have been carried out due to methodological difficulties in its measurement. The involvement of PAF in trigemino-vascular activation, which is believed to be the final event occurring in migraine crises, has been indirectly suggested by the evidence of platelet activation in the model of neurogenic inflammation and supported by experimental studies demonstrating the stimulation of PAF release by certain neuropeptides (2). In particular, substance P but also calcitonin gene-related peptide (CGRP), released by the activated trigeminal endings, seem to be able to stimulate the synthesis of PAF by the endothelium with subsequent activation of blood cells, especially platelets, which are particularly sensitive to this factor compared with other blood cells (3, 4). By inducing activation of human platelets at very low concentrations in the range of 1–10 n
This abnormal response to PAF consists of a specific platelet α-granule hypersecretion in response to this aggregating agent, which is consistent with the finding of an enhanced increase in cytosolic ionized calcium on stimulation with PAF (7–10).
Although this specific platelet hypersensitivity has been related to an aberrant intracellular transduction mechanism intrinsic to this cellular element, attention was also focused on PAF as a putative contributor to platelet activation in migraine.
In this regard, it is conceivable that migraine attacks may be associated with an increased production of PAF in the cerebral circulation, perhaps as a consequence of trigemino-vascular activation.
Until now, no studies have been carried out to verify PAF levels in internal jugular venous blood as a consequence of the pathophysiological mechanisms underlying migraine attacks.
The present research was aimed at verifying the changes in levels of PAF and PAF acetyl-hydrolase (PAF-AH) (EC 3.1.1.47), the enzyme intervening in PAF catabolism, in the internal jugular venous blood of migraine without aura (MO) in-patients examined during attacks. The determination of these levels was carried out at different times during the attacks and their variations were compared with the levels detected at the end of the attacks. In the same samples, the levels of the stable metabolites of nitric oxide, nitrites, and those of the trigeminal neuropeptides (CGRP) and neurokinin A (NKA) have already been determined and the results published (11).
Patients and methods
Patients
The study protocol was approved by the local ethics committee.
After informed written consent, five MO patients (three females and two males) (mean age 42.3 ± 4.6 years) were enrolled. Their number of attacks/month ranged from three to five and the duration of attacks from 8 to 10 h. Three of the five migraine patients had a family history of migraine. Exclusion criteria for patients joining the study were smoking habit, migraine duration < 2 h, and no systemic or concomitant pathologies (renal diseases and cardiovascular diseases including hypertension and epilepsy). Before inclusion in the study two patients had been taking simple analgesics and the other three patients triptans for the symptomatic treatment of their usual attacks with a satisfactory relief of headache. None of the patients had been treated with prophylactic drugs at least 2 months prior to inclusion.
Patients were admitted to the Neurologic Clinic of the University of Perugia as in-patients, and assessed during a spontaneous, non-induced attack occurring within the period of hospitalization (mean hospital stay = 7 days). Diagnosis was made using the current International Headache Society criteria (12).
Before performing central venous access procedure, the internal jugular vein was first examined with a hand-held sonographic transducer to determine the size, patency and location of the internal jugular vein (13). The decision to access the right or left internal jugular vein depended on the location of head pain: in four patients access was to the right jugular vein, in the remaining patient to the left jugular vein. Procedures were carried out under sterile conditions and using sonographic guidance. The internal jugular vein was punctured just cephalad to the clavicle by using a 21 G needle from the peripherally inserted central catheter (PICC) kit (14). Once venous access was obtained, a 0.018-in wire was passed through the needle under fluoroscopic guidance, and the needle was then removed (15). A 4 or 5 peel-away sheath (from the PICC kit) was inserted over the wire, which was then removed. Only one puncture was required for each of the four patients, while two punctures were required for the other migraine patient undergoing the central venous access procedure. In no case was the common carotid artery inadvertently punctured, nor did periprocedural complications occur. Post-procedural chest radiographs showed no evidence of pneumothorax, haemothorax or mediastinal haematoma in any of the migraine patients undergoing central venous catheter insertion.
Internal jugular venous blood samples were taken within 30 min after the onset of the attack (T0), at the first, second, fourth, and sixth hour from the start of the attack, and within 2 h after the end of the attack, when patients were just headache-free. Within the first hour after catheter insertion the head pain was severe enough to be regarded as migraine (score = 3 of pain intensity) in all migraine patients examined.
During the migraine attacks no symptomatic drugs were administered to patients to stop the attacks. Details of the migraine attacks of each patient have been reported in a previously published paper (11). The attacks resembled those typically experienced by our study patients.
Control values in the peripheral blood were obtained from 15 age-matched apparently healthy subjects (nine females and seven males) (mean age 41.9 ± 4.6 years). In particular, they did not suffer from systemic diseases or diseases of the central and peripheral nervous system. They had never suffered from migraine, nor had they experienced any episodic tension-type headache attack in the last 6 months.
We also attempted to verify whether the modification in biochemical variables observed in our migraine patients was the response to catheter insertion through the endothelium of the internal jugular vein. For this purpose PAF and PAF-AH were also measured in the internal jugular venous blood of four patients who had elective internal jugular venous catheters placed for postoperative management in intensive care. Even if the timings of samplings were not exactly the same, these measures could help us to clarify if the PAF and PAF-AH findings obtained in our migraine patients were associated with the migraine attack and not due to trauma from catheter insertion.
PAF and PAF-AH determinations
PAF
Blood samples (4.5 ml) were drawn at different time points of the study from each internal jugular vein into polypropylene tubes containing EDTA (1 mg/ml) and immediately added to 15 ml methanol/chloroform [2 : 1 (v/v)] and stored at − 80 °C until analysis.
Lipids were extracted with a biphasic mixture formed by adding 5 ml each of chloroform and aqueous NaCL(1 mol/l). The chloroform extracts (30 ml) were evaporated to dryness and redissolved in 5 ml chloroform/methanol [4 : 1 (v/v)]. The concentrated extracts, together with a 5-ml rinse, were then applied to small silica columns (particle size 32–62 µm; pore size 60 µm, bed volume 1.6 ml) preequilibrated in chloroform. The columns were washed with 50 ml chloroform/methanol/glacial acetic acid [1 : 1 : 0.04 (v/v/v)] and PAF was subsequently eluted with 40 ml chloroform/methanol/H3PO4]1 : 1 : 0.04 (v/v/v)]. Recovery of PAF was 98–100%. To wash H3PO4 from the eluates, the samples were evaporated to near dryness and redissolved in 15 ml chloroform/methanol (4 : 1 (v/v)]. Nine millilitres of methanol and 12 ml of H2O were then added to form a biphasic mixture and the upper phase was adjusted to pH 6 with concentrated NH4OH. After the mixture was shaken vigorously and the two phases were allowed to reform, the chloroform phase was removed and the upper phase was extracted twice with chloroform. High-performance liquid chromatography (HPLC) purification of PAF was performed using a modified method of Blank and Snyder (16). Samples were purified on an ultrasphere silica column (5 µm, 10 × 200 mm; Beckman, Fullerton, CA, USA) using a two-component solvent system. Solvent A contained isopropanol/hexane [1 : 1 (v/v)] and solvent B consisted of isopropanol/hexane/H2O [23 : 23 : 1 (v/v/v)]. The samples were dissolved in 100 µl of solvent A before injection into a Waters (Milford, MA, USA) HPLC instrument, and PAF was eluted at a flow rate of 5 ml/min with a linear gradient of 50% to 100% B over the first 30 min. The samples were then washed as described above to eliminate H3PO4, concentrated and evaporated to dryness, and finally dissolved in radioimmunoassay (RIA) buffer for determination. PAF RIA kits were purchased from Amersham (Amersham Biosciences Europe, Milan, Italy). Cross-reactivity (%) was < 0.01 for 1-hexadecyl-2-lyso-GPC–lyso-PAF, ∼ 3 for 1-hexadecyl-enyl-2-acetyl-GPC, 0.06 for 1-hexadecanoyl-2-acetyl-GPC, ∼ 4 for 1-hexadecyl- 2-propionyl-GPC, and 0.01 for 1-hexadecanoyl-2-lyso-GPC. Sensitivity was 20 pg/tube. Data were expressed as pg/ml.
PAF-AH activity
A sensitive method developed by Balestrieri et al. (17) was used based on HPLC with fluorometric detection. Briefly, 25 µl of serum were mixed with 25 µl of a 1 mg/ml solution of PAF C:18 in Tris-Tyrode buffer (2.6 m
A Model 342 liquid gradient chromatograph equipped with a Shimadzu Model 160 fluorescence spectrometer and a C-R3A Shimadzu Chromatopack integrating system were used (Shimadzu Italy, Milan, Italy). A 3.9 × 300-mm Nova Pack C18 Waters reversed-phase column, filled with 4 µm average particle size, was also used. The mobile phase was made with a gradient between the head solvent (A), composed of methanol : water [80 : 20 (v/v)] containing 0.25 g/l choline chloride and chloroform (B). The flow rate of the mobile phase was 1 ml/min. The gradient, starting at the time of sample injection, was from 0 to 55% B in 22 min. At the end of each analysis the column was equilibrated for 10 min with solution A. The fluorescence detector was set at an excitation wavelength of 400 nm and at an emission wavelength of 480 nm. The concentrations of derivatized lysophospholipids were calculated by comparing the peak area with that of the internal standard. For details of comparison with the radiochemical assay, precision and sensitivity, see Balestrieri et al. (17). Data were expressed as pmol/µl.
Statistical analysis
All values were expressed as mean ± 2 SD. One-way within-subjects
Results
The levels of PAF measured in the internal jugular venous blood of MO patients assessed ictally already appeared significantly higher in the samples collected at the time of catheter insertion (T0) compared with those measured after the end of the attacks (P < 0.00001) (Table 1). These highest PAF levels were maintained at the first hour (P < 0.00001) and tended to decrease until the fourth and sixth hours, although they were significantly higher than those determined in the headache-free period (P < 0.01) (Fig. 1).

Box plot of mean, SD and standard error (SE) of PAF levels (pg/ml) in the internal jugular venous blood of migraine patients examined within 30 min from the beginning of the attack (T0), at the first, second, fourth hours, and after the end of the ictal period (headache-free period).
Mean (± 2 SD) of PAF (pg/ml) and PAF-AH (pmol/l) levels found at various stages of the attacks, in the internal jugular venous blood of migraine without aura patients examined

PAF levels in the peripheral venous blood of migraine patients taken at the different time points of the study are shown in Table 2. A slight but significant increase in PAF levels emerged in the antecubital fossa venous blood, especially at the time of catheter insertion and at the first hour, but a broader range of variation in peripheral values emerged compared with those measured in the internal jugular venous blood (
Mean (± 2 SD) of PAF (pg/ml) and PAF-AH (pmol/µl) levels found at various stages of the attacks, in the peripheral blood of migraine withour aura patients examined
The highest values of PAF-AH activity in the internal jugular venous blood of MO patients were also found at the time of catheter insertion and at the first hour. They then progressively decreased with the lowest values measured at the fourth and sixth hours (P < 0.0001 and P < 0.00003, respectively) (Table 2) (Fig. 2).
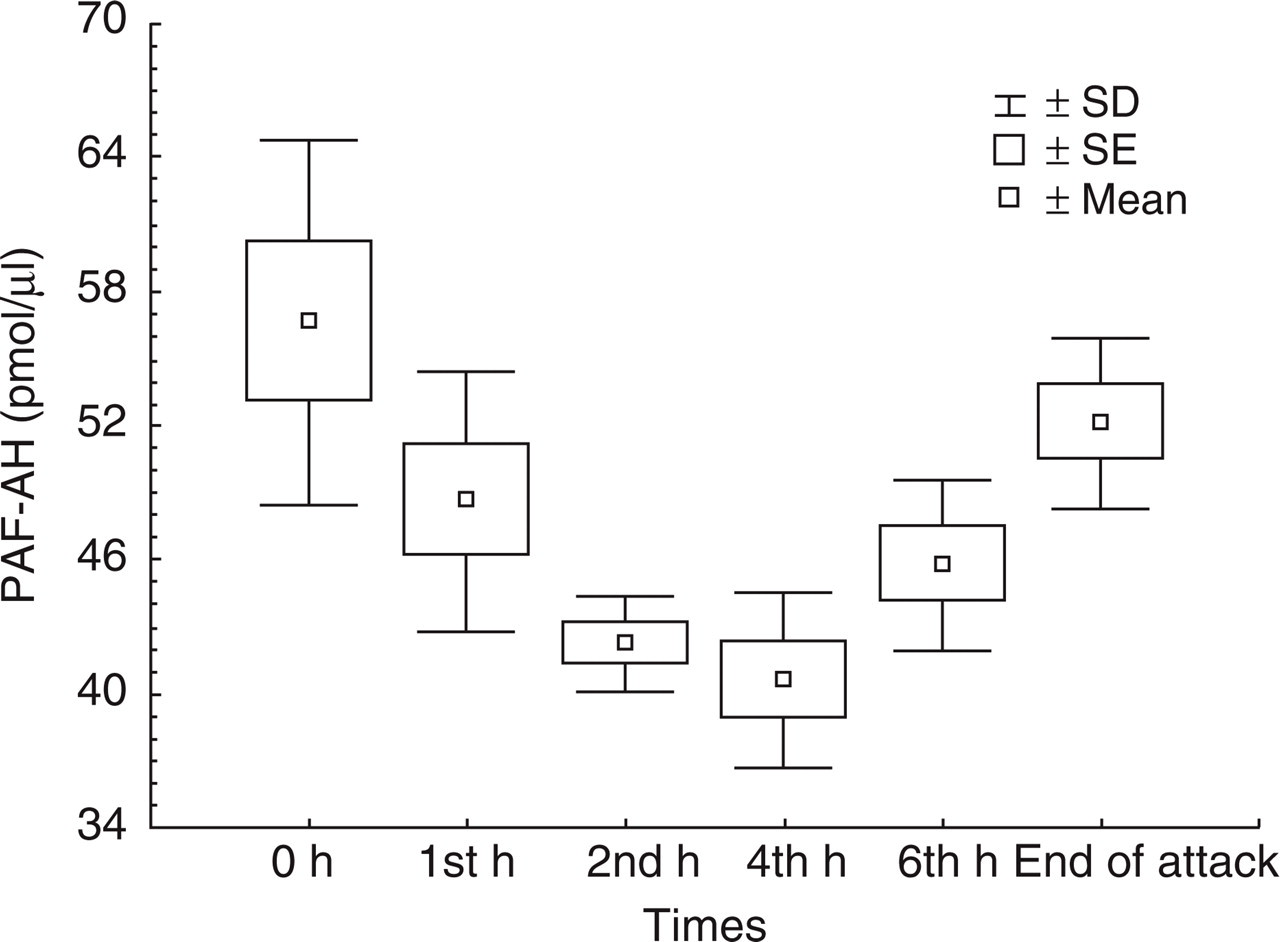
Box plot of mean, SD and standard error (SE) of PAF-AH levels (pmol/µl) in the internal jugular venous blood of migraine patients examined within 30 min from the beginning of the attack (T0), at the first, second, fourth hours, and after the end of the ictal period (headache-free period).
In the peripheral blood PAF-AH showed a similar trend, with a progressive decrease reaching lower values at the fourth and sixth hours after catheter insertion, although the variations observed were less marked than those observed in the internal jugular venous blood (Table 3). No significant difference emerged between the mean values of PAF-AH measured in the internal jugular venous blood of migraine patients at the end of the attacks and the mean values obtained from the peripheral blood of age-matched control subjects (46.7 ± 3.9).
Mean (± 2 SD) of PAF (µmol/l) and PAF-AH levels found at different time points in the internal jugular venous blood from patients without a previous history of migraine having elective jugular vein catheters placed for postoperative management in intensive care

Given that no variations were observed in the PAF and PAF-AH blood levels of patients who had elective internal jugular venous catheter insertion for postoperative management in intensive care, it seems probable that changes found in the internal jugular blood of the migraine patients examined were due to trauma from catheter insertion and not associated with the migraine attack (Table 3).
Discussion
Platelet-activating factor is a phospholipid mediator possessing a wide range of biological activities, which are relevant for the development of numerous inflammatory and thrombotic responses (18). In particular, this acetylated phosphoglyceride is the most powerful aggregating agent for platelets, acting through a specific receptor that is coupled with a G-protein activating a phosphatidylinositol-specific phospholipase C (19).
Its role has also been advocated in migraine, especially during attacks, to explain the activation state and secretion behaviour of platelets of the patients affected. However, no direct measurements of PAF in blood have been made until now.
This study tried to examine the hypothesis regarding PAF involvement in migraine. The determinations of this biologically active phospholipid were made in internal jugular venous blood, which provides more information than peripheral blood regarding the biochemical changes that characterize migraine attacks. In this regard, internal jugular venous blood can more exactly reflect intracranial and biochemical variations, whereas external jugular venous blood can express biochemical changes in both intra- and extracranial cerebral structures.
An increased production of PAF emerged in the internal jugular venous blood of MO patients, which may therefore also be responsible for increased platelet activation and secretion in the cerebral circulation of the same patients.
To exclude the possibility that the variations in PAF levels were not due to the pathogenic events of migraine, but rather to the trauma subsequent to catheter insertion, we measured the same parameters in the jugular blood samples of intensive care patients. We did not find significant variations between the values of the latter patients measured at specific times and those of the migraine patients measured after the end of the attacks, thus, when they were completely headache-free.
Potential sources of PAF production in the cerebral circulation of migraine patients examined during the attacks are represented by endothelial cells, as well as by platelets themselves and mast cells. All these cells are potentially involved in migraine attack, as suggested by the experimental model of neurogenic inflammation (2). The intervention of other cells, especially inflammatory cells, for which PAF has been recognized to play an important role as a chemotactic agent, should, in contrast, be excluded on the basis of the ‘sterile’ nature of trigemino-vascular activation.
Although sensory neuropeptides can stimulate PAF production by endothelial cells, conversely, PAF itself may trigger the release of sensory neuropeptides, particularly CGRP, as suggested by recent experimental evidence (20, 21). This information cannot be extrapolated from our study and additional data are needed to clarify the temporal relationship between sensory neuropeptides, particularly CGRP, and nitric oxide (NO) and PAF release.
The recent observations of an increased reactivity of sensory endings to histamine due to PAF, accompanied by the induction of histamine H1 and PAF receptor mRNA expression in rat trigeminal ganglia, suggest another potential mechanism of PAF in migraine attacks (22).
A further putative effect directly involving the endothelial cells of cerebral vessels could be relevant in migraine: PAF is able to activate the constitutive endothelial NO synthase in these vessels, and this effect is exerted through the induction of the phosphorylation of the above enzyme (23, 24). This may explain the occurrence of the nitrite peak at the first hour in the internal jugular venous blood samples of our migraine patients (11), not only as an effect of sensory neuropeptides on endothelial cells but also as a consequence of a PAF-specific receptor-mediated stimulation.
Several lines of evidence suggest that platelet function is altered in migraine patients. Most studies concern one or few aspects of platelet activity, i.e. response to different aggregating agents, measurement of secretory products from dense bodies or α-granules, oxidative stress, circulating platelet monoamino oxidase and phenolsulfotransferase activity, serotoninergic function and NO production, as well as other cell signalling molecules (25). All these aspects of platelet function were conventionally studied in platelet-rich plasma in most cases and few researches concern whole blood measurements which better reflect the in vivo state, both in patients and controls. In migraine patients results were obtained for the above measurements in headache-free periods (and in this case the exact time from the last attack or the subsequent attack is often not mentioned) or, alternatively, during migraine crises, and this may account for the variability of the results and the difficulty in their comparability. This aspect is further complicated by the wide intrinsic variability of platelet activity tests in normal subjects, variability which has not always been clearly established in the various studies. An attempt to create an index based on the integration of aggregation, secretion, plasma thromboxane and circulating platelet aggregate measurements has been made to account for the changes in platelet activity during acute migraine, but this index appeared at its maximal shortly after cessation of attacks and not at the start, questioning the potential causal role of platelets in migraine pathogenesis (26).
It should be pointed out that, to our knowledge, all studies on platelet function have been carried out on peripheral blood, and no information has been obtained from jugular blood during migraine attacks. Therefore the above data, in particular those obtained ictally, cannot answer the question of the involvement of platelet activation in the cerebral circulation of migraine patients during the crisis, as suggested by the model of neurogenic inflammation.
We hypothesize, on the basis of our results, that the increased production of PAF found may result in persistent platelet activation and hyperfunction in the cerebral circulation during migraine attacks, even in the presence of increased NO production by the endothelium of cerebral vessels as shown by our previous study (11). This platelet activation should, in any case, be substantiated by further experiments involving internal jugular venous blood.
The increase of PAF also seems to be evident, although to a lesser extent, in peripheral blood. There is, however, a wide range of variability of the values, and a direct relationship between this finding and platelet functioning and secretion should be clarified by future research.
Studying changes in the levels and activation states of the enzymes involved in the metabolism of PAF is important for understanding the pathophysiological role of this mediator. We focused our attention on the enzyme PAF-AH which intervenes in its metabolism (27).
The increase of PAF observed in our study in the internal jugular venous blood of MO patients could, in part, be explained by a non-parallel increase in the enzyme involved in its catabolism which, in contrast, shows a decrease that is particularly evident at the second and fourth hours. This trend, although less significant from a statistical point of view, was also observed at the peripheral level.
A potential explanation of this finding is the enzyme saturation as a result of a continuous reaction to the increased PAF levels, or alternatively, unknown substances produced during the migraine attacks antagonizing its activity.
Footnotes
Acknowledgements
The authors thank John A. Toomey for editing the English and Marisa M. Morson for technical assistance.
