Abstract
Many reports indicate that nitric oxide (NO) could be involved in migraine without aura (MWA), an extremely diffuse clinical event. Since monocyte may be a relevant source of NO, we analysed monocyte activation in MWA patients, in a period in which they were free of symptoms. NO basal production by MWA peripheral monocytes was significantly higher than in healthy subjects (91.25 ± 8.6 μ
Keywords
Introduction
Nitric oxide (NO), a gas generated via the
It has recently gained attention as new class of messenger molecule and serves a variety of functions not only in cell-mediated immunity (7) but also in the modulation of vascular-smooth muscle relaxation and vascular dilatation (8). At present there is growing interest in NO as a causative molecule of migraine pain (9). Olesen recently suggested that NO formation in the endothelium of cerebral arteries is a potential initiator of a migraine attack. Migraine attacks are associated with intra- and extracranial arterial dilatation that could be due to NO (10).
NO has therefore already made the leap forward out the shadows onto the main stage of migraine mechanics from the basic fields to the clinical ones (11–15).
Several authors also reported a cross-linking between NO and cyclooxygenase (COX) pathways (16–18). In fact, both NO and COX exist in two major isoforms: the constitutive forms (COX1, NOS1 and NOS3) that are responsible for the production of PGs, and NO involved in physiological functions; and the inducible forms (COX2 and NOS2) that are, instead, rapidly up-regulated upon appropriate stimulation (19, 20).
The enzyme COX2 is more selectively distributed in mammalian tissues than COX1, which is present in almost all tissues and cells (21). NOS1 and NOS3 produce NO for minutes per response episode, whether the stimulus is chemical, such as the neurotransmitter glutamate, or mechanical such as the pulsatile strain in arterial wall. They are constitutively expressed and transiently activated by increase of intracellular calcium/calmodulin complexes.
In contrast, NOS2 can produce NO for as long as 5 days, at least in mouse macrophages, when care is taken to replenish both the inductive stimuli and the
The interaction between NOS and COX could be responsible, at least in part, for the final goal leading to the pain production in migraine.
In our previous work we were able to demonstrate a sure involvement of NO in migraine without aura (MWA) attacks (23), since we reported a marked increase of nitrite serum level in NO donor induced migraine. In addition, NO interacts with many iron-containing enzymes, such as COX2, by modifying their activity (24).
In the light of these findings we analysed in the present study the synthesis and release of NO by peripheral monocytes of MWA patients in a period in which they were without symptoms in order to evaluate the unstimulated NO release.
Since it is still unclear whether a reciprocal modulation of NO and PGE2 synthesis and release exists or not, we also analysed in the present study the PGE2 release by peripheral monocytes.
Patients and methods
Patients
This study was conducted in 10 patients suffering from MWA (five males, five females, mean age 40.5 ± 5.2 years), randomly recruited among the out-patients of the University ‘La Sapienza’ Headache Centre and diagnosed according to the 1988 International Headache Society (IHS) criteria (25). The mean range of migraine duration was 12.4 years (mean 3.5, max 21.6 years). There was no coexistence of tension-type headache in these MWA patients. Eight clinically healthy subjects (C), sex- and age-matched (four males, four females, mean age 33 ± 5 years) were evaluated as normal controls. The study protocol was approved by our Institutional Ethic Board and informed consent was obtained from all patients and from controls. The recommended principles of the Declaration of Helsinki, September 1989, were closely observed during this clinical research study.
Both MWA patients and controls were tested while recumbent in bed at a time when MWA patients had been asymptomatic for at least 5 days and had not taken any medication for at least 10 days.
Preparation of monocyte monolayers
Peripheral venous blood (10 ml), obtained from the two groups of subjects under study, was immediately transported to the laboratory to be processed. Heparinized blood samples were diluted with an equal volume of RPMI 1640 medium (Flow Labs, MacLean, VA). After careful mixing, the diluted blood was underlayered with 10 ml of Lymphoprep (Nyegaard, Oslo, Norway) in a 50-ml sterile conical polypropylene tube (Falcon, Oxnard, CA). The samples were centrifuged for 30 min at 400
Cells incubated with culture medium alone represented the controls.
In some experiments, monocytes were incubated for 24 h in the presence of different concentrations (20–100–300 μ
At the end of the incubation, supernatants from each experiment were collected, centrifuged at 1643
Cell vitality, checked by trypan blue exclusion test, was 98 ± 1.0% before incubation and 95 ± 1.2% after.
NO2 − accumulation
NO2
− accumulation was used as an indicator of NO production in the medium and was assayed by the Griess reaction (28). Briefly, 100 μl Griess reagent (1% sulfamide/0.1% naphthylethylene diamine dihydrochloride/2% H3PO4) were added to 100 μl of each supernatant in triplicate wells of a 96-well plate (Corning, NY). The plates were read using a Dynatech MR5000 ELISA plate reader (Dynatech, Chantilly, VA) at 550 nm, after 10 min of incubation at room temperature. Serial dilutions of NaNO2 were used as standard curve. Results are expressed as μ
Prostaglandin E2 radioimmunoassay
Prostaglandin E2 (PGE2) was measured directly in all culture media by a commercial available radioimmunoassay kit (NEN Du Pont, De Nemours, Cologno Monzese, Italy). The cross-reactivates of the anti-PGE2 antiserum with other prostanoids are < 1% with the exception of PGE1 = 3.7%. The limit of sensitivity for the PGE2 is 1.3 pg/ml. The intra-assay coefficients of variation range from 2% to 7% for PGE2 assay.
Statistical analysis
Results and calculations were done blindly by two different operators. The SAS System was used for the statistical analysis of data. Paired comparison t-test procedure was used to reveal differences between pretreatment and post-treatment values. The data were reported as mean ±
Results
NO synthesis and release by peripheral monocytes
Since NO is a lipid and water-soluble radical gas that reacts in water with oxygen to yield other radicals, we assessed NO synthesis and release by monocytes by measuring supernatant nitrite concentration (NO2
−). Monocytes obtained from both patients and healthy subjects spontaneously release ‘in vitro’ detectable amounts of NO. Moreover, monocytes obtained from MWA patients release in 24 h culture medium a significant higher amount of nitrite than monocytes of healthy subjects (91.25 ± 8.6 μ
Monocyte stimulation with 5 μ
Individual values of monocyte nitrite release in migraine without aura (MWA) patients (n = 10) and healthy controls (n = 8)
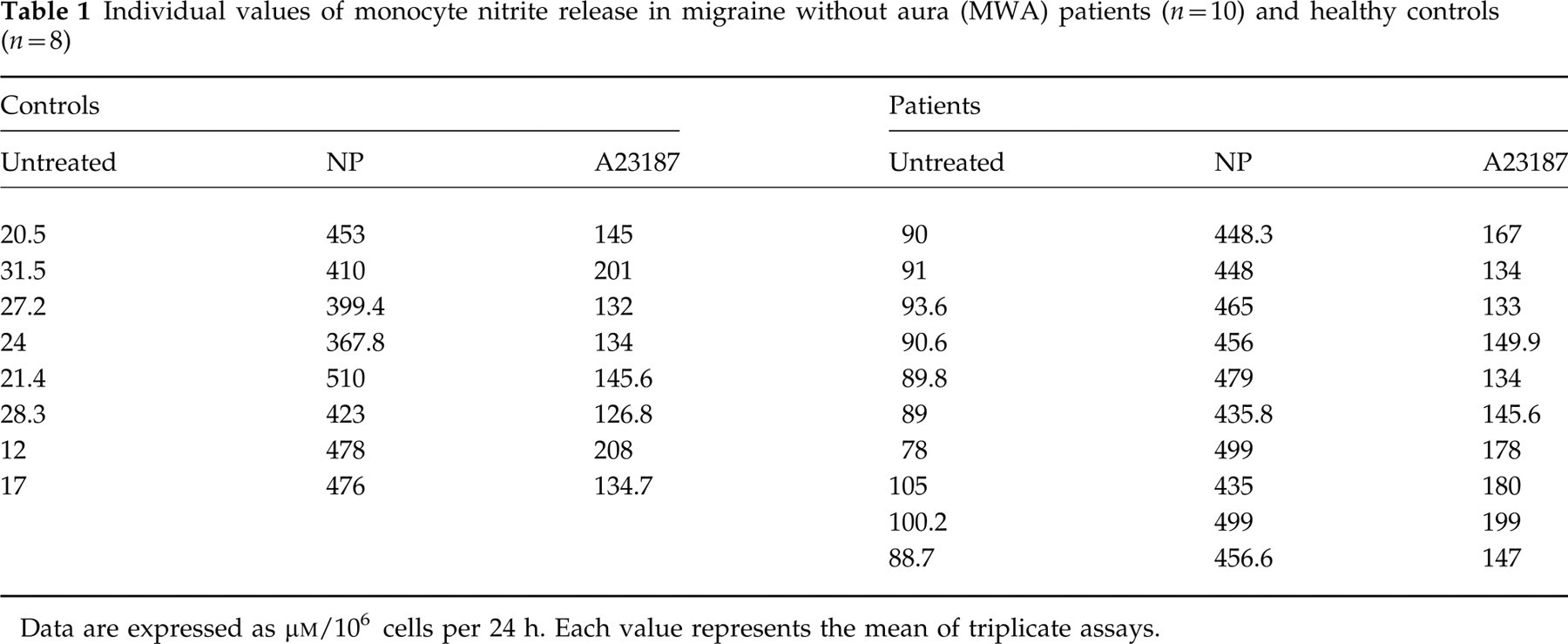
Data are expressed as μ
Interestingly, no significant difference between patient and healthy monocytes was detectable after calcium ionophore and sodium nitroprusside stimulation: in fact cells of both groups under study released NO derivates at the same level (Fig. 1).
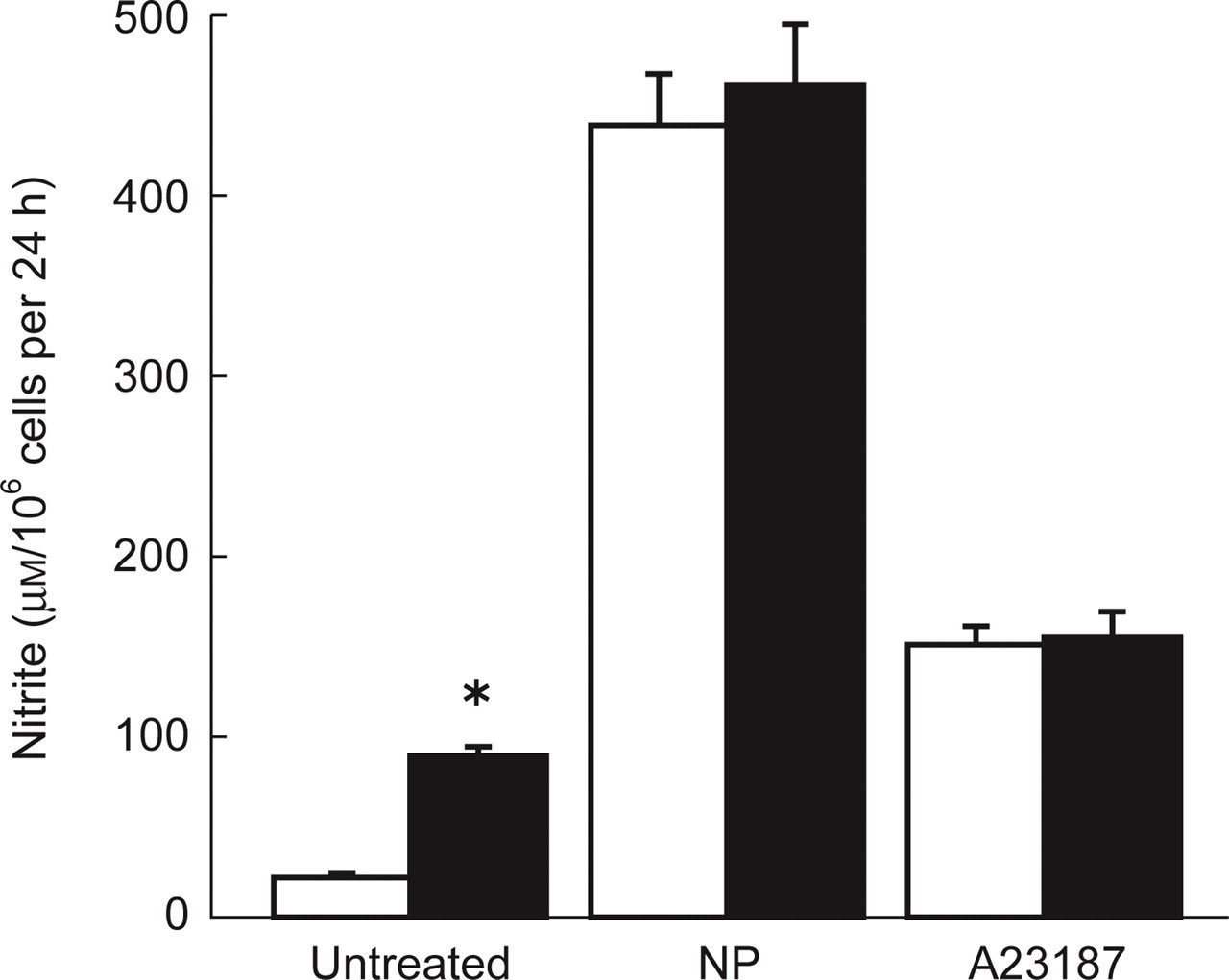
Nitrite accumulation in culture supernatants of monocytes obtained from healthy subjects (control group, □) and migraine patients (▪). Monocytes were incubated for 24 h at 37°C with culture medium alone (untreated) or with 0.4 m
PGE2 synthesis and release by peripheral monocytes
PGE2 synthesis and release from monocytes of both groups under study was assayed in the supernatants of 24 h incubation. Monocytes obtained from MWA patients showed a markedly higher release than healthy subjects (3137 ± 320 pg/106 cells vs. 1531 ± 220 pg/106 cells) (Table 2).
Individual values of monocyte prostaglandin E2 (PGE2) release in migraine without aura (MWA) patients (n = 10) and healthy controls (n = 8)
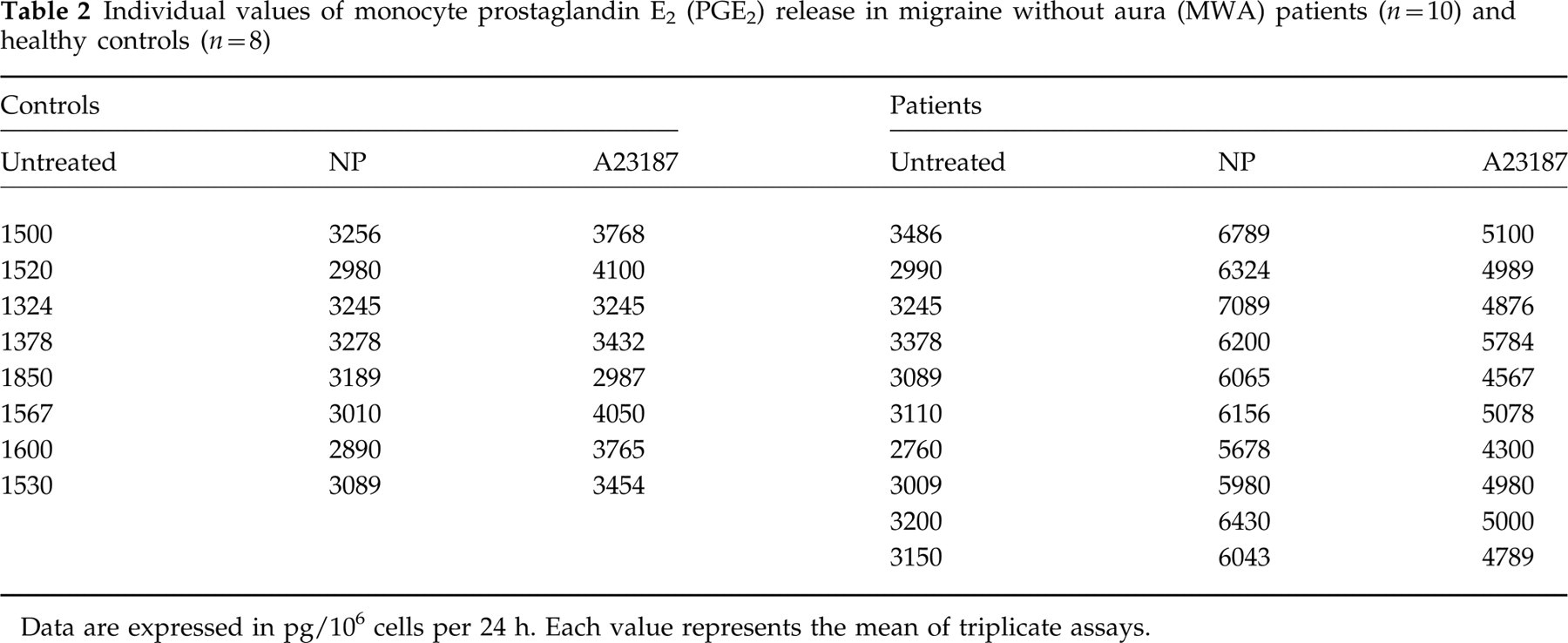
Data are expressed in pg/106 cells per 24 h. Each value represents the mean of triplicate assays.
Stimulation with 5 μ

Prostaglandin E2 (PGE2) synthesis and release by monocytes obtained from healthy subjects (control group, □) and migraine patients (▪). Monocytes were incubated for 24 h at 37°C with culture medium alone (untreated) or with 0.4 m
Effect of L-NAME on nitrite and PGE2 release by monocytes
Incubation of monocytes of both healthy subjects and migraine patients with different concentrations (20–100–300 μ

Nitrite accumulation in culture medium of monocytes obtained from healthy subjects (□) and migraine without aura (MWA) patients (▪) after incubation for 24 h with different concentrations of N-nitro-
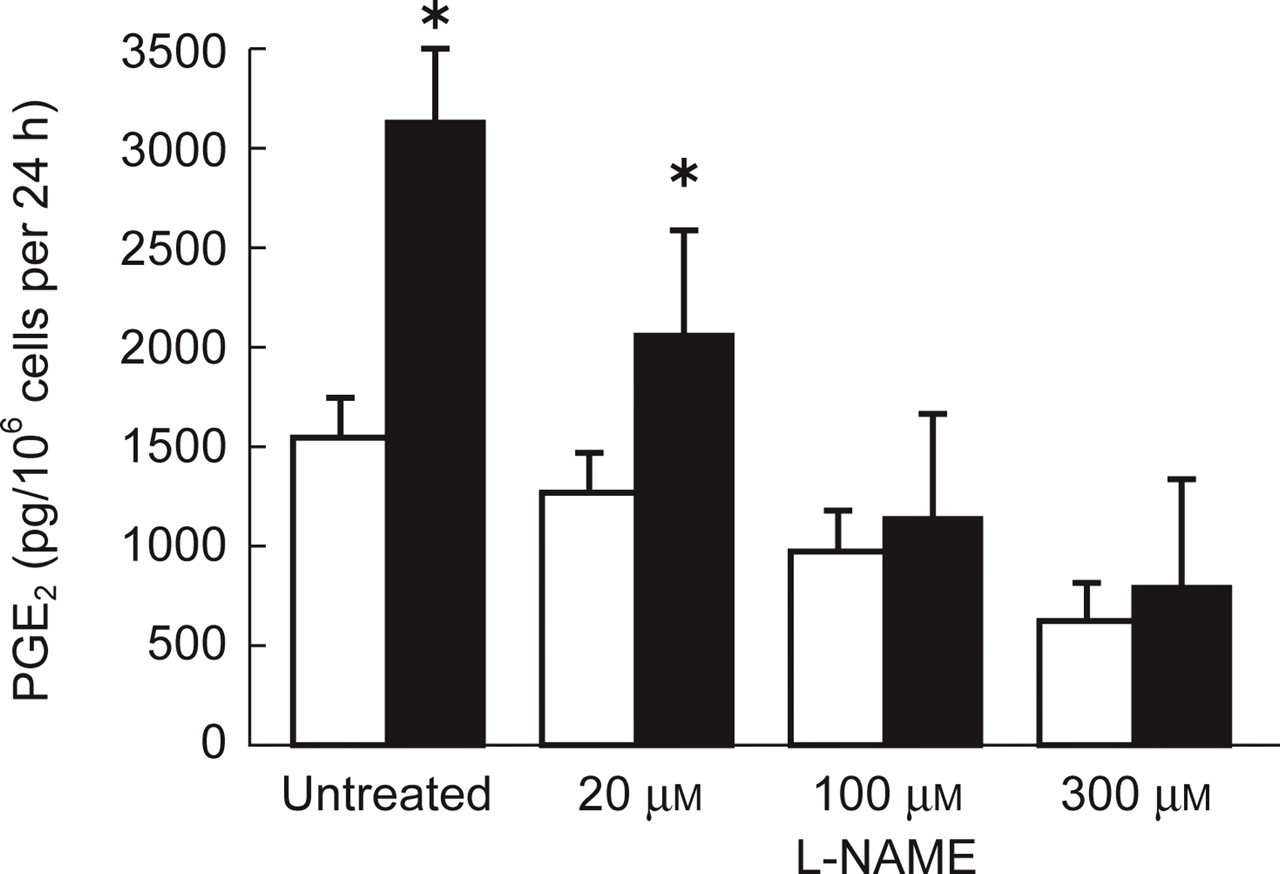
Prostaglandin E2 (PGE2) synthesis and release by monocytes obtained from healthy subjects (□) and migraine without aura (MWA) (▪) patients after incubation for 24 h with different concentrations of N-nitro-
On the other hand, removal of L-NAME from cell cultures and resuspending cells in fresh medium completely restored the synthesis of both the compounds. It is interesting to note that the reduction of both NO and PGE2 release was more pronounced in monocytes of MWA patients than healthy subjects.
Discussion
Although the importance of NO synthesis and release by rodent macrophages is well established, in human monocytes/macrophages it is still controversial.
In most studies, stimulation of human macrophages with bacterial products and/or lymphokines has yielded negative results (29–32). Dugas and co-workers suggested that NO synthase could be stimulated in human monocytes by an IgE-dependent mechanism or by sequential treatments with IL-4 and interferon-gamma (IFN-γ), in some donors (33, 34). Furthermore, other studies carried out on fresh blood-stream monocytes show the release of high levels of nitrite in the culture supernatants (35). The production of NO in human monocytes/macrophages remains therefore undefined, though the antigenic NO synthases' presence is readily demonstrable.
With the present study we demonstrate not only that human monocytes spontaneously release ‘in vitro’ detectable amounts of NO, but also that in MWA patients this cell system is activated, with NO release being higher than in monocytes obtained from healthy subjects.
NO release, in both patients and healthy subjects, is further increased after stimulation with calcium ionophore or after incubation with sodium nitroprusside. This compound is a divalent anion, universally known as a NO-donor. However, it has been demonstrated that sodium nitroprusside is stable in solution, that the NO release is undetectable in absence of light and that sodium nitroprusside can be readily activated to NO by metabolism in vascular smooth muscle (36, 37).
In the light of these data we hypothesize that NO release after SNP stimulation is not only due to a spontaneous chemical degradation to NO, but also requires an active enzymatic presence.
Moreover, after SNP treatment, no difference is detectable between monocytes of the two groups under study, suggesting that NO synthesis and release cannot exceed a certain level. In fact, a larger production of NO may become self-destructive (38–40), so that is not surprising that NO production is somehow limited.
It is now clear that NO plays an important role in both central and peripheral nervous systems. NOS has been detected in all areas of the human brain, suggesting that NO plays a role in neurotransmission, as well as of either nociceptive or anti-nociceptive responses, depending on the tissue level (41). NO also seems to be involved in the regulation of the cerebral circulation (42). It is not clear how exogenous NO could be responsible for the local regulation of blood flow to the brain, as is the case of NOD migraine. However, a metabolic mechanism necessary to convert organic nitrates, such as the clinically used NOD, to biologically active forms exists mainly in the vasculature (43).
These data reinforce the hypothesis of migraine as a NO-derived disturbance. At sites of the previously hypothesized migraine ‘neurogenic inflammation’ several cell types, such as vascular smooth muscle cells, monocytes, endothelial cells and neurones, may have the capacity to generate a high concentration of inducible NO, at levels comparable to the NO released by NOD (44, 45).
It is interesting that even PGE2 release by peripheral monocytes is higher in MWA patients than in healthy subjects. This phenomenon is particularly relevant, since it is known that NO can stimulate or inhibit prostaglandin (PG) synthesis, depending on the cell type under investigation (46). PGE2 synthesis and release remain higher in patient monocytes even after stimulation with both calcium ionophore and sodium nitroprusside. These two compounds are also responsible for an increase of NO release from monocytes.
We can hypothesize that in human monocytes there is a positive feedback between NO and PGE2. Moreover, the incubation of monocytes of both groups under study with L-NAME, a compound known for its inhibiting activity on nitric oxide synthases, caused a marked decrease of both nitric oxide and PGE2 release. Therefore we suppose that the activation of COX, the enzyme leading to prostaglandin synthesis, could be mediated by nitric oxide itself.
Since activated monocytes release a higher amount of oxygen radicals (O2 –, H2O2, OH) in the same circumstances under which they make NO (47, 48), we cannot establish whether PGE2 release could even be mediated by peroxynitrite (OONO–) that is formed by the combination of oxygen radicals and NO. Moreover, ONOO– can exert even stronger pro-oxidant effects (49) and through further reaction might lead to chronic pathological conditions.
On the other hand it has been reported (50) that in a mouse monocytes/macrophages cell line RAW264.7, treated with SIN-1 (3-morpholinosydnonimine), a donor of both O2 – and NO, basal and lipopolysaccharide (LPS)-stimulated production is enhanced and such enhancement appeared to be mainly, if not exclusively, due to PGE2; in fact, an increased expression of COX2 is evident. In contrast, it has been recently demonstrated (51) that NO binding with the haeme-group of COX could result in its inactivation and in subsequent PGE2 decrease.
In the light of our data we can hypothesize that in migraine patient monocytes, COX and NOS pathways are mutually linked and probably interact with each other, and this relationship could be important in the modulation of monocyte activation. These interactions could be responsible, at least in part, for the final goal leading to the pain production in migraine. In any case, further studies are necessary to prove this hypothesis.
Footnotes
Acknowledgements
This study was supported by grants from MURST 60% 1998.
