Abstract
Headache occurs in sickle cell disease (SCD), but its characteristics and frequency have not previously been studied. Our aim was to study patterns of headache in adults with SCD and to correlate its presence with blood flow velocities measured by transcranial Doppler (TCD) and with brain magnetic resonance imaging (MRI) abnormalities. We studied 56 adults with SCD. Twenty-eight patients (50%) had severe and frequent headaches. In 20 patients (35.7%) the headache met the International Headache Society criteria for migraine without aura. Patients with frequent and severe headache presented TCD velocities significantly higher than those without headache, or with milder headache. No correlation was found between headache and abnormalities in brain MRI. A migraine-mimicking headache occurs in SCD but we should not understand it as a primary headache because the blood flow abnormalities secondary to SCD detected by TCD seem to play an important role in these patients.
Introduction
Headache occurs in sickle cell disease (SCD), but its cause, frequency and relationship to transcranial Doppler (TCD) velocities have received little attention in the literature (1). It is a matter of debate whether these patients have migraine and SCD by coincidence or if we should consider the headache as secondary to SCD. A previous study showed that patients with SCD and severe throbbing headache had a higher incidence of cerebral hyperaemia demonstrated by regional cerebral blood flow (CBF) determined by the 133xenon inhalation method (1). Headache in SCD patients may also be a symptom of a serious problem, since they have an increased incidence of intracerebral aneurysm and subarachnoid haemorrhage (2).
Complicated migraine was reported to occur with a higher frequency in people with sickle cell trait (3). Although the genesis of migraine attacks is now thought to be of neuronal origin, alterations of intracranial vessel tone may play a significant role in its pathophysiology (4, 5). Studies focusing on intracranial flow velocities measured by TCD have shown either increased or normal blood flow velocities in patients with migraine (6, 7). It is well established that TCD velocities are also increased in patients with anaemia (8). In SCD patients, high CBF and consequently increased TCD velocities could be a precipitating condition for migraine-like episodes.
The main objective of this study was therefore to determine the frequency and characteristics of headache in an adult SCD population and correlate the presence of headache to blood flow velocities measured by TCD and to abnormalities in brain magnetic resonance imaging (MRI) and intracranial magnetic resonance angiography (MRA).
Patients and methods
Patients
Patients were selected from the adult out-patient clinic of the Department of Haematology at our University Hospital. All patients with haemoglobin SS (HbSS) disease, confirmed by haemoglobin electrophoresis on cellulose acetate, and aged ≥16 years were studied. Data were systematically collected including: demographic features, the presence of stroke risk factors and past medical history of transient ischaemic attack or stroke. The patients underwent a semistructured interview guided by a questionnaire. The questionnaire sought information on lifetime occurrence of headache, location of pain, frequency of attacks, quality and severity of pain, duration of attacks, aggravation by physical activity, presence of autonomic symptoms and relief factors. Patients were asked to recall the above-mentioned features referring to their usual headaches. This information was sufficient to diagnose headache type according to International Headache Society (IHS) criteria (9). Headaches that reached ≥5 points for pain intensity on a 1–10 scale were defined as severe. Patients with at least one episode of pain per month were classified as having frequent headaches. A complete neurological examination was also performed in all patients. This protocol was approved by our ethics committee and all subjects gave written informed consent for participation in the study.
Transcranial Doppler ultrasonography
TCD exams were performed on SCD patients and reviewed by the same author (G.S.S.). If the patient had fever, pain or other acute illness, TCD was deferred until they were well. Attention was focused on the finding of the highest mean flow velocities (FVmax). In each examination FVmax was recorded in the middle cerebral artery (MCA) at depth settings ranging from 40 to 60 mm. One recording was registered in the anterior and posterior cerebral arteries. Vertebral arteries were studied at depth settings ranging from 60 to 80 mm and the basilar artery, from 80 to 100 mm. Although peak-systolic, end-diastolic and time-averaged mean velocities were read from the spectrum, only the time-averaged mean was used in this report because of its higher accuracy (10).
Magnetic resonance imaging
Brain MRI was performed according to a standardized protocol using a 1.5-T device (Sonata Maestro Class, Siemens Medical Systems, Siemens AG, Medical Solutions, Erlangen, Germany). The protocol included: axial and sagittal spin echo T1-weighted images, turbo spin echo T2-weighted images, axial FLAIR and spin echo T1-weighted images after intravenous gadolinium injection.
MRA was performed using a 3-dimensional time-of-flight technique. All portions were photographed in sequence and were large enough for the vascular anatomy to be worked out when maximal intensity projections raised questions.
MR images were evaluated by two neuroradiologists who were unaware of the TCD results. Lesions were broadly defined to include lacunar infarction, encephalomalacia, leucoencephalopathy or atrophy. A lacuna was defined as a shelled-out volume, usually in white matter, that was visible with T1-weighted, T2 weighted and FLAIR sequences. Encephalomalacia was defined as any other ischaemic change such as an infarct, which could be seen as high signal intensity by using a T2-weighted or FLAIR sequence. Atrophy was defined as loss of brain tissue volume (lobar asymmetry, open sulci or ventricular dilation). Leucoencephalopathy was defined as the degeneration or demyelination of white matter, which was seen as abnormally high signal intensity on T2-weighted or FLAIR images. MRA criteria of abnormality included stenosis or apparent occlusion of any vessel, arterial tortuosity or the presence of intracranial aneurysms. Tortuosity diagnosis was based on the presence of dilation of a vessel segment, abnormal increase in length of a vessel segment or obvious bowing of an artery (11).
Haematological studies
Blood samples were collected in EDTA from all participants at the same day of TCD examination. Haematological studies (haemoglobin level, haematocrit, red blood cell, reticulocyte, platelet and white blood cell counts) were performed with a Sysmex E Series (TOA Medical Electronics, Kobe, Japan).
Statistical analysis
Descriptive analyses, including means, standard deviations, medians and interquartile ranges were used to describe patient characteristics. The independent samples t-test was used to compare means between groups for continuous variables. A two-tailed P < 0.05 was considered statistically significant. Dichotomous variables were compared with a χ2 or Fisher’s exact test. Univariate analyses of variables associated with increased TCD velocities and with headache status were performed using linear regression. We estimated the effect of haematocrit on FVmax by multiple linear regression. A baseline model was fitted for FVmax and our possible confounders of age, sex, the presence of intracranial stenosis on MRA and headache status (having severe and frequent headaches or not). Statistical analysis was performed with SPSS 10.0 software (SPSS Inc., Chicago, IL, USA).
Results
Fifty-six adult patients with SCD (mean age 27 ± 10.09 years, range 17–55 years) were studied. Forty-one patients (73.2%) reported headaches. Headache was more common in women than in men (P = 0.035). Haematocrit was similar between the patients with and without headache. Twenty-eight patients (50%) had severe and frequent headaches. The most frequent pain location was frontal (48.8%). Twenty-four patients described their pain as throbbing (58.5%). Nausea was present in 18 patients (43.9%), photophobia in 20 (51.2%) and phonophobia in 24 patients (58.5%). In 20 patients (37.5%) the headache met the IHS criteria for migraine without aura (9). None of the patients had aura. The most frequent treatments used by our patients for headache relief were: dipirone (43.9%), anti-inflammatory drugs (17.1%), paracetamol (12.2%), opioids (7.3%), ergotamine (2.4%) and non-pharmacological measures (17.1%). Only two patients were regularly followed on the headache clinic of our hospital and were headache free using prophylactic therapy (one patient was using flunarizine and the other, amitriptiline) Table 1 summarizes the characteristics of patients with and without headache.
Characteristics of sickle cell disease adult patients with and without severe and frequent headaches

Twenty-six patients in the group without or with mild headache underwent brain magnetic resonance imaging (MRI)/magnetic resonance angiography (MRA) and 24 patients in the group with severe and frequent headache.
Fifty patients underwent MRI and MRA (two patients were not studied because of claustrophobia and four because of metal implanted prostheses). The overall frequency of lacunar infarction, encephalomalacia, leucoencephalopathy or atrophy was 60%. Leucoencephalopathy was the most common finding (48%). MRA was abnormal in 36 patients (72%) and intracranial stenoses were observed in 16% of the patients. Intracranial aneurysms were not observed. There was no correlation between headache and the presence of MRI/MRA abnormalities (see Table 1).
The 28 patients with frequent and severe headaches presented TCD velocities significantly higher than patients without or with milder headaches (P = 0.035) (Fig. 1). The only variables associated with headache status (having severe and frequent headaches) were sex (female) and higher TCD velocities.
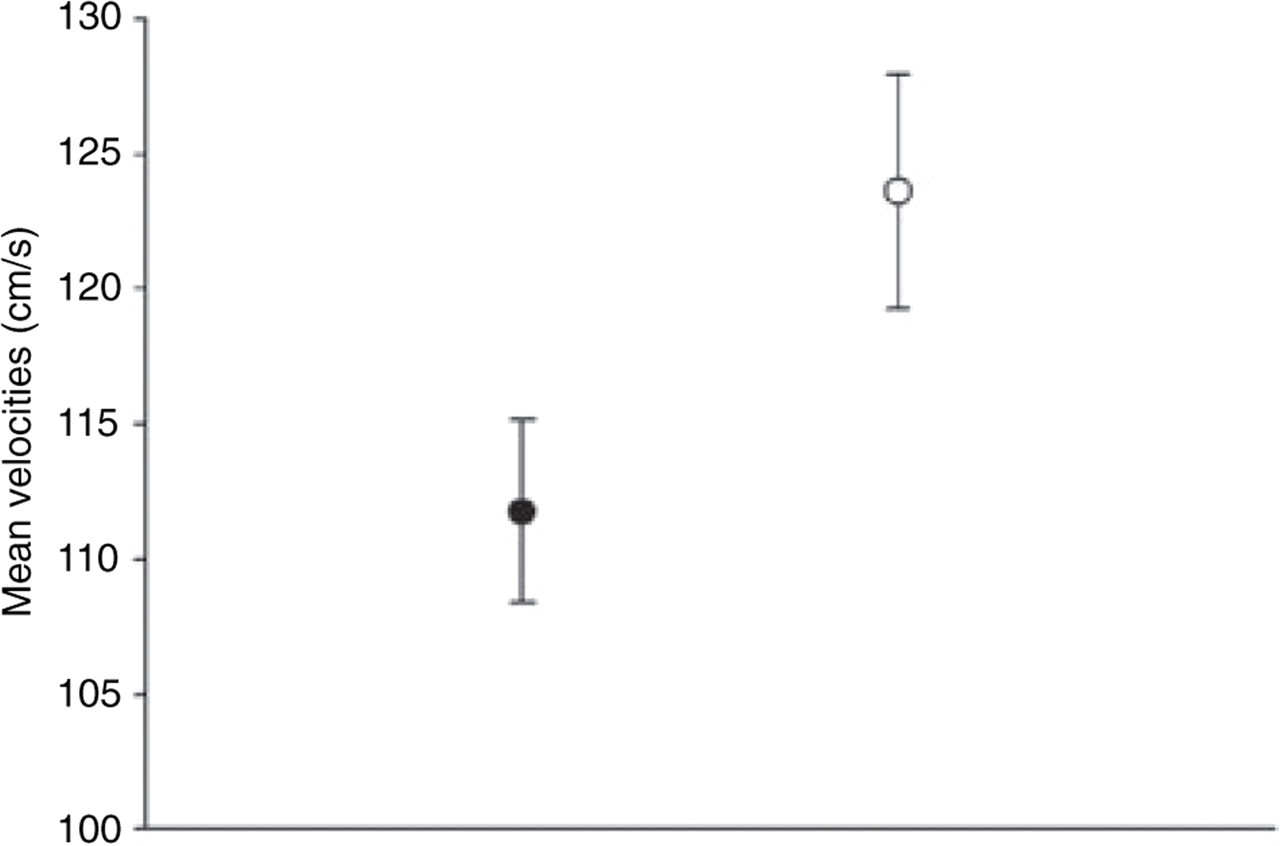
Patients with severe and frequent headaches (s) had significantly higher blood flow velocities (cm/s) than patients without or with milder headaches (•). Data are presented as mean ± SE.
Predictors of TCD velocity were investigated by examining the relationship of age, the presence of intracranial stenosis on MRA, haematocrit, reticulocyte, white blood cell and platelet counts to FVmax. Haematocrit was negatively correlated with FVmax in both groups (P < 0.05). Age, the presence of intracranial stenosis on MRA, reticulocyte, white blood cell and platelet counts were not correlated with TCD velocities. In a multivariate model that included haematocrit, age, sex and headache status (having severe and frequent headaches or not), significantly higher values of FVmax were associated with lower haematocrit and the presence of headache.
Discussion
Our findings indicate that high blood flow velocities in cerebral arteries correlate with the occurrence of headache in SCD patients. Although TCD velocities are not the measurement of CBF, they can reflect flow volume if the angle of insonation remains constant, the perfused territory remains the same and major arterial abnormalities are excluded by neuroimaging as in our patients (12). Blood flow velocities in MCA were directly related to CBF (measured by the 133xenon inhalation method) in a group of 23 patients with SCD (13). There are many factors influencing CBF, the main factor being tissue requirements for oxygen and glucose delivery (14). Haematocrit is a major determinant of oxygen delivery. Severe anaemia causes a measurable increase in total CBF. Once the haematocrit is below 30% there is a reliably detectable increase in CBF and consequently in blood flow velocities. SCD patients have a haematocrit of 18–25%, so their CBF is higher than that of age-matched patients with normal haematocrit (15). The increased CBF observed in patients with SCD may function as a trigger for migraine, explaining the high frequency of headache in our patients (1).
Other conditions that associate cerebral hyperperfusion and headache are: hypertensive encephalopathy, eclampsia and preeclampsia syndromes, post-endarterectomy hyperperfusion syndrome, high-altitude mountain sickness and post arteriovenous malformation (AVM) treatment (16). In the first two situations hyperperfusion is believed to result from the failure of intracranial vessels to compensate adequately for rapid increases of blood pressures greater than those to which the vascular system is normally exposed (17, 18). After treatment of high-grade carotid stenosis or large intracranial AVMs a regional cerebral oedema seems to occur (19, 20). In high-altitude cerebral oedema a combination of factors tends to produce a hyperperfusion condition. Hypoxaemia associated with exposure to high altitudes results in cerebral vasodilation. An activation of the sympathoadrenal axis and secondary hypertension also seem to play a role in high-altitude mountain sickness (21). Fever-related headache can also be associated with dynamic changes in CBF due to shifts in carbonic oxide pressures as the respiratory centres participate in the thermoregulatory reflexes of fever. Pyrogens also induce increases in arachidonic acid metabolites such as the cyclooxygenase-derived thromboxane, prostaglandins and prostacyclin. Prostagladin E2 has vasoactive properties and could be indirectly implicated in any vascular component of fever-associated headache (22). All these situations occur after an acute loss of the autoregulation capacity of CBF. This is not the case in patients with SCD, because they have a permanent and chronic increase in CBF.
A study of migraine prevalence in the USA showed that 17.6% of females and 5.7% of males had one or more migraine attacks per year. In this study migraine prevalence varied considerably by age; it was highest between the ages of 35 and 45 years (23). The frequency of headaches fulfilling migraine criteria in our patients was much higher (37.5%). Structural disease was excluded in our patients, since headache did not correlate with MRI and MRA abnormalities, especially the presence of arterial stenosis since high TCD velocities could be associated with an intracranial vasculopathy that can occur in SCD patients (10, 11).
Sickling causing formation of aggregates of erythrocytes in the microvasculature may lead to local stasis, hypoxia and contribute to worsening the neurogenic inflammation that occurs in migraine. On the other hand, vasoconstriction during a migraine attack can disrupt the steady-state flow of cerebral arteries and induce sickling. As both migraine and SCD are risk factors for stroke in the young adult population, the coexistence of these two disorders may be a cause for concern (24).
Various modes of functional neuroimaging have given insight into the pathophysiology of migraine headache. Studies using 133xenon, TCD and SPECT suggest an initial hypoperfusion state during migraine aura, typically localized in the posterior regions of the cortex. The hypoperfusion state is followed by a prolonged period of hyperperfusion attributed to vasodilation and inflammation of blood vessels (5, 25, 26). Our patients seem to have a chronic elevated CBF and this could be a permanent trigger for migraine-like episodes.
TCD velocities were reported to be either increased or normal in headache-free migraineurs. A decrease in the cross-sectional area of a vessel near to the point of insonation or alterations at the level of cerebral arteriole affecting regional CBF are possible explanations for increased blood flow velocities in migraine. A difference between migraineurs with and without aura regarding cerebrovascular vasoreactivity has also been reported (6, 7). Although migraineurs have been extensively studied with TCD, the method is considered an investigational test for migraine at the present time (27). The higher TCD velocities observed in our patients with frequent and severe headache can be explained by the presence of a decreased haematocrit in migraine-prone subjects.
It is a matter of debate whether our patients had migraine and SCD, or if we should consider the headache as secondary to SCD. We think that a migraine-mimicking headache can occur in SCD but we should not understand it as a primary headache because CBF hyperaemia secondary to SCD and increased TCD velocities seem to play an important role in these patients, but not in migraine. We suggest that a SCD-associated headache be included in the IHS classification as a cause of secondary headache.
Footnotes
Acknowledgements
The authors thank Dr Paulo Monzillo for his expert comments and advice.
