Abstract
Background
Altered neurovascular coupling in migraineurs could be a consequence of impaired function of modulatory brainstem nuclei. The cold pressor test (CPT) should activate brainstem structures. We measured visually evoked cerebral blood flow velocity response (VEFR) to CPT in migraine.
Methods
Twenty-three healthy volunteers and 29 migraineurs participated in the study. We measured arterial blood pressure, end-tidal CO2, heart rate and cerebral blood flow velocity in posterior and middle cerebral artery using transcranial Doppler. VEFR was calculated as cerebrovascular reactivity to photic stimulation before, during and after CPT.
Results
In healthy individuals, there was a significant decrease in peak systolic VEFR from CPT phase to recovery phase (
Conclusions
The absence of the effect of CPT on VEFR in migraine is likely to be a consequence of impaired subcortical modulation of neurovascular coupling.
Keywords
Introduction
In recent years, migraine has been viewed as a neurovascular disorder (1). Neuroradiological studies have shown that patients with migraine have an increased incidence of ischemic-like lesions, especially in the territory of posterior circulation (2). Clinical studies have suggested that migraine is an independent risk factor for ischemic stroke (3). The etiology of ischemic-like lesions in migraine is debated; nevertheless, the impairment of cerebrovascular regulation seems to be an important issue in migraine pathophysiology (4).
Single-photon emission tomography and magnetic resonance imaging studies have shown interictal perfusion changes in patients with migraine (5,6). Additionally, increased interictal cerebrovascular reactivity in patients with migraine has been shown by functional transcranial Doppler, a method with good temporal resolution (7). It has been proposed that altered cerebrovascular reactivity to metabolic demand of active brain tissue in migraineurs is a consequence of impaired function of modulatory brainstem nuclei, which are an important source of perivascular innervation of cerebral microvessels (8). Additionally, impaired function of brainstem structures might also cause dysfunctional pain processing in migraineurs (9). In animals, neurovascular coupling (NVC) is affected by tonic painful stimuli, which in principle activate brainstem structures (10,11).
Our previous study has shown that visually evoked cerebral blood flow velocity (CBFV) response (VEFR) is increased during the cold pressor test (CPT) in healthy humans (12). This finding supports the assumption that during tonic pain NVC is modulated by activated subcortical structures. In view of this finding, we have extended our study to measure VEFR during tonic pain stimulus in patients with migraine. We aimed to substantiate the hypothesis that in patients with migraine the function of subcortical structures, activated during tonic pain, which are also involved in modulation of NVC, is impaired.
Participants and methods
Participants
Descriptive data on patients with migraine.

F: female; M: male; MoA: migraine without aura; MA: migraine with aura; NSAID: nonsteroid anti-inflammatory drug; OCP: oral contraceptive pill; THRT: thyroid hormone replacement therapy.
Experimental design
All measurements were made between 7:00 and 10:00 in the morning. Participants were instructed not to drink caffeine or smoke cigarettes at least 12 hours prior to the examination. The examination room was dark, quiet and warmed to air temperature of 25℃. During the testing individuals were comfortably placed in the supine position on the examination table with head and upper body elevated 45 degrees. All examinations were performed by the same examiner.
Arterial blood pressure (ABP) was recorded continuously on the right hand using Finapres 2300 blood pressure monitor (Ohmeda, Englewood, CO, USA). Heart rate (HR) was recorded with electrocardiography (ECG) using second standard limb lead. End-tidal CO2 (Et-CO2) levels were monitored through a facemask using Oscaroxy capnometer (Datex, Helsinki, Finland). The left arm was free of any measurements and was used in CPT. Participants were asked not to move during the testing and any additional sensory stimuli were painstakingly avoided.
CBFV was measured using two 2 MHz Delica EMS-9U transcranial Doppler ultrasound (TCD) probes (Shenzen Delicate Electronics, Shenzen, China) attached to an individually mounted headpiece to ensure a fixed position during measurements. A temporal acoustic window was used. On the right side the P2 segment of the posterior cerebral artery (PCA) and on the left side the middle cerebral artery (MCA) was insonated. The identification of the vessels followed standard criteria described elsewhere (14).
As a stimulus paradigm, a reversed-pattern checkerboard with inverting frequency of 2 Hz was used, presented on a 90 cm × 120 cm rectangular canvas, placed in front of the participant at a viewing distance of 1.5 meters. Before the recording was started, there was a 10-minute adaptation period with the visual stimulus displayed. The recording protocol consisted of 20 consecutive cycles with a resting period of 20 seconds (OFF period) and stimulation period of 30 seconds (ON period). During the OFF period, participants were instructed to close their eyes; during the ON period, individuals were instructed to fix their eyes on a small red cross in the middle of the canvas displaying a reversed-pattern checkerboard. The changes between periods were signaled automatically by acoustic tones, which also served as triggers for later averaging procedures.
A mixture of water and ice in a plastic container was used for tonic pain testing. The temperature of the mixture was between 3 and 4℃. The participant’s hand was submerged down to the wrist into the ice-cold water at the beginning of the ninth OFF period and removed at the end of 12th ON period. It was then dried and wrapped in a paper towel during further recording.
Data analysis
Analog signals were sampled at a frequency of 5000 Hz. One recording was divided into three phases: the basal phase consisted of the first eight OFF/ON cycles, the CPT phase consisted of the following four OFF/ON cycles during CPT and the recovery phase consisted of the last eight OFF/ON cycles. Mean values of ABP, HR and Et-CO2 were determined over the whole length of one OFF/ON cycle and then averaged across all cycles in a particular phase.
The envelope curve of the CBFV signal was used to obtain peak systolic (PSV), end-diastolic (EDV) and mean (MV) velocities in the PCA. VEFR vas calculated using equation 1:
The recording of heart rate (HR), end tidal carbon dioxide (end-tidal CO2), mean arterial blood pressure (BP), mean arterial velocity in posterior cerebral artery (vPCA) and mean arterial velocity in middle cerebral artery (vMCA) before, during and after cold pressor test (CPT) in a patient with migraine.

Statistical methods
The normality of the distribution of data sets was tested using the Kolmogorov-Smirnov test with Lilliefors’ correction. When a data set showed a normal distribution, one-way repeated-measures analyses of variance (ANOVAs) with Fisher post hoc test were used to compare the means of measurements in the basal, CPT and recovery phases or to compare the means of interphasal differences of the measurements between the two groups of participants. When a data set showed a non-normal distribution, one-way repeated-measures ANOVAs on ranks with Student-Newman-Keuls post hoc test were used to compare the medians of measurements in the basal, CPT and recovery phases or to compare the medians of interphasal differences of the measurements between the two participant groups. Statistical significance was set at
Results
Systemic data
Arterial blood pressure (ABP), heart rate (HR) and end-tidal carbon dioxide (Et-CO2) data for basal, CPT and recovery phase in healthy subjects and migraineurs.

Average mean values of the measurements are shown with their corresponding standard deviations. CPT: cold pressor test.
Resting CBFV data
Average resting peak systolic (PSV), mean (MV) and end-diastolic velocities (EDV) with their corresponding standard deviations are shown for consecutive phases for healthy subjects and patients with migraine.
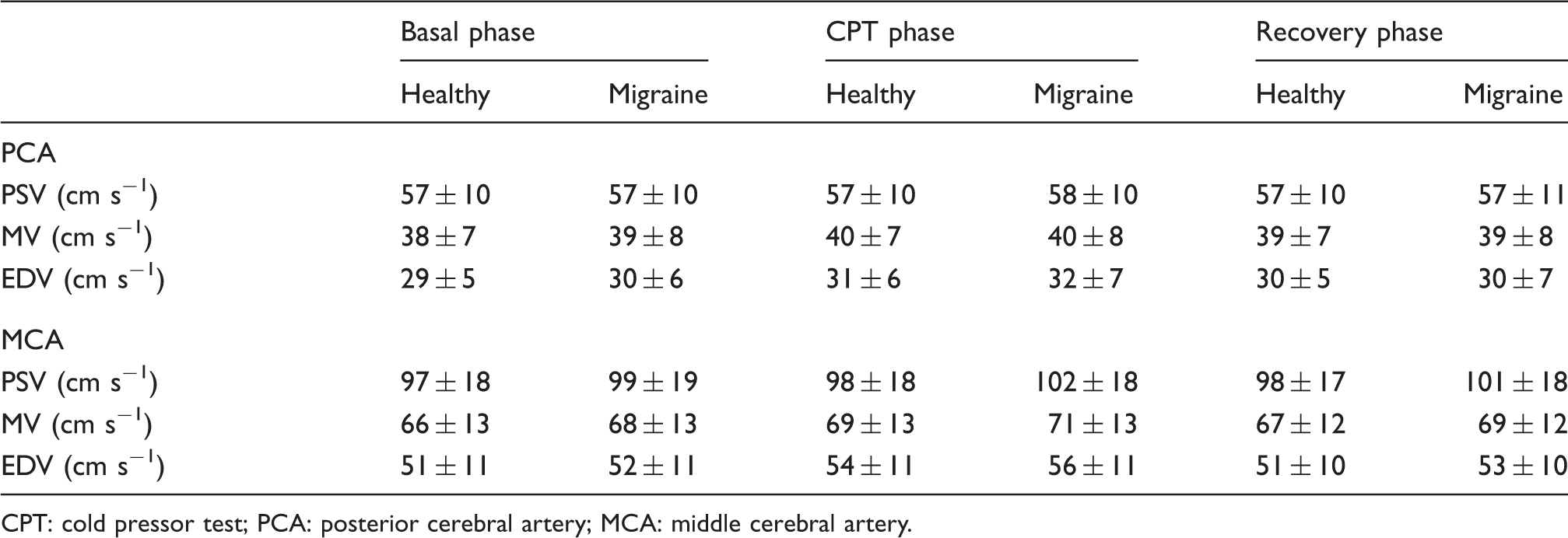
CPT: cold pressor test; PCA: posterior cerebral artery; MCA: middle cerebral artery.
In healthy individuals, there was a statistically significant increase in resting MV and EDV in the CPT phase compared to basal and recovery phase in MCA (
In patients with migraine, there was a statistically significant increase in resting PSV in the CPT phase and in recovery phase compared to basal phase in the MCA (
Evoked CBFV data
Averages of VEFR in consecutive experimental phases of participants are given in Table 4 and Figure 2 for each group of participants. Statistically significant differences between phases are also shown in Figure 2. The comparison of differences in consecutive experimental phases between both groups is shown in Table 5.
The histogram of visually evoked flow responses (VEFR) for (a) peak systolic velocities (PSV), (b) mean velocities (MV) and (c) end-diastolic velocities (EDV) in the posterior cerebral artery (PCA) in healthy individuals (left) and patients with migraine (right). The VEFR is calculated as the percentage increase in the average velocity during the last 10 seconds of the ON period (visual stimulation) compared with the last five seconds of the preceding OFF period (resting). In the basal and recovery phases, eight consecutive cycles were averaged, whereas four consecutive cycles were averaged in the cold pressor test (CPT) phase. *Denotes statistically significant difference between selected phases ( Average peak systolic, mean and end-diastolic visually evoked flow responses (VEFR) in consecutive phases are shown for healthy individuals and patients with migraine with their corresponding standard deviations. CPT: cold pressor test. The comparison of differences in visually evoked flow response (VEFR) in consecutive phases between healthy individuals and patients with migraine. Average mean differences are shown with corresponding standard deviations. NS: not statistically significant (


In healthy individuals, there was a statistically significant decrease in peak systolic VEFR from CPT phase to recovery phase (
Additionally, the differences in consecutive phases in mean and end-diastolic VEFRs between the basal phase and the CPT phase and between the CPT phase and the recovery phase were statistically significantly higher in healthy individuals than in patients with migraine (
Discussion
The main finding of this study was that the difference in VEFR between the basal phase and the CPT phase was statistically significantly smaller in patients with migraine compared to healthy subjects. The effect was statistically significant in mean and end-diastolic VEFR, whereas in peak systolic VEFR there was a statistically nonsignificant trend toward the difference. Moreover, while in healthy human subjects VEFR increased in the CPT phase compared to the basal phase, there was no statistically significant change in patients with migraine.
In our study, basal-phase VEFR was higher in patients with migraine compared to healthy individuals, but the difference was statistically insignificant. Similarly, other studies using TCD have demonstrated increased visually stimulated PCA blood flow velocities in patients with migraine interictally (7,15,16). The explanation of this finding is not straightforward. In principle, increased cerebrovascular reactivity to visual stimulation in patients with migraine could be due to increased cortical excitability and/or impaired NVC.
The results of studies evaluating cortical excitability in patients with migraine are equivocal (17,18). VEP amplitudes were normal in most studies (7); on the other hand, some studies have shown increased or decreased interictal VEP amplitudes in patients with migraine (19,20). The most consistent finding was the absence of normal evoked potential habituation (21). In contrast to healthy participants, in patients with migraine habituation of VEP is inversely proportional to the amplitude of the first stimulation block, which points to a decreased level of preactivation excitability rather than hyperexcitability (22). Consistent with these findings is the assumption that the increase in interictal VEFR in patients with migraine is most probably not due to increased cortical excitability, but more likely due to impaired NVC. Correspondingly, it has been recently shown that VEFR is increased in patients with migraine compared to healthy humans, while there was no difference in simultaneously measured VEP amplitude (7).
In our study, the difference in VEFR between the basal phase and the CPT phase was smaller in patients with migraine compared to healthy individuals. It is unlikely that VEFR reached maximal values in the basal conditions in patients with migraine, rendering VEFR unresponsive to tonic pain, since a previous study has shown that an increasing visual contrast increases VEFR similarly in patients with migraine and healthy humans (7). Therefore, we can assume that the lack of difference in VEFR between the basal phase and the CPT phase in patients with migraine in our study is either the consequence of impaired function of subcortical structures that are involved in modulation of NVC (9) and/or impaired function of the effector of such modulation, i.e. the neurovascular unit (8).
It has been shown that CPT abolishes normal VEP habituation in healthy humans, whereas CPT has no effect on abnormal VEP habituation in patients with migraine (23). This is suggested to be due to impaired function of brainstem serotoninergic projections (24). Accordingly, interictal concentrations of serotonin and its metabolites are decreased in the plasma of patients with migraine (25). Also, a study using positron-emission tomography (PET) in patients with migraine interictally has shown that there is an increase in serotonin 1 A receptor antagonist binding in the posterior parieto-occipital, temporal and limbic cortex, suggesting decreased serotonin concentration in these areas (26). Interestingly, the habituation of VEP normalizes during a migraine attack (27). It is known that during a migraine attack there is activation of the dorsal rostral brainstem area that could correspond to serotoninergic nucleus raphe dorsalis and that at the same time the level of serotonin in the brain decreases (28). On the other hand, serotoninergic projections play an important role in the regulation of NVC (29). Therefore, the lack of effect of tonic pain stimulus on VEFR in patients with migraine in our study might be due to an impaired function of subcortical modulatory structures.
The increased (although not statistically significant) basal VEFR amplitudes in our present study accord with the possibility that the lack of effect of tonic pain stimulus on VEFR in patients with migraine is (at least partially) due to the dysfunction of the neurovascular unit. A study on mice and rats has shown that the pathophysiological substrate of migraine aura, the cortical spreading depression (CSD), is preceded by a wave of arteriolar dilation that propagates with a higher velocity than CSD and reaches areas of the cortex beyond the spread of CSD (30). Furthermore, CSD is accompanied by dissociation of cortical microvessel vasomotor activity from neuronal metabolic demands (31). A recent TCD study has shown that cerebrovascular reactivity to l-arginine in the territory of the PCA is decreased in patients with migraine compared to healthy individuals (32).
The differences in resting period ABP, HR and CBFVs in MCA and PCA between healthy participants and patients with migraine in our present study were statistically insignificant, as are their interphasal differences. This finding accords with previous studies (33). Animal experiments have shown that the increase of ABP during the CPT is related to the activation of the intermediolateral column of the thoracolumbar spinal cord (IML) (34). Most of the excitatory input to the IML comes from the rostral ventrolateral medulla (RVLM) (34,35), although some excitatory input also comes from more rostral subcortical structures (34). For instance, animal studies have shown that the activation of the locus coeruleus (LC) can increases ABP either through α1-adrenergic mediated activation of the IML or through activation of the hypothalamic paraventricular nucleus (36). On the other hand, LC activation can decrease ABP through α2-adrenergic-mediated inhibition of the RVLM, although this effect is normally minimal (36). The activation of the LC is also responsible for the increase in HR during the CPT through the inhibition of the parasympathetic nucleus ambiguus (34,35). Comparable responses of ABP and HR between healthy participants and patients with migraine in our present study implicate a preserved function of subcortical structures involved in the increase of ABP and HR in patients with migraine.
We have found no statistically significant difference in proportion of women using the
A similar study could be performed to test whether a migraine-prophylactic therapy normalizes VEFR during tonic pain. The experimental procedure therefore offers a future possibility of measuring an effect of a prophylactic therapy through physiological response in the clinical setting. The findings of our study also support the neural (as opposed to vascular) theory of migraine as well as appropriate etiological therapeutic interventions that would be aimed at normalizing dysfunctional subcortical structures.
Our present study is somewhat limited by the fact that so far we have no technical possibility of a direct view of subcortical neuronal network activation during tonic pain stimulus. Regrettably, it gives no data on the activity of cortical neurons during visual stimulus. Further imaging studies with the use of advanced technology are needed to address these issues.
In conclusion, the lack of effect of tonic pain on VEFR in patients with migraine is likely to be a consequence of impaired subcortical modulation of NVC and/or altered reactivity of cerebral microvessels.
Clinical implications
Migraine is associated with impaired neurovascular coupling during tonic pain. Normalization of subcortical nuclei function might be an important therapeutic goal in migraine. Our experimental procedure might be used to evaluate the effect of migraine prophylactic therapy.
Footnotes
Funding
This research was partly funded by Slovenian Research Agency.
Conflict of interest
None declared.
