Abstract
Interictal cerebrovascular reactivity and blood flow velocities were tested in 23 patients with migraine without aura and 10 age- and sex-matched healthy controls by using the breath holding index (BHI). The mean systolic, diastolic and mean velocities and pulsatility indices were not different in the controls and patients. The BHI was found to be significantly greater (P = 0011) in the patients (1.64 ± 0.33) compared with the controls (1.26 ± 0.37), showing an exaggerated reactivity to hypercapnia in migraineurs. Reactivity to pCO2 theoretically depends on pre-existing arteriolar tone and thereby on baseline velocity. Our finding of similar blood flow velocities in controls and patients suggests that the underlying cause for this high reactivity may not be an increased vasotonus but an increased sensitivity to changes in blood CO2 levels.
Keywords
Introduction
Although the pathophysiology of migraine still remains unknown there are many studies showing that changes in the diameter of intracranial vessels play an important role in migraine headache. Transcranial Doppler ultrasonography (TCD) is a non-invasive, cheap and simple method for determination of cerebrovascular reactivity, which reflects the capacity of the cerebral resistance vessels to react to changes of arterial CO2 partial pressure with dilatation or constriction, thus showing their residual autoregulatory capacity (1, 2).
The results of TCD studies in migraine reveal conflicting results. Studies focusing on intracranial flow velocities interictally have shown either increased (3, 4) or normal (5) blood flow velocities (BFV) in patients with migraine. Results of studies measuring cerebrovascular reactivity (CVR) in migraine interictally have also been contradictory. Most studies done on migraine without aura (MWOA) or migraine with aura (MA) showed a normal or increased CVR (6–9), but studies showing a reduced vasodilatory response also exist (10–12). The discrepancies in these results can be explained by various factors, including time of measurement with regard to the last migraine attack, inclusion of both MA and MWOA as ‘migraine’ or differences in techniques used for measuring CVR (i.e. hypercapnia, hyperventilation or acetozolamide stimulation).
In this paper we report the results of interictal TCD examination of middle cerebral artery (MCA) BFV, pulsatility indices (PI) and CVR in a group of patients with MWOA.
Subjects and methods
Twenty-three patients (22 women and 1 man) with MWOA diagnosed according to the criteria of the International Headache Society (13), who were completely healthy otherwise and had not used any prophylactic medication for 3 months, were recruited from the neurological outpatients clinic of the Akdeniz University Medical School Hospital and included in the study. Their mean age was 35.1 ± 9.1 years. Attack frequency, duration and severity were assessed by a headache diary the patients filled in. Headache intensity was rated from 0=none to 4=severe. Headache characteristics are given in Table 1. All patients gave informed consented.
Demographic data and headache characteristics in the patient and control groups
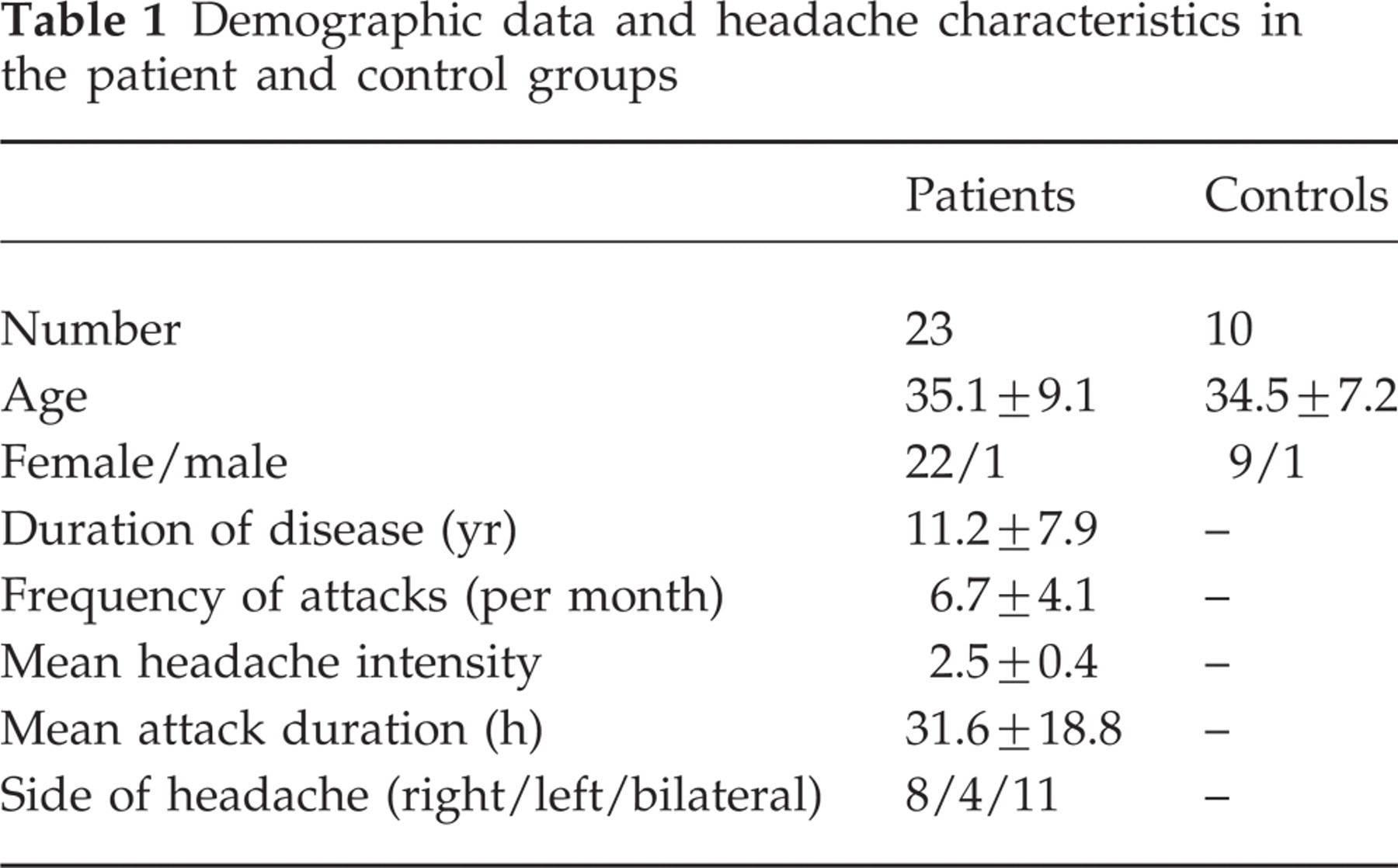
Ten age- and sex-matched healthy controls (nine women and one man) with no history of headache were selected from the hospital staff. The mean age of the control group was 34.5 ± 7.2 years.
TCD measurements were done with a Multidop X-4 TCD instrument (DWL Elektronische Systeme GmbH, Sipplingen, Germany) by using Spencer Headset monitoring equipment and two dual 2-MHz transducers placed on the temporal bone window. Both MCAs were insonated simultaneously at a depth of 40–50 mm and the same depth was used for each patient on the right and left sides.
Baseline systolic (Vs), diastolic (Vd) and mean BFV (Vm) and PI measurements were made after a period of 3 min of normal room air breathing. The PI, which is an useful tool showing the resistance of the distal vessels, was calculated by the formula PI=(Vs−Vd)/Vm automatically by the Doppler device. Vascular reactivity to hypercapnia was measured by using the breath holding index (BHI). This index is obtained by dividing the percentage increase in mean flow velocity occurring during breath holding by the time (s) subjects held their breath. The BHI has been shown to be at least as effective as methods requiring carbon dioxide inhalation, which require invasive monitoring (5, 14). We used a fixed breath holding period for 30 s after 3 min of normal inspiration. The mean BFV at rest was obtained by the continuous recording of 3 min of normal room breathing. The subject was then ordered to hold his breath for 30 s after a normal inspiration and the maximum increase in mean BFV was measured. The BHIs of three consecutive periods of breath holding were averaged for each side to calculate the mean BHI for the right and left sides. The right and left BHIs were averaged to calculate the mean BHI for each individual.
All TCD measurements were done at least 5 days after the last migraine attack and consumption of abortive medication.
Statistical analysis was done by using a SPSS 10.0 for Windows software program. Comparisons between the patient and control groups were done using the Mann–Whitney U-test. Side to side comparisons in the patient group were done using the Wilcoxon signed ranks test. P<0.05 was considered statistically significant for all comparisons.
Results
Mean values of systolic, diastolic and mean BFV, PI and BHI values for patients and controls are given in Table 2.
Transcranial Doppler parameters in the control and patient group and in 12 patients with unilateral migraine attacks

∗ P=0.011 for patients vs. controls.
The difference between the systolic, diastolic and mean BFV and PIs in the patients and controls was non-significant. The BHI was significantly higher (P=0.011) in the patients with MWOA compared with the control group.
When the TCD values of the predominant side of pain were compared to the pain-free side in the 12 patients with right- or left-sided onset of pain, no difference could be demonstrated.
Discussion
We have demonstrated an exaggerated response to hypercapnia in a group of patients with migraine without aura by using the breath holding test. This exaggerated response seems to be symmetrical even in patients with unilateral migraine. The results reported in the literature are contradictory. Differences in techniques used to assess CVR make it difficult to compare the results obtained. In a study of 15 patients with MWOA and 15 with MA, Silvestrini et al. could not show any difference in vasoreactivity and BFVs, either between MA and MWOA or compared with controls (5). Although we used the same technique and breath holding time as them, the BHIs in our patients (mean BHI=1.64) were much higher then the BHI measured in their patients with MWOA (1.27 for the right MCA and 1.18 for the left MCA). This difference could result from a slight methodological difference between both studies. They took the mean BFV over 4 s after breath holding to calculate the CVR but we chose to take the maximum point of increase in BFV, which we think reflects the real maximum vasodilatory capacity after 30 s of breath holding. Similar to our results, they also found no side difference in patients with unilateral migraine. It seems that there is no side difference between the headache side and non-headache side in MWOA interictally as other studies also failed to demonstrate any difference (9). Others have also found an increased CVR in patients with migraine using different methods but they did not analyse patients with MA and MWOA separately, which makes a healthy comparison difficult (6, 7). On the contrary, studies reporting a normal (8, 9) or attenuated vasodilatory response (10–12) also exist. The great differences in inclusion criteria, diagnostic techniques and timing of study with regard to the last migraine attack make it extremely difficult to pool the results obtained. Techniques for assessing CVR, which are easy to perform, cheap and non-invasive, like the breath holding index, could allow more and larger scale studies to be performed to solve this problem.
The results of our study suggest that there is a high reactivity to hypercapnia in migraine without aura. The pCO2 reactivity might theoretically depend on pre-existing arteriolar tone and thereby on baseline velocity. Therefore a higher reactivity could be attributed to interictally increased vascular tone. Interictally elevated BFVs have been demonstrated by some investigators and have been explained by a chronic state of enhanced vasotonus or vasospastic vessel narrowing (3, 6, 15, 16). Our finding of similar blood flow velocities in controls and patients suggests that the underlying cause for this high reactivity may not be an increased vasotonus but an increased sensitivity to changes in blood CO2 levels.
