Abstract
Adenosine is an endogenous neurotransmitter that is released from the brain during hypoxia and relaxes isolated human cerebral arteries. Many cerebral artery dilators cause migraine attacks. However, the effect of intravenous adenosine on headache and cerebral artery diameter has not previously been investigated in man and reports regarding the effect of intravenous adenosine on cerebral blood flow are conflicting. Twelve healthy participants received adenosine 80, 120 μg kg-1 min-1 and placebo intravenously for 20 min, in a double-blind, three-way, crossover, randomized design. Headache was rated on a verbal scale (0-10). Regional cerebral blood flow (rCBF) with 133Xe inhalation and single-photon emission computed tomography (SPECT) and MCA flow velocity (VMCA) with transcranial Doppler, were measured in direct sequence. Six participants developed headache during 80 μg kg-1 min-1 and six during 120 μg kg-1 min-1 compared with none on placebo (
Introduction
Adenosine is a ubiquitous endogenous nucleoside with a wide range of biological effects throughout the human body, playing a role in both vascular and nociceptive systems (1, 2). It is released from many tissues in response to metabolic stress. In the brain, it has been suggested to be a mediator between cerebral metabolism and blood flow (3). Adenosine is released from neurons during hypotension, hypoxia, seizures and cortical spreading depression (4, 5), a neurophysiological phenomenon, which may underlie the migrainous aura. The effect of infused adenosine on human cerebral blood flow (CBF) is controversial, which may reflect differences in adenosine dose or methodology used (6–8).
Evidence suggests that adenosine plays a role in migraine. Plasma levels of adenosine may be elevated in migraineurs during attacks, while interictal levels are similar to healthy controls (9). Furthermore, there is anecdotal evidence that adenosine may precipitate migraine with aura (10). Finally, the non-selective adenosine antagonist caffeine is generally assumed to have an adjuvant effect on migraine and caffeine withdrawal has been suggested as a trigger of migraine (11).
We have previously shown that dilation of the middle cerebral artery (MCA) is usually associated with headache or migraine (12). Adenosine dilates human pial arteries
Methods
Design and participants
A double-blind, three-way cross-over design was used. Placebo (0.9% NaCl), adenosine 80 µg kg−1 min−1 and 120 µg kg−1 min−1 were administered intravenously in a balanced randomized order on one single study day. Syringes with placebo or adenosine (5 mg/ml; Item Development AB, Stocksund, Sweden) diluted with 0.9% sterile saline were prepared and marked 1–3 representing the order of infusion by a nurse not otherwise participating in the study. The final concentration in the syringes reflected the weight of the participant, which allowed the same rate of infusion to be used at all times.
Twelve healthy participants, six men and six women, not suffering from migraine, with a median age of 25 ± 3 years were included (16). Inclusion criteria were: healthy individuals 18–35 years old and weighing 50–100 kg. Fertile women were included only if using either oral contraceptives or intrauterine devices. Exclusion criteria were: history of migraine, close relatives with migraine, episodic tension-type headache more than once a week, history or clinical findings suggesting neurological or cardiovascular disorders, asthma or bronchospasm and daily intake of any kind of medicine except oral contraceptives. A full medical history and examination was performed, and an ECG was recorded to detect pre-existing atrioventricular conductance disturbances of the heart, including Wolf–Parkinson–White syndrome. All participants gave their written informed consent before inclusion. The study was approved by the ethics committee of Copenhagen and the Danish Medicines Agency, and was conducted according to the Helsinki II declaration.
Experimental protocol
The participants arrived at the clinic at 08.00 h, free from headache. The intake of coffee, tea, cocoa or other methylxanthine-containing foods or beverages was not allowed at least 8 h before the start of the study, nor was the use of any medication, except oral contraceptives, for at least four times the plasma half-life of the drug. On arrival, the participants were placed in the supine position, and catheters [Optiva∗2 (18 G); Johnson & Johnson, Ethicon, Italy] were inserted into the right and left antecubital vein for blood sampling and drug infusion, respectively. After completing these procedures, the participant rested 30 min before starting the first round of infusion, using a time and volume controlled infusion pump (Braun Perfusor, Melsungen, Germany). Each infusion lasted 20 min followed by a 40-min wash-out. In each round, baseline values were obtained at time point − 5 min (T−5) for headache and adverse events, middle cerebral artery flow velocity (VMCA), superficial temporal artery diameter, vital signs and plasma adenosine. Infusion of adenosine or placebo was initiated at T0 and maintained for 20 min. VMCA, superficial temporal artery diameter and vital signs were recorded at T5, T15, T30, T45 and T60 after each dose, plasma adenosine at T18, and CBF at T10−15. Corresponding VMCA values at T15 were measured immediately after completing the CBF acquisition protocol. After completing the last round, the participants were discharged from the clinic, and asked to complete a headache diary every hour until 10 h after discharge. They were allowed to take rescue medication of their own choice at any time.
Adverse events
Headache was recorded on a verbal scale from 1 to 10. One represented a very mild headache, including sensation of pressing or throbbing or otherwise altered sensation in the head, 5 a headache of moderate intensity, and 10 the worst possible headache imaginable by the participant (14). Headache characteristics and accompanying symptoms were recorded according to the International Headache Society (16), using a structured questionnaire.
CBF measurement
The examination was performed with the subject in the supine position, in quiet surroundings with eyes closed and ears unplugged. Four markers were drawn on the skin to ensure accurate positioning in each acquisition. End-tidal partial pressure of CO2 (PetCO2) was measured during each examination (Datex Normocap 200, Roedovre, Denmark). CBF was measured with 133Xe inhalation (Ceretronix XAS SM 32C, Randers, Denmark) and single-photon emission computed tomography (SPECT), using a brain-dedicated gamma camera (Ceraspect, DSI, Waltham, MA, USA). The system uses a stationary annular NaI crystal and a fast rotating collimator. A dynamic protocol of 133Xe inhalation using the Kanno-Lassen algorithm was used (17, 18). Calculation of flow in the perfusion territories of the major cerebral arteries was performed by fitting standard vascular regions of interest (ROI). Flow in the territory of the MCA (rCBFMCA) was calculated as mean of the right and left side, since there was no difference between the sides. If significant changes in PetCO2 were found, values were corrected with 2% for each mmHg change (19).
Cerebral artery blood flow velocity
Blood flow velocity in the middle cerebral artery (VMCA) was recorded bilaterally using hand-held 2-MHz probes and transcranial Doppler (TCD; Multidop X, DWL, Sipplingen, Germany). We used hand-held probes, since fixed probes cause headache and discomfort on their own (20). A time-averaged mean over four cycles each comprising approximately four cardiac cycles or 4 s was used as final measure for each time point. Identification of the MCA and marking reproducible fix points were done as previously described (18, 21). PetCO2 was recorded simultaneously with the transcranial Doppler measurements using an open mask that caused no respiratory resistance. If changes in PetCO2 were found, VMCA was corrected with e0.034 for each mmHg change in PetCO2 (22). VMCA measurements at T15 were performed immediately after CBF measurements. Since rCBFMCA equals the product of VMCA and cross-sectional area of the MCA, changes in MCA diameter (Δd) between treatment a and b were calculated as Δd = √[(rCBFMCA(b) × VMCA(b) −1 × (VMCA(a)× rCBFMCA(a))]−1 (23).
Diameter of superficial arteries
Diameter of the frontal branch of the superficial artery was measured using high-resolution ultrasonography, 20 MHz, bandwidth 15 MHz (Dermascan C, Hadsund, Denmark) (24), as previously described (25).
Vital signs
Heart rate and blood pressure were measured using an auto-inflatable cuff (Omega 1400, Orlando, FL, USA). ECG (Cardiofax V, Nihon-Cohden, Japan) was monitored on an LCD screen and recorded on paper at every 15 min.
Biochemistry
Sample collection has been described (26). Briefly, blood collected from the antecubital vein at baseline and at T18 (8 ml/sample) was drawn into vacuum tubes on ice containing a stopping solution [0.2 m
The assay technique has been described (26). Briefly, a Hewlett Packard HP 1100 modular system was used, with a diode array detector. Lyophilized samples (500 µl) were mixed with 1 ml of phosphate buffer (NaH2PO4/Na2HPO4, pH 4), injected in a 1-ml loop, and then eluted with a methanol gradient on a Merck LIChrospher C18 column (0% for 3 min, then 10–25% methanol for 15 min). The intra- and interassay coefficient of variation for nucleosides ranged between 1 and 3%. The limit of detection at 254 nm was 1 pmol in 1 ml of plasma matrix injected. For identification and quantification, retention times and spectra were compared with those of exogenous adenosine and metabolites. Quantifications were made by comparing areas obtained for samples with areas of known quantities of nucleosides.
Reagents
Adenosine (crystallized, 99% pure), dipyridamole, a,b methylene-adenosine-5′-diphosphate (AOPCP), adenosine triphosphate (ATP), 9-erythro(2-hydroxy-3-nonyl) adenine (EHNA), were from Sigma (Saint Quentin Fallavier, France). Deoxycoformycin was from Lederle Laboratories (Paris, France). Heparin was from Sanofi Winthrop (Gentilly, France). Na2-ethylenediaminetetracetic acid (Na2-EDTA) was from Sigma. Bovine serum albumin (BSA) was from Johnson and Johnson Clinical Chemistry (Rochester, NY, USA). The reversed-phase chromatography column (Merck LIChrospher C18, and RP8250 × 4 mm) and other reagents were from Merck (Darmstadt, Germany).
Statistical analysis
Baseline was defined as T−5 before the start of infusion of each dose. Data are presented as mean ± SD, except headache score, which is listed as medians (range). Significant differences between headache scores (peak and AUC) were analysed with a non-parametric analysis of variance (Friedman test). Binary categorical data were analysed with Cochran
Due to the very short lasting effect of adenosine, we did not expect any carryover effect, but period effects may occur. Study design did not allow simultaneous testing for carryover effect and period effect. Assuming no carryover effect, period effect was tested with univariate analysis of variance with the factors: number of infusion and volunteer. Test variables were baseline and response (AUC) for analysing period effects.
Results
Six participants reported a headache score of more than zero during infusion of adenosine 80 µg kg−1 min−1 and six during 120 µg kg−1 min−1 compared with none during placebo. Five participants experienced headache on both doses. Median headache score by those experiencing headache was 1 during both adenosine doses. There was a significant difference between groups in peak headache score (
After discharge from the clinic, six participants developed a mild, bilateral and pressing headache (median for the affected group was 1.5, range 1–6); of these, three took rescue medication. The headache started from 3.5 to 8.5 h after the first active dose. Four of the six participants with late occurring headache had experienced immediate headache during the infusion of adenosine 80 µg kg−1 min−1 and/or 120 µg kg−1 min−1. The two participants with the most pronounced headache (peak headache score of 3 and 6, respectively) did not experience headache during the infusion of either adenosine dose.
Global CBF was 52.14 ± 7.01 ml 100 g−1 min−1 during infusion of placebo, 47.04 ± 5.75 ml 100 g−1 min−1 during adenosine 80 µg kg−1 min−1 and 49.65 ± 11.60 ml 100 g−1 min−1 during adenosine 120 µg kg−1 min−1. There was no significant difference between treatment groups (
VMCA decreased significantly over time after both adenosine 80 and 120 µg kg−1 min−1 (

(a) Mean flow velocity in the middle cerebral artery (VMCA) ± SD after placebo (•), adenosine 80 µg kg−1 min−1 (▪) or 120 µg kg−1 min−1 (▵). Values not corrected for changes in end-tidal partial pressure of CO2. Changes were significant over time for both adenosine doses (
Baseline values ± SD

Baseline values for middle cerebral artery flow velocity (VMCA), end-tidal partial pressure of CO2 (PetCO2), blood pressure (BP) and other variables before the infusion of placebo, adenosine 80 µg kg−1 min−1 or 120 µg kg−1 min−1.
The diameter of the superficial temporal artery increased significantly over time after both adenosine doses (
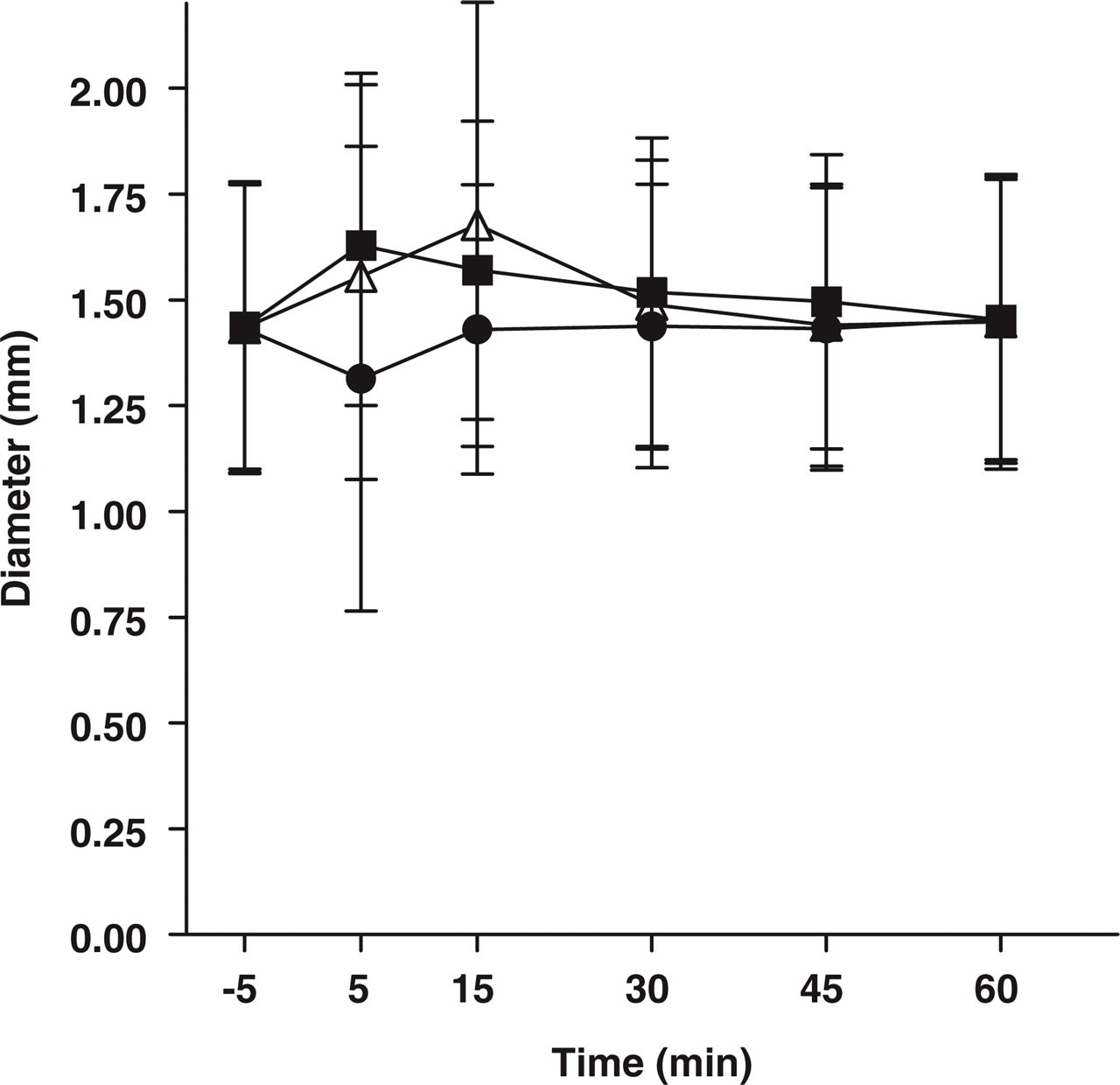
Superficial temporal artery diameter ± SD placebo (•), adenosine 80 µg kg−1 min−1 (▪) or 120 µg kg−1 min−1 (▵) measured with high-resolution ultrasonography. A non-dose-dependent increase in diameter was seen.
Regarding vital signs, there were no significant changes over time or between doses for systolic or mean blood pressure. Baseline values are listed in Table 1. For diastolic BP, there were no changes between doses or over time after placebo (
Baseline values of plasma adenosine levels are listed in Table 1. There were no significant changes between the ratio between plasma levels during infusion of placebo, adenosine 80 µg kg−1 min−1 or 120 µg kg−1 min−1, and baseline (

Mean plasma adenosine concentrations ± SD. Baseline compared with response during the infusion of placebo, adenosine 80 µg kg−1 min−1 (80) and 120 µg kg−1 min−1 (120). Measurements were reproducible, but revealed no changes in adenosine due to the short half-life.
There were no significant period or carryover effects for baseline or response for VMCA, superficial temporal artery diameter, PetCO2, blood pressure or heart rate. There was no carryover effect for CBF or rCBFMCA, but period effects could not be evaluated due to the nature of the data. Carryover effect on plasma adenosine could not be evaluated because of insufficient data, but there was no period effect on baseline.
Discussion
Several vasodilator arteries have been demonstrated to cause headache and migraine concomitant with MCA dilation in both normal participants and migraine patients (12, 18, 27–29), though the role of large cerebral dilation in experimental headache models is presently not clear (15, 25). No previous study has evaluated the effect of adenosine on headache response and diameter of the large cerebral arteries.
TCD measurements of velocity as a stand-alone are difficult to interpret, because it is dependent on both the diameter and rCBF, but can be used to estimate changes in the cerebral arteries in combination with quantitative rCBF. We have investigated the MCA because measurements of this artery are the most precise and reproducible using TCD (21). A previous TCD study showed a ∼ 15% decrease in VMCA using adenosine 140 µg kg−1 min−1 i.v. (30), similar to our findings, but no rCBF was measured. In the same study, mean PetCO2 decreased from 41 to 31 mmHg, which may account for the observed decrease in VMCA (22). In the present study, adenosine likewise caused a marked decrease in PetCO2, which is expected to reduce CBF and consequently VMCA, if the MCA diameter remains unchanged. A correlation between PetCO2 and VMCA has previously been established, allowing the calculation of PetCO2-corrected VMCA values (22). After using this correction, there were no significant changes in VMCA. However, it can be argued that PetCO2 does not always reflect PaCO2, which is the true determinant of CBF. This is the case if lung perfusion and ventilation do not match, e.g. due to bronchospasm or a sudden change in cardiac output. It can also be argued that adenosine potentially has several such effects. However, it has previously been established that i.v. adenosine markedly increases minute ventilation, possibly due to activation of carotid body chemoceptors (30–33), and adenosine 140 µg kg−1 min−1 i.v. decreases mean PaCO2 from 41 to 31 mmHg (31), similar to the observed change in PetCO2 in the present study. We therefore suggest that the observed changes in PetCO2 reflect changes in arterial blood gases caused by hyperventilation, and that the observed changes in VMCA are explained by changes in PaCO2, not MCA dilation.
Data on the effect of adenosine on rCBF are conflicting. In a magnetic resonance imaging and positron emission tomography (PET) study, nine healthy participants received adenosine at a rate of 60–100 µg kg−1 min−1 i.v. causing no significant effect on CBF (6). In another PET study, seven patients scheduled for cerebral arteriovenous malformation surgery were examined during general anaesthesia and controlled ventilation. In six of the patients, PetCO2 was kept as close as possible to 40 mmHg, and very large doses of adenosine (200–500 µg kg−1 min−1) were administered via a central venous catheter. This resulted in a 55% (range 23–85%) mean increase in global CBF, though there was no clear correlation between adenosine dose and CBF changes (7). A 99mtechnetium hexamethylpropyleneamine oxine (Tc-HMPAO) SPECT study, including six healthy participants and six patients with occlusive carotid disease, showed a marked increase of approximately 30% in the cortex and 70–80% in the thalamus and basal ganglia after adenosine 140 µg kg−1 min−1 i.v. (8), but this tracer is only suitable for measuring the relative distribution of rCBF, not absolute rCBF changes (34). The present results support the PET study (6) and together this strongly suggests that doses < 120 µg kg−1 min−1 i.v. do not increase CBF in awake, spontaneously breathing participants. In contrast, a 6–10%, but not statistically significant (
There are at least two possible explanations to why adenosine does not dilate cerebral arteries and arterioles in man, despite the presence of vasodilatory A2 receptors in cerebral artery smooth muscle cells and a dilatory effect on human cerebral arteries
The role of adenosine in headache generation
In a large open-label multicentre study, adenosine 140 µg kg−1 min−1 over 6 min caused headache in 14.2% of more than 9256 patients undergoing cardiac stress testing (38). This study has limited value because there was no control group, and headache is a frequently reported adverse event in both placebo and active groups in most drug trials. The present study used a randomized, double-blind, cross-over design with frequent and systematic scoring of headache. We found significantly more headache after adenosine than after placebo. The headache induced during the infusion was very mild and merely reported to be an altered, pressing or throbbing sensation in the head by four of six on both adenosine doses. Since there was no indication of a dose–response relationship in the present study, it is doubtful that higher doses would have caused significant degrees of headache. Due to need to maintain the infusion for 20 min and the pronounced discomfort experienced by all participants, we estimated that 120 µg kg−1 min−1 was the highest testable dose. Considering the marked side-effects it seems improbable that even higher plasma adenosine levels would occur naturally during a migraine attack. Migraineurs are more sensitive to headache-provoking substances than healthy participants, but in our experience substances that provoke migraine in migraine patients cause a much greater headache response in normal participants than seen in the present study (mean peak headache 2.5–3 vs. 0.5) (15, 25, 39).
We found a surprisingly high number of participants who developed delayed headache several hours after the experiment. For this, however, there was no placebo control, since the study was not designed to address this problem. With other infused substances such as histamine, calcitonin gene-related peptide (CGRP) and glyceryl trinitrate (GTN), a biphasic headache response has been observed in patients with migraine without aura, i.e. an immediate response observed during infusion followed by a secondary headache response 3–8 h later, long after elimination of the test substances (28, 29, 39). This phenomenon has also been demonstrated in normal participants after GTN (39), but less frequently. There was, however, no evident correlation between immediate and delayed headache response, suggesting that the headache observed after finishing the study may not be true delayed headache. Indeed, the patients reporting the highest headache score of 3 and 6, respectively, in the observation period, experienced no headache during adenosine infusion. In summary, circulating adenosine is not a likely mediator of migraine attacks. The question remains, whether the migraine attack could be caused by adenosine released from perivascular nerve terminals or from the brain itself. In this context, it is interesting that intrathecal adenosine had a general analgesic effect, but nevertheless caused mild headache in three of six normal subjects at a dose of 2 mg (40).
In conclusion, intravenous adenosine in the maximal tolerated dose has no dilator effects on the MCA or cerebral resistance vessels, but dilates extracranial arteries. We found little evidence supporting a role for circulating adenosine in the pathogenesis of migraine or headache, but repeating the study with migraine patients may be necessary to preclude an effect in particularly sensitive subjects. It is likewise possible that adenosine released from perivascular nerve terminals or from the brain itself may cause headache.
Footnotes
Acknowledgements
The authors thank Boehringer Ingelheim Pharma KG (Ingelheim am Rhein, Germany) for financing the study, MSc Joergen Holm Petersen, Department of Medical Statistics, University of Copenhagen, for expert statistical advice, and lab technicians Lene Elkjaer and Kirsten Brunsgaard for their technical assistance. S.B. receives a grant from the County of Copenhagen.
