Abstract
Previously an amplitude enhancement of laser evoked potentials (LEPs) was detected during migraine attack: we further examined pain threshold to CO2 laser stimuli and LEPs during attacks, evaluating the effect of almotriptan, lysine-acetylsalicylate and placebo treatment on cutaneous hyperalgesia to thermal stimuli delivered by CO2 laser and on LEP components. Eighteen patients suffering from migraine without aura were analysed. They were divided into three groups of six patients each, randomly assigned to lysine acetyl-salicylate, almotriptan or placebo treatments. The supraorbital zones and the dorsum of the hand were stimulated on both the symptomatic and not symptomatic side in all patients. The LEPs were recorded by 25 scalp electrodes During attacks, the P2 wave was significantly enhanced; the amplitude of the P2 component obtained by the stimulation of the supraorbital zone during the attack on the side of the headache was significantly correlated with the intensity of pain and the frequency of headache. Both almotriptan and lysine acetyl-salicylate significantly reduced the P2 amplitude but they showed no effects on hyperalgesia to laser stimulation; headache relief following therapy was correlated with the reduction of the P2 amplitude. The cortical elaboration of laser-induced experimental pain seemed increased during migraine attack, and the severity of headache was mainly related to the increase of the later LEPs components expressing the attentive and emotive compounds of suffering. Reversion of this process appeared to be primarily responsible for the efficacy of drugs in treating migraine, though both almotriptan and lysine-acetil salicilate seemed to have no effect in reducing sensitization at second and third order nociceptive neurons.
Introduction
Migraine is a disabling disorder. Its severity, as measured by headache intensity and duration, is maintained and recurrence is favoured by central sensitization. It is associated with the sensitization of medullary dorsal horn neurons, measured by mechanical and thermal pain thresholds of periorbital and forearm skin areas during unilateral migraine attacks (1).
One method that has the potential to examine pain-related central nervous system (CNS) processing involves the measurement of cerebral event-related potentials recorded in response to painful stimulation; such nociceptive-evoked responses are thought to reflect a secondary process of noxious input. Clinical neurophysiological studies have shown that infrared CO2 lasers can be used to record nociceptive laser-evoked responses from the human skull vertex, elicited by the selective activation of Aδ afferents in the skin and related closely to the perception of pain (2). Previously, CO2 laser-evoked potentials (LEP) were employed during migraine attacks, using a derivation on CZ to record the greatest LEP waves (3). Cutaneous hyperalgesia to laser stimuli was observed on the hand on the symptomatic side and supraorbital zone on both the symptomatic and nonsymptomatic sides during acute migraine, confirming earlier results (1). In addition, the amplitudes of cortical responses were clearly increased during migraine attacks when the symptomatic side were stimulated at both the head and the hand levels, suggesting that there may be some associated changes in cortical function (3).
Although sensitization may play a role in the initiation and maintenance of pain during migraine, the effects of acute treatment in reducing sensitization phenomena are not clear. Evidence that triptans may reduce trigeminal sensitization has been described in animal models (4, 5). Indirect evidence for the possible effects of acute therapy of migraine on trigeminal sensitization in humans has been provided through the study of trigeminal reflexes: in a previous study the later blink reflex components, elicited mainly by nociceptive afferents, were inhibited by zolmitriptan during migraine attack (6). In another study performed by a nociception specific blink reflex, both zolmitriptan and lysine acetyl-salicylate increased the onset latency and reduced the area of R2 component, which were modified by temporary sensitization of central trigeminal neurons during acute migraine (7). Recently Burstein & Jakubowski (8) showed that in animal model early sumatriptan intervention effectively blocked the development of all aspects of central sensitization: further, in a cohort of migraine patients who experienced attack both with and without cutaneous allodynia, the same authors found that patients who never developed allodynia were highly likely to be rendered pain-free by triptan therapy anytime after the onset of pain while triptan treatment was less effective during allodynic attacks (9).
The aim of the study was to further evaluate the LEPs pattern during acute migraine, using multichannel recording, and to detect the effect of almotriptan, lysine–acetylsalicylate and placebo treatment on both cutaneous hyperalgesia and LEP components.
Methods
Subjects
Eighteen outpatients suffering migraine without aura, according to the International Headache Society criteria (10), were selected, among a group of 50 migraine patients. All patients were diagnosed after six months’ follow-up, during which all the 50patients were submitted to a clinical evaluation of thermal hyperalgesia by CO2 laser during the pain-free phase and during at least 5 migraine attacks, after a range of 2–4 h from the onset of migraine headache Presence or absence of hyperalgesia was determined based on differences between migraine and baseline pain thresholds to thermal stimulation of periorbital skin: subjects who had experienced thermal hyperalgesia (pain threshold to laser stimulus settled at least at 1.5 W below the baseline intensity) in all the five migraine attacks were selected for the further analysis. Patients with general medical, neurological or psychiatric diseases, according to the DSM IV (11), and patients who were taking psycho-active drugs or prophylactic treatment for headache, or who were assessed as overusing analgesic drugs in the last two months, were excluded from the study. The selected migraine patients were all females, aged 28–46 years (mean 38.4 ± 4.5). Both the severity of headache, measured by the MIDAS scale (12), and the frequency of headache, measured in days with headache/month, were computed in the last six months. The patients were instructed to attend a recording session at the onset of a migraine attack without taking triptans or other symptomatic drugs. They were examined by a neurologist with experience in headache and selected for the experimental task, after having given their informed consent, according to the Declaration of Helsinki. The study was approved by the local Ethics Committee of our Department. After the recording session performed over a range of 4–6 h (mean 3.7 ± 2.1 h) from the onset of the attack, each patient was randomly assigned to placebo, almotriptan 12.5 mg or lysine–acetylsalicylate 1800 mg orally, and a recording task was performed two hours after drug administration. The allocation treatment criteria were based on a random consecutive assignment of 1 almotriptan (12,5 mg Solvay Pharma) vs. 1 placebo vs. 1 lysine-acetylsalicilate (1,8 g Sanofy Sintelabo) consecutive patients. Patients were blind regarding the drug assumed. The severity of headache was checked on a scale from 0 (no headache) to 3 (severe headache) (13).
The control task was carried out at least 72 h after the end of the critical migraine phase (mean 111 ± 31.3 h) and well before the next attack (mean 102 ± 10.2 h), verified by the headache diary during a following clinical examination. The main clinical features of the patients are summarized in Table 1.
Main clinical features in migraine without aura patients

value given as mean ± SD.
CO2 laser recording
The subject was seated in a comfortable chair, awake and relaxed with eyes closed in a quiet room kept at 21–23°C. Both the subjects and experimenters wore protective goggles during data acquisition. The pain stimulus was laser pulses (wavelength 10.6 µm) generated by a CO2 laser (Neurolas, Electronic Engineering, Florence, Italy; http://www.elengroup.com). The beam diameter was 2.5 mm, and the duration of the stimulus pulse was 20 ms. Signals were recorded through 25 disk electrodes (impedance below 5000 ohms), referring to the nasion with the ground at Fpz.; another electrode was placed above the right eye to record the EOG. Signals were amplified, filtered (0.5–100 Hz), and stored on a biopotential analyser (MICROMED System Plus; MICROMED, Mogliano Veneto, Italy; http://www.micromed-it.com). The analysis time was 1000 ms with a bin width of 4 ms (512 Hz sampling rate).
Stimulation
The dorsum of both hands and the supraorbital zones ipsilateral and contralateral to the headache were stimulated. Irradiated points were moved slightly between each stimulus, to minimize skin burn, nociceptor fatigue, and central habituation. To define the pain threshold, single stimulus pulses were presented in random order at four to five different intensities, starting from 1.5 W. A series of 20 stimuli for each intensity level was presented at intervals of 20–40 s. The intensities were increased in 1.5 W steps. The subjects were requested to report the quality of sensation after any stimulus presentation, using a verbal analogue scale (VAS) in which 0 indicated no sensation and 10 the most severe pain imaginable. The VAS rate was expressed for all the series of 20 stimuli settled at each intensity level. The pain threshold was in each patient the laser intensity expressed in Watt which determined a level 4 (a pinprick sensation followed by a burning sensation) for at least half of the 20 stimuli.
LEPs analysis
The LEP recordings were analysed blind regarding the clinical conditions and the acute treatment for migraine. Blocks of at least 15 trials obtained by laser stimuli settled at the pain threshold were averaged offline. An artefact rejection algorithm excluded from the average, all runs containing transient exceeding ± 65 mV at any recording channel. Moreover LEPs were identified on the basis of their latency and distribution and five responses were labelled according to Valeriani et al. (14). The N1 component was measured on the temporal derivation (T5 and T6) contralateral to the side of pain; the P2 and N2a components were analysed at the vertex (CZ), and the N2b wave was analysed on FZ. Absolute latencies of scalp potentials were measured at the highest peak of each response component, and the amplitude of each wave was measured from the baseline. First, all patients were considered during the attack and in the pain-free phase: the features of the LEPs and the pain thresholds obtained during the attack and in the attack-free phases were normally distributed and were compared using Student's t-test for paired data. Pearson's correlation test was also employed to relate the main LEPs features with clinical data. The effects of drugs on LEPs and pain theshold were evaluated in the three groups of patients using Student's t-test for paired data, corrected for multiple comparisons: the three groups of patients were also compared employing univariate
Results
The patients assigned to the three treatments were like regarding clinical features, compared by

Pain threshold to laser stimulus (a) Mean values and standard deviation of pain thresholds by laser stimulation in 18 migraine patients. □ attack-free; ▪ attack. Results of Student's t-test for paired data are shown: ∗P < 0.05. (b) Mean values and standard deviation of pain threshold by laser stimulation in the three groups of patients with an attack (▪) or treated by almotriptan (□), lysine–acetylsalicylate ( ) and placebo (
) and placebo ( ). Results of Student's t-test for paired data were not significant.
). Results of Student's t-test for paired data were not significant.
The latencies of all the LEPs waves were unchanged during migraine attack in comparison with the intercritical phase. All the LEP waves appeared increased during the attack in comparison with the attack-free periods, when both the hands and the facial zone were stimulated, but only the later LEP components P2 and N2b showed a statistically significant increment during the critical phase on the symptomatic side, as shown in Figs 2 and 3. Sixteen (88.8%) patients had an amplitude increase of P2 component > 2 SD of the baseline values when the ipsilateral forehead and hand were stimulated. The amplitude of the P2 component obtained by the stimulation of the supraorbital zone during the attack on the side of the headache was significantly correlated with the intensity of pain (r = 0.51, P < 0.05

Features of the LEPs in basal condition and during migraine. Mean values and standard deviations of amplitudes of the main LEP components in migraine patients, obtained by the stimulation of the symptomatic (▪,□) and nonsymptomatic ( ,
, ) sides. (▪,
) sides. (▪, ) attack-free; (□,
) attack-free; (□, ) attack. Results of Student's t-test for paired data are shown: ∗∗P < 0.01: ∗P < 0.05.
) attack. Results of Student's t-test for paired data are shown: ∗∗P < 0.01: ∗P < 0.05.
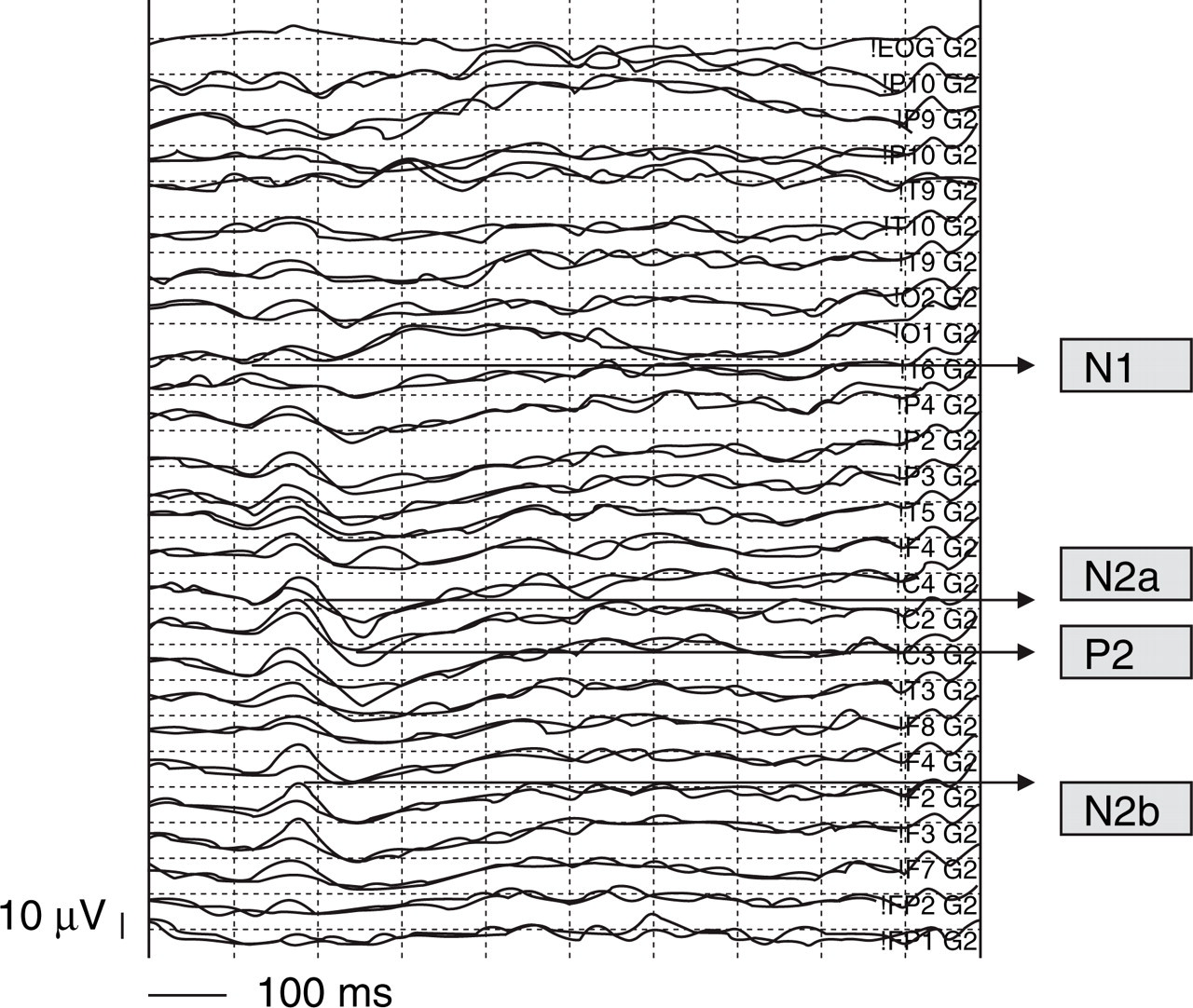
Grand average of LEPs obtained by the stimulation of the symptomatic supraorbital zone across 9 patients, who experienced left migraine; the control task was superimposed. The amplitude difference between the two conditions was outlined in grey colour at CZ, FZ, T6.
The intensity of pain after drugs was measured as a percentage of the basal intensity during the attack, considering the values resulting from the application of the scale for migraine intensity, according to the IHS Committee on Clinical Trials in migraine (13).
It was 19 ± 12.8% after almotriptan, 29 ± 13% after lysine–acetylsalicylate and 54.1 ± 8.3% after placebo;

Mean values and standard deviations of (a) N2b and (b) P2 waves amplitude recorded during the attack (▪, ) and after the treatment (□,
) and after the treatment (□, ). Results of Student's t-test for paired data are shown: ∗P < 0.05.
). Results of Student's t-test for paired data are shown: ∗P < 0.05.

Mean values and standard deviations of (a) N1 and (b) N2b waves amplitude recorded during the attack (▪, ) and after the treatment (□,
) and after the treatment (□, ). Results of Student's t-test for paired data: ns.
). Results of Student's t-test for paired data: ns.
Discussion
In our migraine patients without aura, pain threshold was decreased during the acute phase, as reported previously (1). Though they were selected among patients experienced hyperalgesia, one of them did not show thermal hypersensitivity during the attack. In migraine the sensitization of peripheral nociceptors innervating blood vessels and meninges causes the increase of headache under low mechanical stimuli, while the heat hyperalgesia confirms a sensitization of the skin nociceptors of the face ipsi- and contralateral to the side of pain, extending also to the hand ipsilateral to pain. This phenomenon is explained by central sensitization of medullary dorsal horn neurons with periorbital receptive fields, compounded by additional sensitization of third-order neurons. The pain threshold of the face ipsilateral and contralateral to the side of migraine was significantly reduced with the duration of illness, suggesting that the phenomenon of central sensitization at second and third order nociceptive neurons may get worse in relation to the cumulative number of migraine attacks experienced, though it seemed to have no relation with the intensity of current migraine attack and the frequency of migraine experienced in the last six months.
All the LEP waves appeared increased during the attack in comparison with the attack-free periods, but only the P2 and N2b components obtained by the stimulation of the symptomatic side showed a significant amplitude increase, confirming previous results (3). In healthy volunteers neurogenic sensitization induced by capsaicin reduced LEP amplitudes despite the presence of hyperalgesia (15), so a peculiar pattern of enhanced cortical response to experimental pain was confirmed in acute migraine. In migraine, pain may be subtended by activation of peripheral sensory fibers innervating dura and intracranial vessels, activation of descending pathways that facilitate processing of pain signals, and suppression of descending pathways that inhibit such processing of pain signals in the spinal cord (16). The finding of a prevalent amplitude enhancing of the later LEP responses, which varied as a function of the level of attention, arousal, and anxiety (17, 18), suggests that subjective attentive attitude against the painful stimuli during migraine may increase the effect of the pain transmission disinhibition and further ease pain processing via a facilitation mechanism of cortical origin (19). In a previous evaluation on episodic and chronic migraine patients without aura, in both headache groups, distraction from the painful laser stimulus induced by an arithmetic task failed to suppress the LEPs amplitudes in comparison with control subjects (20). The inability to reduce pain elaboration during an alternative cognitive task indicates a condition of reduced inhibition of cortical areas devoted to pain elaboration and hyper-attention to experimental painful stimuli, which appeared further increased in the acute migraine condition despite the intrinsic trigeminal pain.
The positive correlation between the amplitude of the P2 component obtained by the stimulation of the supraorbital zone during the attack on the side of headache, and the intensity and the frequency of headache may suggest that the cortical hyper-attention to painful stimuli may be responsible for the severity of migraine and for the repetition of migraine attacks under precipitating factors. Such hypothesis deserves to be confirmed in larger migraine series, considering that in the present study the pattern of enhanced LEPs during acute pain was absent in 2 migraine patients.
Both almotriptan and lysine–acetylsalicylate were effective in relieving headache compared with placebo, which however, was able to reduce headache intensity by nearly 50%. No drugs exerted any effect on the hyperalgesia induced by laser stimuli, which persisted for two hours after the treatment, even though the treatment had provided relief from headache. The sensitization of trigeminal nociceptive neurons is a lasting phenomenon. Our patients were studied approximately four hours after the onset of attacks and a further two hours later, for a total of six hours after the initiation of migraine: sufficient to complete the spreading of cutaneous sensitization (1). The drugs were able to reduce headache and LEPs amplitude but were unable to totally stop the central sensitization at the second-order and third order nociceptive neurons levels, which caused hyperalgesia to laser stimuli on the hand and the face. This apparent paradox suggests that pathways required for the normal elicitation of LEP are inhibited by drugs whereas alternative pathways may project the laser-induced hyperalgesic response, according to Treede et al. (21). Our results are only partly in accordance with the most recent findings by Burstein et al. (9), who reported no effect of triptans in reducing allodynia, with a concomitant failure in reducing headache. In our series of patients, drug-induced headache relief was accompanied by reduced elaboration of experimental pain in the cortical areas devoted to the emotional and attentive compounds of pain processing. Both almotriptan and lysine–acetylsalicylate target on peripheral nociceptors and exert central inhibitory effects on trigeminal nociceptive firing to the cortex (22–24); the central inhibitory effect may be responsible for the reversion of LEPs increment due to migraine attack. Though the effect of drugs in reducing trigeminal nociception might not be prompt enough to reduce central sensitization at second- and third-order neurons it was sufficiently effective to reduce trigeminal inputs to the cortex and cortical processing of pain in our series. The reduction of the activity of cortical areas devoted mainly to the attentive and emotive correlates of pain, which may facilitate the nociceptive transmission by the trigeminal and spinal cord neurons, seemed above all responsible for the success of therapy and could explain also the relevant effects of placebo both in reducing headache and LEP amplitudes: furthermore the effect of drugs may be variable also across migraine attacks with allodynic symptoms, producing different results in migraine groups (9).
Our study provides further evidences that the treatment of acute migraine by both triptans and not steroidal analgesic drugs is unable to stop the sensitization phenomena after they are established.
In contrast with the results of Burstein et al. (9), in our series the efficacy of therapy seemed quite independent from the reversion of cutaneous hyperalgesia, as may also be suggested by the evidence of the large spectrum of the clinical efficacy of triptans (25), in relation to the rate of patients who experiences allodynia during migraine attack (16).
Further studies performed in larger migraine series in the early phase of the attack, may clarify if the prevention or the suppression of second and third order neurons sensitization may contribute to increase the efficacy of drugs on headache intensity and to reduce the tendency toward headache recurrence and chronicization.
Footnotes
Acknowledgements
We wish to honour the memory of our Director, Prof Francomichele Puca, who inspired this study.
