Abstract
Background
Transcutaneous external supraorbital nerve stimulation has emerged as a treatment option for primary headache disorders, though its action mechanism is still unclear.
Study aim
In this randomized, sham-controlled pilot study we aimed to test the effects of a single external transcutaneous nerve stimulation session on pain perception and cortical responses induced by painful laser stimuli delivered to the right forehead and the right hand in a cohort of migraine without aura patients and healthy controls.
Methods
Seventeen migraine without aura patients and 21 age- and sex-matched controls were selected and randomly assigned to a real or sham external transcutaneous nerve stimulation single stimulation session. The external transcutaneous nerve stimulation was delivered with a self-adhesive electrode placed on the forehead and generating a 60 Hz pulse at 16 mA intensity for 20 minutes. For sham stimulation, we used 2 mA intensity. Laser evoked responses were recorded from 21 scalp electrodes in basal condition (T0), during external transcutaneous nerve stimulation and sham stimulation (T1), and immediately after these (T2). The laser evoked responses were analyzed by LORETA software.
Results
The real external transcutaneous nerve stimulation reduced the trigeminal N2P2 amplitude in migraine and control groups significantly in respect to placebo. The real stimulation was associated with lower activity in the anterior cingulate cortex under trigeminal laser stimuli. The pattern of LEP-reduced habituation was reverted by real and sham transcutaneous stimulation in migraine patients.
Conclusions
The present results could suggest that the external transcutaneous nerve stimulation may interfere with the threshold and the extent of trigeminal system activation, with a mechanism of potential utility in the resolution and prevention of migraine attacks.
Introduction
Extracranial stimulation (eTNS) has emerged as a treatment option for primary headache (1). A randomized, sham-controlled trial showed that the transcutaneous Cefaly® device (Cefaly® Technology sprl, Herstal, Belgium) had significant efficacy in reducing both migraine attacks and headache days (2). Its beneficial preventive effect in low-frequency migraine was also suggested by a small open study (3) and a prospective large study, showing the safety and efficacy of eTNS (4,5). In addition, growing evidence exists on the efficiency of eTNS in acute migraine treatment (5–7). The most recent device consists of a self-adhesive, supraorbital electrode, which is able to transcutaneously excite the supratrochlear and supraorbital branches of the ophthalmic nerve (V1) located under the skin of the forehead (8). However, the exact mechanism of pericranial nerve neurostimulation is still not clear. The convergence of somatic trigeminal and cervical afferents and visceral trigeminovascular fibers on the spinal nucleus may modulate pain transmission with the effect of resolving and preventing migraine attacks. Few physiological studies have dealt with the question of the mechanisms of action of the Cephaly device. A double-blinded, crossover, sham-controlled study on 30 healthy volunteers showed that high frequency (120 Hz) transcutaneous supraorbital nerve stimulation (eTNS) induces a sedative effect (9). Didier et al. evaluated the action of the eTNS device on the pericranial and neck muscles by quantitative electromyography (EMG) recordings in 23 patients with chronic migraine (10). The EMG amplitude and frequency of the frontalis, anterior temporalis, auricularis posterior, and middle trapezius muscles increased in a significant way after eTNS application (10). These results might suggest an action of eTNS on the inhibitory circuit in the spinal cord with a consequent neuromuscular facilitation. Studies employing Nociceptive Blink Reflex (NBR) and Contact Heat-Evoked potentials (CHEPs) showed that eTNS modulated NBR amplitude and habituation and reduced frontal CHEPs amplitude, an effect consistent with a supra-segmental inhibitory action specific for the trigeminal system (5,11). Functional neuroimaging studies based on Positron Emission Tomography (PET) suggested a modulatory effect at central level, particularly at Orbito-frontal (OFC) and Anterior Cingulate cortex (ACC) levels, exerted by the eTNS used for preventive purposes (12). The modulation of ACC after two months of preventive treatment with Cephaly was also confirmed by a Functional Magnetic Resonance (FMRI) study on 16 migraine without aura patients (13). Laser evoked potentials (LEPs) were used to analyze the status of the nociceptive system (14). They were employed to analyze the mechanism of action of the transcutaneous electrical stimulation (TENS) in chronic pain, on the basis of the changes induced on the pain sensation and on the cortical potentials obtained by the laser stimuli. These studies, one controlled by sham session, confirmed a reduction of LEP amplitude and subjective pain by laser stimuli, and a real neurophysiological TENS-related effect on nociceptive transmission (15,16,17). However, these studies investigated the effects of high frequency TENS on limb stimulation in healthy volunteers, leaving the mechanism of action of this stimulation on the complex dysfunction of the trigeminal system in migraine still unexplained.
In this pilot study, we aimed to test the effects of a single eTNS session on pain perception and cortical responses induced by painful laser stimuli to the right forehead and the back of the right hand in a cohort of migraine without aura patients and healthy controls.
Methods
Subjects: Our sham-randomized-controlled study included 22 consecutive patients, 16 women and six men (mean age 35 years, SD 6.7, range 21–56), selected at the tertiary Headache Center of the Applied Neurophysiology and Pain Unit of Bari University. Twenty-two age- and sex-matched healthy volunteers (16 women and six men, mean age 34.9, SD 5.8, range 20–57, ANOVA F 0.25, n.s.) responded to a call among the University staff. All patients were migraine without aura sufferers who met ICDH-3 beta 2013 criteria (18). The clinical and neurological examinations were normal in all subjects. Analgesics intake in the last 48 h at the time of recording, CNS acting drugs intake (including preventive treatment for migraine) in the last three months, co-morbidity for medical, neurological, or psychiatric diseases, were causes of exclusion. Exclusion criteria for controls was first-degree inheritance for migraine. Migraine patients were recorded during the interictal phase at least 72 h after the last attack and 48 h before the next one, as ascertained by a telephone interview.
Transcutaneous supraorbital nerve stimulation: The eTNS was delivered with a 30 mm × 94 mm self-adhesive electrode placed on the forehead, covering the supratrochlear and supraorbital nerves bilaterally. The eTNS generates biphasic rectangular impulses with an electrical mean equal to zero and with the following characteristics: Frequency 60 Hz, pulse width 250 µs, intensity 16 mA. The eTNS sessions lasted 20 minutes. For sham stimulation, we used the same procedure described above, interrupting the increase of the electrical stimulation intensity after 10 seconds at 2 mA final intensity, which was perceived as a weak paresthesia in all cases.
Laser Evoked Potentials Stimulation procedure: Cutaneous heat stimuli were delivered by a CO2 laser (wavelength 10.6 µm, 2 mm beam diameter (ELEN, Florence, Italy)) on the right back hand and the right supraorbital zone.
The duration of the stimulus was 30 msec. The laser pain threshold (Pth) was established by delivering a series of stimuli at increased and decreased intensities using 0.5 W steps. The pain threshold was the lowest intensity that enabled at least 50% of the stimuli to be perceived as a painful pinprick. The laser intensity was two steps over the Pth, in accord with the procedure suggested by Treede et al. (14). We asked all subjects to rate the laser pain on a VAS (Visual Analogue Scale) after each trial of stimulation. In the 0–100 VAS, the white color, corresponding to 0, means no pain sensation, the intense red, corresponding to ‘100’, means the worst pain conceivable. For each stimulation site and each condition, we delivered one series of 30 laser stimuli, with an inter-series interval of 5 min. To avoid damage to the skin, fatigue or sensitization of nociceptors, we changed the inter stimulus interval pseudo randomly (10–20 s) and shifted the irradiated spot after each stimulus.
Laser evoked potentials recording: During LEP recording, the subjects laid on a couch in a warm, semi-darkened room and were awake and relaxed with their eyes open. We recorded LEPs using 21 surface recording electrodes placed on the scalp referred to the nasion, according to the International 10–20 System, by means of a MICROMED EEG apparatus (Micromed Brain Quick, Mogliano Veneto, Italy). Two additional electrodes were positioned above the eyebrows for the electrooculogram (EOG) recording; the ground electrode was located at Fpz.
Experimental procedure: All subjects were informed about the experimental procedure. The study was approved by the local Ethic Committee (EC) of Bari Policlinico General Hospital, and all participants signed an informed consent. We were not ethically allowed to give public access to our clinical database. Data could be accessible on specific request, after an ethical permission and the deletion of any personal detail.
All the recording sessions were performed in the morning, at the Laser Laboratory of the Applied Neurophysiology and Pain Unit. Control subjects and migraine patients were randomly assigned to real or sham eTNS with 1:1 computer-based assignation, performed by one of the laboratory nurses. The medical staff responsible for LEP analysis as well as the technical staff responsible for LEP recording were blinded in regard to the diagnosis and the type of procedure. For real and sham stimulation, we recorded LEPs in basal condition (T0), during (T1) and just after these (T2). In T1, LEPs were recorded after at least two minutes of the occurrence of a clear sensation of paresthesia and vibration (on average 2.75 ± 0.4 min. after the Cephaly switching on). We applied laser stimuli on the right forehead and also on the right hand in a random order, to be able to distinguish a possible direct effect on the trigeminal branch from a remote and non-topographically specific effect. All subjects were blinded during the entire procedure. All the procedures used in this study were consistent with the Code of Ethics of the World Medical Association (Declaration of Helsinki).
LEP analysis: An investigator who was blinded to the procedure and clinical condition analyzed the LEP recordings, considering 1 second as post stimulus and 100 ms of pre-stimulus time, at a sampling rate of 256 Hz and bandpass filters 0.1–70 Hz. All LEP recordings containing transient signals that exceeded 65 mV or oculomotor artifacts in any recording channel were excluded from the average by an automatic artifact rejection algorithm. Other artifacts were visually inspected. The artifact induced by the Cephaly device was cut off using a 60 Hz digital filter. For each stimulation site, we evaluated the average of at least 21 valid (artifact-free) potentials. The LEPs were identified on the basis of their latency and distribution; three potentials (N1, N2 and P2) were labeled according to the procedure of Valeriani et al. (19). The N1 component was analyzed at T3 referred to Fz; the N2 and P2 components were analyzed at the vertex – Cz referred to nasion (14,19). Considering its importance in migraine pathophysiology, we also evaluated the habituation pattern: For its computation, the sequence of potentials was divided into three blocks and we considered the average of at least five artifact-free consecutive potentials for each block (20,21). Habituation was computed for the N2-P2 complex, given that the N1 is small in amplitude and recognizable only if a consistent number of potentials is averaged. The ratio between the third block and the first block of averaged LEP potentials was considered as the habituation index. Values under 1 indicated habituation (21).
Topographical analysis of LEPs was obtained using standardized low-resolution brain electromagnetic tomography by LORETA software (SAKA 2011 version) (22–24). Numerous studies have supported the usefulness and validity of LORETA in localizing generators of scalp-recorded potentials, including those related to pain processing and modulation (25–28). The randomization procedure for the Statistical non Parametric Maps (SnPM) was applied, with 5000 randomization, according to LORETA software (29).
Statistical analysis: The one way ANOVA with conditions of sham vs. real, phases T0 vs. T1 vs. T2 and group migraine vs. controls as factors, and LEP latencies and amplitudes, laser pain rating, and N2P2 habituation index as variables, was performed separately for face and hand sites. The post-hoc Bonferroni test was also employed to compare the three phases T0, T1 and T2. The statistics were computed by IBM SPSS version 21.
For LORETA analysis, statistical differences between conditions were computed as images of voxel-by-voxel t-values. The localization of the differences in cortical activity was based on the standardized electric current density and resulted in three-dimensional t-score images. In these images, cortical voxels of statistically significant differences were identified by a non-parametric approach, with a 5% probability level threshold determined by 5000 randomizations (22). A randomization procedure was implemented to control for type I errors arising from multiple comparisons (29).
Paired t tests (T0–T1) and (T0–T2) were performed in sham and real conditions in migraine and control groups. Then the comparison (T0–T1 sham) – (T0–T1 real) and (T0–T2 sham) – (T0-T2 real) (T0,T1,T2 were paired, real and sham were independent) was made separately in migraine and control groups, as the Loreta software did not enable comparison of the sham vs. real effect between the two groups.
Results
We report data on 17 patients and 21 controls: In fact, one patient was excluded because an attack occurred just after the experimental session, and the LEPs of four patients and one control were not available due to the excessive presence of ocular artifacts. Eight migraine patients (mean headache frequency: 3.2 ± 0.9 days with headache/month in the last three months, migraine duration 8.8 ± 4.5 years) and 11 controls were finally evaluated in real and nine patients (mean headache frequency: 3.8 ± 0.5 days with a headache/month in the last three months, migraine duration 10 ± 3.5 years, ANOVA F 0.45, n.s.) and 10 controls in sham conditions.
Mean and standard deviations (SD) of laser evoked potentials recorded from the right forehead amplitudes and N2P2 habituation index in migraine patients (M) and controls (C), for the t-SNS Real and sham stimulations. The results of one way ANOVA are reported.

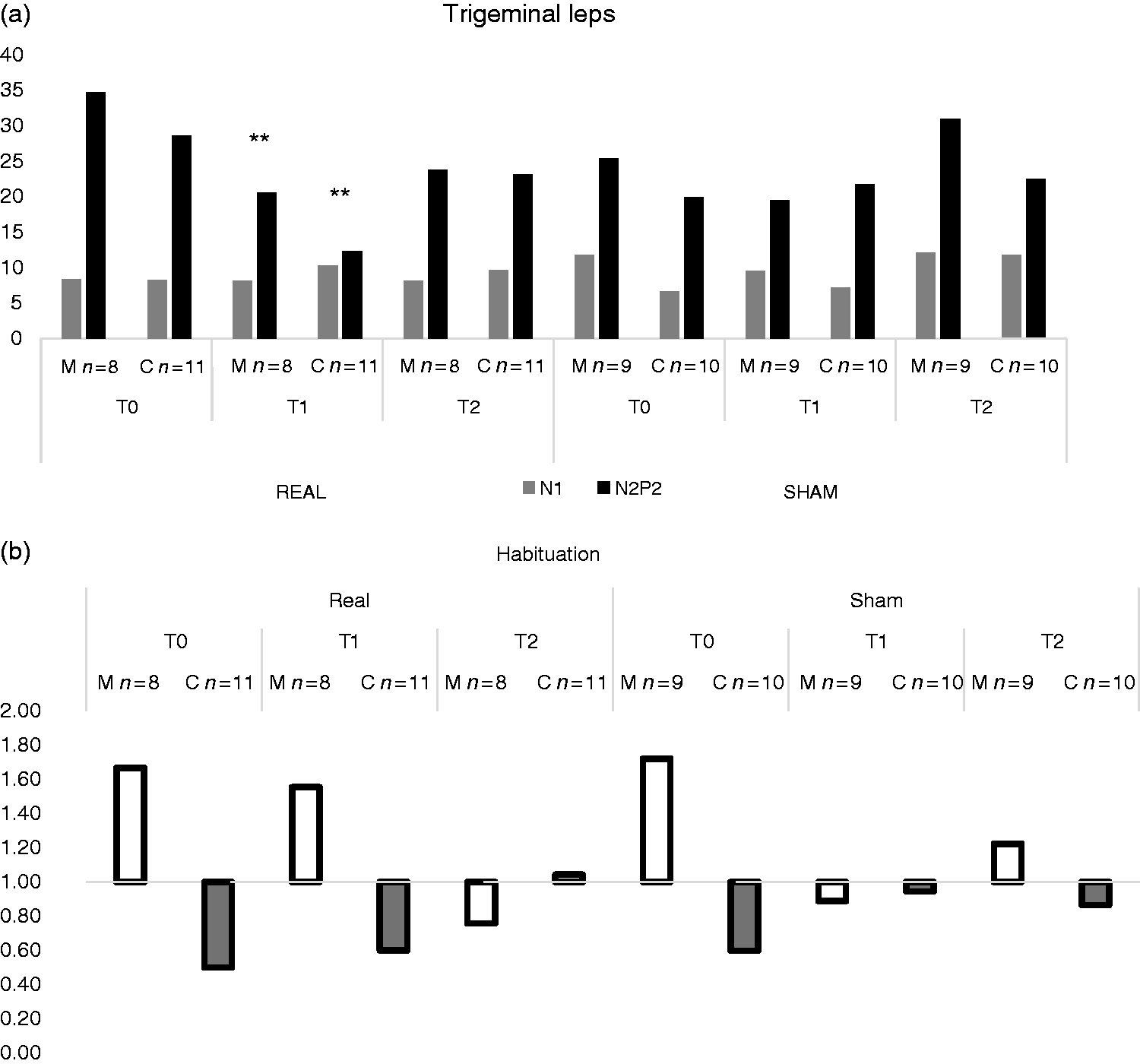
Mean of (a) Laser Evoked Potentials (LEPs) amplitudes and (b) habituation index of N2P2 components from trigeminal stimulation recorded before (T0), during (T1) and after (T2) eTNS by Cephaly device (real) and sham stimulation in migraine patients and controls. Results of Bonferroni test are reported (a) T1 vs. T0 and T2; **p < 0.01, (b) in the migraine group: T0 vs. T1 and T2 *p < 0.05 T0 vs. T1, °°p < 0.01; T0 vs. T2 °p < 0.05.

Laser evoked potentials by right supraorbital stimulation in two representative migraine patients and healthy volunteers, submitted to real eTNS and sham stimulation. Each track is the average of at least 21 artefact free potentials. Black, T0; blue, T1; red, T2. (a) Real stimulation, control subject, 21 years old; (b) real stimulation, migraine patient, female, 22 years old; (c) sham stimulation, control subject, female, 22 years old; (d) sham stimulation, migraine patient, 25 years old.
Mean and standard deviations (SD) of laser evoked potentials recorded from the right hand, amplitudes and N2P2 habituation index in migraine patients (M) and controls (C), for the t-SNS real and sham stimulations. The results of one way ANOVA are reported.

LORETA analysis: Voxel-by-voxel t-values were computed for the comparison of trigeminal LEPs between T0–T1 and T0–T2 phases in migraine patients and controls in real and sham conditions. A significant amplitude reduction of LEPs in the 200–350 msec time range was found in real T1 in patients and controls. This reduction was evident in the bilateral prefrontal regions, anterior cingulate cortex, and right insula in migraine, while the sham stimulation caused a reduced representation of cortical activity induced by laser in the right medial prefrontal gyrus. The comparison between real and sham stimulations (T0–T1 sham) – (T0–T1 real) confirmed significant Loreta values changes in the 250–350 msec range in patients, corresponding to a reduced activation of the anterior cingulate cortex (Broadmann area 24), induced by the real eTNS (Figure 3). In controls, the real stimulation seemed to reduce the LEPs generated in the medial frontal gyrus and anterior cingulate, while no clear difference emerged in the sham condition. However, the real eTNS showed a trend toward a more evident reduction of LORETA sources activation in the medial frontal gyrus and anterior cingulate, which approached statistical significance in respect to placebo (Figure 4).
Statistical Randomized non-Parametric Maps showing locations of significant changes of brain activity related to trigeminal laser stimulation in the time interval 200-350 msec in migraine patients; (a) Paired t test (T0–T1) in group subjected to real eTNS. The critical t values were localized in the medial frontal gyrus and anterior cingulate, Broadmann areas 11 and 24; (b) paired t test (T0-T1) in group submitted to sham eTNS, Broadmann areas 11 and 24; (c) (T0–T1 sham) – (T0–T1 real); critical t values in ACC, Broadmann area 24. For these comparisons, the critical threshold for t value was automatically settled at p < 0.05 (the yellow color indicated a significant reduction, the light blue a significant increase). Statistical Randomized non-Parametric Maps showing locations of significant changes of brain activity related to trigeminal laser stimulation in the time interval 200–350 msec in controls. (a) Paired t test (T0–T1) in group submitted to real eTNS – the critical t values were individuated in the medial frontal gyrus-Broadman area 10; (b) paired t test (T0–T1) in group submitted to sham ETNS; (c) (T0–T1 sham) – (T0–T1 real). For these comparisons, the critical threshold for t value was automatically settled at p < 0.05 (the yellow color indicated a significant reduction, the light blue a significant increase; each scale reports the critical t values for the single comparisons).

Discussion
To our knowledge, this is the first study of the effects of trigeminal eTNS on nociceptive potentials induced by noxious laser stimuli. Our pilot study preliminarily demonstrated that, compared with sham stimulation (placebo), the eTNS with Cefaly® induced a significant but transient decrease in trigeminal late cortical potentials by acute pain in migraine patients and healthy subjects. In the following paragraphs, the main results will be discussed in detail.
The effects of eTNS on the latencies and amplitude of LEPs: The real eTNS did not change the trigeminal or brachial LEP latencies, in accordance with previous studies dealing with the effects of high frequency TENS on LEPs (15–17). Also, the N1 amplitude remained substantially unaffected by real and sham eTNS in patients and controls, a result that does not confirm a peripheral action on a-delta fibers or a central action on early cortical processing of pain stimuli. Despite the limitations related to the analysis of N1 (14), mainly for studies performed in small cohorts, it remains an interesting potential to be investigated. In fact, it represents the first cortical response by a-delta fibers, and it contributes to the neurophysiological examination of the peripheral pathways and the operculoinsular cortex devoted to the sensory-discriminative aspect of pain (30,31).
Only one study reported the effects of TENS on the N1 component (17), consisting of an inhibition of this early response evoked by the limb where the TENS was applied. This could indicate that the nociceptive volley was attenuated before its arrival to the cortex, with a “gate action” at the peripheral level through a ‘‘busy line effect’’ (32). The lack of a possible peripheral effect on the right a-delta trigeminal afferents induced by Cefaly® in migraine and controls could be due to different reasons, such as anatomical and physiological peculiarities of the trigeminal system, or the placing of TENS application in the middle of the forehead, not specifically in correspondence with the anatomic course of the right supraorbital branch stimulated by the laser. This effect was also absent for the N1 wave obtained by stimulation of the hand, which is in line with a lack of action of the transcutaneous electrical stimulation on the first cortical station of laser stimuli coming from outside the anatomical district of TENS application (17). We observed a significant reduction of the N2P2 vertex complex obtained by the right supraorbital stimulation during the real eTNS, with a partial recovery soon after. A slight sham effect on trigeminal LEPs was also present in migraine patients, but it was significantly smaller than that exerted by the real eTNS.
This result could be attributed to a central effect of the real eTNS on the cortical generators of the vertex complex, localized in the bilateral insula and anterior cingulate cortex, which are devoted to the cognitive and emotional compound of pain experience (30). It would be a cognitive effect topographically limited to the anatomical regions stimulated by the eTNS, considering that the LEPs from the hand were not significantly modified by the transcutaneous electrical stimulation directed over the forehead. However, a slight reduction also involved the N2P2 complex evoked from the hand, suggesting a possible extra-cranial effect, at least in a few cases. Despite the Cefaly® device being unable to exert a gating effect on the peripheral trigeminal branch, the clear sensation of electrical superficial vibration over the forehead could have diverted the subject's attention away from the laser stimuli to the face. This effect seemed to be transient and not prolonged beyond the stimulation period, though the N2P2 amplitude recovery occurring after the stimulation session was incomplete in patients and controls. An increase in the number of cases would confirm the possible persistence of the eTNS effect, in line with other studies on the effects of high frequency TENS on LEPs (17). Differently from those studies, the eTNS and the sham stimulations did not change the subjective laser pain sensation in patients and controls. The first studies on LEPs indicated a linear correlation between the vertex N2P2 and the subjective pain sensation (33), but more recently the N2P2 amplitude was attributed to the subjective expression of stimulus salience, more than being a direct correlate of pain perception (34). The same lack of correlation was observed in large groups of normal subjects (21), ultimately confirming that the N2P2 amplitude and the subjective laser pain perception are independent patterns. In the present study, the real eTNS would cause a decrease in the relevance attributed to the contemporary trigeminal laser stimuli. It would result in a deviation of attention away from supraorbital stimuli, which was more relevant in comparison to that exerted by the low intensity placebo stimulation in patients and controls. A clear vibration sensation seems to be necessary to divert the subject's attention away from the trigeminal laser stimuli, thus modulating the cortical regions generating the vertex complex, as reported below.
LORETA analysis: In accordance with the above reported hypothesis of a diversion of attention from the trigeminal stimuli, the topographical analysis showed that the real eTNS reduced the LEPs representation in the cortical prefrontal regions, anterior cingulate and insula, which were also indicated in previous studies with LORETA as the N2P2 wave's generator (25–27,35,36). In migraine patients, the activity in the prefrontal cortex, especially in the medial region, was also reduced by the placebo stimulation, though in a non-significant way. Although the statistical comparison of the N2P2 amplitude did not show relevant differences between groups, the LORETA analysis based on the source reconstruction of main potentials revealed a different effect from eTNS in migraine patients with respect to controls. In fact, in healthy subjects the effect of real eTNS was limited to the medial prefrontal cortex, while the sham stimulation did not exert evident effects. In migraine, the real ETNS caused an inhibition of the anterior cingulate cortex to a greater extent in respect to placebo, an effect present in a limited measure also in controls. The prefrontal and anterior cingulate cortex are pivotal regions in pain control (37). In previous studies, the generator of the trigeminal LEP component localized in the ACC seemed to change its activity during the attack and in chronic patients (38,39), confirming it as a key zone in migraine pain processing. Positron Emission Tomography (PET) studies have indicated that three months of Cefaly® treatment caused a modulation of the metabolism of the orbito-frontal and anterior cingulate cortex in migraine patients (12). In a recent FMRI study in migraine without aura patients, the increased BOLD response in the ACC under noxious heat stimuli was reverted after 60 days of eTNS, an effect correlated with the clinical efficacy (40). The authors suggested a possible action of eTNS on the mechanisms of antinociceptive modulation in the ACC, explaining its preventive anti-migraine efficacy. Here we could confirm a short-term modulation of LEPs originating from the ACC, which can support the effect of eTNS in the acute phase of migraine (5–7). Further studies are needed to confirm that this short-term inhibition exerted by the single stimulation session may cause long-term changes in the nociceptive cortex that are useful for migraine prevention (2–5,39).
Effects on LEP habituation: The effect of real eTNS on trigeminal LEP habituation was not dissimilar to the sham stimulation, indicating that even a low intensity trigeminal stimulation may interfere with sustaining attention toward the laser stimuli, which is the basis of the lack of progressive decline of cortical potentials. Reduced habituation to painful stimuli is a feature of different forms of chronic pain (41,42), which may be reverted by preventive treatments (43,44). The neurostimulation methods act in a complex way on LEPs habituation, as shown by the TDCS studies (45). The amplitude of the N2P2, averaged for the total of the responses, and the ratio between the last and the first responses block, that is, the habituation index, are independent patterns, as shown in previous studies (21). While the real stimulation exerted a global inhibition of the nociceptive cortex, starting from the first series of laser stimuli, the sham eTNS probably produced only a diversion of attention during the laser session, which was able to disrupt the persistence of the high level of cortical activation that causes the LEP reduced habituation. Many studies focused on the biological action of the placebo effect, and the possible utility of sham procedures for clinical therapeutic strategies (46). The effects of treatments on LEP amplitude and habituation may in some way indicate their efficacy in migraine. In previous studies on the effects of pharmacological therapies on LEPs of migraine patients, the reversion of the dis-habituation pattern was linked to the biological properties of the drugs (41,42), while the neurostimulation procedures were accompanied by an evident placebo phenomenon (47), which could be taken into account to explain their clinical efficacy (42). The effect of sham stimulation on reduced LEPs habituation observed in migraine would not be accompanied by a reduction of the amplitude of the averaged potentials, confirming the mutual independence of the two patterns, the first referring to the modulation of attention during the single series of stimulation, the second to a global inhibition of the cortical areas generating the LEPs.
Study limitations
The major limitation of this study is the small number of cases, and the further reduction due to different reasons, such as the artifacts. The sham group presented with a slight reduction in LEPs amplitude which, although not significant, could bias the results in favor of a “real” effect, if one assumes that a significant amplitude decrease is more likely to occur when the initial amplitude is high. To avoid a time-consuming procedure, we did not perform a baseline recording before T0 to assess the effect of time itself. This could exclude changes due to the physiological intra-individual variability. Another potential flaw may be the validity of the placebo procedure, which is always a matter of debate in neurostimulation studies (48). However, the sham method employed here, based on low-intensity stimulation, was effective in modulating LEPs habituation, but inefficacious in reducing the amplitude of the averaged LEPs, a result that could refer to a manipulation of attention caused by a strong placebo effect.
Another important limit is the absence of correlation between neurophysiological results and possible clinical efficacy, which would be the argument of further studies.
Conclusions
This pilot study showed that transcutaneous trigeminal stimulation by the Cefaly® device could reduce the late LEPs recorded from the forehead, influencing the cortical processing of trigeminal stimuli and the cortical areas devoted to the cognitive aspects of pain in migraine patients and controls. The reversion of the neurophysiological effects after the single session of stimulation was incomplete, and worthy of confirmation in larger groups. The strong placebo effect exerted by the device seemed able to modulate the pattern of LEPs reduced habituation in migraine patients, which ultimately is not a bad result, considering the neural mechanism of placebo (37) and the correlation between the reversion of the dis-habituation pattern and the biological activity of different preventive treatments for migraine (43,44). In conclusion, the present pilot study could suggest that eTNS may interfere with the threshold and the extent of trigeminal system activation, with a mechanism of potential utility in the resolution and prevention of migraine attacks.
Footnotes
Article highlights
Supraorbital transcutaneous electrical stimulation was more efficacious than placebo in reducing the late trigeminal laser evoked potentials.
Laser evoked potentials source in the anterior cingulate cortex was inhibited by the real transcutaneous electrical stimulation.
The pattern of reduced LEP habituation was reversed by both real and sham stimulations in migraine patients.
The Cefaly® device may interfere with the extent of trigeminal system activation, with a mechanism of potential utility in the resolution and prevention of migraine attacks.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marina de Tommaso had financial support for conferences and lessons from Allergan, Pfizer, Myalin and Mundipharma Industries.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Bari University Research fund.
