Abstract
This study analysed the organization of central nervous system networks involved in the processing of meningeal inputs in the male, Sprague-Dawley rat. We injected the anterograde tracer, biotin dextran, into areas of the medullary trigeminal nucleus caudalis (Sp5C), which receive inputs from the ophthalmic division of the trigeminal nerve. Double-labelling immunohistochemical studies were then performed to compare calcitonin gene-related peptide (CGRP) or serotonin 1D (5HT1D) receptor distributions in the areas innervated by Sp5C neurons. Dense, topographically organized intratrigeminal connections were observed. Sp5C neurons projected to the commissural subnucleus of the solitary tract, A5 cell group region/superior salivatory nucleus, lateral periaqueductal grey matter, inferior colliculus and parabrachial nuclei. Trigeminothalamic afferents were restricted to the posterior group and ventroposteromedial thalamic nuclei. Some of these areas are also immunoreactive for 5HT1D and CGRP and thus remain potential central targets of triptan molecules and other antimigraine drugs.
Introduction
The pain of headaches is elicited by activation of trigeminovascular primary afferents from the ophthalmic division of the trigeminal nerve, which densely innervate meningeal blood vessels (1–3). In different animal models, it has been shown that meningeal nociceptors activate neurons in the grey matter of the trigeminal nucleus caudalis (Sp5C) and the upper cervical spinal dorsal horn (4, 5). The axons of these neurons in turn convey pain signals to higher brain regions, including parts of the brainstem, thalamus and cerebral cortex.
One of the main outstanding questions is how changes in activities of both first- and second-order neurons and the circuits that they activate within the central nervous system (CNS) can elicit headache pain and its concomitant reactions. With respect to episodic headaches, a number of animal studies have been devoted to the neurobiological mechanisms of migraine (6, 7) due to its higher prevalence and great importance in public health (8). It has been suggested that the pain that occurs during the triggering of migraine attacks involves peripheral sensitization elicited by neurogenic inflammation of meningeal primary afferents (9, 10). Moreover, the ongoing pain that outlasts the initial migraine attack and spreads to widespread areas of the body is due to central sensitization of Sp5C neurons elicited by sustained peripheral inputs (11) and changes in descending modulatory mechanisms (12). Better knowledge of the functional architecture of the CNS circuits involved in trigeminovascular processing could help elucidate the mechanisms underlying the variety of pathophysiological changes that occur during migraine and other headaches.
The aim of the present work was to analyse the organization of the projections in the CNS through which medullary trigeminovascular neurons might elicit the symptoms of headache. For these purposes, the anterograde tracer biotin dextran (BD; (13)) was injected into the Sp5C areas that contain trigeminovascular neurons driven from the ophthalmic division of the trigeminal nerve. In addition, double-labelling immunohistochemical studies were performed to compare calcitonin gene-related peptide (CGRP) and serotonin 1D (5HT1D) receptor distributions in the areas that, according to our BD tracing experiments, receive dense projections from trigeminovascular neurons.
Materials and methods
Animal preparation
All the animal experiments were approved by our local animal care committee and were in accordance with the guidelines of the International Association for the Study of Pain (14). Surgical procedures were performed on 35 male Sprague-Dawley albino rats, weighing 250–300 g, which were anesthetized with chloral hydrate (400 mg/kg, intraperitoneally).
Electrophoretic injections of biotin-dextran into the trigeminal nucleus caudalis
The rats were mounted with their necks in a ventro-flexed position in a stereotaxic device. The atlanto-occipital membrane and the dura were opened. Glass micropipettes (25–40 µm diameter tips) filled with a 10% solution of BD (molecular weight 10 000; Molecular Probes, Eugene, OR, USA) were inserted at an angle of 20–30° to the vertical plane, 2700–4000 µm caudal to the obex. The rostrocaudal location of the injection sites was around 5.3–5.6 mm caudal to the interaural line in accordance with the stereotaxic atlas of Paxinos and Watson (15). In a pilot study, we explored systematically the part of the spinal trigeminal nucleus that contains cells responding to meningeal inputs. All the neurons that responded to meningeal stimuli (Fig. 1C2
) were also activated by tactile stimulation of the periocular region (Sp5C, Fig. 1C3
). According to previous electrophysiological studies of the Sp5C (16), neurons which can be driven from the dura and which have additional, periocular receptive fields, are confined to its caudal, ventrolateral aspect (Fig. 1C1
). Multi-unit activity was recorded through the electrode filled with BD, and once the neurons responding to either meningeal or periocular stimuli were encountered, direct current (2–4 µA; micropipette positive) was applied for 20 s every 30 s for 15–20 min. Such an injection period allowed the injection electrophoretically of a sufficient amount of the anatomical tracer BD to produce anterograde labelling. Following a postoperative survival period of 1 week, the animals were re-anaesthetized, perfused over a 20-min period with 200 ml of warm (37°C) heparinized saline, followed by 500 ml of phosphate-buffered saline solution (0.2

Location and extent of biotin dextran (BD) injection sites (black areas for the two representative cases) in schematic representations of coronal sections of the caudal medulla, 1.600 µm caudal to the obex. Injections were performed following electrophysiological exploration of the most lateral (A) or medial (B) trigeminal nucleus caudalis (Sp5C). As shown in the individual example (C), the ventral Sp5C region (C1, recording site) contains neurons driven from the dura (C2, Aδ and C-fibre responses evoked by 1-mA repetitive pulses of electrical stimuli applied on the surface of the transverse sinus). These neurons have additional, periocular receptive fields (C3, responses evoked following brushing of the periocular skin). Bright field photomicrographs of labelling in a coronal section of the caudal medulla, 1.600 µm caudal to the obex (D) following a BD injection in the medial Sp5C (injection site in B). The images show a high density of labelled terminals in the commissural aspect of the nucleus of the solitary tract (SolC; D1). Contralateral projections were distributed as a diagonal band in the magnocellular and superficial layers of the ventral Sp5C (D2).
The sections were divided into three groups. Two groups were processed for avidin–biotin complex/diaminobenzidene (DAB) histochemistry in order to localize the anterograde labelling in different brain structures; one of these groups was counterstained with Cresyl Violet. When the BD labelling was strong enough, the third group was processed for immunohistofluorescence. In brief, the sections were preincubated for 1 h in TBS containing 0.3% Triton X-100, 0.3% bovine serum albumin (BSA) and 2% normal mouse serum. They were then incubated in the same solution for 2 h with a 1 : 200 anti-biotin antibody conjugated with the red chromogen Cy3 (produced in mice; Jackson, West Grove, PA, USA). After successive rinsings with TBS, the sections were preincubated for 1 h in TBS–BSA–Triton and 2% normal goat serum and were then incubated at room temperature for 12 h either with a 1:10 000 anti-5HT1D primary antibody produced in rabbits (see (17)) or with a 1:10 000 anti-CGRP primary antibody produced in rabbits (Peninsula Laboratories, San Carlos, CA, USA). After 20 min rinsing with TBS, the sections were incubated for 1–2 h with a 1:200 secondary antirabbit antibody (produced in goats; Jackson) conjugated with the green fluorescent chromogen Cy2. The level of fluorescence was controlled under epifluorescence microscopy.
Analysis of anterograde labelling with BD
In all the BD experimental series, the injection sites, labelled fibres, varicosities and boutons were examined under bright and dark field illumination. Each injection site was analysed using camera lucida drawings of coronal sections processed with DAB. Some representative cases including the full extent of the injection sites were grouped on standard drawings of the caudal medulla (Fig. 1A,B). The delineation and nomenclature of brainstem and supramedullary areas was based on the atlas of Paxinos and Watson (15). The areas of interest were also fully identified in our counterstained material.
Computer-assisted reconstruction of BD labelling
In some cases, bright-field photomicrographs of the injection sites and representative labelling were built as digitized grey scale images using computer-assisted reconstruction of labelling. The technique employed has been described in detail elsewhere (18).
Analysis of the anterograde labelling with BD combined with CGRP or 5HT1D labelling
Digitized images were captured with an Olympus FV300 confocal microscope. Each image was made by assembling several z-series captured in 1-µm steps. Images of the anterograde labelling and CGRP or 5HT1D labelling were simultaneously captured in the same section of tissue and overlaid in a single file, and then imported into Photoshop (Adobe Systems, San Jose, CA, USA) for additional processing.
Results
The data reported here are based on experiments in which the BD injection sites were located in the most lateral (n = 10; Fig. 1A) or medial (n = 10; Fig. 1B) region of the Sp5C that contained the majority of neurons driven from the dura with additional periocular receptive fields (Fig. 1C). Two sites were selected on the basis of their small sizes and locations. These covered the superficial laminae and magnocellular layers of the Sp5C, where the majority of neurons driven from the dura were located (16). The selected injections covered the caudal aspect of the ventrolateral Sp5C, spreading to the upper cervical 1 (C1) segment.
As detailed below, the main findings of this study were that both the lateral and medial Sp5C projected to common areas in the brainstem and thalamus. Qualitative analyses showed only slight differences in the density of projections. The lateral Sp5C more densely innervated some rostral brainstem areas, including the lateral periaqueductal grey (PAG) matter and adjacent external inferior colliculus. Moreover, a close relationship between Sp5C projections and CGRP or 5HT1D labelling was observed only in some brainstem areas.
We will present successive, representative cases of labelling following BD injections in the most lateral or medial Sp5C, starting from the most caudal part of the brainstem up to the most rostral labelled diencephalic structures. In addition, we will describe the distribution in brainstem areas of CGRP or 5HT1D immunoreactivity and their topographical relationships with regard to Sp5C projections.
Ascending projections following anterograde tracing from the Sp5C
The distribution and density of caudal brainstem projections were similar from the lateral and the medial Sp5C. Figure 1 illustrates labelling from the medial site (Fig. 1D). Bilateral projections were confined to the commissural aspect of the nucleus of the solitary tract (Fig. 1D1 ) and covered all its caudo-rostral extent. Contralateral projections crossed the midline and were distributed as a diagonal band of varicosities intermingled with fibres of passage in the magnocellular layer, spreading in the superficial layers of the ventral Sp5C (Fig. 1D2 ). Sparse labelling was also observed bilaterally in the subnucleus reticularis dorsalis (SRD). These projections extended more rostrally also, including the ventral aspect of the trigeminal nuclei oralis and principalis, bilaterally (Fig. 2). Labelling also spread to the superior salivatory nucleus (SSN)/A5 cell group region within its full rostro-caudal extent.

Bright field photomicrographs of labelling in coronal sections of the brainstem following biotin dextran (BD) injection in the lateral trigeminal nucleus caudalis (Sp5C). The images show a high density of labelled terminals in the ventral part of the ipsilateral and contralateral trigeminal nuclei oralis (Sp5O) and principalis (Pr5) 10.8 mm and 9.8 mm posterior to Bregma, respectively. Labelling also spreads to the superior salivatory nucleus/A5 cell group region (SSN/A5) throughout its rostro-caudal extent.
More rostrally, dense labelling was observed in the parabrachial area, including the dorsolateral and central lateral subnuclei (Fig. 3). Labelling was also observed in other parabrachial areas, including the ventrolateral, internal lateral, external lateral, and superior lateral subnuclei. Dense labelling, notably from the lateral Sp5C (injection site in Fig. 1A) was observed contralaterally, within the lateral PAG matter and adjacent external inferior colliculus (Fig. 3).

Bright field photomicrographs of terminal labelling in coronal sections of the dorsal (PBdl) and central lateral (PBcl) parabrachial nuclei, between 9 and 9.6 mm posterior to Bregma. Labelling spreads rostrally within the lateral periaqueductal grey matter (LPAG) and external inferior colliculus (ECIC), 7.6 mm posterior to Bregma, following biotin dextran (BD) injection in the lateral Sp5C (injection site in Fig. 1A).
Diencephalic labelling was distributed contralaterally, including in the posterior triangular thalamic (PoT) nucleus, the posterior thalamic nucleus and the lateral/dorsal aspect of the ventral posteromedial (VPM) thalamic nucleus (Fig. 4).

Bright field photomicrographs of coronal sections of the diencephalon showing contralateral labelling within the posterior triangular thalamus (PoT, 4.8 mm posterior to Bregma), posterior thalamic nucleus (Po) and the ventral posteromedial thalamic nucleus (VPM), 4.4 mm posterior to Bregma, following biotin dextran (BD) injection in the lateral trigeminal nucleus caudalis (Sp5C) (injection site in Fig. 1A).
Brainstem projections from the Sp5C combined with CGRP or 5HT1D labelling
As shown in Fig. 5, the projections from the Sp5C were intermingled with CGRP and 5HT1D immunoreactive fibres in the commissural subnucleus of the nucleus of the solitary tract (Fig. 5A1,A2 ) and the contralateral Sp5C (Fig. 5A3,A4 ). More rostrally, efferent projections were intermingled only with CGRP immunoreactive fibres within the ipsilateral trigeminal nucleus oralis (Fig. 5B), and both CGRP and 5HT1D fibres were intermingled with efferents from the Sp5C within the external lateral parabrachial area (Fig. 5C).
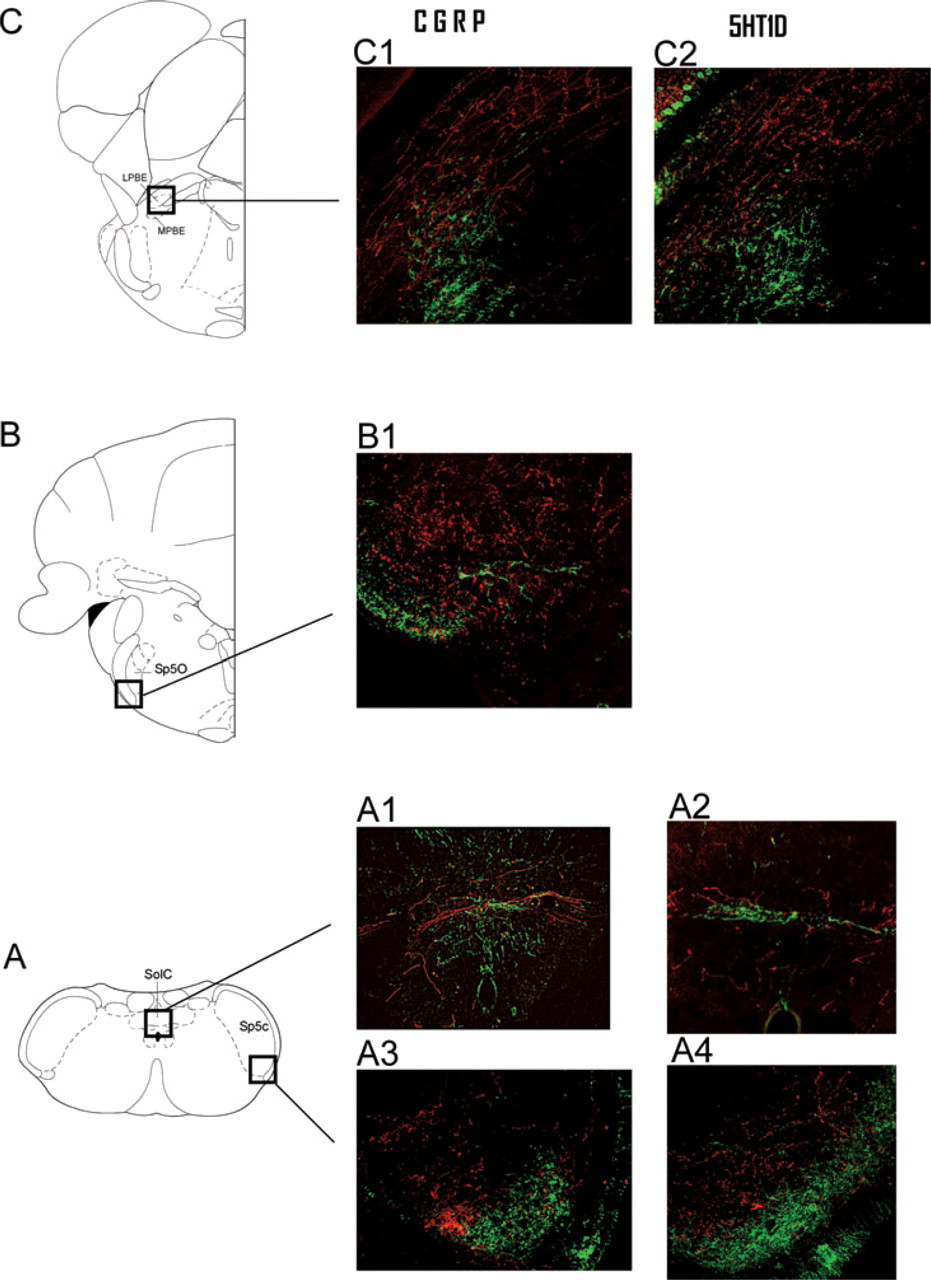
Confocal fluorescent images of anterograde labelling with biotin dextran (BD) (red) and calcitonin gene-related peptide (CGRP) or 5HT1D receptor labelling (green) in coronal sections of the brainstem (14.6, 11 and 9.1 mm posterior to Bregma, respectively), following injections of BD in the trigeminal nucleus caudalis (Sp5C) (injection site shown in black in Fig. 1A). Note that the projections from the Sp5C were intermingled with CGRP and 5HT1D immunoreactive fibres in the commissural aspect of the nucleus of the solitary tract (A1, A2) and the contralateral Sp5C (A3, A4). More rostrally, Sp5C projections were intermingled only with CGRP fibres within the ipsilateral trigeminal nucleus oralis (B1). Both CGRP and 5HT1D fibres were intermingled with efferents from the Sp5C within the external lateral parabrachial area (C1, C2).
Discussion
Interpretation of the results of the present study must take account of some technical caveats. It was not possible to inject into an Sp5C area that contained neurons that responded only to meningeal inputs, since the majority of these cells have additional, periocular receptive fields (16, 19). Our injections included large areas of the ventrolateral Sp5C, since trigeminovascular neurons are widely distributed in the superficial, most lateral and more medial, deeper laminae of the medullary dorsal horn. There was a lack of significant difference between the supramedullary projections from superficial and deeper Sp5C, and the pattern of ascending projections was similar to that observed in anterograde tracing studies of superficial spinal and medullary dorsal horns (20–22). Most trigeminovascular neurons labelled by injections of BD in our study were within the superficial laminae, which constitute a semicircular shape of superficial dorsal horn cells extending deeply into the ventrolateral aspect of Sp5C. The semicircular distribution of lamina I cells around the dorsal horn has been explained by developmental studies, which have shown that these cells migrate following a ventromedial rotation of the entire dorsal horn, from an early lateral superficial band to a semicircular position around the dorsal horn. This occurs with the arrival of small-diameter afferents that contact these neurons (23, 24). We obtained similar patterns of labelling in preliminary studies employing other anterograde tracers.
Moreover, the question of the relationship between the distribution of Sp5C projections and CGRP or 5HT1D fibres must be interpreted with caution, since our approach did not provide evidence that efferents from the Sp5C make contacts with these fibres. However, such a close relationship was observed in only a few, restricted CNS areas.
Trigeminal projections from brainstem neurons driven from the ophthalmic division: a substrate for central sensitization phenomena
As reported previously (25), ventrolateral Sp5C projections to the contralateral trigeminal brainstem are confined to its ophthalmic primary afferent projection area. This study has shown that such projections spread rostrocaudally from the caudal spinal trigeminal nucleus to the dorsal horns at levels C1–C3. Moreover, the Sp5C projects to the ventral region of the ipsilateral oral and principal nuclei over their whole rostro-caudal extent. Such a precise rostro-caudal somatotopic organization of the connections between the different divisions of the ipsilateral spinal trigeminal nuclei has been observed both in animals (19, 26, 27) and in man (28).
The functional significance of these topographically organized, intratrigeminal connections is not clear. Ipsilateral inputs from trigeminovascular neurons to rostral trigeminal nuclei could contribute to the amplification of nociceptive outputs to supramedullary structures via the oral and principal subdivisions, since these regions convey orofacial inputs to brainstem and thalamic areas (29–31). Contralateral projections could provide inputs that specifically modulate the activity of medullary and spinal dorsal horn cells driven from the ophthalmic division of the trigeminal nerve. Such inputs could become effective following long-lasting noxious stimulation of meningeal nociceptors and thus contribute to the central sensitization that occurs following long-lasting migraine attacks (11). This phenomenon is manifest as an increase in the sizes of the receptive fields and in the levels of spontaneous and evoked activities of Sp5C neurons (16). In patients, it could elicit the cutaneous allodynia that extends outside the referred pain area to the skin over the contralateral head and ipsilateral forearm (32). Central sensitization increases with time and diminishes the actions of triptans due to the increase in excitability of Sp5C neurons (33). Interestingly in this respect, intratrigeminal connections are closely associated with CGRP and/or the 5HT1D receptor, which are a main target of triptan molecules that bind to and diminish neuropeptide release (34–36). These contralateral projections could also mediate bilateral c-fos labelling that spreads from the ophthalmic division of Sp5C to C2/C3 following sustained noxious stimulation of the dura (37).
Brainstem neurons driven from the ophthalmic division project to supramedullary regions involved in autonomic and limbic functions
Sp5C projections to the ventrolateral medullary nucleus reticularis parvocellularis are distributed within the region that includes the A5 cell group region and the SSN, a cluster of parasympathetic preganglionic neurons located in a narrow area between the trigeminal oral and the facial nuclei, that project to the pterygopalatine ganglion (38, 39). Spencer et al. (39) have also reported labelling of brainstem trigeminal neurons following retrograde, transneuronal tracing from the pterygopalatine ganglion with pseudorabies virus. The pterygopalatine ganglion provides parasympathetic innervation via cholinergic and vasoactive intestinal peptide fibres of the anterior cerebral artery, lacrimal glands, nasal and palatine mucosa. Overall, these studies suggest that dural nociceptive inputs conveyed by Sp5C neurons may regulate parasympathetic preganglionic outflow via the SSN and elicit cerebrovascular changes that in turn induce protein extravasation and release of inflammatory mediators that sensitize and/or activate meningeal nociceptors. Such a vicious circle could progressively increase the parasympathetic tone, as observed during migraine attacks (40).
In addition to autonomic reactions, first- or second-order targets of trigeminovascular neurons could participate in adaptative changes that occur during headache pain. These functions include motivational, affective-defensive adaptations elicited by the paraventricular hypothalamic nucleus and PAG (41, 42); the latter region has been shown to modulate specifically the activities of Sp5C neurons (43). The affective-emotional/chemosensitive reactions, such as taste aversion, could be mediated via the dense parabrachial projections to the central nucleus of the amygdala (44, 45). Interestingly, the lateral parabrachial area that receives trigeminovascular projections is immunoreactive to both CGRP and 5HT1D. However, a close relationship between trigeminal afferents and 5HT1D or CGRP labelling does not demonstrate specific processing of headache pain inputs, since both substances are distributed in nociceptors throughout the body, including superficial laminas of the dorsal horn and Sp5C areas innervated by the three branches of the trigeminal nerve (17).
Parabrachial projections to other hypothalamic areas such as the ventromedial nucleus (46) could mediate sympatho-adrenal stress responses. Finally, exaggerated arousal reactions, such as those that elicit phonophobia, could be mediated by inputs conveyed from the Sp5C to the inferior colliculus, an area that integrates both acoustic and somatosensory trigeminal inputs (47).
Brainstem neurons driven from the ophthalmic division: projections to the thalamo-cortical system
Our data confirm previous retrograde (48) and anterograde (20, 49) tracing studies, which have shown that restricted contralateral areas within the posterior group and the ventroposteromedial thalamic nucleus are the main targets of trigemino-thalamic afferents in the rat. Sp5C neurons driven from the ophthalmic division do not innervate the ventral posterior parvicellular nucleus, a thalamic area targeted by mandibular/maxillar afferents usually considered to relay gustatory/intraoral messages (20). Labelled terminals were observed in the PoT, a caudal thalamic nucleus that conveys nociceptive inputs to the secondary somatosensory cortex and tactile and nociceptive inputs to the insular cortex and amygdala (50). More rostrally, labelled terminals were distributed in the Po and mainly in the lateral/dorsal aspect of the VPM thalamic nucleus, confirming the somatotopic organization within this nucleus. Indeed, the lateral VPM receives afferents from the ventral (ophthalmic branch) Sp5C, in contrast to intermediate and medial regions that receive intermediate (maxillar branch) and dorsal (mandibular branch) Sp5C afferents, respectively (48). Both Po and VPM convey tactile and nociceptive inputs to the primary and secondary somatosensory cortices (51) and could participate in the sensory-discriminative aspect of pain. Accordingly, a recent study has shown that neurons located mainly in the dorsal aspect of the VPM are activated by stimulation of the superior sagittal sinus (52). These data are also in agreement with human functional imaging studies that have shown activation of the VPM and dorsal thalamus following noxious thermal stimulation of the face (28) and also during headache (53, 54). However, this somatotopic organization cannot solely account for processing of pain from different trigeminal regions. Endogenous, top–down modulating mechanisms probably play an important role. In addition to the well-known bulbospinal descending controls, a number of studies have suggested that corticofugal modulation could participate in several types of pain that occur in the absence of detectable organic lesions (55, 56), including migraine (57) and probably temporo-mandibular joint disorder (TMD). TMD and headache patients have increased psychological distress, including somatization and catastrophizing (58); the latter has been correlated with activity in cortical regions that modulate affective, attention and motor aspects of pain (59).
Moreover, inputs to the frontal cortices via the medial thalamic nuclei could be conveyed by the Sp5C projections to the SRD, which in turn convey heterotopic nociceptive inputs to the ventromedial, parafascicular and other intralaminar nuclei (60, 61). Additional inputs to the frontal cortex could be conveyed by the parabrachial nucleus, internal lateral aspect, which provides nociceptive inputs via the paracentral thalamic nucleus (62).
Conclusions
In conclusion, our study has shown an ascending brainstem trigeminovascular projection system that carries a spectrum of information relevant for homeostasis. Indeed, as meningeal afferents belong to the interoception system, pain from cephalic blood vessels and from the viscera share common mechanisms (63). The similarity between our data and CNS projections from the superficial dorsal horn laminae fits with the proposal that trigeminovascular neurons provide afferent inputs necessary for interoception and accordingly participate in homeostatic processes (64).
Some brainstem areas are also immunoreactive to 5HT1D receptor and CGRP and thus remain additional, central targets of triptan molecules and other antimigraine drugs. This central network may also affect certain consciously complex behavioural activities such as those involving food intake as well as the pathways that influence the sensory-discriminative and affective aspects of pain via its connections with the cerebral cortex and limbic system. As a whole, this is a fundamental network that provides ongoing feedback mechanisms necessary to keep functioning within a homeostatic range and whose dysfunction leads to a number of adaptative changes, such as those that occur during headache pain.
Footnotes
Acknowledgements
The authors are grateful to Dr S. W. Cadden for advice in the preparation of the manuscript. We thank A. H. Ahn and A. I. Basbaum (University of California at San Francisco, USA) for the generous gift of the 5HT1D antibody. This research was supported by grants from Institut UPSA de la douleur, ECOS-Sud Programme no. C04 S01, European Union Programme Alban scholarship no. E04D027959CL to R.N., Proyecto FONDECYT 1050099 to L.C.
