Abstract
Objective
To investigate the role of NN414, a selective KATP channel opener for the Kir6.2/SUR1 channel subtype found in neurons and β-pancreatic cells, in inducing migraine attacks in individuals with migraine without aura.
Methods
Thirteen participants were randomly allocated to receive NN414 and placebo on two days separated by at least one week. The primary endpoint was the difference in the incidence of migraine attacks after NN414 compared with placebo. The secondary endpoints were the difference in the area under the curve for headache intensity scores, middle cerebral artery blood flow velocity (VMCA), superficial temporal artery diameter, heart rate and mean arterial pressure.
Results
Twelve participants completed the study, with two (16.6%) reporting migraine attacks after NN414 compared to one (8.3%) after placebo (p = 0.53). The area under the curve for headache intensity, VMCA, superficial temporal artery diameter, heart rate and mean arterial pressure did not differ between NN414 and placebo (p > 0.05, all comparisons).
Conclusion
The lack of migraine induction upon activation of the Kir6.2/SUR1 channel subtype suggests it may not contribute to migraine pathogenesis. Our findings point to KATP channel blockers that target the Kir6.1/SUR2B subtype, found in cerebral vasculature, as potential candidates for innovative antimigraine treatments.
Introduction
Migraine affects approximately 15% of the global population and remains a significant health challenge (1). Despite substantial advancements in migraine research, the exact mechanisms underpinning this incapacitating disorder are not fully delineated. Studies focusing on human migraine models have shown that vasoactive peptides, acting through G-protein-coupled receptors (GPCR) on the cell surface, can trigger migraine attacks (2). This pivotal insight has led to the development of disease-specific therapies (3). However, given the partial efficacy of currently available migraine preventive treatments (4), exploring downstream molecules as potential targets represents a promising strategic shift in migraine management.
Adenosine 5′-triphosphate-sensitive K+ (KATP) channels are expressed throughout the trigeminovascular system, including the vascular smooth muscle cells (VSMCs) of cranial vessels, neurons of the trigeminal ganglion (TG), and the trigeminal nucleus caudalis (TNC) (5,6). These channels play instrumental roles in neuronal excitability and in modulating nociceptive processing (7). Experimental human studies demonstrated that administration of KATP channel opener levcromakalim induced headache associated with a prolonged dilation of cranial arteries in healthy participants (8) and triggered migraine attacks in individuals with migraine (9). These findings position the KATP channel as a potential novel target for migraine treatment (10). However, further exploration is needed to pinpoint the precise locations of KATP channel sites and to ascertain the functions of its different subunits.
NN414, a selective opener of KATP channels, is known to preferentially target KATP channels present in neuronal and β-pancreatic cells, but not those in VSMCs (11). Given this selective action, we hypothesized that the activation of KATP channels by NN414 would neither trigger migraine attacks nor cause significant changes in cerebral hemodynamics. To test this hypothesis, we conducted a randomized, double blind, placebo-controlled study with individuals with migraine without aura.
Methods
Participants
Between February 2022 and January 2023, we recruited 13 individuals diagnosed with migraine through the Danish online recruitment platform www.forsoegsperson.dk. After initial contact, each participant was further screened at the Danish Headache Center via telephone interview conducted by a trained physician (LK), followed by a physical examination. Due to unpublished data from a Novo Nordisk study indicating a potential elevation in liver enzymes following extended exposure to NN414 (personal communication), we implemented an additional precautionary step of ensuring normal liver function among participants via blood sample analysis. We collected two blood samples from each participant: one before the start of the study and another within a 48-hour period following the study's end. Both samples underwent same-day analysis at Rigshospitalet’s biochemical laboratory. Prior to enrolment, all participants provided written informed consent. Inclusion criteria for the study were as follows: i) age between 18 and 60 years, ii) weight ranging from 50 to 90 kg, and iii) sufficient contraceptive method for female participants, iv) diagnosis of episodic migraine without aura according to the International Classification of Headache Disorders 3rd edition (ICHD-3) (12), with migraine attacks occurring no more than five days per month. Exclusion criteria included: i) diagnosis of another primary or secondary headache disorder (except tension type headache occurring on less than five days per month) ii) daily intake of medication (except contraceptive pills), iii) pregnancy or lactation, iv) a history or presence of serious somatic or psychiatric disease. The study was approved by the Regional Health Research Ethics Committee of the Capital Region (H-20009266) and the Danish Data Protection Agency. It was conducted in accordance with the Declaration of Helsinki of 1964, as well as its later revisions, and was registered at ClinicalTrials.gov (NCT04744129).
Study design
In a randomized, double-blinded, placebo-controlled, crossover design, participants with episodic migraine without aura were randomly allocated to receive 1000 mg of NN414 (2 × 500 mg oral capsules, AXON MEDCHEM) or placebo (two capsules without active substance) on two separate study days, with at least one week between each administration (Figure 1). The Hospital Pharmacy of the Capital Region of Denmark prepared the study drug and placebo, ensuring identical appearance of the NN414 and placebo capsules. The randomization code was kept sealed in the clinic during the study and was not accessible to the investigators until the study was completed and the data were analyzed.

Panel I: The Study Design. Thirteen participants, all diagnosed with migraine without aura, took part in a randomized, double-blind crossover study. Each participant was allocated to receive NN414 or placebo in two separate sessions, with a minimum washout period of one week between the two study days to eliminate carry-over effects. Twelve participants completed both study days. Panel II: Overview of the Experimental Timeline. The figure presents a detailed timeline of the experimental procedures. This includes an initial in-hospital phase of 150 minutes, followed by a 9.5-hour outpatient phase during which participants maintained a headache diary. Key measurements taken during the experiment include diameter of the Superficial Temporal Artery (STA), the mean velocity of the middle cerebral artery (VMCA), Mean Arterial Blood Pressure (MABP), Heart Rate (HR), and Blood Glucose Level (BGL).
Experiments were conducted in a quiet room with standardized temperature and low lighting. Participants were instructed to arrive non-fasting, at least 48 hours headache-free, and to refrain from consuming coffee, tea, cocoa, alcohol, and tobacco for at least 12 hours prior to study onset. Female participants were tested for pregnancy on both study days. Upon arrival, a venous catheter was inserted in the antecubital vein to collect blood samples. After a 30-minute rest, baseline measurements of mean arterial blood pressure (MABP), heart rate (HR) (ProPac Encore; Welch Allyn Protocol) and blood glucose levels (BGL) were taken. Following this, the study drugs, either NN414 or placebo, were administrated (T0). Vital signs were continuously monitored and recorded at 10-minute intervals. Blood glucose levels were measured again 30 minutes after NN414/placebo administration (T30) and subsequently every 10–15 minutes (Figure 1).
Headache and associated symptoms
A blinded investigator (LK) collected data on headache/migraine characteristics at baseline (T0) and every 10 minutes after the administration of NN414 or placebo (T0) for 150 minutes using a standardized questionnaire. Headache intensity was recorded on a numerical rating scale (NRS) from 0–10 (0; no headache, 1; very mild headache [including a pressing or throbbing feeling], 10; worst imaginable headache). Headache characteristics included pain localization, pain quality, aggravation by physical activity, associated symptoms (nausea, photophobia, and phonophobia) and whether the headache mimicked participants’ usual migraine attack. After discharge from the hospital, participants were instructed to complete a headache diary every hour for up to 12 hours following drug administration (Figure 1). The diary included headache characteristics and accompanying symptoms, any use of rescue medication, adverse events (AEs) and premonitory symptoms such as unusual fatigue, yawning, neck stiffness and mood swings. Participants were allowed to treat a headache with over-the-counter rescue medication or their usual migraine specific treatment at any time.
Migraine-like attack criteria
Migraine attacks provoked in an experimental setting are not spontaneous and cannot fulfill the ICHD-3 criteria for migraine (12). Thus, provoked attacks were defined as either headache that fulfills criteria C and D for migraine without aura according to ICHD-3 or headache mimicking the patient’s usual migraine attacks and treated with rescue medication (13).
STA diameter
The diameter of the frontal branch of the Superficial Temporal Artery (STA) was measured with a high-resolution ultrasonography unit (20 MHz, bandwidth 5 MHz, Dermascan C; Cortex Technology, Hadsund, Denmark) at baseline, immediately before administration of NN414/placebo (T0) and every 20 minutes until T140 during the in-hospital period (14,15). All measurements were performed on the left side. For each time point, four measurements of STA diameter were recorded in the same spot, and the mean of each time point was calculated.
MCA velocity
The mean velocity of the middle cerebral artery (VMCA) was recorded bilaterally using transcranial Doppler (TCD; Doppler Box) with handheld 2-MHz probes. End-tidal CO2 was recorded simultaneously with TCD recordings, using an open mask that caused no respiratory resistance (ProPaq Encore; Welch Allyn Protocol). VMCA was measured at baseline, immediately before administration of NN414 or placebo (T0) and every 20 minutes until T140. For each time point, four measurements of the VMCA were recorded on each side of the head, and the overall mean of every time point was calculated.
Data analysis and statistics
Data are presented as mean ± standard deviation (SD) except for headache intensity scores, which are presented as individual data sets and median scores. Baseline was defined as T0 before the oral administration of NN414 or placebo.
Sample size calculation was based on the difference between two paired groups reporting migraine attacks after NN414 and placebo. We assumed that 60% of the trial participants would develop migraine after NN414, and 10% after placebo (16). Significance level was set at 5%, while the risk of type 2 error was set to 20% with 80% power. We calculated a sample size of 19 participants using the “power_mcnemar_test” function and “MASS” package in R statistical software (version 4.2.1).
To prevent unnecessary participant exposure to study procedures we tested whether the enrolment could be terminated due to futility once 13 participants were enrolled. Ordinary test statistics (z-score) were calculated by an investigator not involved in the experimental process. The z-score value was below the information fraction critical value, indicating that participant enrolment could be terminated without compromising the statistical power of the study. The functions “gsDesign” and “glm” were used in R statistical software (version 4.2.1). All participants who completed both study days were included in the analysis.
The primary endpoint was the difference in incidence of migraine attacks between the two experimental days. Secondary endpoints were differences in the area under the curve (AUC) for the following parameters between NN414 and placebo day: headache intensity score (T0-T12h), HR (T0-150min), MABP (T0-150min), diameter of STA (T0-140min) and VMCA (T0-140min). Binary paired data such as the incidence of migraine attacks, head pain, associated symptoms, and adverse events were analyzed using McNemar’s test. AUC was calculated using the trapezium rule (17), and differences between NN414 and placebo were analyzed using the paired t-test or the Wilcoxon signed ranked test, depending on the data distribution. Statistical analysis and graphs were performed using GraphPad Prism 8.3.0 (San Diego, CA, USA). The significance level was set at 5% (P < 0.05, two-tailed) for all tests and no adjustments were made for multiple comparisons.
Data availability
The corresponding author can provide the data supporting the findings reported in this study upon reasonable request.
Results
Patients
Thirteen participants with migraine were randomly assigned to receive either NN414 or placebo, and 12 of them completed both study days and were included in the analysis (Figure 2). The study included 10 women (83.3%) and two men (16.7%) with a mean age of 31.4 years (range 21–56 years) and a mean body weight of 69.3 kg (range 59–87 kg). Mean VMCA data from two participants were excluded from the analysis due to technical issues.

CONSORT flow chart of the study.
Migraine attacks and headache
Two (16.6%) participants reported migraine attacks after NN414 administration compared to one (8.3%) after placebo (P = 0.53). Five (41.6%) participants reported headache following NN414 administration compared to four (33.3%) after placebo (P = 0.67). The AUC for headache intensity (T0-T12h) did not differ between the NN414 (450.8 ± 848.5) and placebo (189.6 ± 330.4) day (P = 0.68) (Figure 3).

Panel I: Headache intensity. The Numerical Rating Scale (NRS) scores were used to assess headache intensity from baseline until 12 hours following the administration of either NN414 or placebo. Dotted lines represent the individual headache scores of each participant and bold lines the median headache intensity on NN414 (A) day and placebo (B) day. No significant difference was observed in the Area Under the Curve (AUC) for headache intensity between the NN414 day (450.8 ± 848.5) and the placebo day (189.6 ± 330.4) (P = 0.68). Panel II: Superficial Temporal Artery (STA). Mean changes in the diameter of the STA from baseline up to T140 after the administration of NN414 (red) and placebo (blue). The Area Under the Curve (AUC) for STA diameter from 0 to 140 minutes did not exhibit any significant difference between the NN414 administration day and the placebo administration day (176.6 ± 44.7 vs 186.5 ± 45.53, P = 0.09). Panel III: Velocity of the Middle Cerebral Artery (VMCA). Mean changes in the VMCA from baseline to T140 after the administration of NN414 (red) and placebo (blue). The AUC for VMCA from 0 to 140 minutes was not significantly different on the NN414 administration day compared to the placebo administration day (10058 ± 1702 vs 10117 ± 1360, P = 0.78).
Following NN414 administration, the median time to headache onset was 130 minutes (range 90–480 min) compared to 275 minutes (range 40–5480 min) after placebo. The median maximum headache intensity was 3 (range 1–10) after NN414 compared to 1.5 (range 1–3) after placebo. The median headache duration was 360 minutes (range 20–570 min) following NN414 compared to 365 minutes (range 180–590 min) after placebo.
There was no difference in the incidence of nausea (three [25%] vs two [16.6%], P > 0.99), photophobia (two [16.6%] vs two [16.6%], P > 0.99), or phonophobia (two [16%] vs 0, P = 0.47), between NN414 and placebo (Table 1).
Clinical characteristics of headache and associated symptoms in migraine patients after NN414 or placebo (0–12 hours observation period).
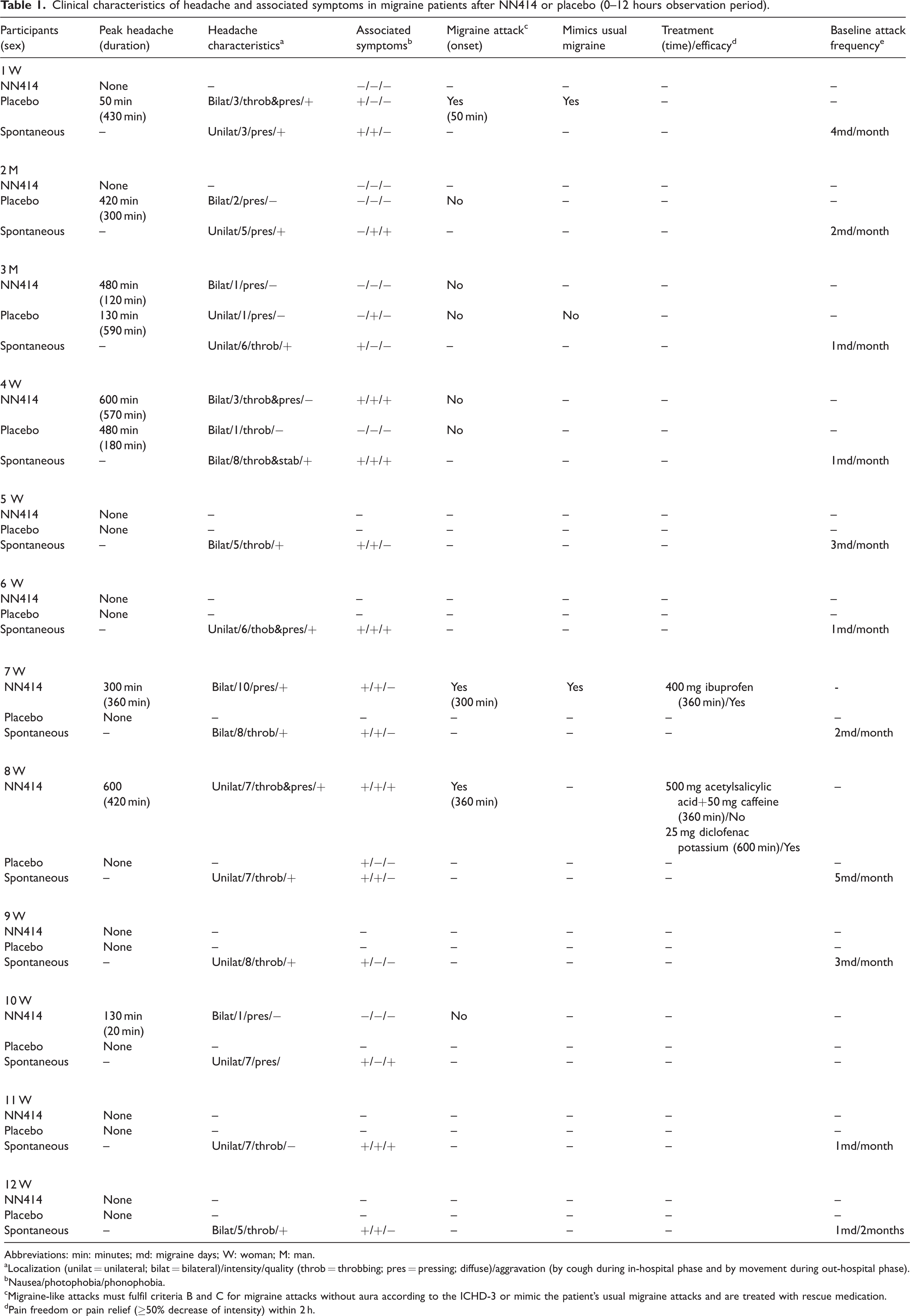
Abbreviations: min: minutes; md: migraine days; W: woman; M: man.
Localization (unilat = unilateral; bilat = bilateral)/intensity/quality (throb = throbbing; pres = pressing; diffuse)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Nausea/photophobia/phonophobia.
Migraine-like attacks must fulfil criteria B and C for migraine attacks without aura according to the ICHD-3 or mimic the patient’s usual migraine attacks and are treated with rescue medication.
Pain freedom or pain relief (≥50% decrease of intensity) within 2 h.
Cerebral and systemic hemodynamics
We found no difference in the AUC (T0-T140) for STA diameter (176.6 ± 44.7 vs 186.5 ± 45.53, P = 0.09) (Figure 3) and mean VMCA (10058 ± 1702 vs 10117 ± 1360, P = 0.78) (Figure 3) between NN414- and placebo. In addition, there was no difference in the AUC (T0-T150) for MABP (11499 ± 1211 vs 11355 ± 971.3, P = 0.36) and HR (9238 ± 1208 vs 9212 ± 770.4, P = 0.89) between the NN414 and the placebo days (Figure 3).
Blood glucose levels
No significant difference was found in the AUC for blood glucose levels between the NN414 and placebo day during the in-hospital phase (T0-T140) (591.8 ± 68.41 vs 580.1 ± 48.81, P = 0.44) (Figure 4). However, a post hoc analysis revealed a significant increase in blood glucose levels from T40 to T120, after NN414 administration compared to placebo (335 ± 41.37 vs 306.9 ± 39.08, P = 0.03).
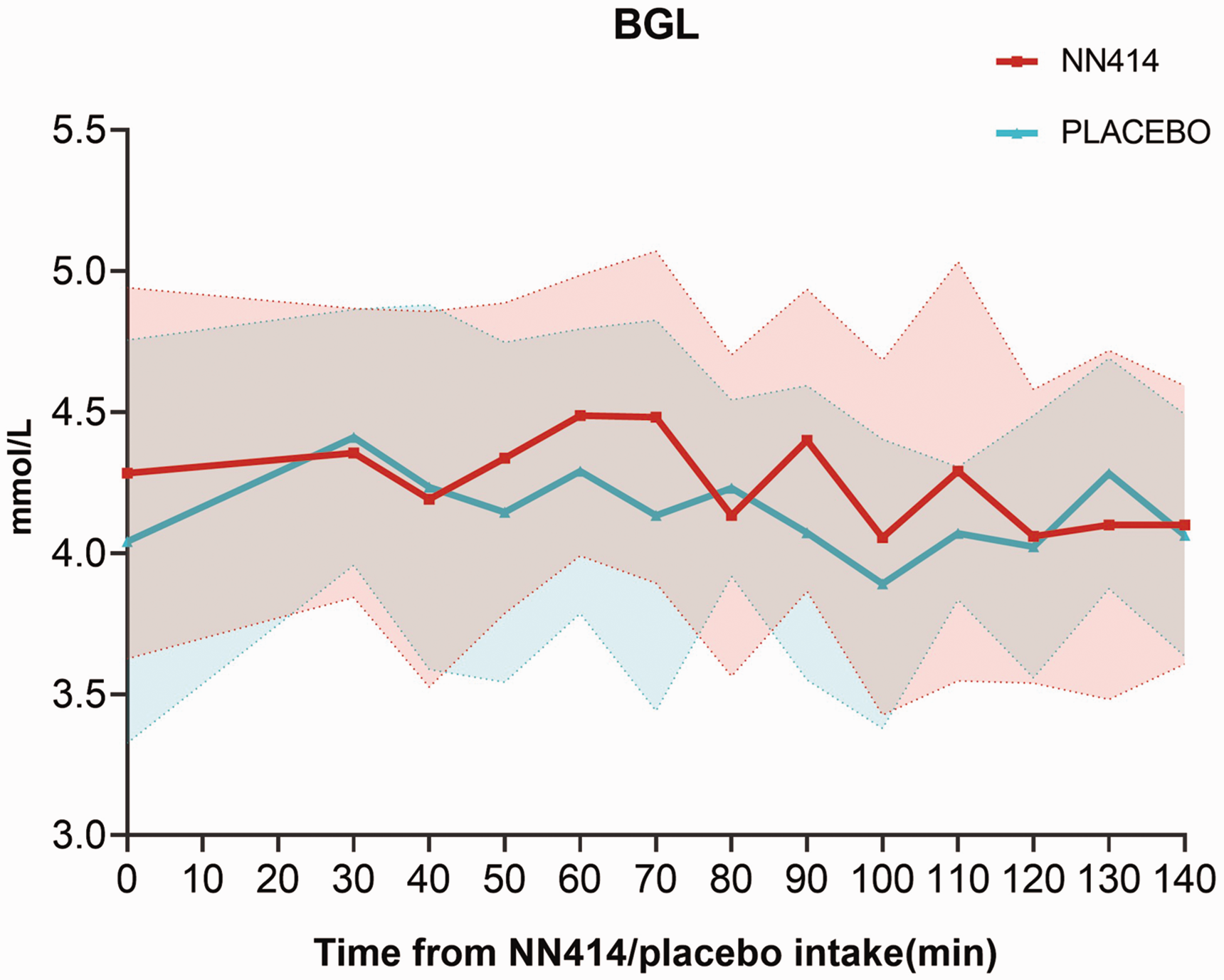
Blood glucose levels (BGL). Mean change in BGL after the administration of NN414 (red) or placebo (blue), from baseline to T140 min. There was no difference in the AUC (0–140 min) for BGL on the NN414 day compared to the placebo day (591.8 ± 68.41 vs 580.1 ± 48.81, P = 0.44).
Adverse events
Three participants reported adverse events on the NN414 day and three on the placebo day. All adverse events were mild and resolved within 12 hours. There was no serious adverse event. All adverse events are presented in Table 2. Two patients required rescue medication following NN414 administration, while none required it after placebo (Table 1).
Number of patients with reported adverse events (0–12 hours observation period).

Data are number (%).
Discussion
The main finding of our study was that the KATP channel opener NN414 did not cause headache or migraine attacks in individuals with migraine without aura. Moreover, NN414 did not alter the cerebral or systemic hemodynamics.
KATP channels are composed of four pore-forming subunits from the inward rectifier potassium (Kir) channel family (two isoforms: Kir6.1 and Kir6.2) and four regulatory sulfonylurea receptor (SUR) subunits (three isoforms: SUR1, SUR2A and SUR2B) (18,19). The various Kir6.x and SUR subunits’ compositions yield distinct channels featuring different morphologies, functionalities, and unique tissue expression (20). The activation of these channels results in potassium efflux from the cell and membrane hyperpolarization, which triggers a tissue-specific cellular response (21,22). NN414, a diazoxide analogue, is a selective opener of the Kir6.2/SUR1 subtype KATP channel (11,23), which is primarily expressed in neurons and β-pancreatic cells (24,25). The administration of NN414 activates these channels and reduces insulin production in β-pancreatic cells, subsequently increasing blood glucose levels (11). Previous randomized, placebo-controlled trials have evaluated the effects of oral NN414 in glucose regulation and metabolism (26,27). One study indicated that a single dose of NN414 is rapidly absorbed after oral intake, reaching peak plasma concentration between 0.5 h to 2 h post-dose in healthy male participants (26). In our study, a single dose of 1000 mg of NN414 led to a significant increase in the AUC for blood glucose levels from T40 to T120 post-administration compared to placebo.
Kir6.2/SUR1 channels are expressed in the TG, the TNC, and cortical and subcortical neurons (24,25,28,29). The activation of KATP channels by NN414 has been shown to promote burst firing in substantia nigra dopamine neurons, both in vitro and in vivo, suggesting that the Kir6.2/SUR1 subunit is involved in neuronal firing (30,31). Our study found that the administration of NN414 did not trigger migraine attacks in individuals with migraine, which suggests that the Kir6.2/SUR1 subtype is unlikely to be involved in migraine pathophysiology. Rodent studies supporting this hypothesis have shown that the intraplanar injection of KATP channel opener diazoxide could mitigate nociceptive behavior induced by bradykinin, thermal and mechanical stimuli (32). Interestingly, NN414 enhanced the analgesic effects of morphine, indicating a potential anti-nociceptive role of the Kir6.2/SUR1 channels subtype (33). Whether NN414 can cross the blood–brain barrier (BBB) is uncertain. Given its lipophilic nature and small molecular weight (291.78 Da) a direct action of NN414 on neuronal KATP channels is possible.
Previous human studies have demonstrated that administration of KATP channel agonist levcromakalim induced headache and dilation of extracerebral and cerebral arteries in healthy individuals (9,34). In addition, it has been found to trigger migraine attacks in people with migraine, both with and without aura (8,35). Moreover, the action of several other migraine triggering molecules is dependent on KATP channel activation (36,37). These findings emphasize the pivotal role of KATP channels in migraine pathophysiology and highlight the potential for therapeutic intervention targeting these channels. Levcromakalim is a KATP channel opener with high affinity for the Kir6.1/SUR2B channel subtype, which is primarily expressed in the VSMCs of cranial arteries (6,38). The opening of vascular KATP channels leads to potassium efflux, hyperpolarization, and vasodilation (39). Given that all headache and migraine provoking substances dilate extracerebral and cerebral arteries (2), vasodilation in response to KATP channel activation may play a crucial role in migraine pathophysiology.
Interestingly, recent research demonstrated that levcromakalim-induced cephalic hypersensitivity in mice only occurred via systemic and not intracerebroventricular administration (7). Furthermore, compared to placebo, intradermal and intramuscular injections of levcromakalim in trigeminal and extra-trigeminal areas did not evoke more pain in healthy participants (40). These observations indicate that the direct activation of the KATP channels in peripheral and central neurons may not play a significant role in initiating headache and migraine pain. Instead, it appears that the vascular Kir6.1/SUR2B subtype could be responsible for the migraine triggering effects of levcromakalim. This is further supported by a study which showed that levcromakalim induced cephalic hypersensitivity in mice, while mice lacking the Kir6.1 subunit in VSMCs did not develop tactile hypersensitivity upon levcromakalim administration (7). Of note, KATP channel blocker glibenclamide, which has a higher affinity for the Kir6.2/SUR1 rather than the Kir6.1/SUR2B subunit, blocked levcromakalim-induced trigeminal pain and vasodilation in rodent models without altering the basal vascular tone (36,37,41). However, both pre- and post-treatment with glibenclamide failed to attenuate levcromakalim-induced headache and vasodilation in healthy participants (42–44). This highlights the importance of the Kir6.1/SUR2B channel subtype in migraine.
Conclusions
The present data support the notion that the Kir6.1/SUR2B subtype, rather than the Kir6.2/SUR1, is a key channel in the signaling pathways that trigger migraine attacks. Hence, the development of selective Kir6.1/SUR2B antagonists might represent a promising new strategy for treating migraine and advancing our understanding of the disease.
Article highlights
Activation of neuronal KATP channels does not induce migraine attacks. Neuronal KATP channels are unlikely to contribute significantly to the signaling pathways responsible for initiating migraine attacks. We propose that the KATP channels expressed in the cerebral vasculature could offer potential avenues for innovative antimigraine treatments.
Footnotes
Acknowledgements
The authors thank all participants who took part in the study, as well as Dr. Janu Thuraiaiyah for her valuable assistance during the study's conduction.
Author contributions
LK, MMK, FMA and MA initiated the study and contributed to study design; protocol development; participant enrolment; data acquisition, data processing, analysis, statistics, and interpretation; and drafting and revision of the article. ZAZ and SA contributed to participant enrolment, data acquisition, statistical analyses, and critical review of the article.
Declaration of conflicting interests
LK, MMK, SA and ZAZ report no conflicts of interests. FMA has received honoraria or personal fees from AbbVie, Eli Lilly, Lundbeck, Novartis, Pfizer, Teva for lecturing and/or participation in advisory boards; serves as president of Danish Headache Society and board member of the European Headache Federation; serves as junior associate editor for Acta Neurol Scand, Front Neurol, Front Res Pain, and Headache Medicine; serves as junior associate editor for Cephalalgia and Cephalalgia Reports; serves as member for the editorial board of Journal Headache Pain. MA reports receiving personal fees from AbbVie, Amgen, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals during the conduct of the study. MA also reports serving as associate editor of The Journal of Headache and Pain, and associate editor of Brain.
Funding
The research leading to these results has received funding from Lundbeck Foundation (R155-2014-171). MA was also supported by the Lundbeck Foundation Professor Grant (R310-2018-3711) and MMK by the Research Fund of Rigshospitalet (E-23327-04).
