Abstract
Background
Migraine and vestibular migraine are disorders associated with a heightened motion sensitivity that provoke symptoms of motion-induced nausea and motion sickness. VM affects ∼3% of adults in the USA and affects three-fold more women than men. Triptans (selective serotonin receptor agonists) relieve migraine pain but lack efficacy for vertigo. Murine models of photophobia and allodynia have used injections of calcitonin gene-related peptide (CGRP) or other migraine triggers, such as sodium nitroprusside (SNP), to induce migraine sensitivities in mice to touch and light. Yet, there is limited research on whether these triggers affect motion-induced nausea in mice, and whether migraine blockers can reduce these migraine symptoms. We hypothesized that systemic delivery of CGRP or SNP will increase motion sickness susceptibility and motion-induced nausea in mouse models, and that migraine blockers can block these changes induced by systemically delivered CGRP or SNP.
Methods
We investigated two measures of motion sickness assessment [motion sickness index (MSI) scoring and motion-induced thermoregulation] after intraperitoneal injections of either CGRP or SNP in C57BL/6J mice. The drugs olcegepant, sumatriptan and rizatriptan were used to assess the efficacy of migraine blockers.
Results
MSI measures were confounded by CGRP’s effect on gastric distress. However, analysis of tail vasodilatations as a surrogate for motion-induced nausea was robust for both migraine triggers. Only olcegepant treatment rescued tail vasodilatations.
Conclusions
These preclinical findings support the use of small molecule CGRP receptor antagonists for the treatment of motion-induced nausea of migraine, and show that triptan therapeutics are ineffective against motion-induced nausea of migraine.
Keywords
Introduction
Motion sickness and motion-induced nausea, as well as motion sensitivity, are symptoms of migraine and especially vestibular migraine (VM) (1–7). In addition, preponderance for motion sickness in childhood correlates with migraine occurrence in adulthood. Common vertiginous symptoms of VM include vertigo lasting seconds to days, dizziness, imbalance, a disorienting sense of space and anxiety to motion (4). Treatment of migraine typically involves achieving two primary endpoints: pain freedom at two hours and the absence of the most bothersome symptom (MBS), which is typically nausea, vomiting, photophobia or phonophobia. Although the most frequent MBS is photophobia and affects 50% of migraine patients, the second most frequent MBS is nausea and affects 30% of these patients (8).
VM patients show lower motion perception thresholds compared to healthy controls and also show enhanced susceptibility to motion sickness (3). Interestingly, Wang and Lewis (9) found that, in VM patients, but not in migraine or healthy controls, the residual sensory conflict between gravitational (otolith) and rotational (semicircular canal) cues correlated with motion sickness susceptibility.
The diagnostic criteria for human motion sickness have recently been updated (10) and are included in the international classification of vestibular disorders. The major symptoms of motion sickness include facial pallor, nausea, vomiting, gastric awareness and discomfort, sweating, and hypothermia. Motion sickness-related hypothermia is broadly expressed phylogenetically in humans, mice, rats and musk shrews (11,12). However, rodent models of motion sickness have been constrained because rodents do not have an emesis reflex. Pica-eating behavior is considered to be an alternative to vomiting, yet, pica has not been shown to be a sensitive measure of motion sickness (13). Instead, piloerection, tremor, fecal and urinal incontinence have been used in scoring criteria called the motion sickness index (MSI) to quantify the degree of motion sickness-like behavior experienced by rats and mice to emetic stimuli (13,14). In addition, thermoregulatory changes can also be used to assess motion sickness, which, in the mouse model, involves a decrease in head temperature (hypothermia) and transient tail-skin vasodilation early in the onset of provocative motion (12). Tail vasodilatations to provocative motion have been reported to precede emesis events in the musk shrew, serving as an early index for motion-induced nausea in experimental rodent models.
Despite the high prevalence of motion-induced nausea in VM, the underlying mechanisms have yet to be defined. It is accepted that migraine involves the neuropeptide calcitonin gene-related peptide (CGRP). CGRP is up regulated during migraine attacks (15,16), infusion of CGRP can induce migraine (16) and antibodies that block CGRP or its receptor can effectively treat most migraines (17,18). Yet, it is unclear whether CGRP signaling antagonism can also alleviate motion-induced nausea of VM. An anti-CGRP monoclonal antibody is being assessed as a clinical trial (galcanezumab; NTC#04417361) for vestibular migraine treatment. Yet, there is strong evidence in favor of anti-CGRP signaling therapy for treating VM because a prospective observational cohort study observed significant vertigo reduction and headache frequency in 50 VM patients treated with various anti-CGRP monoclonal antibodies (19).
Animal models of migraine have used injections of CGRP or other migraine triggers such as sodium nitroprusside (SNP), which generates nitric oxide and stimulates the release of CGRP, to induce allodynia responses to touch (20,21) and light-aversive behavior (22,23). However, it is not known whether these triggers can induce motion-induced nausea in preclinical models. It is also not known whether migraine blockers used in preclinical models of touch and light sensitivities can also block motion-induced nausea and motion sensitivity characterized in VM. The selective serotonin receptor (5-HT1B/D) agonists, typically known as triptans, are known to decrease plasma CGRP and inhibit. Sumatriptan administration reduced CGRP levels in the external jugular vein and provided pain relief in migraine patients (24). To better understand CGRP’s role in VM motion-sickness susceptibility, we investigated two measures of motion sickness assessment (MSI scoring and motion-induced thermoregulation) after systemic injections of either CGRP or SNP in the wild-type C57BL/6J mouse. In the present study, we found that MSI measures based on gastric distress were lessened by CGRP antagonism. Yet, this was driven by CGRP’s effect in causing diarrhea, which was not observed after SNP administration. However, motion-induced thermoregulation was a robust model of motion sickness. CGRP receptor antagonism by olcegepant was effective in relieving CGRP effects on motion-induced nausea, whereas triptan therapies were not efficacious. Our studies provide a strong premise that antagonizing CGRP signaling will be effective for treating motion-induced nausea of VM because it has been shown to be highly effective for typical migraine.
Methods
Animals
C57BL/6J mice were obtained from Jackson Laboratories (JAX #664) and were housed under a 12 hour day/night cycle under the care of the University Committee on Animal Resources (UCAR) at the University of Rochester. Mice are housed with ad libitum access to food, water, bedding and enrichment materials. In total, 213 mice (106 male/107 female) were tested, and all studies were powered sufficiently to detect male/female differences. Prior to behavioral testing, mice were equilibrated in the testing room controlled for an ambient temperature between 22 and 23°C for at least 30 minutes. Mice were tested around 2.3 to 6 months of age. This age range correlates to 20–30 years in humans and is within the range that migraine symptoms most likely occur in patients (25). Different cohorts of mice were used to test motion sickness indices and motion-induced nausea. Although all mice were assessed for intraperitoneal (IP) vehicle and IP CGRP effects in MSI and motion-induced thermoregulation experiments, mice were randomized and staggered when assessing IP CGRP co-delivered with either IP olcegepant, sumatriptan or rizatriptan. Analyses of thermoregulation recordings were performed blinded as animal IDs numbers were de-identified from treatment groups.
Testing occurred from 9:00 am to 4:30 pm during the day cycle to control for behavioral and thermoregulatory changes that may arise from circadian rhythms. For motion-induced nausea testing, mice were screened for instances of patchy fur (alopecia) and were not included in this study (26).
Drug administration
Injections were performed IP with a fine 33-gauge insulin syringe. Dulbecco phosphate-buffered saline served as the diluent and as the vehicle control. Migraine blockers used were the CGRP-receptor antagonist olcegepant and the selective serotonin receptor agonists sumatriptan and rizatriptan. The concentrations are: 0.1×, 0.5× and 1.0× CGRP were prepared at 0.01, 0.05 and 0.1 mg/kg (rat ɑ-CGRP; Sigma-Aldrich, St Louis, MO, USA), 1.0× olcegepant was prepared at 1.0 mg/kg (BIBN4096; Tocris, Bristol, UK), 1.0× sumatriptan was prepared at 0.6 mg/kg (Sigma-Aldrich), 1.0× rizatriptan was prepared at 0.6 mg/kg (Sigma-Aldrich), and 0.1×, 0.5× and 1.0× SNP (Sigma-Aldrich) were prepared at 0.25, 1.25 and 2.5 mg/kg. After injection, animals were placed in a separate cage from their home cage to recuperate from injection stress. Mice were tested approximately 20–30 minutes after delivery of either vehicle, CGRP, SNP, CGRP or SNP co-administered with a blocker, or drug controls. Animals were gently handled and anesthesia was not needed. All animal procedures were approved by the University of Rochester’s (IACUC) and performed in accordance with the standards set by the National Institutes of Health.
Off-vertical axis rotation (OVAR)
Prior human and rodent studies have used OVAR to assess the otolith-ocular reflex and assess the semicircular canal-otolith interaction (27–29). Constant velocity OVAR at a tilt can be disorienting and promote motion sickness in human participants (30,31) and further studies in mice have shown that provocative rotation leads to pica and observations of urination, piloerection and tremor (32). In the present study, a two-cage rotator (cage dimensions: 12.7 × 8.9 cm) was use for 20 minutes (60 rpm, 45° tilt from the vertical, 20 cm from the axis of rotation) as an OVAR vestibular challenge.
MSI testing
We adapted the protocol of Yu et al. (13) that previously assessed the drugs scopolamine and modafinil on mitigating motion sickness indicators in rats and mice. We incorporated OVAR (described above) as the vestibular challenge (VC) in this test. We evaluated MSI in mice at the end of the following time points: (i) a five-minute baseline (pre-injection/pre-VC); (ii) 20–30 minutes after drug injection (post-injection/pre-VC); and (iii) five minutes after the OVAR (post-injection/post-VC). Mice were placed in a testing box separate from their home cage to observe for indicators of motion sickness detailed in Figure 1a. A MSI score was determined by the summation of the indicators.

Calcitonin gene-related peptide (CGRP) and sodium nitroprusside (SNP)’s effects on MSI: female (pink) and male (blue) C57BL/6J mice were tested for motion sickness indicators. Treatments were delivered intraperitoneally (IP): vehicle control (open circle/open square), CGRP (closed circle), SNP (closed square), olcegepant + CGRP (top half-filled circle) and sumatriptan + CGRP (bottom half-filled circle). (a) Assay timeline is illustrated and (b) and a table of criteria and MSI scoring. Motion sickness index (MSI) was computed by a summation of criterion at the appropriate scoring and involved measuring feces, piloerection, urination, and tremor. MSI was recorded at three different time points. The vestibular challenge involved off-vertical axis rotation (60 rpm, 45° tilt from the vertical) for 30 minutes. (c, d) The first bar graph is MSI calculated pre-injection, comparing the second part of bar graph which is post-vestibular challenge (c) to CGRP and CGRP+blockers, and (d) to SNP injection. (e,f). Feces weight (g) comparing post-vestibular challenge to pre-condition to CGRP and CGRP + blockers (e) and SNP injection (f). Two-way analysis of variance was analyzed in males and in females. F-statistics and p-values can be found for all MSI conditions in supplementary Table 1. Significance is listed as * = p < 0.05, ** = p < 0.01, *** = p < 0.001, and **** = p < 0.0001.
Certain actions were taken to normalize the weight distribution of MS indicators during reporting. Fecal granules (Fg) were counted separately at each time point. The unit weight per Fg after vehicle injection was used to back-calculate the granule number in cases where the weight of fecal incontinence (g) was instead measured; for example, in cases of gastric distress. This calculation was necessary during CGRP testing because mice experienced diarrhea, which made it difficult to count fecal.
Urination was assigned a 1.2 score and was only counted once throughout the time points. Piloerection was measured as either mild (0.6) or severe (1.2), and tremors were assigned a 1.2.
Motion-induced thermoregulation testing
We adapted the protocol of Tu et al. (12) who first noticed these thermoregulatory changes when measuring the temperatures of the heads, bodies, and tails of mice to provocative motion. In this study, head and tail temperatures of C57BL/6J mice were measured for a total 45 minutes using a FLIR E60 IR camera (model: E64501; Teledyne FLIR, Wilsonville, OR, USA). This camera is connected to a tripod and is positioned approximately 43 cm above an open, plexiglass box (mouse box) used to house an individual mouse during testing. Both the tripod and mouse box are securely attached to the shaker’s base. Briefly, baseline measurements were recorded for five minutes prior to the provocative motion (–5 to 0 minutes). The provocative motion was an orbital rotation (75 rpm, 2-cm orbital displacement), and mice were recorded for 20 minutes (0–20 minutes). After 20 minutes, the provocative motion was turned off, and mice were recorded for an additional 20 minutes to measure recovery to baseline (20–40 minutes) as shown in Figure 2. Head and tail temperatures were measured after data retrieval using the software FLIR Tools+. Tail and head temperatures were measured within predefined field of views: square region (3 × 3 mm) for tail, and circular region (10 × 10 mm) for head. Tail measurements occurred 2 cm from the base of the tail and head measurements occurred at the center of the head image, in between the mouse’s ears. Infrared imaging data was collected every minute during baseline measurements, and every 2 minutes during and after the provocative motion. We quantified thermoregulatory changes to provocative motion by comparing changes in tail vasodilatations, and we approximated the magnitude of the head hypothermia based on second-order curve fit estimates. Transient increases in the tail temperature of the mouse to provocative motion are referred to as Δ tail vasodilatations (oC) and were computed by subtracting the tail temperature at time t = 0 minutes (rotation ON) from the max tail temperature measured during the first 10 minutes of the rotation (0 ≤ t ≤ 10). To facilitate quantification and comparisons of the delta tail vasodilatations between treatment groups, a threshold of 1.5°C was imposed upon the data to make it a binary outcome measure. Tail temperature changes equal to or greater than +1.5°C were designated a delta tail vasodilation and those less than +1.5°C did not meet the criteria.
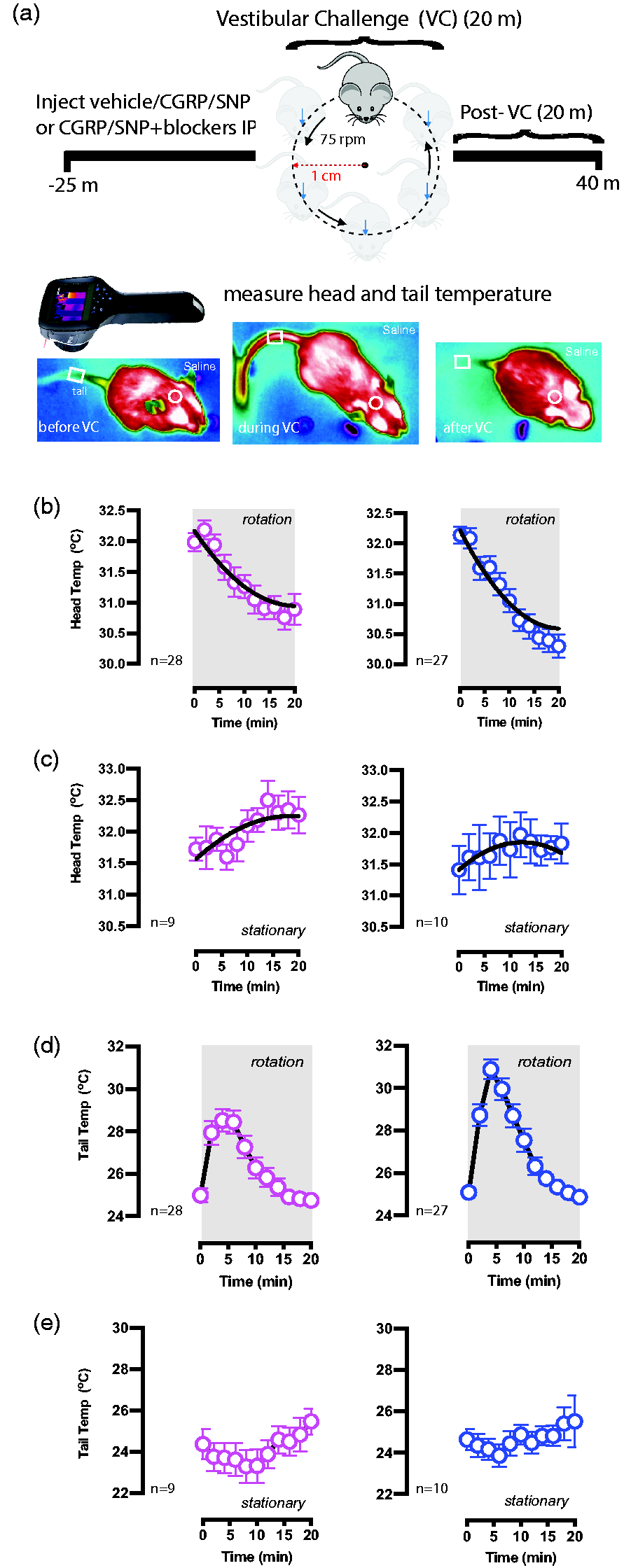
Stationary and provocative motion testing after intraperitoneal (IP) vehicle control. (a) Using a FLIR E60 infrared camera, head and tail temperatures for all conditions are recorded before, during and after a provocative twenty-minute orbital rotation acting as the vestibular challenge. Infrared recordings are 45 minutes in duration. (b–e) Sample sizes are depicted in the bottom left for female (pink) and male (blue) Continued.C57BL/6J mice. After administering vehicle (saline), (b, c) head and (d, e) tail temperatures were compared during (c, e) stationary testing or (b, d) during the provocative rotation. Head temperatures for stationery and VC testing were fit to the second-order curve (B2X2 + B1X + B0) and delta head temperatures were computed by subtracting temperature at time t = 20 minutes from t = 0 minutes. Head curves compute similar hypothermia between sexes but different from stationary tests. During the first 10 minutes of the provocative rotation, delta tail vasodilatations were computed to be 4.34 ± 0.34°C for females and 5.44 ± 0.42°C for males. Mice during stationary testing did not experience Δ tail vasodilatations, and had delta tail changes of −1.1 ± 0.8°C for females and 0.2 ± 0.6°C for males.
Prior to assessing the efficacy of migraine blockers on thermoregulatory changes as a result of provocative motion, control experiments were performed to assess (i) test–retest reliability of motion-induced thermoregulation and (ii) stationary testing (no rotation) after vehicle, 1.0× CGRP and 1.0× SNP injections. Additionally, we tested motion-induced thermoregulation at 0.1× and 0.5× concentrations of CGRP and SNP to compare with 1.0× effects.
Power and statistical analysis
All studies used both male and female mice with numbers determined using a priori G*Power software for power analyses (https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower). The number of each sex needed for MSI was calculated to be four animals based on power analysis using an alpha of 0.05, with 95% power and 5% significance with effect size d = 2.9; based on findings from n = 260 mice with motion sickness testing (13). The number of each sex needed for motion-Induced thermoregulation was calculated to be three animals (core temperature) or four animals (tail temperature) based on power analysis using an alpha of 0.05, with 95% power and effect size d = 17.33 (based on findings from n = 6 mice with nausea-like thermoregulation testing for core body temperature). We also tested for tail temperature increase after provocative motion effect size d = 4.0 (12). However, all studies were overpowered, allowing for clear sex differences to be detected.
Statistical analyses were conducted in Prism, version 9.0 (GraphPad Software Inc., San Diego, CA, USA). Multivariate analysis of variance (ANOVA) values were analyzed for MSI and motion-induced thermoregulation testing and are described in detail in the Results. Second-order curve fitting were used to generalize trends in head temperature profiles and approximate the magnitude of head hypothermia, a single value with no standard error, computed by subtracting the curve’s predicted head temperature at t = 20 minutes from t = 0 minutes. For test–retest reliability of head and tail profiles, Pearson’s correlation coefficient is listed as r (df), where r is the coefficient and df is the degrees of freedom. Head temperature curves are computed by second-order order curve fitting and an R2 value is provided. Bonferroni multiple comparisons test was the primary post hoc analytical tool, except in cases where Dunnett’s multiple comparisons test was used to compare specific treatments to vehicle control responses. Values are reported as the mean ± SEM unless noted otherwise. p < 0.05 was considered statistically signiifcant for all analyses.
Results
CGRP and SNP’s effects on motion sickness index
We investigated the effects of CGRP administration (n = 9 male/10 female) on motion sickness indicators (MSI) and whether the CGRP receptor antagonist olcegepant or the 5-HT1D receptor agonist sumatriptan could mitigate MSI according to the timeline shown in Figure 1a and MSI indices as shown in Figure 1b. In females, two-way ANOVA analyzed MSI outcomes between treatments (CGRP vs. CGRP co-delivered with blockers) and between pre-injection versus post-VC time points (Figure 1c). Significant effects were observed as a result of treatment (F2.150,19.35 = 9.938; p < 0.001) and post-VC (F1.000,9.000 = 308.2; p < 0.001). A Bonferroni post-hoc test showed that this increase was present for every treatment (p < 0.001 for all comparisons). Post-VC MSI was higher after 1.0× CGRP (p < 0.002) and after 1.0× CGRP + sumatriptan (p = 0.1, trending) than vehicle, but significant differences were not observed between 1.0× vehicle and 1.0× CGRP + 1.0× olcegepant. This analysis was also conducted in males and the factors of treatment (F3,30 = 19.09; p < 0.001) and post-VC effects (F1,30 =634.8; p < 0.001) were also strong. A Bonferroni post-hoc test showed post-VC MSI was higher than pre-injection MSI for every treatment in males (p < 0.001). Similar to females, post-VC MSI after 1.0× CGRP and 1.0× CGRP + 1.0× sumatriptan in males was significantly higher than after vehicle (p < 0.001 each), although this difference to vehicle was not seen with 1.0× CGRP + 1.0× olcegepant. In Figure 1d, MSI outcomes after IP injection were compared to post-VC MSI and two-way ANOVA in females and males showed similar trends as was established in the analysis portrayed in Figure 1c.
MSI outcome is driven by feces criteria
A closer observation of MSI outcomes showed that the main driver of MSI is the magnitude of fecal incontinence (Figure 1e,f) and so two-way ANOVA was used to assess differences in fecal incontinence after treatment and across time points in males and females. Consistent in both sexes, Bonferroni post-hoc analyses comparing post-VC fecal incontinence outcomes indicated high fecal excretion after 1.0× CGRP (p < 0.001) and after 1.0× CGRP + 1.0× sumatriptan (p < 0.001) compared to vehicle tests, but not in mice treated with 1.0× CGRP + 1.0× olcegepant (p = 0.99). Diarrhea is a frequent observation after 1.0× CGRP and 1.0× CGRP + 1.0× sumatriptan but not after 1.0× CGRP + 1.0× olcegepant. Mice treated with SNP did not exhibit diarrhea or increased fecal granules across all time points or compared to vehicle (Figure 1f). In addition, piloerection, urination and tremor did not significantly differ between CGRP or SNP-treated groups (data not shown). A detailed breakdown of F-values and p-statistics is found in supplementary Table 1. These results suggest that computing an index for motion sickness based on fecal incontinence, urination, piloerection and tremor is problematic as a result of how biased the index is towards fecal excretion. Thus, we used a different method to assess motion sickness based on motion-induced thermoregulation.
Hypothermia and tail vasodilatations occur during provocative motion and not stationary testing
Figure 2a shows the experiment for assessing motion-induced thermoregulation. After a five minute baseline recording, mice (n = 28 males/27 females) are challenged with provocative motion (t = 0 minutes, rotation = ON) and exhibited gradual hypothermia until the end of the motion (t = 20 minutes, rotation = OFF). Second-order curve fits calculated similar magnitudes of the mouse’s head hypothermia to between males and females (Figure 2b). In addition to the hypothermia, mice exhibited transient but significant increases in tail temperature during the first 10 minutes of the provocative motion. The Δ tail vasodilatations were quantified for females (4.34 ± 0.34°C) and males (5.44 ± 0.42°C) (Figure 2d). To ensure that the hypothermia and the tail vasodilatations were physiological responses to the provocative motion stimulus, a separate group of mice (n = 9 females/10 males) was tested similarly but for stationary testing. No observed hypothermia or transient vasodilatations were seen in mice of either sex during the stationary tests (Figure 2c,e).
CGRP and SNP’s effects during stationary testing
To exclude the possibility of thermoregulatory changes that may be a result of the treatments (CGRP and SNP) and not the rotation, a secondary stationary test was performed on mice (n = 9 males/9 females) who were tested with 1.0× CGRP (Figure 3a,d) and 1.0× SNP (Figure 3b,e). No differences were seen in the head or tail temperatures after 1.0× CGRP or 1.0× SNP testing compared to vehicle, and hypothermia or delta tail vasodilatations were not observed.
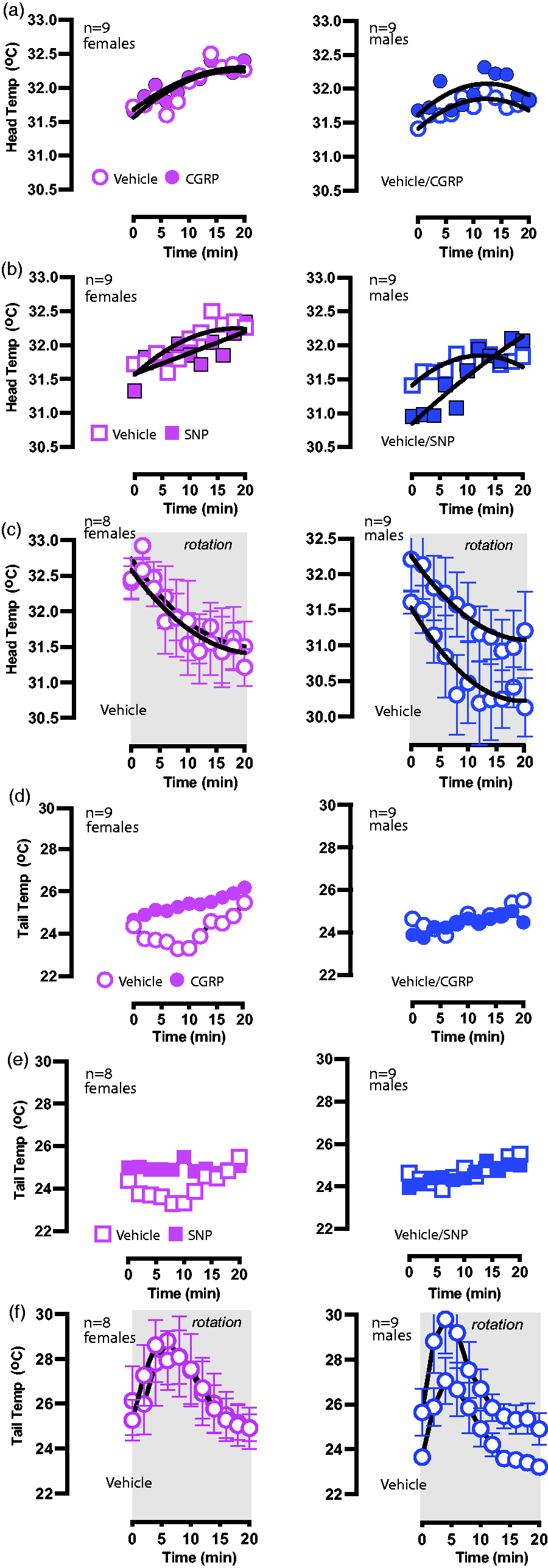
Stationary testing after intraperitoneal (IP) calcitonin gene-related peptide (CGRP)/sodium nitroprusside (SNP) andContinued.test-retest of IP vehicle control. Sample sizes are listed in the upper left corner. (a, b) Head temperature curves were recorded during stationary testing after IP delivery of vehicle, 1.0× CGRP and 1.0× SNP. (c, f) A different cohort of mice was used to assess the test-retest reliability of head and tail temperatures during provocative rotation. (c) When assessing test–retest reliability, no significant differences were seen in the approximate magnitude of hypothermia in females (test vs. retest: –1.2°C vs. –1.2°C) or males (test v.s retest: –1.3 vs. 1.2°C). (d, e) Tail temperatures were compared between stationary tests for vehicle, 1.0× CGRP and 1.0× SNP as similarly performed with head temperatures, and no significant differences were seen. (f) Similar to head temperatures, test–retest of tail measurements during provocative rotation showed no significant differences in delta tail vasodilatations in either female (test vs. retest: 4.00 ± 0.80°C v.s 4.20 ± 0.80°C) or male (test vs. retest: 4.90 ± 0.80°C vs. 4.90 ± 0.90°C).
Test–retest reliability during provocative motion
When assessing IP vehicle delivery, male and female mice exhibited hypothermia during the provocative rotation and this response was repeatedly observed in the retest (n = 9 males/8 females). In Figure 3c, second-order curve fits were generated for test-retest head curves and strong fits were seen for female (Pearson’s r23 = 0.87, p < 0.0001) and for male (Pearson’s r23 = 0.89, p < 0.0001). The magnitude of hypothermia measured in the head temperatures was similar between sexes and reproducible during the test-retest. In Figure 3f, similar tail temperature profiles were observed during test-retest for females (Pearson’s r23 = 0.92, p < 0.0001) and for male (Pearson’s r23 = 0.95, p < 0.001). Delta tail vasodilatations were observed to have similar magnitudes for females (mean vasodilationtest: 4.0 ± 0.8°C, mean vasodilationretest: 4.2 ± 0.8°C) and for males (mean vasodilationtest: 4.9 ± 0.8°C, mean vasodilationretest: 4.9 ± 0.9°C).
CGRP and SNP affect motion-induced thermoregulation in C57/B6J mice
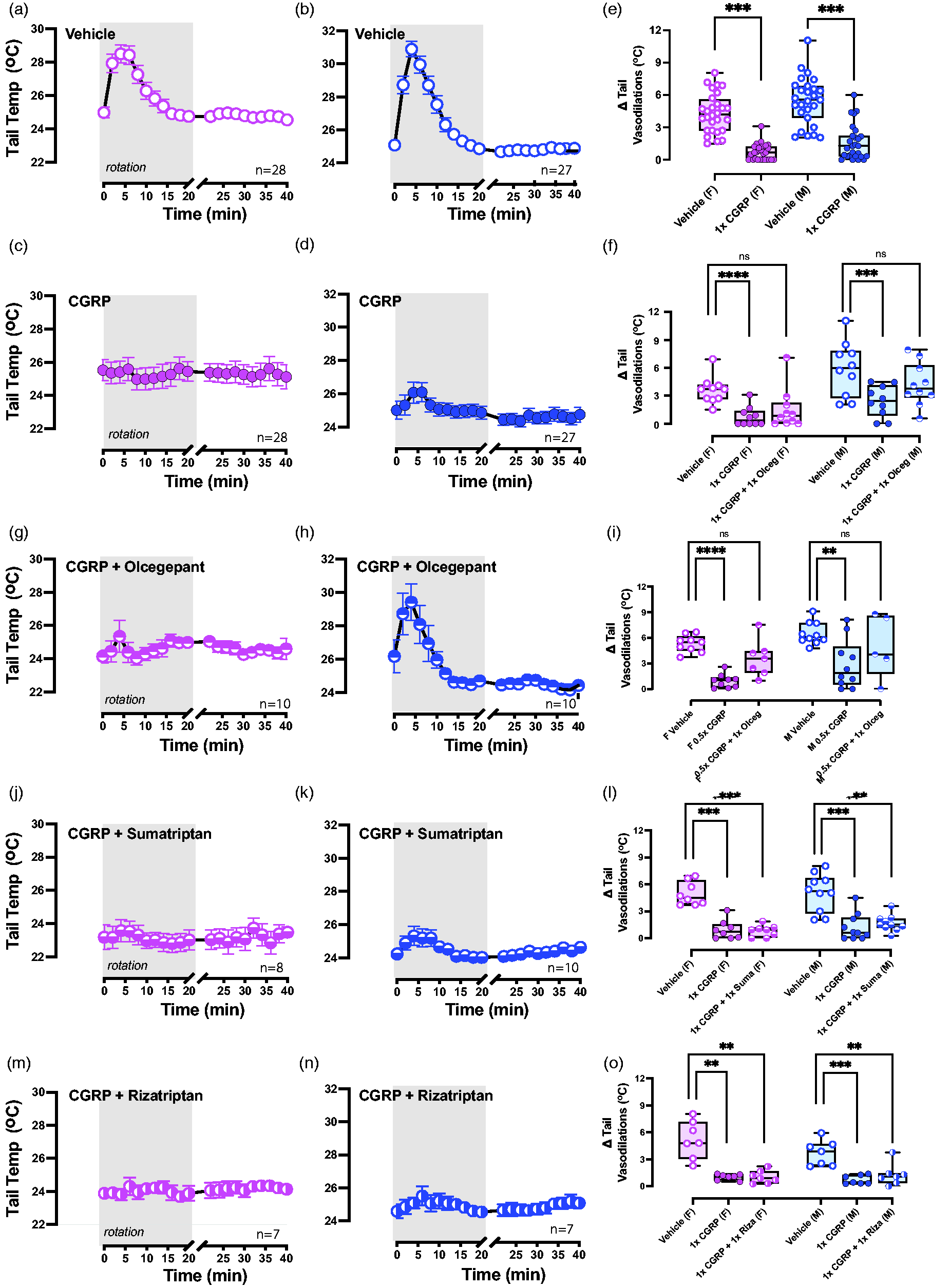
As seen in Figure 4a,b, second-order curve fits were used to compute the recovery of head temperatures back to baseline values after the hypothermia after 1.0× CGRP delivery (28 females/27 males). A general trend of extended recovery times was observed as a result of 1.0× CGRP delivery (female: 24.0 minutes, R2 = 0.73; male: 23.7 minutes, R2 = 0.83) compared to their vehicle control responses (female: 20.7 minutes, R2 = 0.76; male: 19.0 minutes, R2 = 0.87). We observed no differences in the magnitude of head hypothermia between CGRP and vehicle control tests in either sex. Figure 4e,f shows tail temperature profiles after 1.0× CGRP, and two-way ANOVA showed effects of biological sex (F1,53 = 6.79, p = 0.01) and IP CGRP (F1,53 = 251.7, p < 0.001). 100% of mice exhibited normal delta tail vasodilatations after vehicle control administration but a majority of mice (89% female, 59% male) exhibited diminished delta tail vasodilatations (Bonferroni, p < 0.001) after 1.0× IP CGRP administration compared to IP vehicle (Figure 4e). Because of the diminished delta vasodilatations and the extended recovery times detected after CGRP testing, it is inferred that 1.0× CGRP impacted a mouse’s natural response to the provocative motion and their nausea response. In parallel, a different group of mice was used to assess SNP’s effects on motion-induced thermoregulation (8 females/8 males) and temperature profiles were analyzed. When measuring head temperatures, we did not observe any differences in recovery time or in the magnitude of head hypothermia (Figure 4c,d). However, similar to CGRP, two-way ANOVA indicated IP SNP impacted tail vasodilatations (F1,14 = 38.8, p < 0.001) but saw no effects of biological sex (F1,14 = 0.002, p = 0.97). Bonferroni post-hoc analysis indicated 100% of females (p < 0.001) and 75% of males (p < 0.01) exhibited diminished vasodilatations after 1.0× SNP (Figure 4g,h).

Calcitonin gene-related peptide (CGRP)/sodium nitroprusside (SNP) effects on hypothermia to provocative motion and dose-dependent changes in tail vasodilatations. Sample sizes are shown in the upper right corner. Mice allocated for assessing CGRP’s effects on head and tail temperatures were tested for (a, e) vehicle and then (b, f) 1.0× CGRP. When assessing head temperature recovery, the second-order curve fits extrapolated recovery times for vehicle control (female: 20.7 minutes, male: 19.0 minutes) and intraperitoneal (IP) CGRP (females: 24.0 minutes, male: 23.7 minutes). The Δ tail vasodilatations observed after vehicle treatment significantly diminished after 1.0× CGRP. A separate mice cohort was allocated for assessing SNP’s effects on head and tail and was tested for (c, g) vehicle control and later (d, h) 1.0× SNP. Although head temperature recovery did not differ between vehicle and 1.0× SNP groups, delta tail vasodilatations were diminished after 1.0× SNP. (i, j, l, m) Dose-dependent changes in tail temperatures were observed for both IP CGRP (i, j) and IP SNP (l, m). For CGRP testing, 0.1×, 0.5× and 1.0× concentrations were prepared at 0.01, 0.05 and 0.1 mg/kg. For SNP testing, 0.1×, 0.5× and 1× concentrations were prepared at 0.25, 1.25 and 2.5 mg/kg. (k) Two-way mixed effects model assessed the factors (i) sex (male vs. female) and (ii) CGRP dilutions (0.1×, 0.5× and 1.0× CGRP). Compared to vehicle responses, significant differences were observed at 0.5× CGRP for females (p < 0.001) and for males (p = 0.03). These differences were more strongly observed at 1.0× CGRP (p < 0.001 for both sexes). 0.1× CGRP did not have a strong effect. Sample sizes are listed: vehicle (34 females/37 males), 0.1× and 0.5× CGRP (9 females/10 males), 1.0× CGRP (28 females/27 males). (n) Two-way mixed effects model assessed the factors (i) sex (male vs. female) and (ii) SNP dilutions (0.1×, 0.5× and 1.0× SNP). Compared to vehicle responses, significant differences were observed at 1.0× CGRP for females (p = 0.015) and for males (p = 0.0002). Significant differences were not seen at 0.1× and 0.5× SNP. All 8 females/8 males were tested across dilutions. Significance is listed as * = p < 0.05, ** = p < 0.01, and *** = p < 0.001.
Dose-dependent response of CGRP or SNP on tail vasodilatations
Although 1.0× concentrations of CGRP and SNP were used for the motion sickness assay in the present study and these concentrations are based on previous studies assessing these migraine triggers, it was unclear whether lower doses would also elicit observed responses. To answer this question, a dose-dependent response curve was tested in mice (n = 10 males/9 females) and delta tail vasodilatations were analyzed for vehicle, 0.1× and 0.5× of IP CGRP (Figure 4i,j) or IP SNP (Figure 4l,m) and these mice were compared with the original groups of mice tested for vehicle and 1.0× CGRP (Figure 4k) or to SNP (Figure 4n). A two-way mixed effects model and Bonferroni post-hoc assessed the factors: biological sex and CGRP/SNP dilutions. 0.1× CGRP had no significant effect on delta tail vasodilatations in either sex. However, differences were observed when comparing delta tail vasodilatations between vehicle and 0.5× CGRP in females (p < 0.001) and in males (p = 0.03). When assessing SNP’s effects (n = 8 females/8 males), no significant differences were seen at 0.1× and 0.5× SNP compared to vehicle control (Figure 4n), but a significant difference was seen when comparing vehicle to 1.0× SNP in females (p = 0.01) and in males (p = 0.0002). The greatest effect of CGRP and SNP on delta tail vasodilatations was observed at their 1.0× concentration and these concentrations were used to assess effects of migraine blockers.
Olcegepant but not triptans protect against CGRP’s effects on tail vasodilatations
Administration of 1.0× CGRP reduced delta tail vasodilatations in almost all mice (Figure 5e) and so it was hypothesized that mice would regain their healthy response to provocative motion with IP treatment of either CGRP + olcegepant (n = 10 males/10 females), CGRP + sumatriptan (n = 10 males/8 females) or CGRP + rizatriptan (n = 7 males/7 females) and different groups of mice were used. Prior to assessing CGRP + blockers, groups of mice were assessed for blocker-only effects by testing vehicle + 1.0× olcegepant (n = 8 females/10 males), vehicle + 1.0× sumatriptan (n = 7 females/10 males) or vehicle + 1.0× rizatriptan (n = 7 females/7 males), but no differences were seen compared to their vehicle control, and so the blockers, by themselves do not have a secondary effect on tail vasodilatations (supplementary Figure 1).
Olcegepant, but not sumatriptan or rizatriptan, protects against calcitonin gene-related peptide (CGRP)’s effects on tail vasodilatations. Sample sizes are labeled in the bottom right corner. Tail temperatures were recorded after intraperitoneal (IP) Continued.delivery of (a, b) vehicle, (c, d) 1.0× CGRP, (f, g) 1.0× CGRP + 1.0× olcegepant, (i, j) 1.0× CGRP + 1.0× sumatriptan and (l, m) 1.0× CGRP + 1.0× rizatriptan. Bonferroni post-hoc indicated diminished tail vasodilatations after 1.0× CGRP in all mice (p < 0.001 in either sex). (h) Dunnett’s post-hoc comparisons suggested that 1.0× CGRP + 1.0× olcegepant protected against CGRP-induced effects, more so in males than females. (i) Mice treated with 0.5× CGRP but exhibited diminished tail vasodilatations were treated with 0.5× CGRP + 1.0× olcegepant and recovered their response. (l, o) Mice were not protected by 1.0× CGRP + 1.0× sumatriptan or by 1.0× CGRP + 1.0× rizatriptan because Δ tail vasodilatations were still diminished compared to their vehicle response (vehicle vs. CGRP + sumatriptan: p < 0.001 in females and p = 0.004 in males; vehicle vs. CGRP + rizatriptan: p = 0.003 in females and p = 0.004 in males). Significance is listed as * = p < 0.05, ** = p < 0.01, *** = p < 0.001, and **** = p < 0.0001.
Tail temperature profiles were recorded and displayed for vehicle and drug treatments (Figure 5a–d,f,g,j,k,m,n). After quantifying Δtail vasodilatations, one-way ANOVA was used to assess CGRP + blocker effects in the designated groups, and Dunnett’s post-hoc analysis was used to compare treatments to vehicle. F-values and ANOVA p-values are provided in Table 1. We observed olcegepant blocked CGRP-induced changes in males because tail vasodilatations resembled IP vehicle (Figure 5h) and this protective effect was trending in females (p = 0.06). By contrast, triptans did not have a protective effect (Figure 5l,o). Mice after 1.0× CGRP + 1.0× sumatriptan still exhibited diminished Δ tail vasodilatations compared to their IP vehicle test (p < 0.001 in females and p = 0.004 in males). A similar observation was seen in mice after treatment of IP 1.0× CGRP + 1.0× rizatriptan because tail vasodilatations were still significantly diminished in both sexes compared their IP vehicle test (p = 0.003 in females and p = 0.004 in males). In the dose-dependent response study, mice that experienced reduced vasodilatations as a result of the 0.5× CGRP dose were later tested with 0.5× CGRP + 1.0× olcegepant, and 86% of females and 80% of males were rescued after delivery of 1.0× olcegepant (Figure 5i). These findings show that olcegepant blocked the effects of CGRP and allowed for a majority of mice to exhibit their normal response to the provocative motion by blocking CGRP’s effects, whereas both triptans were not effective.
Diminished tail vasodilatations (<1.5°C) indicate a disrupted response to provocative rotation during motion-induced thermoregulation.
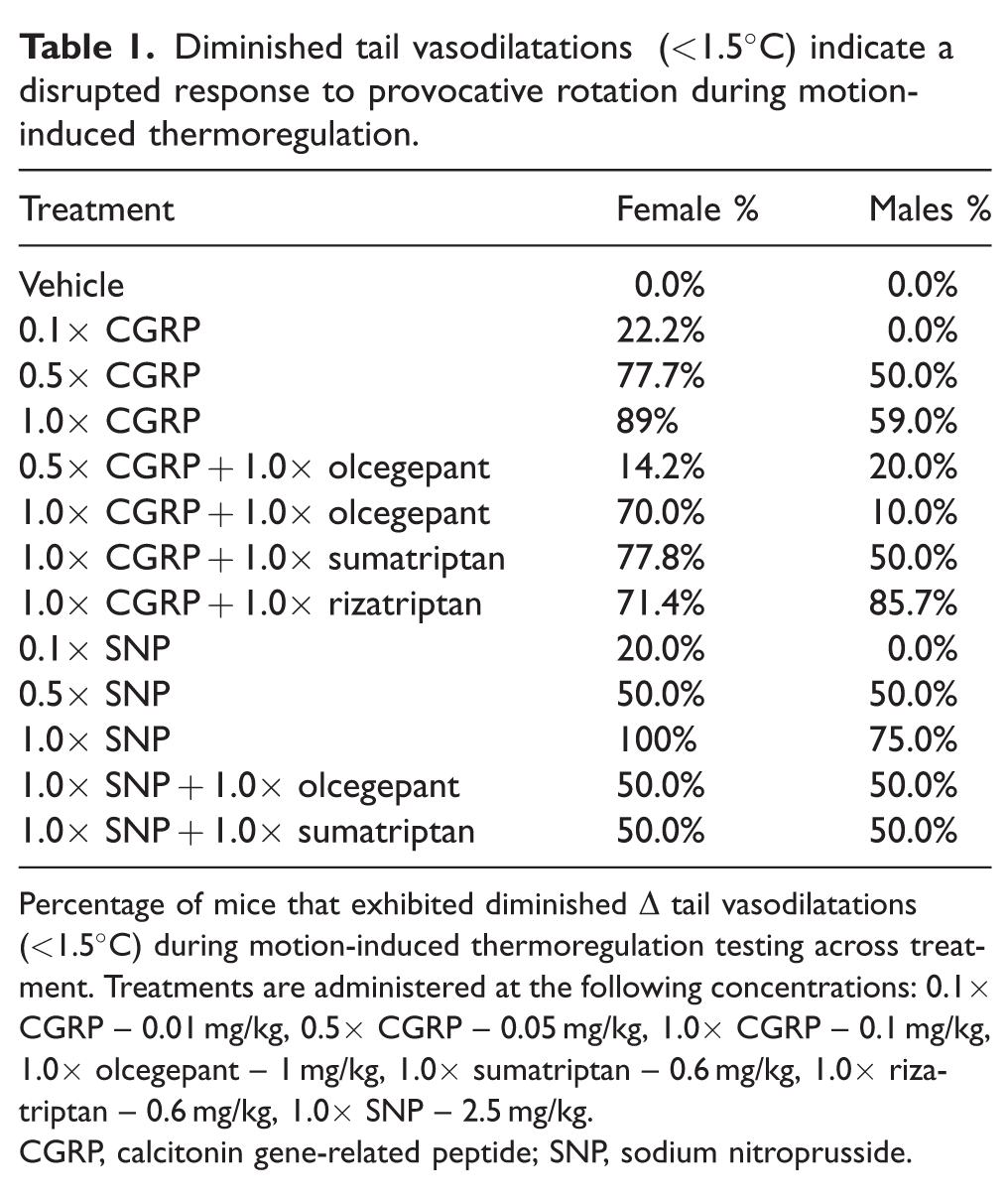
Percentage of mice that exhibited diminished Δ tail vasodilatations (<1.5°C) during motion-induced thermoregulation testing across treatment. Treatments are administered at the following concentrations: 0.1× CGRP – 0.01 mg/kg, 0.5× CGRP – 0.05 mg/kg, 1.0× CGRP – 0.1 mg/kg, 1.0× olcegepant – 1 mg/kg, 1.0× sumatriptan – 0.6 mg/kg, 1.0× rizatriptan – 0.6 mg/kg, 1.0× SNP – 2.5 mg/kg.
CGRP, calcitonin gene-related peptide; SNP, sodium nitroprusside.
SNP’s effects on tail vasodilatations are blocked by olcegepant
The effects of olcegepant and sumatriptan on tail vasodilatations were also assessed against 1.0× SNP (n = 8 males/8 females) and tail temperatures are depicted (Figure 6a–d,f,g,i,j). To conserve mice, the same mice were treated with 1.0× CGRP + 1.0× olcegepant and four days later with 1.0× CGRP + 1.0× sumatriptan. One-way ANOVA with Dunnett’s post-hoc analysis was preformed and F-values and statistics are provided in Table 1. In females and males, robust delta tail vasodilation responses were observed in 50% of females and 50% males after 1.0× CGRP + 1.0× olcegepant (Figure 6h) and these percentages were also seen when mice were later tested with 1.0× CGRP + 1.0× sumatriptan (Figure 6k). Post-hoc analyses in both sexes indicated no significant difference in tail vasodilatations for either SNP + blocker hen compared to vehicle. These mice were not further tested with blockers at lower doses of SNP. Table 1 depicts the percentage differences in the prevalence of diminished tail vasodilatations observed across all treatments (CGRP/SNP + blockers) for both male and female mice.

Olcegepant and sumatriptan block sodium nitroprusside (SNP)’s effects on tail vasodilatations. (a–d, f, g, i, j) Similar to calcitonin gene-related peptide (CGRP) testing, tail temperatures were recorded after intraperitoneal (IP) delivery of vehicle, 1.0× SNP, 1.0× SNP + 1.0× olcegepant, and 1.0× SNP + 1.0× sumatriptan, and sample sizes are labeled in the bottom right corner (e, h, k). Dunnett’s post-hoc comparisons showed that SNP diminished delta tail vasodilatations compared to vehicle control in both sexes, yet no difference between vehicle vs. 1.0× SNP + 1.0× olcegepant, or vehicle vs. 1.0× SNP + 1.0× sumatriptan. Significance is listed as * = p < 0.05, ** = p < 0.01, and *** = p < 0.001
Discussion
The present study evaluated motion sickness and motion-induced nausea using the MSI and motion-induced thermoregulation assays; however, these symptoms comprise few of the many vertiginous symptoms that arise with VM and so this one study cannot fully characterize the VM disease. Interestingly, in VM patients, it has been previously determined that motion-sickness scores directly correlated with the VM symptom severity (2,33).
We show that systemic CGRP injections can induce motion sickness in wild-type C57BL/6J mice, and this motion sickness can be strongly antagonized by the CGRP receptor blocker, olcegepant. Previous studies assessing CGRP’s effects on light-aversive behavior (photophobia) and cutaneous hypersensitivity (allodynia) in rodents indicate that triptans are effective in reducing these surrogate behaviors (34,35) but, interestingly, in the present study, sumatriptan and rizatriptan did not show significant efficacy in blocking CGRP’s effects in motion sickness and motion-induced nausea.
A systematic review of various therapies’ efficacy for VM indicated that effective treatments include vestibular rehabilitation, antiepileptic drugs, calcium channel blockers, antidepressants, beta blockers and selective serotonin reuptake inhibitors (SSRIs) (7). However triptans (5HT1B/1D agonists), such as sumatriptan and rizatriptan which are commonly used to treat migraine pain, are viewed to be ineffective at treating VM’s nausea and motion sensitivity (26–38). Triptans are also associated with inducing nausea in a subset of patients (39). Moreover, a clinical trial of rizatriptan therapy for VM patients (NCT#T02447991) showed no significant benefit of rizatriptan therapy, which is consistent with our preclinical findings. In addition, serotonin receptor agonists for 5-HT1F, known as ditans, have recently gained focus for migraine. Ditans are similar in mechanism to triptans but can penetrate the blood brain barrier and do not have vasoconstrictive properties like triptans (40). However, ditans are correlated with dizziness, nausea, fatigue, vertigo and reduced alertness, thereby reducing their use case for treating VM (40–43).
We have used two different murine motion sickness assays: (i) MSI and (ii) motion-induced thermoregulation. In both assays, provocative motion caused distinct changes, but MSI was too weighted on gastric distress. This bias reduced its validity as an assay because gastrointestinal complications do not directly imply motion sickness. However, the motion-induced thermoregulation model is more suitable for investigating motion sensitivity as it involves studying motion-induced thermoregulation caused specifically by a vestibular stimulus: the constant orbital rotation. In addition, the motion-induced thermoregulation model was robust for assessing CGRP and SNP’s effects. Similar rodent studies suggest tail vasodilatations are a precursor to emesis and removal of toxins (12). Our data showing CGRP’s blunting of tail vasodilatations to provocative motion suggests that motion-induced nausea may be aggravated with CGRP and that olcegepant is protective but not triptans. This finding is in agreement with clinical findings showing that a two-hour infusion of CGRP caused gastrointestinal hyperactivity and nausea, and pretreatment with a CGRP receptor antagonist ameliorated these clinical symptoms but pretreatment with sumatriptan did not (44). Moreover, a clinical meta-analysis of anti-CGRP signaling treatments for nausea in episodic migraine provided strong support for their effectiveness (45). Interestingly, we also observed that female mice were more severely affected by systemic CGRP than their male counterparts, which correlates with preclinical studies showing stronger tactile sensitivity in female mice when challenged with durally applied CGRP (46).
CGRP signaling and murine motion-sickness indices
Other studies have shown in human subjects and in mouse models that CGRP causes diarrhea by disrupting peristaltic intestinal activity and promoting ion and water secretion in the intestinal lumen, and that CGRP signaling antagonism can reverse these effects (47–49). Thus, it is expected that CGRP-induced diarrhea would cause noticeable increases in the MSI calculation. The OVAR rotation further increased the MSI regardless of CGRP or SNP, highlighting the effect of a strong vestibular perturbation in increasing MSI outcomes.
Interestingly, the motion-induced thermoregulation model revealed differences between vehicle and CGRP/SNP injected mice. Differences in motion-induced nausea after CGRP administration were blocked by a CGRP receptor antagonist (olcegepant) but not by triptans (sumatriptan or rizatriptan). We also showed that, in the stationary tests, there are no thermoregulatory changes in the absence of provocative motion. Although male and female mice both showed disrupted thermoregulatory responses to CGRP injections, dose–response experiments at 0.05 and 1 mg/kg CGRP show that olcegepant can rescue CGRP-induced nausea in male yet olcegepant did not fully rescue all female mice. (Table 1). This sexual dimorphism is similar to higher incidence of migraine and VM in female patients (4,50,51) and reports of sexual-dimorphic effects of CGRP both in dura-induced pain, spinal cord neuropathic pain and preclinical surrogate of allodynia or touch sensitivities (46). To this end, other studies have demonstrated a sex difference in the expression of CGRP receptor components in the trigeminal nucleus (52).
The present study is the first to provide preclinical evidence for the use of CGRP antagonists in treating motion hypersensitivities that occur in VM and should be paired with findings from pertinent clinical trials for validation. We provide data showing that the motion-induced thermoregulation assay is sensitive to the known migraine triggers, CGRP and SNP, and also show the effectiveness of different migraine drugs (‘gepants’ and triptans) with respect to reducing nausea responses observed in this mouse model. Future experiments will assess whether mice over- or under-expressing the CGRP receptor will be more or less susceptible to changes in their thermoregulatory responses to provocative motion, and if migraine blockers can provide protection. These experiments will achieve the greater goal of elucidating the role of CGRP signaling in the symptoms of nausea and motion sickness that occur in migraine and VM.
Clinical implications
Enhanced motion-induced nausea is an observed symptom in migraine and especially VM. We used two different murine motion sickness assays (thermoregulation and MSI), but MSI was too weighted on gastric distress to be effective. Differences in motion-induced thermoregulation after systemic CGRP were blocked by olcegepant but not by triptans. Olcegepant was more effective in rescuing CGRP-induced thermoregulatory changes in male compared to female mice.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231223971 - Supplemental material for Calcitonin gene-related peptide receptor antagonism reduces motion sickness indicators in mouse migraine models
Supplemental material, sj-pdf-1-cep-10.1177_03331024231223971 for Calcitonin gene-related peptide receptor antagonism reduces motion sickness indicators in mouse migraine models by Shafaqat M Rahman and Anne E Luebke in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231223971 - Supplemental material for Calcitonin gene-related peptide receptor antagonism reduces motion sickness indicators in mouse migraine models
Supplemental material, sj-pdf-2-cep-10.1177_03331024231223971 for Calcitonin gene-related peptide receptor antagonism reduces motion sickness indicators in mouse migraine models by Shafaqat M Rahman and Anne E Luebke in Cephalalgia
Footnotes
Acknowledgments
We acknowledge assistance with data collection and analysis from Vedat Duzgezen, Stefanie Faucher, Elana Fine, Catherine Hauser, Benjamin Liang and Blaze Strangio. This work was fully supported by NIH R01DC017261 (AEL).
Declaration of conflicting interests
The authors declare that there are no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
