Abstract
To determine whether painful stimulation of the temple would induce nausea, ice was applied to the temple for 30 s, three times at 4-min intervals in 23 migraine sufferers and 22 age- and sex-matched controls. On one occasion, the ice was applied in the presence of residual motion sickness induced by optokinetic stimulation. On another occasion, the ice application was not preceded by optokinetic stimulation (the baseline condition). In the baseline condition, nausea had developed in migraine sufferers but not controls by the third application of ice. In the presence of residual motion sickness, each painful stimulus intensified nausea and headache in migraine sufferers whereas symptoms were minimal in controls. Changes in frontotemporal pulse amplitude were monitored with photoelectric pulse transducers. The extracranial blood vessels dilated in migraine sufferers but not controls before the first application of ice in the baseline condition, presumably due to anticipatory anxiety. In contrast, the ice application did not provoke extra-cranial vasodilation in either group after optokinetic stimulation. The findings show that susceptibility to nausea and stress-induced extracranial vascular hyper-reactivity are associated with the migraine predisposition. They also suggest that head pain might intensify gastrointestinal disturbances during attacks of migraine.
Introduction
Susceptibility to gastrointestinal disturbances persists between attacks of migraine. For example, migraine sufferers are unusually vulnerable to motion sickness (1–5) and to the emetic effect of dopamine agonists such as apomorphine (6) and piribedil (7). Furthermore, recurrent abdominal pain often alternates with or accompanies headache in children (8). Thus, gastrointestinal disturbances may increase vulnerability to migraine.
Children with a history of migraine are more likely to vomit after a mild head injury than other children (9), suggesting that head pain also triggers gastrointestinal disturbances. To investigate this possibility, head pain was induced during motion sickness in migraine sufferers and controls (5). Nausea and other symptoms of motion sickness were generated by the visual illusion of movement (an optokinetic effect). During optokinetic stimulation, an ice block applied to the temple induced a deep, aching pain and intensified nausea whereas pain evoked by immersing the hand in ice water had no effect on nausea. Participants withdrew from optokinetic stimulation when they thought that they were about to vomit. Head but not hand pain boosted the withdrawal rate in migraine sufferers, even though hand pain was judged to be more intense than head pain. Thus, head but not hand pain appeared to intensify gastrointestinal discomfort, at least in migraine sufferers.
Since neurons in the trigeminal nucleus caudalis project to nuclei in the solitary tract (10, 11), some trigeminal fibres may converge on solitary tract neurons that respond to emetic stimuli; if so, head pain could induce nausea directly. The aim of the present study was to investigate this possibility in the presence and absence of residual motion sickness induced by optokinetic stimulation. If convergence of trigeminal nerve impulses onto neurons that respond to emetic stimuli triggers nausea directly, then head pain in the absence of motion sickness should trigger nausea; in addition, the presence of residual nausea after optokinetic stimulation may intensify this effect. On the other hand, if some non-specific aspect of optokinetic stimulation intensifies nausea during painful stimulation of the head, then head pain would not be expected to induce nausea in the absence of optokinetic stimulation.
The second aim was to investigate extracranial vascular responses to painful stimulation of the temple. Painful stimulation of one side of the face (12) and hand (13) generally provokes greater bilateral extracranial vasodilation in migraine sufferers than controls. The generalized nature of the response suggests that it is triggered by an emotional reaction to noxious stimulation. Surprisingly, head pain did not evoke extracranial vasodilation in migraine sufferers during optokinetic stimulation (5), suggesting that the brainstem disturbances that mediate symptoms of motion sickness inhibited extracranial vascular responses. Alternatively, some non-specific effect of motion sickness provocation might have inhibited responses. For example, sensory conflict during optokinetic stimulation might have disrupted stress-induced extracranial vasodilation by diverting attention away from pain.
To distinguish between these two possibilities in the present study, extracranial vasodilation to head pain was investigated in the presence and absence of residual motion sickness. Loss of the response after optokinetic stimulation would support the hypothesis that brainstem disturbances during motion sickness inhibit extracranial vascular reactivity. On the other hand, if the sensory conflict provoked by optokinetic stimulation disrupts stress-induced extracranial vasodilation, then the response should return after optokinetic stimulation.
Methods
Participants
The migraine sample consisted of 18 women and five men (mean age ± SD, 41 ± 11 years) who met International Headache Society criteria (14) for migraine with aura (three subjects) or migraine without aura (20 subjects) and who averaged two or more attacks of migraine per month. The three migraine with aura subjects were included in the present sample because their attacks were similar to those of other subjects apart from the occasional presence of a visual aura. Participants did not take prophylactic medication for migraine and had no other major medical conditions. The control group consisted of 17 women and five men (mean age 39 ± 12 years) who reported less than 12 mild headaches per year that did not meet diagnostic criteria for migraine and that were relieved, when necessary, by analgesics. Each participant provided informed consent for the procedures, which were approved by the Murdoch University Human Research Ethics Committee.
Experiments were carried out when participants were free from headache for at least 4 days. They abstained from medication and alcohol for at least 24 h, and from food, drink and cigarettes for at least 2 h before testing. Women were tested between menstrual periods.
Procedures
The procedures were carried out in a laboratory maintained at 22 ± 1.5°C. Photoelectric pulse transducers (photoplethysmographs; Grass Instrument Co., Quincy, IL, USA) were attached to the frontotemporal region approximately 4.5 cm rostral and 8.5 cm medial to the ears in the vicinity of anterior branches of the superficial temporal artery. A black elastic headband was stretched slightly and placed over the transducers to screen out changes in background illumination that would otherwise have interfered with the signal. Pulses recorded from the left and right temples were sampled at 100 Hz by an MP100 Biopac Systems Analogue/Digital data acquisition system and displayed on a computer monitor with AcqKnowledge software (Biopac Systems, Goleta, CA, USA). Beat-by-beat changes in pulse amplitude were later calculated off-line using AcqKnowledge software. Changes in pulse amplitude reflect changes in cutaneous bloood flow (15).
Measures were obtained during two sessions separated by at least 3 weeks. On each occasion, an ice block with a surface area of 12.25 cm2 was applied to the temple near the point of emergence of the superficial temporal artery in front of the ear for 30 s. The participant held onto the ice block with a short stick that was embedded in the ice. The ice was applied to the usual side of headache in migraine sufferers and to right (nine subjects) or left side (13 subjects) in controls. Two minutes before the ice was applied, participants rated the intensity of nausea and headache between 0 and 10, where 0 corresponded to no nausea or headache, and 10 corresponded to extreme nausea or headache. Shortly after the ice was removed, participants rated headache, nausea, and the intensity of pain during the application of the ice. In particular, participants were asked to distinguish between the pain induced locally by the ice and headache elsewhere. The ice was applied two more times at 4-min intervals.
On each occasion, the ice applications began 4 min after participants sat quietly for 5 min to establish a physiological baseline. At the end of the 5-min period, participants were given the ice block in a cup and were asked to place it on their temple when instructed to do so. On one occasion, the first ice application began 22–24 min after participants withdrew from optokinetic stimulation that was designed to provoke symptoms of motion sickness (4, 5). During this procedure, the participant sat on a stationary chair with his or her head and shoulders inside a drum that revolved 10 times per minute for 15 min or until the participant was about to vomit. An illusion of movement was created by black and white stripes on the internal surface of the drum that moved past the participant's stationary visual field. The conflict between visual and vestibular cues provoked symptoms of motion sickness. On the other occasion, the ice application was not preceded by optokinetic stimulation (the baseline condition). For 23 of the 45 of the participants, optokinetic stimulation preceded the ice application in the first session.
Data reduction and statistical analysis
Pain ratings were investigated in a 2 × 2 × 3 [Group (migraine, control) × Session (baseline condition, ice after optokinetic stimulation) × Trials (the three trials)] analysis of variance. Analyses of nausea and headache ratings contained an additional factor of Ice Stimulation (before vs. after ice application).
Pulse amplitude was measured in 30-s blocks before, during and after each painful stimulus. Since pulse transducers do not measure vessel calibre or blood flow in absolute terms, changes in pulse amplitude were expressed as the percent change from the level recorded during the last 30 s of baseline. Vascular responses were investigated in 2 × 2 × 3 × 2 × 3 [Group (migraine, control) × Side (ipsilateral or contralateral to painful stimulation) × Ice Stimulation (before, during, and after painful stimulation with ice) × Session (baseline condition, ice after optokinetic stimulation) × Trials (the three trials)] analyses of variance. The
Results
Ratings
Ratings of ice-induced pain were greater in migraine sufferers than controls [main effect for Group, F(1,43) = 7.94, P < 0.01], irrespective of whether or not the test was preceded by optokinetic stimulation (Fig. 1). In particular, none of the interactions involving Group, Trial or Session achieved statistical significance.
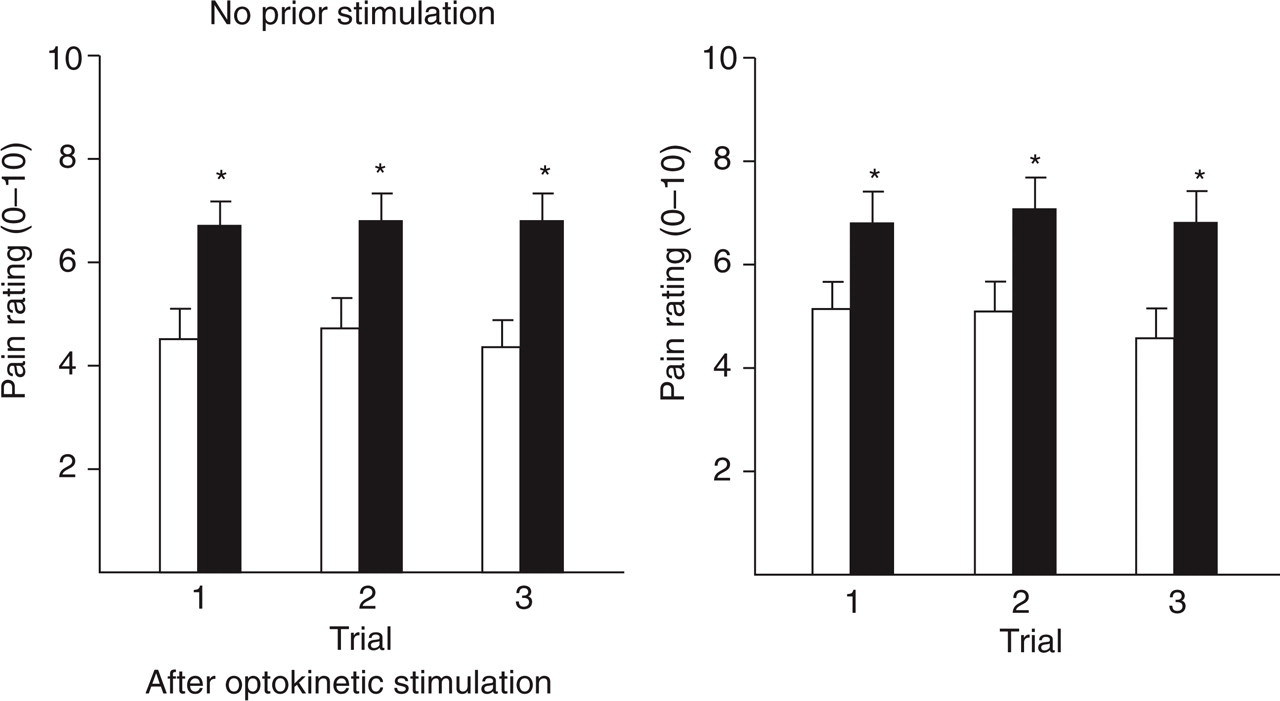
Pain ratings (± SEM) to ice applied to the temple with no prior stimulation (the baseline condition) and after optokinetic stimulation. Pain ratings were greater in migraine sufferers (▪) than in controls (□) (∗P < 0.05).
Nausea increased over the three ice trials in migraine sufferers but was minimal in controls [main effect for Group, F(1,43) = 6.94, P < 0.05; Group × Trial, F(2,42) = 3.77, P < 0.05] (Fig. 2). In addition, nausea was greater in migraine sufferers when the test was preceded by optokinetic stimulation than in the baseline condition [Group × Session, F(1,43) = 5.58, P < 0.05].

Intensity of nausea (± SEM) before and during application of ice to the temple, with no prior stimulation (the baseline condition) and after optokinetic stimulation. Nausea was minimal in controls throughout testing (○), but increased over trials in migraine sufferers (•). Nausea was greater in migraine sufferers than in controls (∗P < 0.05).
As shown in Fig. 3, headache was minimal in controls throughout the experiment, but increased after each application of ice in migraine sufferers [main effect for Group, F(1,43) = 15.5, P < 0.001; Group × Ice Stimulation, F(1,43) = 11.4, P < 0.01]. On average, headache ratings were greater when the test was preceded by optokinetic stimulation than in the baseline condition [main effect for Session, F(1,43) = 7.50, P < 0.01].
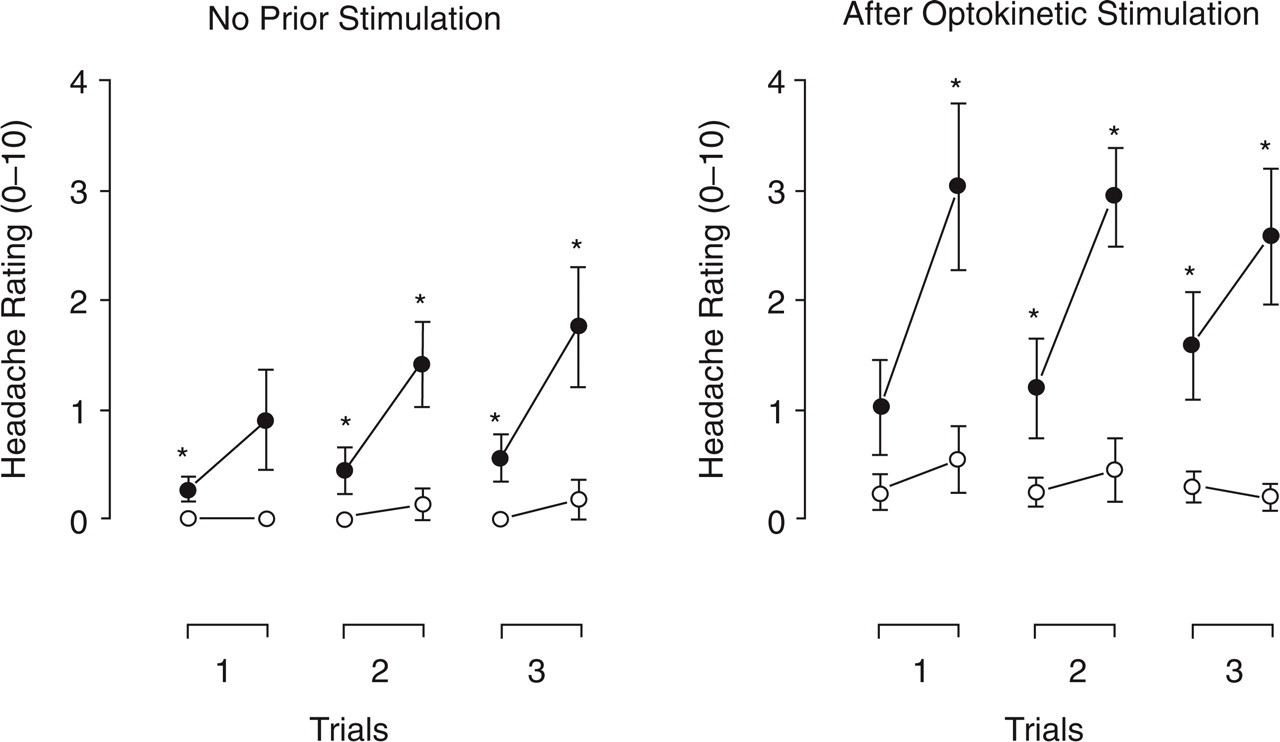
Headache ratings (± SEM) before and during application of ice to the temple, with no prior stimulation (the baseline condition) and after optokinetic stimulation. With no prior stimulation, headache was minimal in controls (○), but increased over trials in migraine sufferers (•). Applying ice to the temple induced greater headache in migraine sufferers than in controls (∗P < 0.05).
Extracranial vascular reactivity
Pulse amplitude increased bilaterally in migraine sufferers over the three trials in the baseline condition, but did not change in either group when ice was applied after optokinetic stimulation [main effect for Group, F(1,43) = 7.89, P < 0.01; Group × Session, F(1,43) = 7.05, P < 0.05; main effect for Session, F(1,43) = 16.1, P < 0.001; Session × Trial, F(2,42) = 8.58, P < 0.001]. The increase in the baseline condition started even before ice was applied for the first time (Fig. 4). Responses did not differ between sides and were not related specifically to ice stimulation (none of the effects involving Side or Ice Stimulation achieved statistical significance).
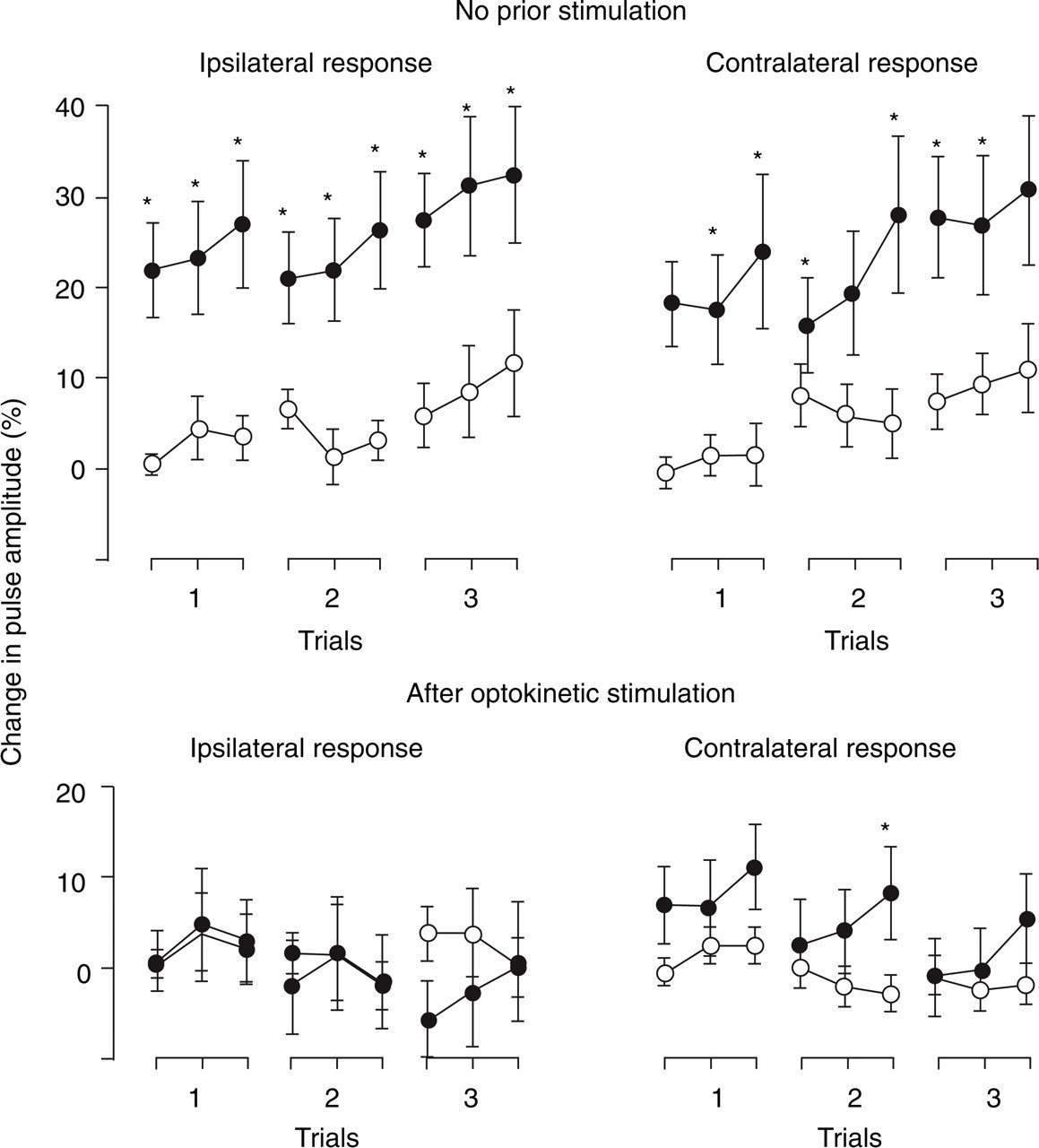
Change in blood flow in the frontotemporal region (± SEM) before, during and after application of ice to the temple, with no prior stimulation (the baseline condition) and after optokinetic stimulation. With no prior stimulation, increases in blood flow were greater in migraine sufferers (•) than in controls (○) both ipsilateral and contralateral to the application of ice (∗P < 0.05). After optokinetic stimulation, increases in blood flow were small and generally did not differ between groups.
Discussion
In the baseline condition, nausea had developed in migraine sufferers but not controls by the third application of ice to the temple. The site of painful stimulation appears to be important because three painful immersions of the hand in ice water did not induce nausea in migraine sufferers or controls (13). Similarly, applying ice to the temple enhanced nausea during optokinetic stimulation and increased the withdrawal rate in migraine sufferers, whereas immersing the hand in ice water did not, even though hand pain was greater than head pain (5). Thus, head pain appears to be a more potent stimulus for nausea than limb pain, at least for migraine sufferers.
Possible triggers for nausea include the trigeminal nerve discharge associated with head pain, or anticipatory anxiety or distress associated with repetitive painful stimulation of the head. Various forms of visceral pain are associated with nausea and vomiting (17–19), presumably because visceral afferents project to emetic circuits in the solitary tract (20). A robust relationship between nausea and affective disorders such as anxiety and depression (21) suggests that negative emotions also influence nausea (22). However, it seems unlikely that pain-related fear or distress mediated nausea in the present study, because intensely painful stimulation of the hand did not provoke nausea in the same group of subjects (5, 13).
Hyperexcitability of trigeminal neurons persists interictally in migraine sufferers (23–25), and spreads from meningeal afferents to second-order neurons in the trigeminal nucleus caudalis during attacks of migraine (26–29). Impulses that converge upon sensitized second-order trigeminal neurons (e.g. from the special senses or from scalp tissues supplied by trigeminal nociceptive afferents) appear to augment headache and produce symptoms such as photophobia (12) and scalp tenderness (30). In the present study, stimulation of trigeminal nociceptive afferents induced headache and nausea more readily in migraine sufferers than in controls, and optokinetic stimulation enhanced these symptoms. Hyperexcitability of trigeminal and/or emetic neurons in migraine sufferers could account for headache and nausea during and after optokinetic stimulation (4, 5), for the synergistic effect of motion sickness symptoms on symptoms induced by painful stimulation of the temple, and for hyperalgesia to painful stimulation of the temple.
Three painful stimuli were required to elicit nausea in migraine sufferers in the baseline condition, whereas nausea increased incrementally from the first to the third painful stimulus after optokinetic stimulation. Presumably residual activity in emetic or trigeminal circuits boosted the nauseating effect of head pain after optokinetic stimulation. The incremental increase in nausea suggests that head pain rapidly sensitized emetic neurons. Delayed habituation (31, 32) or a frank facilitation of responses to repetitive stimulation (12, 33) appears to be characteristic of migraine. Thus, it is tempting to speculate that an inhibitory deficit that enhances the excitability of trigeminal or emetic neurons increases the risk of migraine.
Sensitivity to mechanical stimulation of the forehead increased after optokinetic stimulation in nauseated subjects (4), suggesting that neurons responsive to emetic stimuli facilitate central trigeminal discharge. If so, nausea and headache could build up in a positive loop during attacks of migraine. In the present study ice-induced pain did not increase in migraine sufferers with residual symptoms of motion sickness, but pain ratings were probably limited by a ceiling effect.
Scalp vessels typically dilate more readily in migraine sufferers than controls during short periods of stressful stimulation (34–40). In the present study, anticipation of pain apparently triggered extracranial vasodilation in migraine sufferers because pulse amplitude increased before the first application of ice. This anticipatory response disappeared during optokinetic stimulation (5) and had not returned 20 min later (the present study). The source of the inhibitory influence on extracranial vasodilation is uncertain, but might involve disruption of brainstem vasomotor activity during motion sickness. Body temperature falls during motion sickness (41) in association with increases in skin blood flow in the limbs (42) and face (5). Since cold-induced cutaneous vasoconstriction is attenuated during motion sickness (43), loss of extracranial vascular reactivity during motion sickness may be due to sympathetic inhibition.
In summary, the present findings indicate that head pain triggers nausea and headache in migraine sufferers in the interictal period, possibly because of hyperexcitable emetic neurons or hyperexcitable neurons that project from the trigeminal nucleus caudalis to the nucleus of the solitary tract. If hyperexcitability in these neural circuits is characteristic of migraine, reducing this excitability may decrease susceptibility to recurrent attacks. The findings also show that stress-induced extracranial vasodilation is greater in migraine sufferers than in controls. Dilation of scalp vessels appears to be a source of pain in at least some attacks of migraine (44–46). The link between stress-induced extracranial vascular hyper-reactivity and extracranial vasodilation during attacks of migraine requires further investigation.
Footnotes
Acknowledgements
This study was supported by grants from the National Health and Medical Research Council of Australia and the UK Migraine Trust. We thank Ms Shiree Hassard for technical assistance.
