Abstract
The need for experimental migraine models increases as therapeutic options widen. In the present study, we investigated SB-220453 for efficacy in the glyceryltrinitrate (GTN) human experimental migraine model. SB-220453 is a novel benzopyran compound, which in animal models inhibits neurogenic inflammation, blocks propagation of spreading depression and inhibits trigeminal nerve ganglion stimulation-induced carotid vasodilatation. We included 15 patients with migraine without aura in a randomized double-blind crossover study. SB-220453 40 mg or placebo was followed by a 20-min GTN infusion. Headache, scored 0-10, was registered for 12 h, and fulfillment of International Headache Society (IHS) criteria was recorded until 24 h. Four subjects had a hypotensive episode after SB-220453 plus GTN but none after GTN alone. The reaction was unexpected, since animal models and previous human studies had shown no vascular or sympaticolytic activity with SB-220453. The study was terminated prematurely due to this interaction. GTN was consistent in producing headache and migraine that resembled the patients' usual spontaneous migraine. Nine patients had GTN on both study days. Peak headache score showed a trend towards reduction after SB-220453 compared with placebo (median 4 vs. 7, P = 0.15). However, no reduction was seen in the number of subjects experiencing delayed headache (8 vs. 8), number of subjects reporting migraine (6 vs. 8), migraine attacks fulfilling IHS criteria 1.1 or 1.7 (6 vs. 7) or IHS 1.1 alone (4 vs. 5). SB-220453 had no significant pre-emptive anti-migraine activity compared with placebo in this human model of migraine. Interaction between SB-220453 and GTN was discovered. This is important for the future development of the compound and underlines the usefulness of experimental migraine models.
Keywords
Introduction
Due to the already available effective migraine treatments, it has become increasingly difficult to do early proof of efficacy studies in spontaneous migraine attacks. Patients are reluctant to leave home and to postpone treatment until arrival at a hospital. A human experimental model of migraine allows the administration of an experimental drug under planned circumstances and with many parameters standardized, such as blood samples for safety and pharmacokinetics, time of drug administration in relation to the onset of migraine, and timing in the menstrual cycle. The only human migraine model that at present holds some promise is the provocation of migraine attacks using intravenous infusion of glyceryltrinitrate (GTN) (1). This model has been extensively studied (2) with the aim of harvesting new knowledge about the mechanisms of headache and migraine attacks, but has only a few times been used to test drug effects. One study in migraine patients found no effect of the histamine receptor blocker mepyramine compared with placebo (3). In another study, subcutaneous sumatriptan 6 mg was significantly more effective than placebo in healthy volunteers (4). Recently we have shown the efficacy of sodium valproate in preventing GTN-induced headache and migraine (5).
SB-220453 is a novel benzoylamino benzopyran compound (6), which has shown activity in animal models of migraine, where it inhibits cortical spreading depression (CSD), neurogenic inflammation, induced carotid dilatation and decreases cortical nitric oxide concentrations during CSD (7–9). SB-220453 exhibits a broader spectrum of activity than 5-HT1B/1D agonists by a novel mechanism of action related to an unknown stereo-specific binding site. Triptans show no affinity at this site (6, 10). In animal studies SB-220453 has no cardiovascular (CV) effects and is largely devoid of central nervous system adverse effects, even at very high doses (10). SB-220453 has been given to more than 900 migraine patients and healthy volunteers, but efficacy data are not yet available. No direct CV effects have been seen, and SB-220453 has been well tolerated at all doses.
The aim of the present study was to evaluate the efficacy of SB-220453 and placebo given just before a GTN challenge, both on immediate and delayed migraine headache.
Patients and methods
Patients
Fifteen patients, who met the diagnostic criteria of the International Headache Society (IHS) for migraine without aura, were included in a two-centre study; eight patients (seven female, one male; mean age 34 years, range 26–51) were recruited from the out-patient headache clinic at Glostrup University Hospital (University of Copenhagen, Denmark), and seven patients (six female, one male; mean age 46 years, range 41–52) were recruited among a migraine panel associated to SmithKline Beechams Clinical Research Unit (Addenbrooke's Centre for Clinical Investigation, Addenbrooke's Hospital, Cambridge, UK). Inclusion and exclusion criteria are shown in Table 1. The study followed a double-blind, randomized, placebo-controlled, two-period crossover study design.
Inclusion and exclusion criteria

Study procedures
Patients were all scheduled to have two study days separated by at least 11 days. Patients were re-scheduled to another study day if they had taken any 5-HT1B/D agonists within 48 h, ergotamine or similar medication within 72 h, had experienced a migraine attack within the previous 48 h or had any headache prior to dosing with SB-220453/placebo. Prophylactic migraine drugs were not allowed 28 days prior to study day. Alcohol was not permitted for 24 h prior to dosing and until 48 h postdose on both study days. Patients were required not to eat or drink grapefruit-containing products from 24 h predose until they left the unit, since this can interfere with the plasma concentration of SB-220453. Caffeine intake was restricted on both study days. Each migraine patient was randomized to receive a single oral dose of 40 mg of SB-220453 nanoparticulate tablet and matching placebo tablet on two separate study days. The tablet was given in an overnight semifasting state; a total of 400 ml of water was allowed. On both study days the patients received an infusion with GTN 0.5 µg/kg per min for 20 min in the cubital vein by a volume directed pump (IVAC P2000, Alaris Medical Systems, Basingstoke, Hampshire, UK). The infusion was given 60 min after the dose of SB-220453/placebo. The supine position was assumed during the GTN infusion and until at least 1 h after the start of the infusion. Breakfast was provided at 1 h poststart of the GTN infusion, after which food and fluid were unrestricted. A random urine drug screen was performed on at least one of the study days.
Headache parameters
The headache was scored on a numerical verbal scale from 0 to 10, where 0 represents no headache, 1 represents a very mild headache, including a feeling of pressing or pulsation (prepain), 5 represents a moderate headache and 10 represents the worst possible headache. Headache characteristics (unilateral/bilateral, quality and aggravation) and accompanying symptoms (nausea/vomiting, phono- and photophobia) were recorded for comparison with the IHS diagnostic criteria for migraine without aura. Headache parameters and accompanying migraine symptoms were recorded in the clinic before study start and until 2 h after start of the infusion. Afterwards, the patient used a diary card at home to register headache until 12 h after infusion (see Fig. 1). Distinction was made between an immediate headache occurring within 60 min of the start of infusion and a delayed headache occurring from 60 min and up to 12 h after the infusion. The patients were contacted by telephone 24 h after the infusion to register migraine up to 24 h after infusion and any possible adverse events.

Headache diary. Used by patients at home to register headache until 12 h after infusion.
Vital signs were obtained prior to start of study procedures and were followed until 2 h after start of the GTN infusion. A follow-up clinical examination, including vital signs, ECG, laboratory observations and evaluation of adverse events, was scheduled between 10 and 14 days after the last study day.
Approval was obtained from regulatory bodies and ethical review committees in both Cambridge and Copenhagen. Informed consent was obtained and signed by each patient. The trial was conducted in accordance with the Declaration of Helsinki and conformed to Good Clinical Practice.
Statistical analysis
Due to the exploratory nature of the study, no formal sample size calculation could be made. Target inclusion was 20 migraine patients with completion of 16 patients. This sample size in previous studies with the GTN model has been sufficient and would provide point estimates of the effect of SB-220453 in this model and variability estimates which would assist in the design of future studies. Randomization was done by SmithKline Beecham and the code was not known to the investigators until after the database was closed.
Due to the small number of patients completing the study, no statistical analysis was conducted on the number of patients developing a migraine attack. However, comparison of peak headache was investigated with a paired two-tailed Wilcoxon test. For all analyses, a P-value < 0.05 was considered statistically significant.
Results
The study was stopped prematurely due to a possible interaction between GTN and SB-220453. Therefore, only 15 patients entered the study. Nine patients received GTN on both study days (six in Copenhagen, three in Cambridge). Two patients from Cambridge withdrew informed consent after the first headache provocation with GTN (one placebo and one SB-220453). Four patients had only one headache provocation (two placebo and two SB-220453) before the study was stopped by the investigators. The results are presented in Table 2.
Headache characteristics and associated symptoms in nine patients receiving GTN on both study days and six patients only completing one of the study days

Headache was scored on a numerical verbal scale from 0 to 10.
International Headache Society (IHS) headache criteria: according to IHS criteria the delayed developed ‘migraine’ headache fulfilled two of the following four headache characteristics: unilateral, pulsating, moderate/severe, aggravation by physical activity (patients 1 and 3 fulfillment within 60 min on placebo day).
GTN infusion terminated after 16.5 min.
GTN infusion terminated after 8 min.
GTN infusion terminated after 17 min.
GTN infusion terminated after 13 min.
Fulfilled IHS criteria during GTN infusion, headache less then 4 h. No associated symptoms after 60 min.
Bilateral, moderate pain, not pulsating, not known if aggravated by physical activity.
Fulfilled criteria for atypical migraine IHS 1.7.
Rescue medication taken for headache/migraine before patient fulfilled IHS criteria.
The administration of GTN was consistent in producing both immediate and delayed headache and migraine that resembled the patients’ usual spontaneous migraine. Migraine fulfilling IHS criteria 1.1 occurred on 13 of the 24 days where GTN was dosed (54.2%), with a difference being noted between sites (Copenhagen 11 out of 14, 78.6%; Cambridge two out of 10, 20%). In the nine patients where comparison between SB-220453 and placebo could be made, peak headache score showed no reduction in immediate headache after SB-220453 (mean 3.0 vs. 3.4, median 3 vs. 3) (P = 0.55). There was a tendency for delayed peak headache to be lower after SB-220453 (mean 3.8 vs. 5.0, median 4 vs. 5) (P = 0.11); the same was true for overall peak headache from 0 to 12 h (mean 4.6 vs. 5.8, median 4 vs. 7) (P = 0.15), see Table 2 and Fig. 2. However, no significant reduction of headache score could be detected. No reduction was seen in the number of subjects experiencing delayed headache (8 vs. 8) or migraine attacks fulfilling IHS 1.1 for migraine without aura (4 vs. 5). Number of subjects reporting migraine (6 vs. 8), severity of accompanying migraine symptoms and number of patients taking rescue medication were all numerically somewhat lower after SB-220453, but no definitive conclusions can be made due to the low numbers.
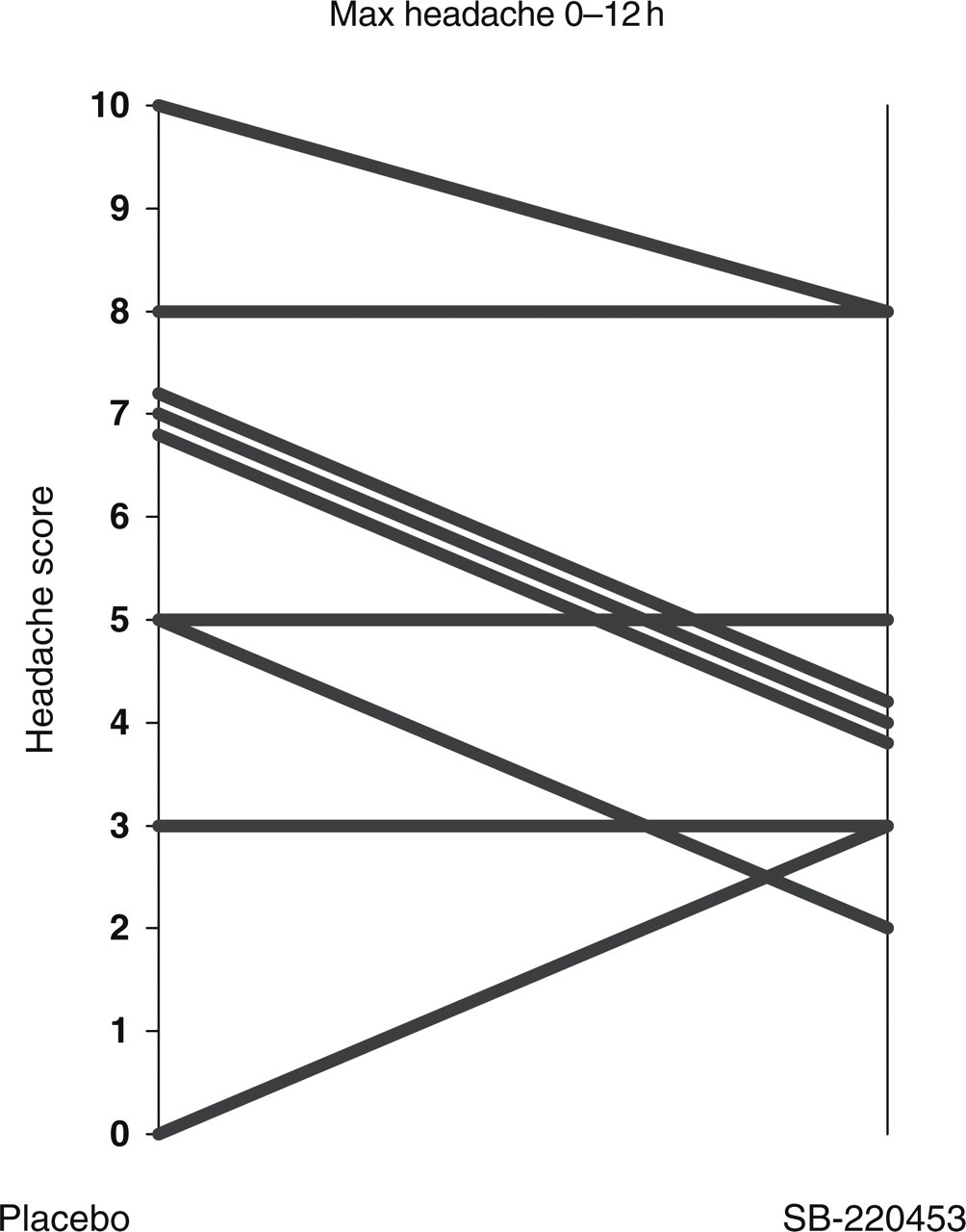
Maximum (peak) headache 0–12 h after glyceryltrinitrate challenge. Median reduced after SB-220453 from 7 to 4 (P = 0.15).
Hypotensive episodes – interaction between SB-220453 and GTN
GTN was dosed twice to nine patients and once to six patients. Of those dosed once, three had placebo and three SB-220453. Overall GTN and SB-220453 were given on 12 occasions and GTN and placebo were given on 12 occasions. Of the 24 occasions where GTN was given, four episodes of clinically observable hypotension occurred, all when GTN was given together with SB-220453. In Cambridge two patients had hypotensive episodes, one very mild and one where the infusion was continued after the patient reported being uncomfortable which resulted in a short period of unconsciousness. The patient regained consciousness after stopping the infusion of GTN and tilting the patient's head down. Both patients in Cambridge with hypotensive episodes were dosed only once and therefore not with GTN alone. In Copenhagen, the infusion of GTN was stopped in two patients due to hypotension. They both had SB-2204453 on the day of the hypotensive episode. The first patient, 16.5 min into the 20-min infusion, had a drop in systolic blood pressure to 82 mmHg and reported being uncomfortable. The infusion was terminated and the legs were raised. The blood pressure (BP) normalized and the discomfort disappeared within 2 min. The patient continued in the study and subsequently on the second study day had placebo and GTN without any signs of hypotension. The second patient, a 33-year-old female, had no signs of hypotension when GTN was given with placebo. On the second study day, when GTN was given with SB-220453, the patient reported discomfort after 8 min of infusion. The infusion was terminated. Heart rate had risen to 102 and then dropped to a minimum of 37. The patient developed a generalized tonic-clonic seizure involving all four extremities, turning of eyeballs, and urinated. The seizure lasted 45 s. 2 min before the seizure vital signs were: BP 105/65, pulse 69. BP was not measurable during the seizure due to the contractions. After the seizure, vital signs were normalized (2 min, BP 97/58, pulse 51; 7 min, BP 106/73, pulse 64). The patient had normal post-seizure symptoms, such as confusion, amnesia and tiredness. She had a childhood history of a few epileptic attacks described as mild faintness, some with tonic-clonic features. There had been no attacks and she had taken no antiepileptic drugs for the last 19 years. Further investigation revealed that the patient had a strong family disposition to epilepsy and avoided attack-provoking situations such as flashing light and large crowds. The seizure was thought to be secondary to a vaso-vagal attack with hypotension and a low seizure threshold. The study was stopped prematurely due to the hypotensive episodes.
Discussion
In this first attempt to use the GTN model for the testing of new potential migraine compounds, we encountered a completely unexpected observation when the test drug and GTN were concurrently administered. The test drug, in an extensive preclinical screening programme, had been completely devoid of vascular effects. Nevertheless, there appeared to be a synergism between the compound and GTN resulting in BP decrease, which in two cases led to serious adverse events and prompted termination of the study before the full number of patients had been included. Therefore, the number of fully treated patients was too small to obtain the desired power and any effects would only be significant if they were of a very large magnitude. This proved not to be the case. Despite this disappointing result, we find that the study is of importance because it provides a range of methodological observations of great importance for the future development of the GTN model of migraine.
Present results and side-effects
Headache results
The data showed a small reduction in peak headache, accompanying migraine symptoms, number of patients with self-reported migraine and migraine fulfilling IHS criteria, when SB-220453 was compared with placebo. The number of patients completing the study and the reduction were too small to show any significance. However, a large treatment effect of SB-220453 in the pre-emptive treatment of GTN-induced headache/migraine can with some caution be ruled out. Studies evaluating the efficacy of SB-220453 on spontaneous migraine attacks are ongoing but results have not yet been published. We conclude that SB-220453 appears to have no pre-emptive efficacy compared with placebo in a GTN-induced migraine model.
Differences between centres
In the present study there appear to be marked differences between the two centres with regard to patient selection and their response to GTN treatment. Thus, the patients in Copenhagen developed headache and migraine fulfilling IHS criteria for migraine without aura after GTN infusion in 11 (79%) of 14 dosing days. This is consistent with results from earlier studies (1). In contrast, at the Cambridge centre, only two out of 10 (20%) developed IHS migraine after GTN infusion. A possible explanation could be the recruitment of patients. At the Cambridge centre no previous clinical migraine studies had been performed and the experience with migraine was limited. This could have adversely affected diagnostic precision despite the fact that efforts were made to standardize the study procedures. It is also possible that, at the Cambridge centre, patients may have taken rescue medication earlier than in Copenhagen, i.e. before they fulfilled the IHS criteria. The more successful migraine provocation in Copenhagen was not due to selection of GTN-sensitive patients, since none of the patients had previously been exposed to GTN. In conclusion, extensive and adequate training is required before the GTN model can be used in multicentre trials.
Interaction, safety of GTN
Hypotensive episodes after infusion of GTN 0.5 µg/kg per min in 20 min have been observed in approximately 10% in previous studies (1, 3) (and unpublished results). We have challenged more then 100 migraine patients and healthy volunteers with this dose of GTN. In previous studies no serious adverse events occurred, incidents having been mild hypotension and discomfort. In no case has unconsciousness or any kind of seizure been observed and no interventions other than termination of the infusion and raising the legs have been necessary. In the present study, four patients had a hypotensive episode and they all had SB-220453 together with GTN. Two of the patients with a hypotensive episode after SB-220453 and GTN were dosed with placebo and GTN on a different day, where they showed no tendency to hypotension. There can be no other explanation of the hypotensive episodes than an interaction between SB-220453 and GTN. This was totally unexpected, since SB-220453 had shown no clinical vasoactivity or sympaticolytic activity in previously performed animal and human studies.
SB-220453 has been shown to be without human coronary vasoconstrictory properties, in contrast to what is seen in triptans (11). This made SB-220453 a treatment candidate for patients with coronary disease where triptans are contraindicated. These patients would, however, have a high risk of experiencing side-effects due to the possible interaction between SB-220453 and GTN. It is clearly of great importance that the interaction between SB-220453 and GTN was discovered under closely controlled conditions and very early in the development of the compound. The mechanism of hypotension is unknown, but it could possibly be caused by reduction of the normal GTN-induced increase in sympathetic activity. This finding indicates that SB-220453 may have a broader range of neuronal inhibition than previously recognized.
Methodological considerations
In the present use of the GTN model, the test drug/placebo was given before the GTN challenge that induced headache and migraine. This design was chosen because the test drug is an oral formulation with peak concentration after approximately 1 h and a long half-life. Furthermore, the design would mimic pre-emptive treatment. Interest has increased in treating patients before they develop migraine (pre-emptive treatment). Many migraine patients have premonitory symptoms occurring hours to days before the attack. Recently we demonstrated that such patients could accurately predict the onset of a migraine attack (12). A drug that is effective given pre-emptively therefore has considerable interest.
In daily practice, most migraine patients treat their migraine headache as soon as they feel a migraine attack is imminent, and before they suffer all the accompanying symptoms. Recent data analysing protocol violators in migraine trials suggest that it might be advantageous to treat the headache when it is mild (13–15). In an experimental model, there is an even greater wish to use rescue medication early. This can abort the attack before it fulfils IHS migraine criteria. Since many patients take rescue medication before they fulfil IHS criteria for migraine without aura (IHS classification 1.1), this, used alone, is an insensitive endpoint, as also illustrated by the present results. Asking patients to refrain from taking anti-migraine medication before they fulfil IHS criteria may lead to non-compliance and impel the patient to describe and experience symptoms before they are actually present. Several other endpoints may be of interest: (i) peak headache, (ii) area under the headache curve (total accumulated headache), (iii) number of patients developing delayed headache, (iv) patient-reported migraine attacks, (v) accompanying migraine symptoms, (vi) use of rescue medication, (vii) fulfillment of IHS criteria 1.7; all of the above with rescue medication taken into account.
Rescue medication has influence on most outcome parameters. It affects the area under the headache curve in an unpredictable way. If, for example, the active treatment reduces migraine pain or postpones the development of migraine without relieving headache completely, this could lead to a longer-lasting headache that is not severe enough to require rescue medication. In contrast, migraine could develop quickly on the placebo day, and be treated effectively with rescue medication resulting, paradoxically, in a lower area under the headache curve. Rescue medication taken on both study days can adversely affect a crossover comparison if the intake is different in relation to the progression of the attack. This can lead to a more severe attack with higher peak headache on one of the study days only due to the timing of rescue medication. These endpoint difficulties make it important to look at more than one endpoint and to evaluate the effect of a drug in the GTN model in a broader way.
Future use of the GTN model
The present study is one of a series examining various uses and modulations of the GTN model. Major factors are whether migraine patients or human volunteers are studied, whether experimental drugs are administered before GTN, after GTN but before development of delayed headache, during delayed headache/migraine, or during a prolonged infusion of GTN, and whether the drug is given as acute use, pre-emptively or as chronic prophylactic use. The present study has shown the importance of studies using the GTN model being done in a headache centre where physicians are familiar with the model and with diagnosing and treating headache. Special care is necesary in taking the patients’ history and in the physical and neurological examination. Patients with past or present neurological abnormalities other than migraine should not participate in GTN studies. The dose of GTN should be carefully chosen, taking the properties of the test drug into account. Proper precautions should be made to anticipate any adverse events and interactions. Thus, patients should be continuously monitored during the study, and particularly during GTN infusion. We find that the GTN model is a safe and reliable method of inducing headache and migraine. The endpoints should be chosen carefully and the present study has shown that due to early use of rescue medication, IHS criteria for 1.1 migraine without aura may be a rather insensitive endpoint. Using peak headache and area under the headache curve as well as patient-reported migraine is not without problems, but should be considered in future studies using the GTN model.
Footnotes
Acknowledgements
This study was sponsered by SmithKline Beecham (SB-220453/65) (now GlaxoSmithKline).
