Abstract
The need for experimental models is obvious. In animal models it is possible to study vascular responses, neurogenic inflammation, c-fos expression etc. However, the pathophysiology of migraine remains unsolved, why results from animal studies not directly can be related to the migraine attack, which is a human experience. A set-up for investigations of experimental headache and migraine in humans, has been evaluated and headache mechanisms explored by using nitroglycerin and other headache-inducing agents. Nitric oxide (NO) or other parts of the NO activated cascade seems to be responsible for the induced headache and migraine. Perspectives are discussed.
Introduction
Along with the fast development of biomedicine, the need for experimental models has increased. The optimal model is an animal model; however, there exist no animal models of migraine. Migraine attacks are difficult to study because of their attack-wise appearance.
During the migraine attack, changes in cerebral blood flow, large cranial arteries and CGRP output have been demonstrated (1 –6). These changes vary throughout the attack; this is why it is crucial to relate changes to the appropriate phase in the migraine attack. This is only possible during a controlled set-up.
Possible models of experimental headache
Nitroglycerin (NTG) and histamine have previously been used to induce migraine and cluster headache attacks, mostly in attempts to use the drugs as a diagnostic test for migraine (7, 8). After a single sublingual dose of NTG, an immediate, short-lasting, bilateral, pulsating headache has been described in healthy subjects. In migraine patients and subjects with a family history of migraine a delayed migraine-like headache occurred several hours afterwards (9). Cluster headache attacks can be induced, but only during cluster periods (10).
During three different doses of intravenous histamine, migraine patients experienced a more severe and pulsating headache than tension-type headache patients and healthy controls (11). The induced headache was blocked by the H1 antagonist mepyramine and to a lesser degree the H2 antagonist cimetidine. During histamine-induced headache in healthy controls the cerebral blood flow is unchanged, as in migraine attacks without aura (12).
Prostacyclin, prostaglandin E1 (PGE1), reserpine and m-chlorophenylpiperazine (m-CPP) are all able to induce headache. Reserpine and m-CPP induce a migraine-like headache several hours after exposure in migraine patients or subjects with a family history of migraine. However, they both have undesirable side-effects (for review see 7).
In 1986 Jes Olesen initiated the work in human experimental headaches. We decided to focus on NTG-induced headache. The goal was to build up a human experimental headache/migraine model, which was safe, robust and not too difficult to manage.
Why use nitroglycerin?
NTG is well tolerated, has a very short half-life, crosses the blood-brain barrier and the side-effects are known and acceptable. NTG is de-nitrated to nitric oxide (NO) and vasoactive S-nitrosothiols. Both activate cytoplasmic guanylate cyclase resulting in increased intracellular guanosine 3′, 5′-monophosphate (cGMP), an increase in cytosolic calcium and vasodilatation (13). NO is involved in the regulation of vessel tone, inflammatory responses, cell communication, non-adrenerge non-cholinerge (NANC)-nerve activity, central sensitization and pain transmission. Nitric oxide synthase (NOS) is responsible for NO generation and is localized in nerve fibres surrounding the adventitia in large cerebral blood vessels.
Basic characteristics of the nitroglycerin-model in healthy subjects
First the NTG-induced headache was described in healthy subjects (14). Headache intensity was registered using a numerical verbal rating scale from 0 to 10, 1 representing a very mild headache (including feeling of pressing or pulsating, pre-pain), 5 a moderate headache, and 10 the worst possible headache. Headache characteristics were recorded according to the IHS criteria for migraine without aura. Because of a very variable bioavailability after sublingual administration, NTG was infused intravenously. The dose-response relationship between headache and NTG was investigated in 10 healthy subjects with no history of migraine. NTG-doses were infused for 10 min, and increased successively from 0.25–0.5–1.0–2.0 μg/kg/min. To estimate headache reproducibility, a retest was performed after at least 1 week. Headache developed in 9 of 10 subjects. The intensity was mild to moderate. The non-responding subject was the same on both study days. A reproducible ceiling effect in headache score at 0.5 μg/kg/min was found from the dose-response curves.
The day to day variation in headache intensity was acceptable. Of a total of 50 paired infusions 25 did not differ in maximal headache response, 20 differed by 1 headache score (out of 10 possible), 4 differed by 2 and only 1 differed by 3 headache scores from day to day. Also the headache characteristics were reproducible. In all subjects the headache was bilateral on both study days. Headache quality was pulsating in seven subjects, pressing in two and did not vary between days.
Vascular responses during NTG-headache in healthy subjects
To follow NTG activity on the arteries, diameters of the temporal and radial arteries were measured using high frequency ultrasound (Dermascan CR, Cortex technology, 20 MHz centre frequency and 15 MHz bandwidth) (15). The NTG-induced dilatation of temporal and radial arteries showed a similar time-profile and ceiling effect as the NTG-induced headache (16). The maximal response at 0.5 μg/kg/min was not due to early developed tolerance, as no tolerance was observed in headache intensity and arterial responses after 7 h NTG infusion (17).
Combining measurements of cerebral blood flow (CBF) (SPECT, Tomomatic 232) and blood velocity (transcranial Doppler, TCD) (EME), it is possible to indirectly estimate changes in diameters of the large cerebral arteries. Changes in cerebral blood volume (CBV) were followed using SPECT with dual energy window facilities (18). During NTG infusion (0.5 μg/ kg/min) headache rapidly increased (median headache score 4), blood velocity in the middle cerebral artery (MCA) decreased by 20% and rCBV increased by 13%, while rCBF remained constant. Sixty minutes after infusion, no subject experienced headache. rCBV was normalized but blood velocity in the MCA remained low (19). A longer lasting effect on the intracranial than extracranial arteries and the venous system is thus indicated. This could be due to an increased release of perivascular vasodilatory peptides or transmitters from trigeminal and parasympathetic nerve endings surrounding the vessels. However, plasma levels of CGRP were not increased during NTG-induced headache in healthy subjects, in spite of moderate headache intensity (20).
Which molecule or compound induces the headache?
NO or other parts downstream in the activated NO-cGMP cascade, because:
The long-acting nitrate, 5-isosorbide-mononitrate (5-ISMN), gives rise to NO formation but its other metabolites are different from those of NTG. In healthy subjects a close relation between 5-ISMN plasma levels, headache and arterial dilatation was found after 15-, 30-, 60-mg 5-ISMN and placebo (21). Therefore, NTG-induced headache must be induced by the action of NO and not by the other metabolites of NTG.
N-acetylcysteine (NAC) enhances the cardiac vasodilatory effects of NTG, presumably by stimulating the NO production. NAC also potentiated the NTG-induced headache and dilatation of the temporal artery (22).
The NTG-induced headache could also be secondary to NTG-induced histamine release. Pre-treatment with mepyramine, a H1 antagonist able to block histamine-induced headache (9), did not change the NTG-induced headache and arterial responses (23). Histamine causes release of NO via endothelial H1 receptors, and NO may therefore be the mediator in both NTG-and histamine-induced headache.
Is the NTG-model useful in migraine drug development?
The value of the NTG-model in migraine drug development is dependent on the response to migraine-specific drugs and the power of the model. Therefore sumatriptan, the first selective 5HT1B/1D agonist highly effective in treating migraine attacks, was administered subcutaneously in 10 healthy subjects, followed by a 20-min infusion of NTG 0.12 μg/kg/min. Sumatriptan significantly decreased the NTG-induced headache response to (median) 1.5 headache score (range 0–5) vs. 4 headache score (range 2–6) after placebo (Fig. 1, 24). The study demonstrated that headache relief could be detected in a small sample size and that the immediate NTG-induced headache must share some mechanisms with migraine.
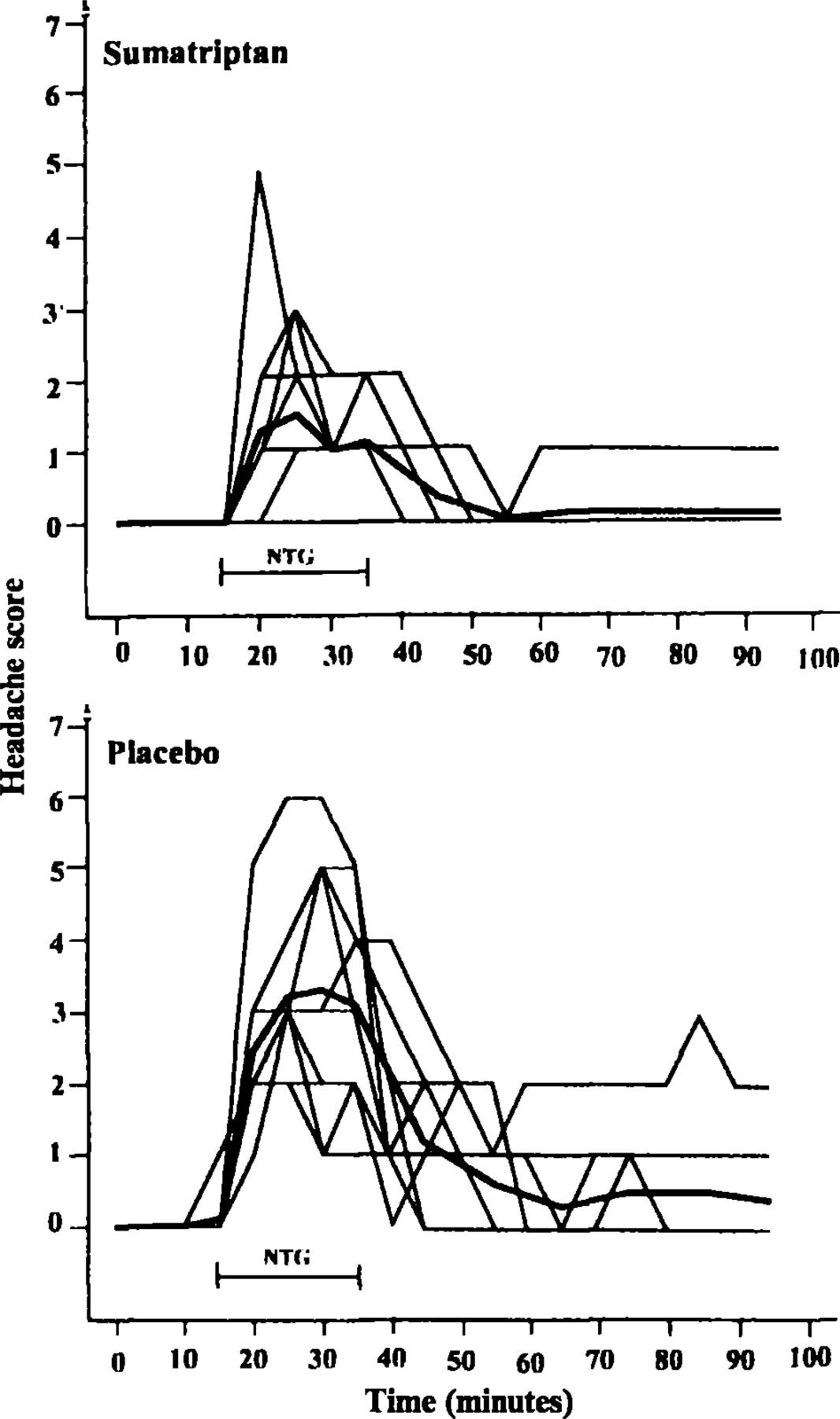
Headache during NTG infusion after pre-treatment with sumatriptan 6 mg s.c. (left) and placebo (right). Individual headache responses are shown and the bold lines represent the mean of 10 subjects. Sumatriptan significantly decreased the NTG-induced headache (24).
NTG-induced headache, migraine and vascular responses in migraine patients
The headache responses to increasing doses of NTG (0.015, 0.03, 0.25 and 0.5 μg/kg/min) were studied in 17 patients with migraine without aura, 17 healthy subjects and nine patients with tension-type headache (25). Changes in mean blood velocity in the MCA were measured with TCD (26). The NTG-induced headache was more severe in migraine patients (Fig. 2) and they also showed a faster and greater MCA dilatation than healthy controls. In migraine patients without aura, an immediate headache of mild to moderate intensity was observed during a single NTG infusion. After a mean delay of 5.5 h, a more severe headache developed. This headache was very similar to the patients' genuine migraine attacks (27). Thus, migraine patients are supersensitive to NO, and NO also triggers a process leading to a migraine attack.
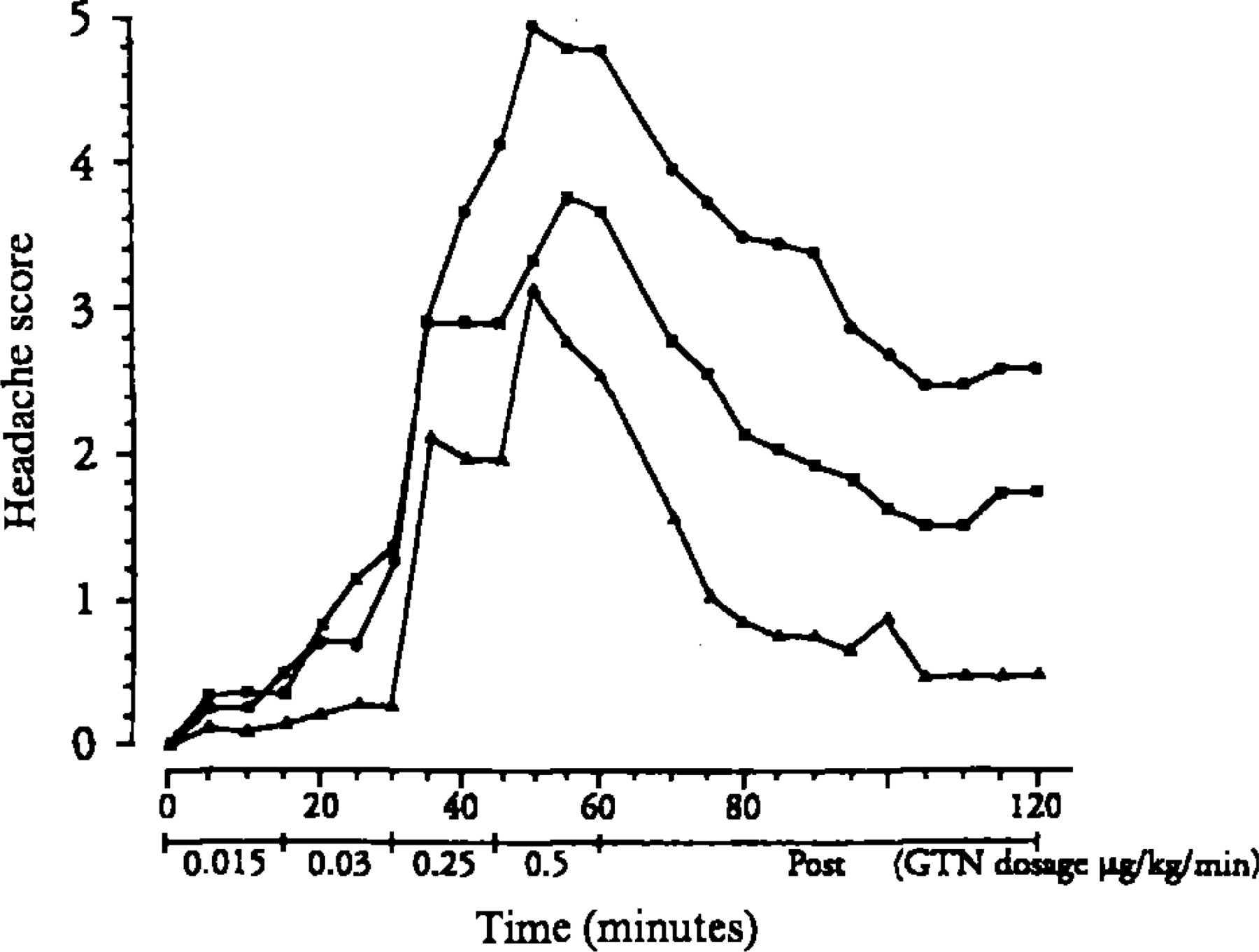
Headache responses after increasing doses of NTG in migraine patients (•, n = 17), tension-type headache patients (▪, n = 9) and healthy controls (▴, n = 17). The NTG-induced headache was significantly most severe in migraine patients (25).
Other compounds used in the experimental set-up and basic mechanisms
In migraine patients without aura histamine induces both an immediate and delayed headache and a dilatation in the MCA, of the same magnitude and following the same time profile as during NTG infusion (28). The histamine-induced headache, migraine and dilatation of the MCA could be blocked by the H1 antagonist mepyramine, whereas the NTG-induced headache, migraine and vascular responses were unaffected by pre-treatment with mepyramine also in migraine patients (29). Thus, the induced migraine after histamine must be triggered by NO released from the endothelium.
The surprisingly long latency period between NTG/ histamine exposure and the fully developed migraine attack, indicates that activation of NO — or steps in the NO-cGMP cascade — initiates a rather slow ongoing process, which in migraine patients results in a migraine attack. The NTG-induced migraine can be blocked by steroids, which inhibit iNOS, the enzyme responsible for the slow inducible NO production (30).
During migraine attacks, increased levels of CGRP have been found in blood obtained from the external jugular vein (6). To clarify if CGRP was able to induce headache, CGRP was given intravenously to 12 migraine patients without aura. Also CGRP induced both an immediate and delayed headache, with a similar time profile as after NTG and histamine infusion; however, the induced headaches were milder (31).
Dipyridamole is a phosphodiesterase (PDE) 5 inhibitor, which increases the level of cGMP. It also inhibits adenosine re-uptake, leading to increased cAMP. Eight of 12 healthy subjects experienced a mild immediate headache-after intravenous dipyridamole (0.142 μg/ kg/min). Five subjects had a mild delayed headache with maximal headache 3 h after primary exposure. The CBF was unchanged and blood velocity in the MCA decreased, corresponding to a dilatation of 5.6% (32). The development of selective PDE inhibitors is ongoing, and with these new compounds, modulation of the NO-cGMP cascade and the effects on the induced headache and migraine can be studied.
Perspectives
The results from studies of NO-induced headache/ migraine have given rise to a lot of new ideas (33, 34). Can the NOS inhibition treat a migraine attack? Yes! (35) However, chronic tension-type headache also decreased after NOS inhibition (36). Can tolerance to NTG prevent chronic cluster headache attacks? No, unfortunately not (37). Are some parts of the activated NO-cGMP cascade more important than others? Do all PDE inhibitors induce headache and migraine? Is CGRP the key molecule?
Studies are ongoing for evaluating the model as a tool in early stage development of migraine prophylactic drugs. The set-up offers the possibility to study all phases of a migraine attack and to explore migraine pathophysiology by comparing induced responses in migraine patients and healthy subjects. At last, use of the model enables a more direct interaction between human and animal studies. Knowledge about differences in responses due to species, topography and other factors, will increase the biomedical relevance of results obtained from animal studies.
