Abstract
Prophylactic drug trials in migraine are long-lasting and expensive and require long-term toxicology information. A human migraine model would therefore be helpful in testing new drugs. Immediate headache and delayed migraine after glyceryltrinitrate (GTN) has been well characterized. We have recently shown that sodium valproate has prophylactic effect in the GTN model. Here we report our experience with propranolol in this model. Nineteen subjects with migraine without aura and 16 sex- and aged-matched healthy subjects were included in a two-centre randomized double-blind cross-over study. Fourteen migraine subjects and 14 healthy subjects completed the study and results from comparison of the 28 subjects are reported. Randomly propranolol 160 mg or placebo were each given daily for 14 days to both migraine and healthy subjects. A 20-min intravenous infusion of GTN 0.25 mg/kg per min was administered on a study day at the end of both pretreatment periods. Headache was registered for 12 h after GTN infusions. Its intensity was scored on a numerical verbal rating scale from 0 to 10. Fulfilment of International Headache Society (HIS) criteria was recorded for 24 h. Radial and superficial temporal artery diameters and blood velocity of both middle cerebral arteries were measured. All migraine subjects developed headache after GTN. No reduction of overall peak headache was found after propranolol (median 5, range 0-7) compared with placebo (median 5, range 0-10) (P = 0.441). Eight of the 14 completing migraine subject developed IHS 1.1 migraine after GTN, two subjects on both days, three subjects only after placebo, and three subjects only after propranolol. No reduction of GTN-induced migraine was found after propranolol compared with placebo (5 vs. 5, P = 1.000). All healthy subjects developed headache after GTN. No reduction of overall peak headache was found after propranolol (median 2, range 1-5) compared with placebo (median 1, range 1-7) (P = 0.315). Two subjects fulfilled IHS criteria 1.1 for migraine without aura after propranolol but not after placebo. The fulfilment was short lasting and did not require rescue medication. Headache after GTN was more pronounced in migraine subjects than in healthy subjects both with (P = 0.003) and without pretreatment with propranolol (P = 0.017). We found that 2 weeks of propranolol constricted the radial artery in healthy subjects but not in migraine subjects. GTN-induced vasodilatation abolished this difference. Mean maximum blood flow velocity in the middle cerebral artery was higher in healthy subjects than in migraine patients (P = 0.003-0.033) and unaffected by propranolol. We observed no effect of propranolol on GTN-induced headache and migraine. This could indicate that GTN induces migraine at a deeper level of the pathophysiological cascade of migraine than the prophylactic effect of propranolol. Propranolol does not constrict cerebral arteries, which therefore cannot be part of its mechanism of action in migraine.
Keywords
Introduction
Migraine is a common disorder with a 1-year prevalence of 10% of the population. Despite the effectiveness of the triptans in acute treatment, there is still need for good prophylactic treatment of migraine in many patients with frequent or long-lasting attacks. It is estimated that 2% of the population and 5–10% of migraine patients may benefit from prophylactic treatment (1).
The intermittent and unpredictable nature of migraine makes it difficult to study. Animal models are useful in studying basic migraine mechanisms, but are of little use in determining the effect of new drugs on migraine frequency or migraine pain. Prophylactic trials in migraine are difficult and expensive, requiring 3 months of treatment and long-term toxicology studies (2). A valid human migraine model would therefore be helpful. Glyceryltrinitrate (GTN)-induced headache has been extensively investigated in both migraine patients and healthy subjects (3–5). Besides an immediate headache during infusion, migraine patients experience a delayed headache after GTN that resembles the patients’ spontaneous migraine attacks and fulfills International Headache Society (IHS) criteria (6). The prophylactic effect of propranolol and other β-adrenoceptor antagonists (β-blockers) in migraine is well established (7). We have previously shown the efficacy of sodium valproate in preventing GTN-induced headache and migraine (8). We therefore studied the effect of propranolol prophylaxis on GTN-induced headache and migraine, in order to validate our model further. In addition, we studied a number of vascular reactions in cerebral and peripheral arteries besides the effect of high-intensity light on urinary catecholamine excretion.
Exposure to high-intensity light has previous been reported to increase urinary noradrenaline excretion with a small decrease in urinary adrenaline. In contrast, migraine patients had a marked decrease in urinary noradrenaline with an increase in adrenaline (9). This change was reversed by 10 days of treatment with propranolol (10). We tried to replicate this finding in order to validate it as a surrogate marker for prophylactic efficacy.
Materials and methods
Subjects
Nineteen migraine subjects who met the diagnostic criteria of the IHS for migraine without aura (11) and 16 healthy subjects were included in this two-centre, randomized, double-blind, placebo-controlled crossover study (see Fig. 1). One patient discontinued due to side-effects of propranolol (nausea, dizziness and emesis). One patient discontinued due to severe headache after headache provocation on the placebo study day. Three patients were excluded due to headache/migraine prior to headache provocation on one of the study days. Three of these patients were replaced in the Copenhagen centre without breaking the randomization code. One healthy subject discontinued due to a pulmonary embolism not related to the study and one healthy subject withdrew consent after the first study day for personal reasons. Fourteen migraine subjects and 14 healthy subjects completed both study days. The subjects were recruited between February 1998 and September 1999. At Glostrup University Hospital (University of Copenhagen, Denmark) subjects were recruited from the out-patient headache clinic and through advertising among the hospital staff. At GlaxoWellcome Clinical Pharmacology (Northwich Park Hospital, London, UK) subjects were recruited through local general practitioners and hospital consultants and through advertising. Migraine subjects and healthy subjects were matched for gender and for age within 5 years (see Table 1).

Double-blind, cross-over study design and subjects according to treatment. Three migraine subjects excluded due to migraine within 48 h prior to study procedures were replaced without breaking the randomization code.
Demographic characteristics

Mean and range. Patients and controls were sex and age matched.
∗In Copenhagen three excluded females were replaced.
Inclusion criteria were: age between 18 and 65 years; body mass index within the range 19–29 kg/m2; weight range female 50–90 kg, male 55–95 kg; good general heath determined by medical history, physical examination, ECG, spirometry and urine and blood screening tests including hepatitis B and C. Females with child-bearing potential could participate provided they had a negative pregnancy test and used a reliable contraceptive. Migraine subjects should fulfil the criteria of IHS 1.1 for migraine without aura (11) with an attack frequency between one per month and one per week for at least 1 year. Exclusion criteria included: known allergy to the drugs used; blood donation within the previous month; breastfeeding; migraine prophylaxis within 2 months of study start; previous proven inefficacy of propranolol prophylaxis; alcohol or drug overuse (urine screen for drugs of abuse); regular use of prescribed or over-the-counter medication except oral contraceptive pill and usual acute migraine treatment; participation in a study with a new molecular entity during the previous 4 months or any trial within 3 months. Healthy subjects were not included if they had more than one tension-type headache per month. Furthermore, they were challenged with sublingual nitroglycerin 0.5 mg (2 × 0.25 mg; Nycomed, Roskilde, Denmark; 0.50 mg; Cox Pharmaceuticals, Barnstable, UK) and only those developing headache were included. Migraine subjects were included without nitroglycerin challenge. The investigator on the two study days was kept blinded to whether the subjects were healthy or migraine subjects.
Study procedures
Subjects were all scheduled to have two separate study days and randomized to receive propranolol LA 160 mg (Beta Progane; Tillomed, St Neots, UK) or matched placebo tablets as a single daily oral dose for 13 days prior to the study days. The tablets were self-administered by the subjects in the morning. No dose titration or adjustment to weight was made. The subject registered the date and time of tablet intake every day in a diary, and attended a control visit after 7 days of medication (diary control, adverse event questioning, vital signs). The two study days were separated by at least 28 days in order to obtain a wash-out period of a minimum of 14 days. Alcohol was not permitted for 24 h prior to study procedures and until 48 h postdose. An alcohol breath test was performed on both study days. Subjects were required to refrain from smoking while in the unit. Standard breakfast was provided prior to start of the study procedures, fluid was unrestricted. Caffeine intake and strenuous exercise were restricted in relation to the study days. A canula (venflon) was inserted in the cubital vein for infusion and safety. On both study days the subjects received an intravenous infusion with GTN 0.25 µg/kg per min for 20 min in the cubital vein by a volume directed pump [IVAC P2000; Alaris Medical Systems (formerly Ivac Medical Systems), Hampshire, UK]. Glyceryltrinitrate [5 mg/ml; SAD, Copenhagen, Denmark; 5 mg/ml Mayne Pharma (formerly Faulding Pharmaceuticals), Paramus, NJ, USA] was diluted with saline to a 50-ml 10 µg/ml solution. Supine position was assumed 30 min prior to GTN infusion and until at least 3 h after the start of the infusion. The start of GTN infusion was designated time 0. Migraine patients were excluded from the study if they had taken any 5-HT1B/D agonists within 48 h, ergotamine or similar medication within 72 h, had experienced a migraine attack within the previous 48 h or had any headache prior to study procedures. Unscheduled prophylactic migraine drugs were not allowed during the whole study.
Headache parameters
Headache was scored on a numerical verbal rating scale from 0 to 10, where 0 represents no headache, 1 represents a very mild headache, including a feeling of pressing or pulsation (prepain), 5 represents a moderate headache, and 10 represents the worst possible headache (4). Headache characteristics (unilateral/bilateral, quality and aggravation by physical activity) and accompanying symptoms (nausea/vomiting, phono- and photophobia) were recorded in order to compare with the IHS diagnostic criteria for migraine without aura (11). Headache parameters and accompanying migraine symptoms were recorded in the clinic before study start and until 3 h after start of the infusion, by a physician in the clinic using a standardized questionnaire. Afterwards, the patient used a diary card at home to register headache until 12 h after infusion. Distinction was made between an immediate headache occurring during the 20-min infusion or within 40 min after, and a delayed headache occurring from 60 min and up to 12 h after start of the infusion. Fulfilment of the IHS diagnostic criteria for migraine without aura was recorded up to 24 h after infusion. The patients could take usual acute migraine medication any time after the headache provocation.
On the day before GTN challenge, subjects from Northwick Park Hospital were exposed to a change from low-intensity light (dim daylight and sunglasses for 90 min) to high-intensity light (160-W fluorescent light for 90 min). Urine samples were collected for measurement of catecholamines and analysed for noradrenaline, adrenaline and dopamine by high-performance liquid chromatography.
Haemodynamics
At Glostrup Hospital the mean maximal velocity in both the right and left middle cerebral artery (MCA) was measured simultaneously by transcranial Doppler (TCD) (DWL Multi Drop ×; DWL Electronische Sys., Singen, Germany) prior to the GTN infusion and at the time points 5, 10, 15, 20, 30, 45, 60, 120 and 180 min (for further description of procedures see 12, 13). Diameters of the superficial temporal artery and the left radial artery were measured with ultrasound (Dermascan C; Cortex Technology, Hadsund, Denmark) (14) at the same time points as TCD. The superficial temporal artery was measured on the patient's usual migraine headache side or on the hand-dominant side if the headache was usually double-sided. End-expiratory CO2 was measured simultaneously during all TDC measurements.
Vital signs were obtained prior to start of study procedures and followed until 3 h after start of the GTN infusion. A follow-up clinical examination, including vital signs, ECG, laboratory observations and evaluation of adverse events, was scheduled between 10 and 14 days after the last study day.
Approval was obtained from The Danish Medicines Agency and The Scientific Ethical Committee for the County of Copenhagen, and in the UK from the Ethics Review Committee at Northwitch Park Hospital. Signed informed consent was obtained from each patient. The trial was conducted in accordance with the Declaration of Helsinki amended in Somerset West, South Africa 1996, and conformed to rules for Good Clinical Practice (GCP).
Statistical analysis
Due to the exploratory nature of the study, no formal sample size calculation was made. Target inclusion was completion of 16 migraine patients and 16 healthy volunteers equally distributed between the two centres. This sample size would provide point estimates of the effect of propranolol in this model and variability estimates which would assist in the design of future studies. Randomization was performed by GlaxoWellcome and the code was not known to the investigators until after the database was closed. Comparison of the number of subjects experiencing headache/migraine attacks on the two study days was investigated with a sign test and peak headache was investigated with paired two-tailed Wilcoxon test. The vascular changes were analysed over time with MANOVA. For all analyses, a P-value < 0.05 was considered to be statistically significant. Catecholamines were analysed with t-test (paired and independent). The statistical programs SAS version 6.12 (SAS, SAS Institute, Cary, NC, USA) and SPSS version 11 (SPSS, Chicago, IL, USA) were used to analyse the data.
Results
In both migraine and healthy subjects blood pressure and heart rate decreased during the 13 days of propranolol. The mean changes in migraine and healthy subjects were, respectively: systolic blood pressure 121–109 and 116–107 mmHg; diastolic blood pressure 77–66 and 78–65 mmHg; pulse 66–60 and 70–61 beats/min. This was an expected finding and no formal statistical analyses were carried out.
Sixteen migraine subjects and 16 healthy subjects were given GTN infusion after pretreatment with propranolol or placebo. Fourteen migraine subjects and 14 healthy subjects completed both study days and crossover comparisons were possible. One migraine and one healthy subject completed only the placebo study day and one migraine and one healthy subject completed only the propranolol study day.
Headache and migraine
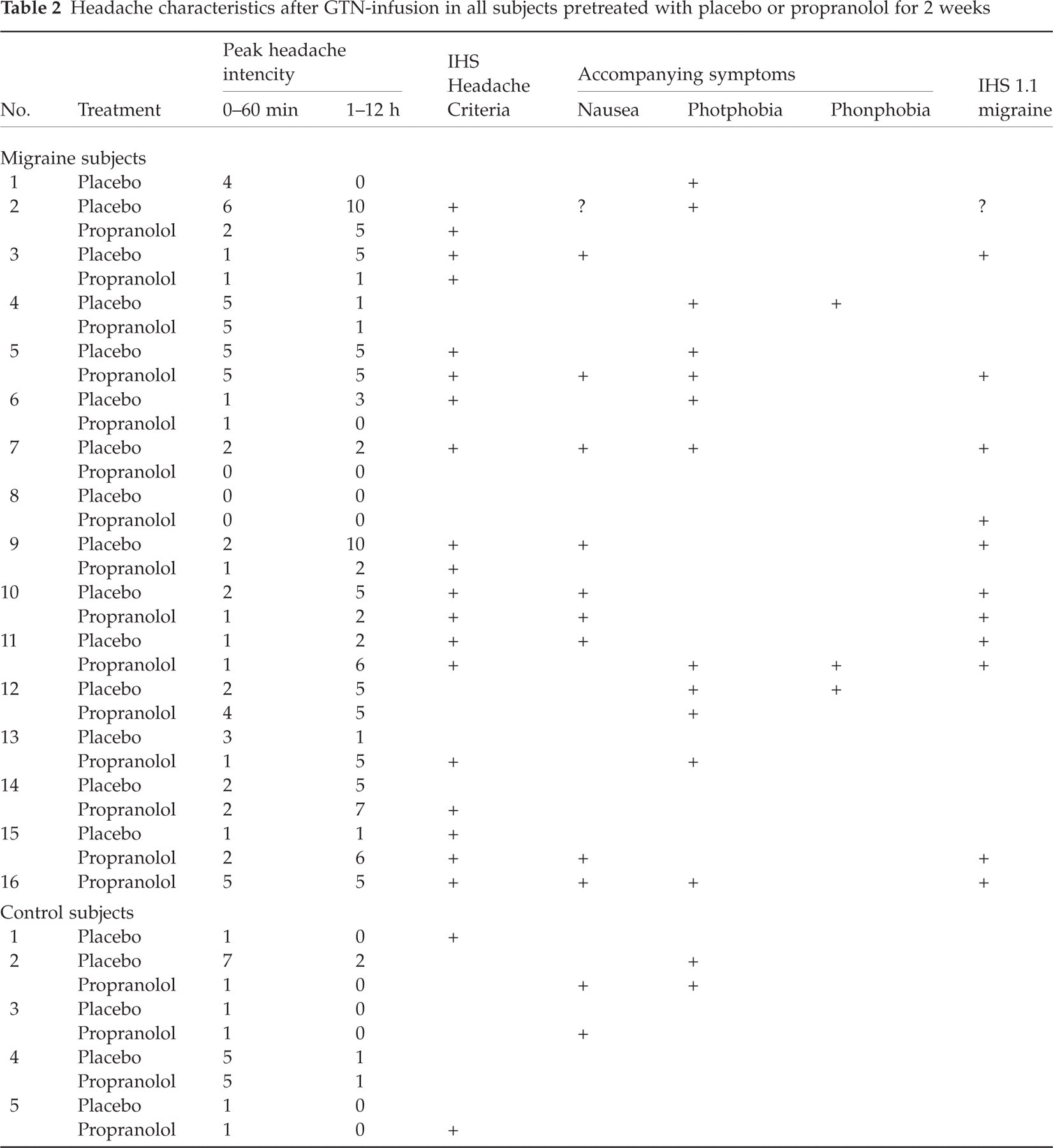
Headache intensity, characteristics, associated symptoms and fulfilment of IHS criteria for migraine are presented for all GTN-treated subjects in Table 2. To illustrate the time course of headache, the mean headache over time is plotted in Fig. 2.
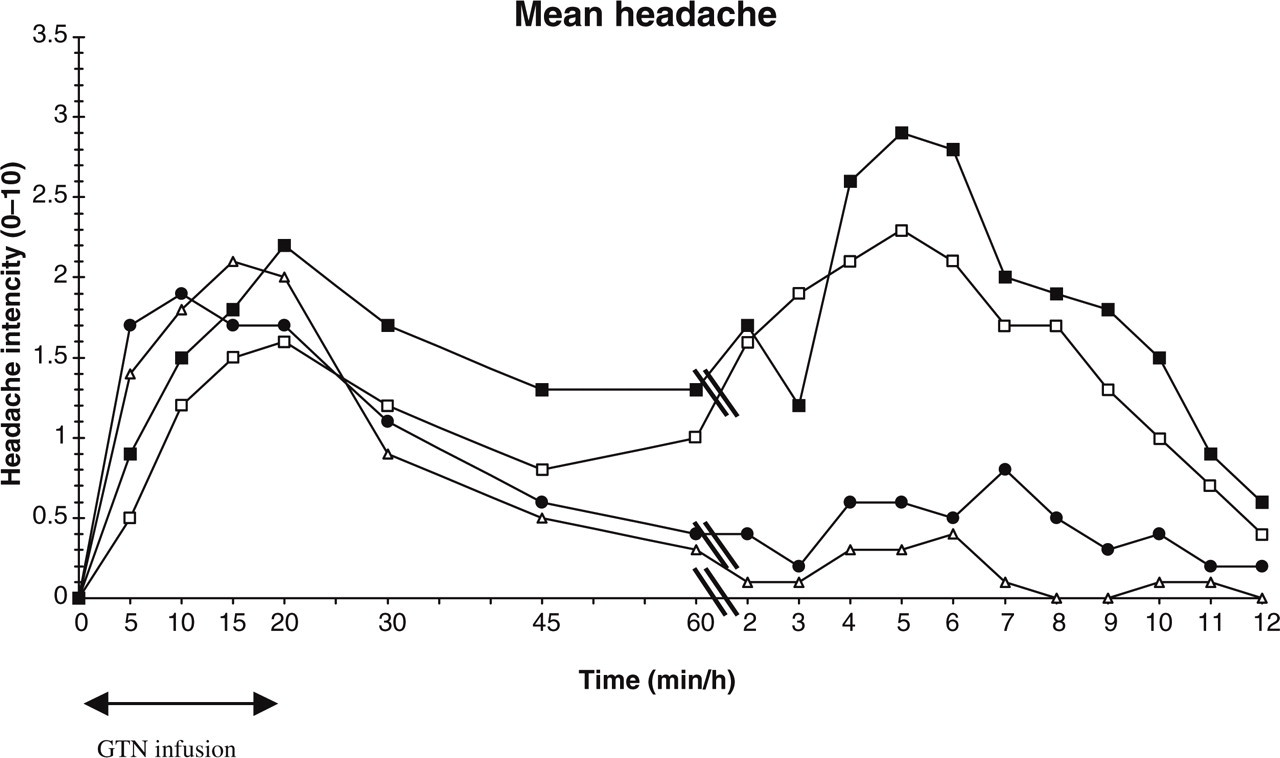
Mean headache over time, immediate (0–60 min) and delayed (up to 12 h). •, Control placebo; ▵, control propranolol; ▪, migraine placebo; □, migraine propranolol.
Headache characteristics after GTN-infusion in all subjects pretreated with placebo or propranolol for 2 weeks

As part of the inclusion criteria, all healthy subjects in the study developed headache after sublingual nitroglycerine. Intravenous GTN infusion of 0.25 µg/kg per min for 20 min was successful in inducing headache in all healthy subjects. Delayed headache (1–12 h) occurred on the placebo day in 10 healthy subjects and after propranolol in six healthy subjects (P = 0.125, sign test). Median peak headache was 1 (range 1–7) after placebo vs. median 2 (range 1–5) after propranolol (P = 0.315, Wilcoxon).
Two healthy subjects (no. 10 and 13) developed a headache that fulfilled IHS criteria for migraine without aura on the propranolol study day. However, the headache was mild or short lasting and did not require medical treatment.
After placebo, GTN induced IHS migraine in five of 15 migraine subjects (33%) compared with six of 15 after propranolol (40%). In a crossover comparison, two migraine subjects had migraine on both study days, three only on the placebo study day, and three only on the propranolol study day (P = 1.000, sign test). One migraine subject who completed only the propranolol study day had migraine on that day.
GTN induced migraine in nine of 16 migraine patients (56%) on at least one of the study days.
Of the 14 migraine subjects who completed both study days, 13 had both immediate and delayed headache at least on the placebo day. Two of these did not have delayed headache after propranolol.
Median peak headache was 5 (range 0–10) after placebo vs. median 5 (range 0–7) after propranolol (P = 0.441, Wilcoxon).
No difference in numbers with headache was seen between migraine and healthy subjects in immediate headache (0–60 min), either on the placebo day (93% vs. 100%) or on the propranolol day (87% vs. 93%). The same was seen for peak headache (see Table 3). A marked difference was seen in delayed headache (1–12 h) between migraine and healthy subjects both on the placebo day (87% vs. 66%) and on the propranolol day (80% vs. 40%). The difference is highly significant for headache intensity (see Table 3).
Peak headache intensity score (0–60 min and 1–12 h); median difference

Vascular data on migraine and healthy subjects are given in Tables 4 and 5, and Fig. 3.

Vascular measurements. Top: mean maximal flow velocity (cm/s) in left (a) and right (b) middle cerebral artery. Bottom: mean diameter of radial artery (c) and temporal superficial artery (d). Values shown are mean ± SEM. •, Control placebo; ▵, control propranolol; ▪, migraine placebo; □, migraine propranolol.
Statistical analysis of difference between propranolol and placebo for vascular measurements

LS-mean. Artery diameter as peak. Flow velocity as throughout 60 min.
Superfic. temp., Superficial temporal artery diameter; radial, radial artery diameter; left MCA; left middle cerebral artery blood flow velocity; right MCA, right middle cerebral artery blood flow velocity.
Statistical analysis of difference between migraine and healthy subjects for vascular measurements

LS-mean. Artery diameter as peak. Flow velocity as throughout 60 min.
Superfic. temp., Superficial temporal artery diameter; radial, radial artery diameter; left MCA, left middle cerebral artery blood flow velocity; right MCA, right middle cerebral artery blood flow velocity.
No change in end-expiratory CO2 was seen during measurements of the vessels.
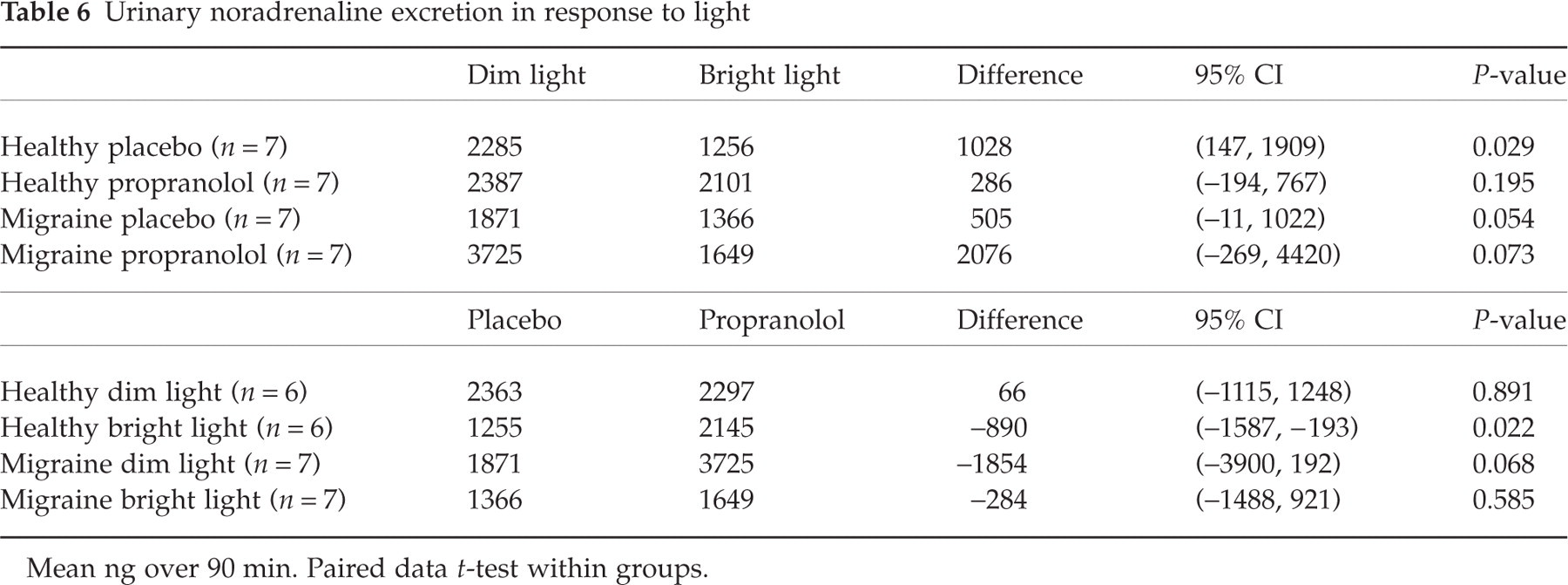
Urinary catecholamine excretion of noradrenaline (NA), adrenaline (AD) and dopamine (DO) in response to high-intensity light in both migraine patients and healthy subjects and the effect of propranolol are shown in Tables 6, 7 and 8. Large individual variation was found in catecholamine excretion and no difference was found between migraine patients and healthy subjects in the response to high-intensity light after placebo [mean difference NA 523 ng, 95% confidence interval (CI) − 411, 1457, P = 0.240; AD − 331 ng, 95% CI − 724, 62, P = 0.090; DO 2769 ng, 95% CI − 4957, 10494, P = 0.432] and after propranolol (mean difference NA − 1789 ng, 95% CI − 4139, 560, P = 0.113; AD 4 ng, 95% CI − 313, 305, P = 0.979; DO − 6209 ng, 95% CI − 15 097, 2679, P = 0.144).
Urinary noradrenaline excretion in response to light
Mean ng over 90 min. Paired data t-test within groups.
Urinary adrenaline excretion in response to light

Mean ng over 90 min. Paired data t-test within groups.
Urinary dopamine excretion in response to light
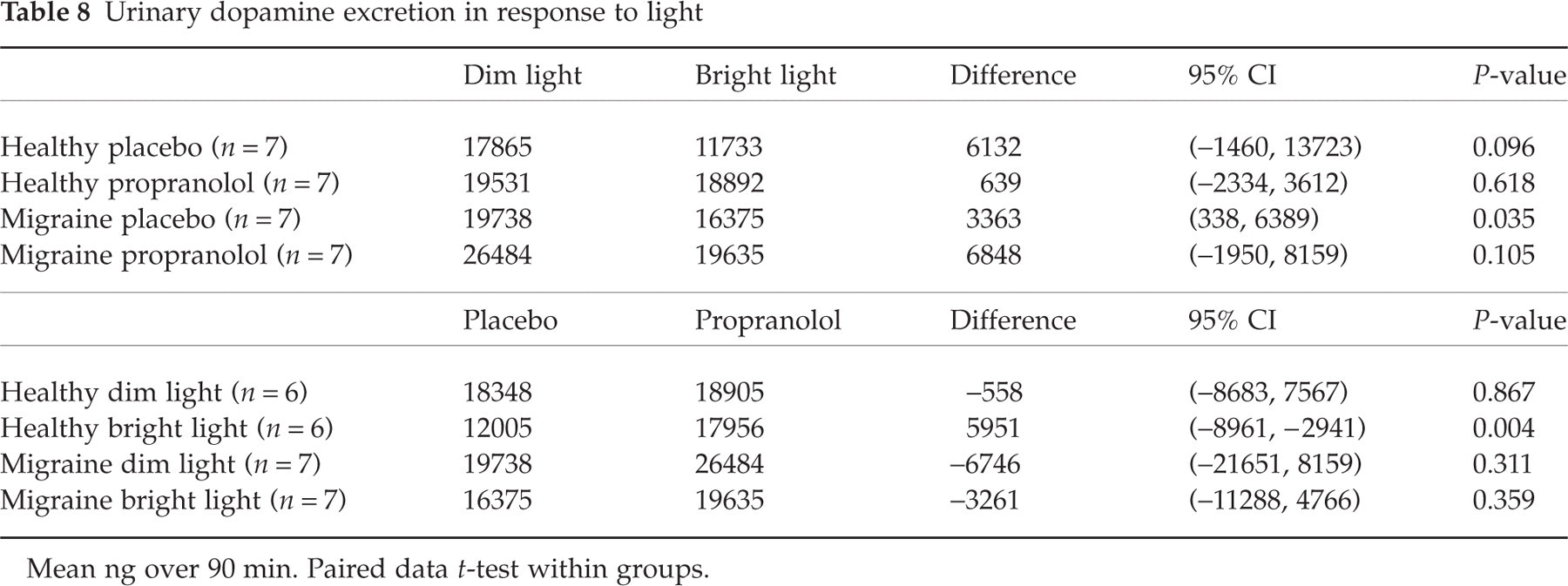
Mean ng over 90 min. Paired data t-test within groups.
Discussion
Headache results
In our previous studies, a dose of GTN 0.5 µg/kg per min induced migraine fulfilling IHS criteria for migraine without aura in 80% of migraine patients (6). In the present study we did not want to overrule the weak prophylactic effect of propranolol and therefore used a dose of 0.25 µg/kg per min. The immediate headache response associated with GTN infusion was almost identical in both healthy and migraine subjects and similar to previous results when taking into account the small differences in dosing regimes (4, 5, 15). The delayed headache was more pronounced in the migraine group compared with the healthy subjects in both severity and number of subjects with a highly significant difference, as also seen in previously published studies (16). However, complete fulfilment of IHS criteria for migraine without aura after GTN infusion was 33% on the placebo day and 40% on the propranolol day, which is lower than previously seen with the 0.5-µg/kg per min dose (6). This made fulfilment of IHS criteria a rather insensitive endpoint. The reason for the low number of migraines is probably the lower dose of GTN and early intake of rescue medication.
The results showed no sign of efficacy for propranolol in the GTN migraine model on any of several outcome parameters such as number with IHS migraine, immediate or delayed peak headache.
The extracranial arteries
Prior to GTN infusion the radial artery in healthy subjects showed a smaller diameter after 2 weeks of propranolol than after placebo. This has not previously been reported in vivo with direct measurement of the arterial diameter, though peripheral vasoconstriction is a well-known initial response to propranolol and other β-blockers (17). A similar numerical but insignificant reduction was seen in the superficial temporal artery. In migraine patients no significant vasoconstriction could be found in the radial artery or the superficial temporal artery, even though they were both numerically reduced.
During GTN infusion the radial and the superficial temporal arteries dilatated and slowly returned to baseline after termination of the infusion (see Fig. 3). The GTN infusion abolished any baseline difference that propranolol might have caused. No differences in response to GTN were found in either migraine or healthy subjects and the arteries were dilatated by GTN to the same extent after both placebo and propranolol (see Tables 4 and 5).
The intracranial arteries
The MCA did not show any baseline change of mean maximal blood flow velocity (V mean) after pretreatment with propranolol, in contrast to the constriction of the radial artery. In fact, a non-statistically significant numerical reduction in V mean was seen both in healthy subjects (left − 3.4%, right − 3.6%) and in migraine subjects (left − 7.1%, right − 7.1%). Since cerebral blood flow is unaffected by GTN (18) and by propranolol (19), a reduced velocity in MCA reflects vasodilatation of MCA.
During GTN infusion, V mean decreased in both MCAs without any difference between pretreatment with propranolol or placebo. Thus, propranolol probably does not constrict MCA.
Surprisingly, we found V mean in both MCAs to be highly significantly lower in migraine subjects than in healthy subjects. The difference was consistent and found at baseline as well as after GTN infusion. It persisted after pretreatment with propranolol. The lower velocity may represent a lower vascular tone in migraine patients. Other studies, some with higher numbers of patients and healthy subjects, have not found this difference (20, 21). However, subjects were not, as in the present study, systematically and to the same extent sex and age matched and no measurements of CO2 were performed, which is known to influence V mean (13, 22). We interpret the result with caution due to the fact that we measured TCD in only eight migraine subjects and eight healthy subjects.
Catecholamine
In contrast to previously reported increases in NA excretion in response to high-intensity light (9), we found a decrease. We could not reproduce the previously reported difference in catecholamine response to high-intensity light between migraine patients and healthy subjects, nor a normalization of response after treatment with propranolol (10). Our data therefore do not support previous suggestions that the prophylactic effect of propranolol may be due to a normalization of an abnormal catecholamine response in migraine patients. The relatively small number of subjects requires caution in interpretation of the results, especially in comparison between groups.
Possible mechanism of action of propranolol in migraine prophylaxis
The dose of propranolol used in the present study is effective in migraine prophylaxis (7, 23). However, in the present study we could not show a prophylactic effect on GTN-induced migraine. In clinical practice, the prophylactic effect of propranolol is not maximal after 2 weeks of treatment. We cannot rule out that this is in part the reason why we did not see any prophylactic efficacy and that a longer period of propranolol treatment before GTN challenge might have shown effect. A longer period with drug administration would, however, limit the advantage of a model, necessitate long-term toxicology, a longer study period and be time consuming. This could limit the use of the model. In a recent study with the same study design, we found that sodium valproate could prevent GTN-induced headache/migraine in migraine patients (8). The present study may therefore indicate that propranolol has a different mechanism of action in preventing migraine and that this effect is overruled or bypassed by the nitric oxide (NO) donor GTN. Propranolol is known to modulate vascular tone and studies suggest that propranolol may act by blocking an endothelial β-adrenergic receptor activating nitric oxide synthase (NOS) or by blocking an endothelial 5-HT2B receptor also coupled to NOS (24). Apparently the only common feature of the various β-adrenergic receptor blockers effective in migraine prophylaxis is that they are devoid of intrinsic activity (agonistic activity on the receptor) (7). If the migraine prophylactic effect of propranolol is to inhibit the formation of NO, then propranolol should not protect against headache induced by GTN, which represents an exogenous source of NO that is independent of NOS. Our finding in the present study, that propranolol has no prophylactic activity in GTN-induced headache and that propranolol-induced changes in vascular measurements were overruled by GTN, supports this hypothesis. We suggest that the prophylactic efficacy of propranolol has a different site of action than sodium valproate. In addition, our data suggest that the efficacy of future prophylactic drugs in the GTN model depends on their mechanism of action. There may also be different mechanisms involved in different patients, so new drugs may not be effective in all patients or all attacks depending on the causative mechanism. The ineffectiveness of a well-proven migraine prophylaxis in the present study limits the usefulness of the model, and must be considered in future testing of new migraine prophylactic drugs.
Footnotes
Acknowledgements
Laboratory technicians Kirsten Enghave and Bente Leisner are thanked for their skilful help in measuring the vessel response. Thanks to Anthony Gibson for providing statistical support. This study (RESB1023) was sponsored by GlaxoWellcome (now GlaxoSmithKline).
