Abstract
In this study the human glyceryltrinitrate (GTN) model of migraine was for the first time used to test the effect of a prophylactic drug. We chose to test valproate due to its well documented effect as a migraine prophylactic drug. Efficacy of this compound would support the usefulness of the model in prophylactic antimigraine drug development. Twelve patients with migraine without aura were included in a randomized double blind crossover study. Valproate 1000 mg or placebo was given daily, each for a minimum of 13 days. On the last treatment day of each arm a 20 min intravenous infusion of GTN (0.25 μg/kg/min) was given. Headache was registered for 12 h after the infusion and headache intensity was scored on a scale from 0 to 10. Fulfillment of IHS criteria was recorded for 24 h. The middle cerebral arteries were evaluated by transcranial Doppler and the diameter of the superficial temporal and radial arteries were measured with high frequency ultrasound. GTN evoked migraine fulfilling IHS criteria 1.1 in 6 patients after placebo and in 2 patients after valproate (P = 0.125). Including additionally 3 patients on placebo and 1 patient on valproate who felt they had suffered a migraine attack, but who had as associated symptoms only photophobia or phonophobia, a significant reduction in the number of patients with induced migraine after valproate was seen (P = 0.031). Median peak headache intensity was 1 (range 0-9) after valproate compared to 4.5 (range 0-8) after placebo (P = 0.120). Pretreatment with valproate as compared to placebo reduced the velocity in both middle cerebral arteries after GTN (left P = 0.021, right P = 0.031). No effect of valproate was seen in the diameter of the superficial temporal artery (P = 0.781) or the radial artery (P = 0.367) before or after GTN. The study indicates that a prophylactic effect of valproate may be demonstrated using the GTN human migraine model. Although, all headache parameters were reduced after valproate compared to placebo, only one parameter was statistically significantly reduced probably because of the small number of patients. The size of the effect was similar to that of valproate in clinical trials. The GTN model may therefore be a valid tool for testing new prophylactic antimigraine drugs.
Keywords
Introduction
Because of the spectacular success of the triptans, the major focus in migraine drug development has in recent years been on acute treatment. The need for prophylactic drugs has certainly been reduced by the triptans, but it is still very considerable, and no migraine specific prophylactic drugs have so far been developed. One of the problems in prophylactic drug development for migraine is that there is no good in vitro or in vivo experimental model with reasonable sensitivity and validity in predicting prophylactic antimigraine activity. Therefore, early and cost effective proof of concept studies in humans are important. Unfortunately, several factors make proof of concept studies rather long lasting and expensive. First of all, treatment periods must be between 3 and 5 months including run in phases, necessitating long-term toxicology studies (1). Secondly, large numbers of patients are necessary in order to get enough power.
A relevant human experimental migraine model, which proved to be sensitive and valid in predicting prophylactic antimigraine activity, would obviously be of value in the development of prophylactic antimigraine drugs. It has been shown that the nitric oxide (NO) donor glyceryltrinitrate (GTN) induces attacks in migraine sufferers that are indistinguishable from their spontaneous attacks (2). This model is by far the best studied experimental human migraine model (3–5). In the present study, we use a novel modification of the model adapted for the testing of prophylactic drugs. We pretreated patients with valproate or placebo in a double-blind crossover trial and, at the end of 2 weeks of pretreatment, the patients were challenged with an infusion of GTN in order to evaluate the effect of the pretreatment on headache/migraine response to GTN. The aim of the study was partly to gain methodological information about such a modification of the GTN model, and partly to validate this use of the model by testing the effect of a recognized prophylactic drug, sodium valproate. In clinical trials, the prophylactic effect of valproate is modest with approximately 50% of patients experiencing 50% reduction in attack frequency (6, 7). A similar efficacy of valproate in the GTN model would support the role of NO as a key molecule in migraine as well as the usefulness of this model in drug development.
Patients and methods
Patients
Twelve patients, who fulfilled the diagnostic criteria of the International Headache Society (IHS) for migraine without aura, were included in this double blind randomized placebo controlled two period crossover study. One patient withdrew informed consent before the first study day for personal reasons. This patient was replaced without breaking the randomization code. The patients were recruited between December 1998 and September 1999 from the outpatient headache clinic at Glostrup University Hospital, University of Copenhagen, Denmark (10 female, 2 male; mean age 36.3 years, range 23–62 years). Inclusion criteria were:
migraine without aura, according to the criteria of IHS 1.1 (8) with 1–6 attacks per month for at least 1 year;
age between 18 and 65 years;
body mass index within the range 19–29 kg/m2;
weight range female 45–90 kg, male 50–95 kg;
good general health determined by medical history, physical examination, ECG and urine and blood screening tests including test for hepatitis B and C.
Females of child-bearing potential could participate provided they had a negative pregnancy test and used reliable contraceptive.
Exclusion criteria included:
tension type headache more than 2 days a month;
known allergy to the drugs used;
blood donation within the previous month;
breastfeeding;
migraine prophylaxis within 2 month of study start;
previous proven inefficacy of sodium valproate prophylaxis;
alcohol or drug overuse (urine screen for drugs of abuse);
regular use of prescribed or over-the-counter medication except oral contraceptive pill and usual acute migraine treatment.
Study procedures
Patients were all scheduled to have two separate study days and randomized to receive sodium valproate 1000 mg (Deprakine®, Sanofi Winthrop) or identical placebo tablets as a single daily oral dose for a minimum of 13 days, prior to the study days (Fig. 1). The tablets were self-administrated by the patient before bedtime. The patient registered the date and time of tablet intake each day in a diary and attended a control visit after 7 days of medication (diary control, AE questioning, vital signs). The two study days were separated by at least 28 days in order to obtain a wash out period of a minimum of 14 days. Patients were rescheduled to another study day if they had taken any 5-HT1B/D agonists within 48 h, ergotamine or similar medication within 72 h, had experienced a migraine attack within the previous 48 h or had any headache prior to study procedures. Rescheduled patients continued medication with valproate or placebo until the study day (maximum another 6 days of medication). Alcohol was not permitted for 24 h prior to study procedures and until 48 h postdose. An alcohol breath test was performed on both study days. Patients were required to refrain from smoking while in the unit. Unscheduled prophylactic migraine drugs were not allowed during the study. Standard breakfast was provided prior to start of the study procedures, fluid was unrestricted. Caffeine intake was restricted in relation to the study days. A cannula (Venflon®) was inserted in the cubital vein for infusion and safety. On both study days the patients received an intravenous infusion with glyceryltrinitrate (GTN) 0.25 µg/kg/minute for 20 min in the cubital vein by a volume directed pump (IVAC P2000, Hampshire, UK). Glyceryltrinitrate (SAD 5 mg/ml) was diluted with saline to 50 ml solution of 10 µg/ml. Supine position was assumed 30 min prior to GTN infusion and until at least 3 h after the start of the infusion. The start of GTN infusion was designated time zero.

Study design, double blind crossover.
Headache parameters
Headache intensity was scored on a numerical verbal rating scale from 0 to 10, where 0 represents no headache, 1 represents a very mild headache, including a feeling of pressing or pulsation (prepain), 5 represents a moderate headache and 10 represents the worst possible headache (3). Headache characteristics (unilateral/bilateral, quality and aggravation by physical activity) and accompanying symptoms (nausea/vomiting, phono- and photo-phobia) were recorded in order to compare with the IHS diagnostic criteria for migraine without aura (8). Headache parameters and accompanying migraine symptoms were recorded in the clinic before study start and until 3 h after start of the infusion, by a physician in the clinic using a standardized questionnaire. Afterwards, the patient used a diary card at home to register headache until 12 h after infusion. Distinction was made between an immediate headache occurring during the 20 min infusion or within 40 min after and a delayed headache occurring from 60 min and up to 12 h after start of the infusion. Fulfillment of the IHS diagnostic criteria for migraine without aura was recorded up to 24 h after infusion. The patients could take their usual acute migraine medication any time after the headache provocation.
Haemodynamics
Mean maximal blood velocity in both the right and left middle cerebral artery was measured simultaneously by transcranial Doppler (TCD) (DWL, Multi Dop X) prior to the GTN infusion and at the time points 5, 10, 15, 20, 30, 45, 60, 120 and 180 min (for further description of procedures see References) (9–11). Diameters of the superficial temporal artery and the left radial artery were measured with ultrasound (Dermascan C, Cortex Technology, Hadsund, Denmark) (12) at the same time points as described above for TCD. The superficial temporal artery was measured on the patient's usual migraine headache side or on the hand dominant side if the headache usually was double sided or alternating in side. End-expiratory CO2 was measured simultaneously during all TCD measurements.
Vital signs were obtained prior to start of study procedures and followed until 3 h after start of the GTN infusion. A follow-up clinical examination, including vital signs, ECG, laboratory observations and evaluation of adverse events, was scheduled between 10 and 14 days after the last study day.
Approvals were obtained from The Danish Medicines Agency and The Scientific Ethical Committee for the County of Copenhagen. Signed, informed consent was obtained by each patient. The trial was conducted in accordance with the Declaration of Helsinki amended in Somerset West, South Africa 1996 and conformed to rules for Good Clinical Practice (GCP).
Statistical analysis
Due to the exploratory nature of the study, no formal sample size calculation was made. Target inclusion was completion of 12 migraine patients. This sample size would provide point estimates of the effect of valproate in this model and variability estimates which would assist in the design of future studies. Randomization was performed by GlaxoWellcome and the code was not known to the investigators until after the database was closed. Comparison of the number of patients experiencing headache/migraine attacks on the two study days was investigated with a sign test and differences in peak headache intensity were investigated with paired 2-tailed Wilcoxon test. The vascular changes were analysed over time with
Results
Headache and migraine
Twelve migraine patients completed the study. Sodium valproate or placebo was administered for a minimum of 13 days and a maximum of 17 days prior to GTN infusion. The GTN infusion was successful in producing headache and 11 patients developed both immediate and delayed headache on at least the placebo study day. The response to GTN was identical on the valproate pretreatment and the placebo pretreatment day with regard to headache and migraine in six patients (Fig. 2). Of these, one did not develop headache at all. Two patients took rescue migraine medication at the onset of delayed headache, where they reported start of usual migraine, but did not fulfil IHS migraine criteria on either study day. Two patients reported migraine that fulfilled IHS criteria 1.1. on both study days. One patient reported usual migraine on both study days that fulfilled IHS criteria 1.1 except having only photophobia and not phonophobia before taking rescue medication. Of the remaining 6 patients, 4 fulfilled IHS criteria 1.1 after placebo but not after valproate, and 2 patients reported migraine only after placebo and not after valproate, but did not quite fulfil IHS 1.1 criteria since they had either phonophobia or photophobia and not both.

GTN induced migraine attacks. ▪ IHS 1.1 migraine,  IHS 1.1 migraine except only photo- or phono-phobia, □ Early rescue both days,
IHS 1.1 migraine except only photo- or phono-phobia, □ Early rescue both days,  No headache.
No headache.
After valproate a numerical reduction compared to placebo was seen in number of patients with GTN evoked migraine fulfilling IHS criteria 1.1 (2 vs 6 of 12; P = 0.125; Fig. 2). A significant reduction was found in the number of patients reporting migraine fulfilling IHS criteria 1.1 or fulfilling 1.1 except only having photophobia or phonophobia before taking rescue medication. (3 vs 9 of 12; P = 0.031). All patients reporting migraine or fulfilling IHS criteria after valproate did the same after placebo. Peak headache intensity showed a nonsignificant reduction after valproate compared to placebo (Wilcoxon test): in the first 60 min (median 1, range 0–4 vs median 1, range 0–5; P = 0.084), in the delayed phase 1–12 h (median 1, range 0–9 vs median 4.5, range 0–8; P = 0.120), and overall 0–12 h (median 3, range 0–9 vs median 5, range 0–8; P = 0.138). To illustrate the time course of headache intensity mean headache is used in Fig. 3.
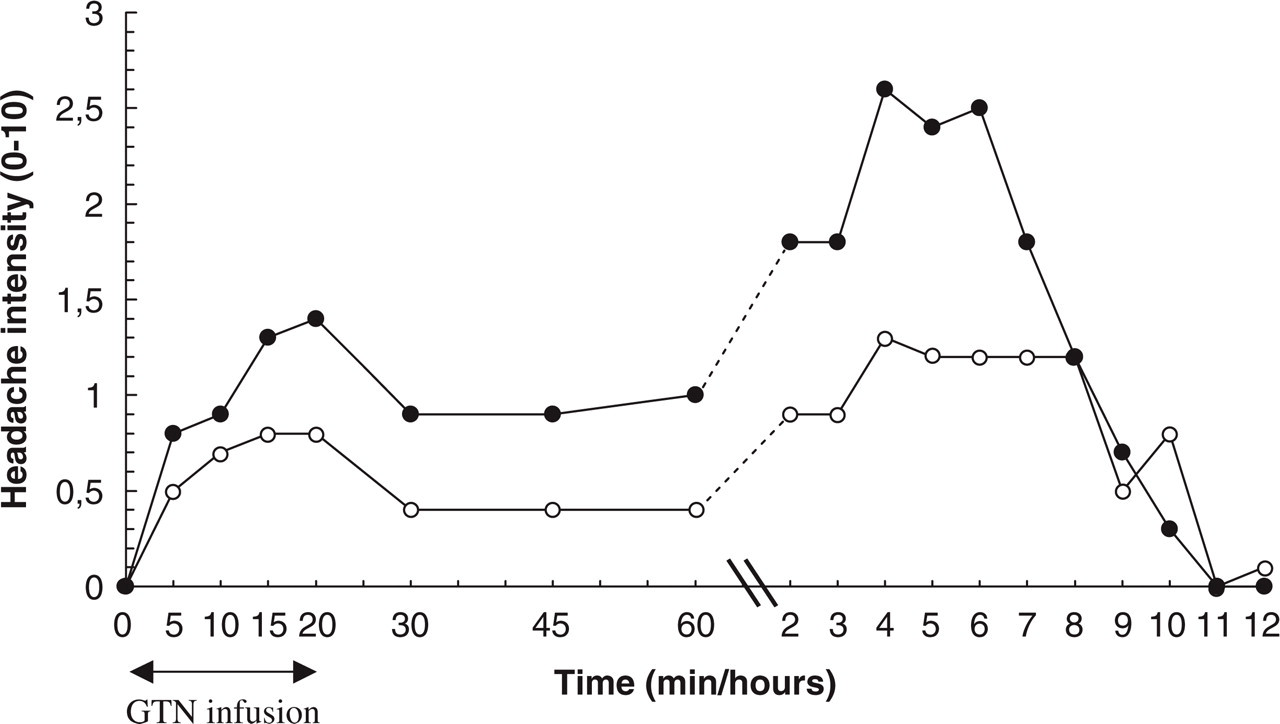
Mean headache intensity over time, immediate (0–60 min) and delayed (up to 12 h). • placebo, ○ valproate.
Vascular measurements
Extra-cranial arteries
Before GTN infusion, there were no baseline differences between valproate and placebo in the mean diameter in both the radial artery (2.15 mm vs 2.16 mm; P = 0.895) and the superficial temporal artery (1.03 mm vs 1.10 mm; P = 0.245). Both arteries dilated during GTN infusion and slowly returned towards baseline after end of the infusion (Fig 4c,d). No differences in response to GTN were found between pretreatment with valproate compared to placebo. During the GTN infusion the mean maximal diameter of the radial artery was 2.94 mm after valproate pretreatment compared to 2.88 mm after placebo pretreatment (P = 0.367), and of the superficial temporal artery it was 1.54 mm compared to 1.52 mm (P = 0.781). Analysis of the weighted mean over the 3-h period after GTN infusion, did not show any difference of GTN's dilating properties between pretreatment with valproate compared to placebo in the radial artery (2.47 mm vs 2.43 mm; P = 0.629) or the superficial temporal artery (1.23 mm vs 1.19 mm; P = 0.522).

Vascular measurements. Mean maximal flow velocity in left (a) and right (b) middle cerebral artery. Mean diameter of radial artery (c) and temporal superficial artery (d). Values shown as mean ± SEM. • placebo, ○ valproate.
Cerebral arteries
Mean maximal blood flow velocities of the left and right middle cerebral artery (MCA) were measured simultaneously. At baseline a small, nonsignificant reduction after pretreatment with valproate was observed on both sides (Left 76.75 cm/s vs 79.75 cm/s (−3.8%); P = 0.319. Right 71.92 cm/s vs 77.08 cm/s (−6.7%); P = 0.122). The GTN infusion reduced the MCA velocity on both study days (peak reduction after pretreatment with valproate: left −28.0%, right −28.0%; after placebo: left −26.3%, right −27.8%). After GTN the reduced MCA velocity was lower after pretreatment with valproate than after placebo (Left 55.29 cm/s vs 58.79 cm/s (−6.0%); P = 0.021. Right 51.75 cm/s vs 55.66 cm/s (−7.0%); P = 0.031) (Fig. 4a,b).
No changes in end-expiratory CO2 were seen during measurements of the vessels.
Discussion
It is important to emphasize that this study aimed at validating the GTN model and to evaluate methodological aspects of importance in the future use of the model to test novel prophylactic agents in migraine. It was not powered to detect effects of the size normally seen with current prophylactic migraine drugs with a high degree of certainty.
The optimal dose of glyceryltrinitrate (GTN)
We have previously shown that GTN in a dose of 0.50 µg/kg/minute induced migraine fulfilling IHS criteria in 80% of migraine sufferers (2). In the present study, we used a dose of 0.25 µg/kg/minute, which resulted in 50% of the patients developing migraine fulfilling IHS 1.1 on the placebo day. However, in 75% of the patients, GTN induced headaches fulfilling the migraine criteria except for associated phono or photophobia and reported by the patients as migraine. Rescue medication was taken by 83% of the patients on the placebo day, and by 58% after valproate possibly reducing the number of patients fulfilling all of the IHS criteria. We selected a smaller GTN dose, because it might be easier to demonstrate a prophylactic migraine action, if the patient was not flooded with excess amounts of GTN and thereby nitric oxide. High doses of GTN could theoretically over stimulate the patients leading to migraine despite prophylactic therapy. The optimal dose of GTN should be high enough to provoke migraine without overruling the prophylactic effect of the test drug and without producing serious hypotensive episodes. In the present study, valproate and GTN were well tolerated by all patients and no hypotensive episodes were observed. The properties of the test drug must be taken into account when deciding the GTN dose. In the present study of valproate, the GTN dose of 0.25 µg/kg/minute seemed adequate.
Headache results
All the headache parameters were reduced after valproate compared to placebo. The size of the effect was similar to that of valproate in spontaneous migraine. The primary endpoint used in this study, IHS criteria 1.1 for migraine without aura, is a very hard endpoint since the patients were allowed to treat their headache/migraine before fulfilling IHS criteria 1.1. This may be the reason why the number of patients with IHS migraine after valproate was not significantly reduced. However, a significant reduction was seen in patients reporting migraine and fulfilling IHS 1.1, with the exception of only having phono or photophobia and not both before taking rescue medication.
The reduction in peak headache intensity failed to be significant in the present study. This can be due to the previously mentioned use of rescue medication and the small number of patients. Migraine attacks, especially headache intensity, vary in severity and are likely to be influenced by intake of rescue medication. This difference in attack severity and the use of rescue medication can blur the overall headache results in small studies like the present. The two patients that on both study days experienced migraine fulfilling IHS criteria, and therefore had no prophylactic efficacy of valproate, treated the induced migraine late on the valproate pretreatment study day, leading to a higher peak headache intensity. In the 10 patients who did not have migraine on both study days, a significant reduction in peak headache intensity took place (median 5, range 0–8 vs median 1.5 range 0–5; P = 0.021).
Overall, the results of the present study after pretreatment with valproate were all numerically positive and roughly of the same magnitude as with spontaneous migraine attacks. The results therefore support the validation and utility of this human migraine model for use in prophylactic studies. Valproate has in open studies been reported to have acute effect on migraine attack (13). However, no large randomized trial has yet confirmed this. In contrast, an evaluation of valproate against prochlorperazine in the acute treatment of migraine showed that valproate was less efficacious (14). In the present study valproate was administered up till the study day and not after GTN. The effect is almost certainly prophylactic rather than acute.
Arterial responses
Valproate is primarily an antiepileptic drug and the prophylactic efficacy in migraine has been discovered fortuitously (15). There is no evidence in favour of any vascular effects of the drug, but this question has not been studied in any depth. In the present study, we took the unique opportunity to study the effect of valproate on the middle cerebral artery, the superficial temporal artery and the radial artery in a double blind fashion. No baseline difference was detected. The response to GTN was similar to what we previously have seen (11). However, we found that pretreatment with valproate enhanced the GTN induced decrease in velocity in the middle cerebral artery in contrast to the superficial temporal and radial artery response, where no difference was found. This was observed in both MCA’s, which makes it unlikely that it is an accidental finding (Fig. 4). GTN has previously been shown not to change rCBF (16). Therefore, a change in flow velocity is due to a change in artery diameter when GTN is given alone (placebo day), whereas valproate has been shown to reduce global CBF by 14.9% (17). The larger GTN induced decrease in velocity after valproate can be a result of an additional decrease in rCBF and/or an enhanced dilatation of MCA. Even though we found no significant baseline difference in the MCA's velocities after valproate, it appears that there is a nonsignificant reduction already at baseline. A decrease in CBF by valproate could explain the velocity difference we found after the GTN infusion. We evaluate the finding with caution and stress that the data does not determine whether, besides reducing CBF, valproate diminishes the central vessel's ability to resist/withstand the dilating effects of GTN.
Prophylactic properties of valproate
The mechanism of valproate in migraine prophylaxis is unknown. Involvement of gamma-aminobutyric acid (GABA) regulation, with elevation of GABA as well as reduction of central trigeminal activation has been suggested (18). The results of the present study suggest involvement of nitric oxide (19). To our knowledge this is the first work in humans where valproate has been succeeded by a nitric oxide donor. In experimental models of induced seizure some studies have suggested involvement of NO, but the results are conflicting (20). In a seizure model in mice the NO donor molsidomine enhanced the protective activity of valproates against seizure. A nitric oxide synthase inhibitor could not reverse this effect and molsidomine had no effect alone on seizures. Molsidomine elevated free plasma valproate. This could explain the enhanced effect of valproate (20). In the present study we found a tendency toward less immediate headache after infusion of GTN when comparing valproate to placebo (P = 0.084), see Fig. 3. However, the induced migraine attacks occurred delayed with respect to the GTN infusion, were indistinguishable from spontaneous attacks and, due to GTN's short half life in man, occurred at a time without any significant GTN present.
Methodological considerations
The present study demonstrates that it is possible to use the GTN model to test prophylactic migraine drugs and that it has a reasonable power to detect clinically relevant effects with a relatively low number of subjects. In the crossover design used in the present study, each patient was stimulated with GTN twice, once after pretreatment with placebo and once after valproate. The study was not easy to undertake, because few patients consent to have migraine attacks induced by GTN. If a panel of paid volunteer migraine patients were collected, who were willing to put up with such hardship for a reasonable payment, then perhaps future studies would be facilitated. One major concern for patients participating in migraine provocation is the use of abortive medication in the event of migraine. In the present study patients were allowed any kind of rescue at the time they wanted, since we did not find it ethically acceptable to provoke headache without the possibility of abortive medication. Participating in a study where the outcome is headache and migraine, the patients had an expectation of getting headache and migraine. This proved to make the patients very attentive toward a possible onset of migraine and may be why many patients took abortive medication very early and before they fulfilled IHS migraine criteria. In future studies patients could be asked to refrain from abortive medication until they fulfilled migraine criteria or patient reported migraine rather than IHS migraine, could be chosen as an outcome parameter.
The study demonstrates that it is probably not wise to use fulfillment of the IHS criteria 1.1 (migraine without aura) as the only endpoint. It may be better to accept fulfillment of IHS migraine criteria together with patient reported migraine if rescue medication is taken. In a study with parallel groups, strict endpoints are important to have valid data comparison. However, in a crossover study, where patients are given the same challenge with GTN on two separate study days, patient reported migraine could be an acceptable outcome parameter, because patients presumably apply the same criteria for migraine on the two study days. This makes patient reported migraine feasible to use as an endpoint in crossover trials whereas in a parallel group design this would be less acceptable.
Intuitively, the area under the headache curve or peak-headache seems to be more powerful parameters than IHS migraine, because they are quantitative and can be measured in all patients. We discovered certain problems with these parameters, the most important being that patients take rescue medication at variable times. If a patient on placebo gets severe headache/migraine after GTN, the patient may take rescue medication very early and abort the headache. On the active arm the same patient may get a much less pronounced headache not requiring rescue medication. That headache may then sum up over several hours to a higher area under the headache curve. Similarly, peak headache may be influenced by the time of intake of effective rescue medication. In the present study peak headache showed a numerical but nonsignificant efficacy of valproate.
In clinical practice the onset of prophylactic migraine activity after valproate may not occur until after one month of treatment and it may take more than one month to get the maximum response. Ideally, we should have pretreated our patients for 2 months before challenging with GTN. Such a long challenge would, however, be difficult in a crossover design, and might also be considered ethically unacceptable because of the side-effects of valproate and/or the long lasting placebo arm. A long trial period would also eliminate many of the advantages of using a migraine model. At the other extreme, we might have given just one dose of valproate and placebo in a ‘microprophylactic’ experiment. The chosen treatment time of 2 weeks represents a compromise between these two extremes and, as mentioned above, proved to be effective.
Advantages of using the present model
The major advantage of the present model is that it allows the testing of drugs with only 2 weeks toxicology. Thus, a potential prophylactic migraine agent can be tested at a very early stage of development. It is also a big advantage that sufficient power seems to be obtainable by a smaller number of patients than in a traditional prophylactic trial. Based on the present study results a total of 20–30 patients are necessary. Furthermore, in the present experimental paradigm extensive testing of safety parameters, blood values and effects of drugs on the cerebral and extracerebral circulation can be tested at the same time as data on efficacy. Clearly, efficacy in the present model is no proof of efficacy in spontaneous migraine, but it implies a high likelihood of clinical efficacy. Conversely, lack of efficacy is no proof that the drug cannot be effective in clinical migraine. The negative predictive value of this experimental paradigm is probably smaller than the positive predictive value. This is because spontaneous migraine can probably be induced via a number of different mechanisms, whereas GTN always induces migraine by the same mechanism. Only future studies can demonstrate whether all prophylactic migraine drugs work in the present model, or whether only drugs with a CNS site of action are effective. It also remains to be shown whether the present model can be used for doze finding in prophylactic studies and how such a doze finding study should best be designed.
Conclusion
This is the first study in which the GTN model of migraine has been modified to test the efficacy of a known prophylactic drug in migraine. GTN induced fewer migraine attacks when patients were pretreated with valproate than with placebo. Accepting less than complete fulfillment of IHS migraine criteria the effect was statistically significant. This indicates that the model is able to show efficacy of valproate and thus may be valid for testing new prophylactic migraine drugs. The sensitivity might be increased with a minor increase in subject size. At the same time, the vascular effects of prophylactic drugs may be studied noninvasively and safety data can easily be collected. More studies with other prophylactic drugs are needed and multiple variations of the present model should be tested in future studies.
Footnotes
Acknowledgements
Laboratory technicians Kirsten Enghave and Bente Leisner are thanked for their skilful help in measuring the vessel response. Thanks to Anthony Gibson for providing statistical support. The study was sponsored by GlaxoWellcome (now GlaxoSmithKline), RESB 1027.
