Abstract
Botulinum toxin A has been suggested to be effective in the prophylactic treatment of migraine. However, only very few randomized, double-blind, placebo-controlled studies are available. We designed such a study with a specific focus on different injection sites. Sixty patients with a migraine according to the criteria of the International Headache Society were randomly assigned to receive either placebo in the frontal and neck muscles, or to receive 16 U botulinum toxin A in the frontal muscles and placebo in the neck muscles, or to receive in total 100 U botulinum toxin A in the frontal and neck muscles. The observation period was 3 months. In both treatment groups, 30% of patients showed a reduction of migraine frequency in month 3 by at least 50% compared with baseline, in the placebo group 25% of the patients showed such a reduction (P = 0.921). There were no significant differences between the three study groups with respect to reduction of migraine frequency, number of days with migraine, and the number of total single doses to treat a migraine attack. In the post hoc analysis, the reduction of all accompanying symptoms was significantly higher in the 16 U treatment group compared with the placebo group. In the 100 U treatment group significantly more adverse events occurred compared with the placebo group. All adverse events were mild and transient. Our study did not show any efficacy of botulinum toxin A in the prophylactic treatment of migraine. Only accompanying symptoms were significantly reduced in the 16 U but not in the 100 U treatment group. Future studies should focus on the efficacy of botulinum toxin A in specific subgroups of patients, on the efficacy of repetitive injections, and on other injection sites.
Introduction
Botulinum toxin A has been suggested to be effective in the prophylactic treatment of different types of headache. However, conflicting results have been reported to date (1–3). For the prophylactic treatment of migraine, one placebo-controlled and well-designed study has been published showing the efficacy of botulinum toxin A in those patients receiving a small dose (25 U) but not a larger dose (75 U) of Botox® in the pericranial muscles (4). Two smaller placebo-controlled studies, not yet published, showed a significant decrease of migraine intensity but not of migraine frequency (5) and a decrease of migraine frequency (6) by botulinum toxin A 12 weeks after injection (5).
In addition, some open retrospective studies have been published showing a positive effect of botulinum toxin A on migraine intensity and frequency using different injection sites and different doses of botulinum toxin A (7–10). Some of these studies, however, had a small sample size, no standardized injection procedures, no statistical analysis, or no full description of the treatment procedure (2), and thus do not give further scientific evidence about the efficacy of botulinum toxin A.
We therefore performed another randomized, double-blind, placebo-controlled, single-centre, parallel group study on the efficacy of different doses of botulinum toxin A (Botox®) in the prophylaxis of migraine with a specific focus on different injection sites. The study protocol was designed according to the recommendations of the International Headache Society (IHS) (11).
Methods
Patient selection
We enrolled 60 patients with migraine without or with typical aura according to the criteria of the IHS (12). Patients had to be between 18 and 65 years of age and to suffer from migraine with an average frequency of two to eight attacks per month in the preceding 3 months. The diagnosis of migraine had to be established for at least 1 year with an onset before the age of 40. Other types of headache, in particular tension-type headache, were allowed up to 10 days per month. Patients had to be able to distinguish between the different headache types. Exclusion criteria were pregnancy or lactating women, other types of migraine except migraine without aura and migraine with typical aura, any type of dystonia, any neuromuscular disease, any type of substance addiction including drug-induced headache, treatment with drugs affecting the neuromuscular junction, and changes of drug treatment with a possible migraine prophylactic efficacy within the last 3 months. After giving written informed consent, the patients were asked to keep a headache diary and to return to the clinic after a baseline of 4 weeks. The study was approved by the Ethics Committee of the Faculty of Medicine, University of Münster, Germany.
Treatment procedure
When the baseline diary confirmed the inclusion criteria, the patients were allocated to one of three treatment groups by a computer-generated randomization list. One investigator (J.V-H.), who was not involved in the remainder of the study procedure, received a sealed envelope with the randomization number. The envelope contained the respective treatment group and was sealed again after preparing the syringes for injection. The patients were treated with either 100 U botulinum toxin A in the frontal and neck muscles, or with 16 U botulinum toxin A in the frontal muscles and placebo (0.9% NaCl) in the neck muscles, or with placebo in all muscles. The doses of botulinum toxin A for the different muscles in the different treatment groups are given in Table 1. Patients were then asked to keep a diary and to return to the clinic after another 3 months. In the diary, the patients had to register the days with migraine and the intensity of migraine, the days with other headaches, accompanying symptoms, acute medication for treating migraine attacks, and any adverse events.
Doses and injection sites of botulinum toxin A in the different treatment groups (the dose is given for one side, maximum amount of botulinum toxin A 100 U Botox®)
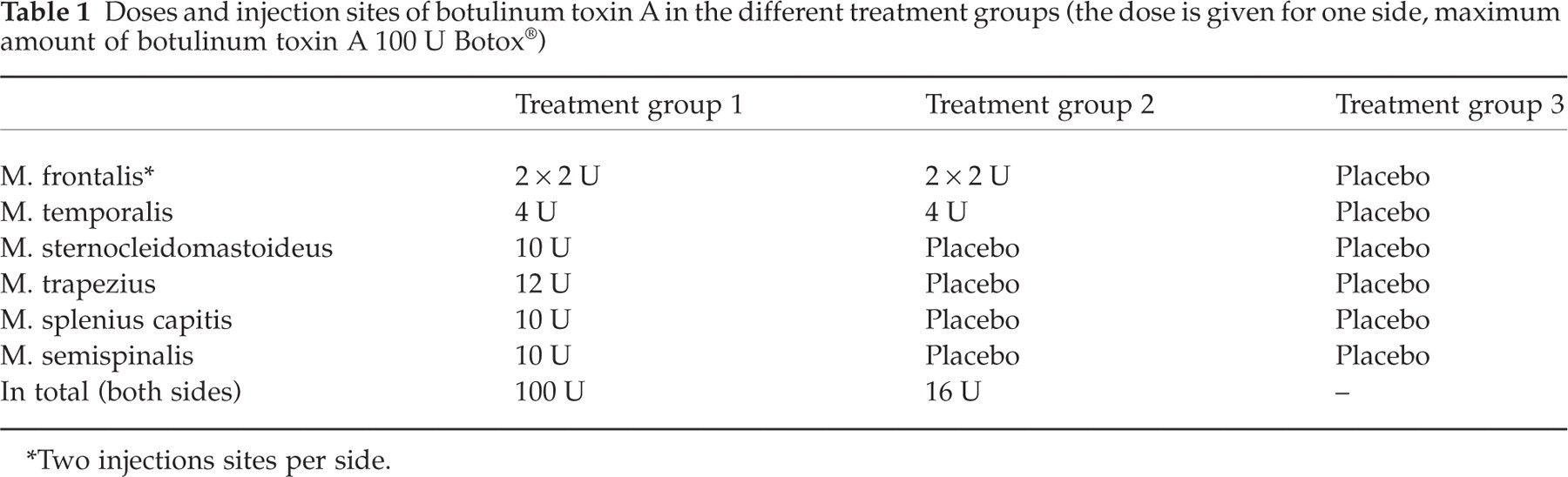
∗Two injections sites per side.
After the baseline month and at the end of the study period, patients filled in the German version of Beck's Depression Inventory (BDI; maximum score 63) and the German version of the Headache Disability Inventory (HDI; maximum score 100) (13).
Statistical analysis
The primary efficacy parameter was the number of patients with a reduction of migraine frequency of at least 50% in month 3 compared with the baseline month. Secondary efficacy parameters were the reduction of migraine frequency, the reduction of days with migraine headache, the reduction of days with moderate or severe migraine headache, the reduction of accompanying symptoms (sum of the presence of photophobia, phonophobia, nausea, and vomiting in each individual patient), the reduction of the total single doses of acute anti-migraine drugs, the improvement of the HDI and the BDI score, and the total number of possibly or probably drug-related adverse events.
All analyses were performed on data from the intent-to-treat population which was identical to the per-protocol population. The statistical power was strong enough to detect differences of > 30% between placebo and verum for the primary efficacy parameter. We used non-parametric tests with the χ2 test for qualitative data (Fisher's exact test if applicable) and the Kruskal–Wallis test for quantitative data (Mann–Whitney U-test as post hoc test). The significance level was set at P = 0.05.
Results
All 60 patients finally enrolled in the study could be followed up for the complete study period of 3 months. All patients were Caucasian. Eighteen patients were on prophylactic migraine medication (10 β-blockers, five flunarizine, two valproic acid, one amitriptyline). The demographic data of the three different treatment groups are presented in Table 2. There were no significant differences between these groups except for significantly more patients with migraine with aura in group 2 compared with group 1. The rate of patients with at least 50% reduction of migraine frequency (primary efficacy parameter) was 30% in the group receiving 100 U, 30% in the group receiving 16 U, and 25% in the group receiving placebo (P = 0.921).
Demographic and clinical data of the three different treatment groups presented as arithmetic mean with simple standard deviation in parentheses or as percentage

Statistical comparison between the three treatment groups by Kruskal–Wallis test.
The data of the secondary efficacy parameter are presented in Table 3. There were no significant differences between the three treatment groups with respect to the reduction of migraine frequency, number of days with migraine, number of days with moderate or severe migraine, or number of acute drugs for the treatment of migraine attacks. The only significant difference that could be observed was in the sum score of all accompanying symptoms. In the group receiving 16 U botulinum toxin A, but not in the group receiving 100 U, the accompanying symptoms were significantly reduced by 29% in month 3 compared with a reduction of 5% in the group receiving placebo (P = 0.048; post hoc test).
Data of the secondary efficacy parameters in the three treatment groups presented as arithmetic mean with simple standard deviation in parentheses

∗P = 0.048 for the post hoc analysis of the relative reduction comparing the 16 U treatment group and the placebo group.
The significance levels are given for the comparison between the three treatment groups regarding the absolute reduction in month 3 compared with baseline (Kruskal–Wallis test).
The BDI and the HDI scores are given in Table 4. No significant differences between the three treatment groups could be observed for these efficacy parameters.
Data of the Beck's Depression Inventory (BDI) and the Headache Disability Inventory (HDI) scores at baseline and after month 3 presented as arithmetic mean with simple standard deviation in parentheses

The significance levels are given for the comparison between the three treatment groups regarding the absolute reduction after month 3 compared with baseline (Kruskal–Wallis test).
The adverse events which were judged by the investigators to be possibly or probably related to the study drug are presented in Table 5. The total number of adverse events was significantly higher in the group receiving 100 U compared with the group receiving placebo (P < 0.05). All adverse events were mild and transient. There was no serious adverse event.
Adverse events possibly or probably related to the study drug as judged by the investigators (absolute figures)

Discussion
Our study could not demonstrate any significant efficacy of botulinum toxin A on migraine frequency or severity or on the psychosocial impact of migraine. This is in concordance with the findings of two previous placebo-controlled studies. In the study of Brin et al. (5), a significant improvement of migraine intensity but not of migraine frequency by botulinum toxin A could be observed. In the study of Silberstein et al. (4), botulinum toxin A showed a significant impact on migraine frequency in a low-dose (25 U) but not in a high-dose (75 U) treatment group. We cannot explain the different results of the studies concerning the low-dose treatment group. It might be that a purely frontal injection of 16 U botulinum toxin A, as in our study, is not effective, whereas a higher dose of 25 U, as in the study of Silberstein et al. (4), is helpful. Interestingly, it was also the low-dose treatment group in which we were able to observe our only significant finding: an improvement of the accompanying symptoms in this treatment group compared with the placebo group. It seems that the total dosage of botulinum toxin A, at least in the ranges used in the published studies (up to 100 U Botox®), is less important, and that other parameters such as injection sites and patient selection are more important in order to yield a positive result from such a study. In the low-dose treatment, pleasant cosmetic effects on the facial muscles without unpleasant effects on the neck muscles cannot be ruled out. This effect might undermine the blinding of the study medication and might be the reason why in this treatment group the accompanying symptoms, which are highly subjective, were lower than in the other groups.
Our data clearly contradict the findings in open case series showing a good efficacy of botulinum toxin A in the prophylactic treatment of migraine (7–9). We interpret these differences as a result of the different study designs. In most of the open case series, individual injection sites and doses were used. The placebo effect, which is very high in open studies on migraine, also has to be considered. A similar discrepancy between placebo-controlled and open designs can be observed in the studies on the prophylactic treatment of tension-type headache with botulinum toxin A. In the double-blind and placebo-controlled studies, no efficacy could be demonstrated for botulinum toxin A (14–16), whereas in the open case series sometimes impressive reductions of tension-type headache were observed (17–19).
We enrolled consecutive migraine patients without any selection bias except the setting of a supraregional specialized headache clinic. In particular, we did not focus on migraine patients known to be refractory to prophylactic treatment. The demographic data of our patients confirm that we enrolled a typical migraine sample in headache clinics. The placebo rate of 25% for the primary efficacy parameter is also typical of studies on migraine treatment (11). Therefore, we believe that our study reflects the typical situation of other published migraine prophylaxis studies.
The number of patients with concomitant tension-type headache was not significantly different between the three treatment groups. In the low-dose treatment group, however, fewer patients had tension-type headache than in the placebo group. In order to exclude any bias caused by the different numbers of patients with concomitant tension-type headache, we compared the migraine patients with and without tension-type headache at baseline and did not observe any significant difference with respect to migraine parameters such as accompanying symptoms, duration, and frequency.
The adverse events reported by the patients and judged by the investigators as possibly or probably related to the study drug were all mild and transient in nature. We observed the typical adverse events known from other studies on botulinum toxin A, such as ptosis and neck weakness. Botulinum toxin A appeared to be a safe drug in the doses applied in our study.
Our study does not provide any final evidence against the efficacy of botulinum toxin in the prophylactic treatment of migraine. Its statistical power was too weak to detect minor differences (i.e. up to 30% for the primary efficacy parameter) between placebo and botulinum toxin. The optimal dose and the optimal injection sites resulting in possible efficacy remain to be determined. In the study by Silberstein et al. (4), only frontal injection sites were used. We added neck injection sites, which did not improve the results. It might be that higher doses injected only in frontal muscles are more effective. However, we observed some typical side-effects in the frontal muscles even after 16 U and suggest care with higher doses. In addition, frontal muscle weakness can result in cosmetic changes which might have an impact on migraine by indirect psychosocial mechanisms. Furthermore, our study does not indicate that other types of botulinum toxin are ineffective in headache treatment. Currently, studies with botulinum toxin B are underway to elucidate the efficacy of this botulinum toxin subtype in migraine and tension-type headache.
In conclusion, our study does not support the hypothesis that botulinum toxin A is effective in the prophylactic treatment of migraine. However, it might be that other injection sites and other doses of botulinum toxin A are effective in a defined subgroup of patients. Furthermore, our study gives some evidence that a low dose of botulinum toxin A might have a mild effect on the accompanying symptoms of migraine. Further studies should elucidate these specific mechanisms of botulinum toxin A rather than focus on the reduction of migraine frequency. Three placebo-controlled and double-blind studies have now shown that there is no major impact of botulinum toxin A on migraine attack frequency. However, the future role of botulinum toxin A in the treatment of migraine remains to be determined.
Footnotes
Acknowledgements
The study was initiated by the investigators and supported by an unrestricted grant of Pharm Allergan, Germany.
