Abstract
Our aim was to evaluate the safety and efficacy of botulinum toxin type A (BoNTA; BOTOX®) for prophylaxis of episodic migraine. In this double-blind, placebo-controlled study, patients were randomized to 225, 150 or 75 U of BoNTA or placebo after a 30-day placebo run-in for three 90-day treatment cycles. The primary efficacy end-point was the mean reduction from baseline in the frequency of migraine episodes at day 180 in the placebo non-responder stratum. All groups (N = 495) improved, with no significant differences. At day 180, the frequency of migraine episodes was reduced from baseline means of 4.3, 4.7, 4.7 and 4.4 by 1.6, 1.7, 1.5 and 1.4 for BoNTA 225 U, 150 U and 75 U and placebo, respectively. The primary end-point was not met. Treatment-related adverse events were transient and mild to moderate. BoNTA treatment was safe and well tolerated but did not result in significantly greater improvement than placebo in this study. Several factors may have confounded the results.
Introduction
Migraine is a primary headache disorder (1) affecting approximately 18% of women, 7% of men and 4% of children worldwide (2–5) and is one of the world's leading causes of disability (6). Migraineurs commonly experience restricted activity, decreased productivity and missed work or school days (2, 3, 5, 7). Migraine-related disability may lead to reduced health-related quality of life and increased depression (8, 9). The worldwide burden of migraine to society is substantial, with upwards of billions of dollars spent in direct costs (utilization of healthcare resources) and indirect costs related to disability, reduced productivity and missed work days (5, 7, 10–13).
Prophylactic treatment of migraine is recommended to reduce the frequency of migraine episodes and reliance on analgesic headache pain medications (4, 10, 14). Effective migraine prophylaxis may also decrease healthcare resource utilization, patients' disability and the risk of disease progression. However, the few available and/or approved oral drug treatments are often associated with limited efficacy and intolerable side-effects (1, 4, 15–17). Thus, there is an unmet therapeutic need for effective, tolerable migraine prophylactic drug treatments.
Botulinum toxin type A (BoNTA; BOTOX®, Allergan, Inc., Irvine, CA, USA) is a purified protein that blocks the presynaptic exocytosis of acetylcholine at the neuromuscular junction inhibiting muscle contractions (18). Focally administered intramuscular injections of BoNTA have provided safe and effective therapeutic treatment of disorders characterized by excessive muscle contractions such as cervical dystonia (19) and poststroke-related spasticity (20). Preclinical in vitro and in vivo data have shown that BoNTA also blocks the release of nociceptive mediators such as substance P, glutamate and calcitonin gene-related peptide from presynaptic peripheral neurons (18, 21–23) and on that basis may be effective in the treatment of pain disorders including headache (21, 24, 25). The results of several large, randomized, double-blind, placebo-controlled clinical trials conducted in over 1000 headache patients have demonstrated that BoNTA significantly reduces the frequency of headache episodes from baseline compared with placebo in the prophylactic treatment of chronic daily headache (CDH) (26–28). Results of randomized, double-blind, placebo-controlled clinical trials evaluating prophylactic treatment of episodic migraine have been mixed; one trial demonstrated significant reductions in frequency of episodes (29), but another failed to show a separation from placebo in migraine frequency (30). Minimal side-effects and low discontinuation rates due to adverse events were observed in all of these studies.
The current study was a Phase 2 clinical trial that was part of a series of exploratory trials designed to investigate the safety and therapeutic effect of BoNTA as prophylactic treatment for headache disorders. Specific goals were to identify a responsive patient population, a safe and efficacious dose, and a well-defined treatment regimen. The current study focused on an episodic migraine population who were not receiving concurrent migraine prophylaxis treatment; the other exploratory studies in the series are reported elsewhere (31, 32).
Methods
Objective
The objective of this study was to evaluate the safety and efficacy of multiple treatments of BoNTA compared with placebo for the prophylactic treatment of migraine headaches in an episodic-migraine population.
Study design
This was a randomized, double-blind, placebo-controlled, parallel-group, multicentre clinical study of multiple treatments of BoNTA in the management of patients with International Headache Society (IHS)-defined migraine headache, with or without aura. The study was conducted at 37 study centres in nine countries (one centre in Belgium, six in Croatia, one in Denmark, three in Finland, six in France, five in Germany, two in Norway, one in Switzerland and 12 in the UK) with an overall maximal duration of participation for each patient of 11 months, including a 30-day baseline period, followed by a 30-day placebo run-in period and a 9-month treatment period encompassing three treatment cycles (Fig. 1). Prior to study initiation, investigators obtained Independent Ethics Committee approval. The study was conducted in compliance with the ethical principles that have their origins in the Declaration of Helsinki regarding biomedical research on human subjects and with informed consent regulations.

Study design.
Participant characteristics
Inclusion criteria
Eligible patients were men or women aged 18–65 years who suffered from an average of at least three moderate to severe untreated migraine episodes per month [defined by IHS 1988 International Classification of Headache Disorders (ICHD-I)] or at least three treated migraine episodes of any severity per month. Patients had to have ≤15 headache days per month as confirmed by a headache diary during the baseline period. Patients' migraine episodes must have occurred for at least 1 year prior to enrolment and be first diagnosed before age 50 years. Patients had to have a stable medical condition and acceptable blood haematology and chemistry results, and be willing and able to give written informed consent. Patients were required to discontinue headache prophylactic medications for at least 3 months immediately prior to the initiation of the baseline period and had to be willing and able to stay on current medications (other than headache prophylaxis) during the course of the study, as well as comply with study instructions including the use of a daily electronic telephone diary capture system.
Exclusion criteria
Patients were excluded if they had any medical condition or used any agent that may have put them at risk with exposure to BoNTA (e.g. neuromuscular disorders or agents that might interfere with neuromuscular function), or if they had an infection or skin problem at any of the injection sites or a known allergy or sensitivity to the study medication or its components. Patients were also excluded if they had an inadequate response to three or more prophylactic treatments after an adequate trial as determined by the investigator, a Beck Depression Inventory score of >24, or psychiatric problems that, in the investigator's opinion, were severe enough to interfere with study participation or results. Patients were also excluded if they had had previous therapy with botulinum toxin of any serotype, had been injected with anaesthetics or steroids into the study-targeted muscles during the 30 days immediately prior to initiation of the baseline period, or if, in the opinion of the investigator, they were overusing or abusing symptomatic medication, alcohol or drugs. Concurrent chronic use or chronic use in the 3 months prior to the screening period of muscle relaxants (e.g. Flexeril®, benzodiazepines, etc.) was prohibited. Patients who were concurrently participating in another investigational study or who had participated in such a study in the 30 days immediately prior to baseline period were not permitted to participate. Patients who had an uncontrolled systemic disease or any condition that in the investigator's opinion might have put the patient at significant risk, might have confounded the study, or might have interfered significantly with the patient's participation in the study were also excluded. Females who were pregnant, nursing, or planning a pregnancy during the study or who were unable or unwilling to use a reliable form of contraception during the study were excluded.
Treatment schedule and protocol
Patients were screened during a 30-day baseline period during which data were collected daily using an electronic telephone diary (ClinPhone, Nottingham, UK) to capture specified characteristics of their migraine episodes and headache pain medication use. Following the baseline period, patients meeting the inclusion/exclusion criteria were treated (single-blind) with placebo in a minimum of seven muscle areas (Table 1), with each patient receiving a total of 20 injections within these areas. The injection protocol used was a symmetrical fixed-site, fixed-dose approach similar to that described by Blumenfeld and colleagues (33).
BoNTA injection sites, doses and volume of injection

Botulinum toxin type A (BoNTA) or placebo was injected bilaterally into the identified pericranial muscle groups at the indicated units (U)/volume (ml).
Headache-specific characteristics were then recorded for 30 days, after which patients were classified as placebo responders (PR) if they had at least a 50% reduction in migraine attack frequency during the placebo run-in period compared with baseline or if they had less than three moderate-to-severe untreated migraine episodes or less than three treated migraine episodes of any severity at the end of this period. All other patients were considered placebo non-responders (PNR).
Following the placebo run-in period, patients within each stratum (PNR and PR) returned to be randomized to the first of three identically masked BoNTA or placebo treatments (day 0). The effects of treatment were analysed separately for each stratum and also for the pooled population. Patients were injected with either 225 U BoNTA, 150 U BoNTA, 75 U BoNTA or placebo using the same injection protocol as the placebo run-in period (Table 1). Patients were to be injected with the same dose and volumes in the same muscle areas and sites for all treatments and, whenever possible, treatments for each patient were to be performed by the same physician throughout the study. Patients received additional treatments at day 90 and day 180 and returned for follow-up visits at 30-day intervals following each treatment through day 270. Throughout the study, patients were required to record daily headache symptoms and use of acute headache pain medications using the electronic data-capture system. If a patient exited the study at any visit prior to day 270, all exit procedures and evaluations were to be completed at that visit.
Study treatment
Each vial of BOTOX® contained 100 U of Clostridium botulinum toxin type A, 0.5 mg albumin (human) and 0.9 mg sodium chloride in a sterile, vacuum-dried form without a preservative. Each vial of placebo contained 0.9 mg sodium chloride in a sterile, vacuum-dried form without a preservative. All vials were to be reconstituted with 0.9% sterile, preservative-free saline as the diluent. The vials were reconstituted using the volume of diluent assigned to each randomization number to attain specific BoNTA doses of 225 U, 150 U or 75 U (1.3 ml, 2.0 ml or 4.0 ml, respectively). Therefore, blinding was maintained as each treatment was a final volume of 4 ml.
Blinding and randomization
The placebo run-in was a single-masked treatment whereby the investigator, but not the patient, knew that the treatment administered was placebo. Starting at day 0, this was a double-blind study, and neither the investigator nor the patient knew which treatment was administered.
Patients were randomized within each stratum (PNR or PR) to treatment (BoNTA 75 U, BoNTA 150 U, BoNTA 225 U or placebo) in a ratio of 1 : 1 : 1 : 1. An individual with no other study involvement reconstituted the study medication and drew the study drug into the syringes for administration. The syringes were then given to the investigator for injection.
Prior and concomitant therapy
The use of any concurrent medication, including acute headache treatments, was recorded along with the reason the medication was taken during the study. Patients taking concurrent routine therapies had to maintain a stable dose and dose regimen during the study. However, use of prophylactic headache medication was prohibited.
Efficacy measures
The primary efficacy measure was the mean change from baseline in the frequency of migraine episodes of any severity in a 30-day period for the PNR group, determined from data recorded by patients in the telephone diary. The primary visit for determination of efficacy was day 180, with the evaluation reflecting the prior 30-day period. Baseline for the efficacy measures was defined as the frequency of migraine headache episodes during the first 30 days of the study. A difference of 1.5 headaches between BoNTA and placebo in the mean change from baseline in the frequency of migraine episodes of any severity in a 30-day period at day 180 was considered clinically significant. This definition was decided arbitrarily, guided by the study investigators and advisers.
Secondary efficacy variables included the proportion of patients with a decrease from baseline of ≥50% migraine episodes in a 30-day period and the proportion of patients with a decrease from baseline of two or more migraine episodes in a 30-day period. Other efficacy measures included: the number of days with headache or migraine episode (in a 30-day period); the frequency of moderate-to-severe migraine episodes (per 30-day period); the number of days that acute headache pain medication was used during the study (in a 30-day period); the Migraine Disability Assessment Scale (MIDAS) administered at days −60, 90, 180 and 270; and the Headache Pain-Specific Quality of Life Questionnaire administered at days −60, 0, 60, 90, 180 and 270.
Safety measures
Safety was assessed by reports of adverse events, physical and neurological examinations, and clinical laboratory tests. At each visit following treatment at day 0, adverse events were recorded and documented with information regarding the date of onset, resolution date (if applicable), severity (mild, moderate or severe), duration, frequency, relationship to study treatment, action taken regarding study treatment, treatment required (if any) and outcome.
Statistical analyses
As planned prior to unmasking the treatment code, the 'as-treated' population for both the safety and efficacy analyses included all patients receiving their treatment at day 0, regardless of subsequent injections. They were analysed in the responder/non-responder stratum indicated by baseline and placebo run-in data. Pooled data from both strata were also analysed. Multiple comparison adjustments were made by using the O'Brien–Fleming group sequential method to set the significance level at 0.048 for the primary analysis at day 180 (34). All other hypothesis tests used a type I error of α = 0.05 to determine statistical significance, except that treatment-by–subgroup interactions were examined at the 0.10 level.
For the frequency of migraine episodes at baseline and the change from baseline in frequency in the non-responder stratum, comparisons among and between treatment groups were performed using Kruskal–Wallis and Wilcoxon rank sum tests (35). If there were significant baseline differences among treatments in the primary variable, a baseline covariate was to be included in an analysis of covariance of the ranks of the variable. The secondary efficacy variables (the incidence of patients with a decrease from baseline of ≥50% migraine episodes and the incidence of patients with a decrease from baseline of two or more migraine headache episodes) were analysed using the Pearson's χ2 tests. Other continuous efficacy variables were summarized for each 30-day period. Pairwise comparisons between treatments were made using Wilcoxon rank sum tests. Quality of life, as addressed by the MIDAS survey and the Headache Pain-Specific Quality of Life questionnaire, were analysed among treatment groups with the Kruskal–Wallis test.
Results
Patient demographics and baseline characteristics
Of the 954 patients screened and assessed over the baseline period, 515 were considered eligible for participation and entered the placebo run-in phase. At the end of the placebo run-in period (day 0) a total of 495 patients [mean age 43.2 years, range 18–65 years; 435/495 (87.9%) female] were classified as either PNR [322/495 (65.1%)] or PR [173/495 (34.9%)]. Twenty patients (3.9%) were withdrawn from the study for a variety of reasons, including not meeting the minimum threshold for headache symptoms and incomplete diary data. Subsequently, patients within each stratum were randomized to receive BoNTA 225 U, 150 U or 75 U or placebo treatment. Within the PNR stratum, 85, 82 and 83 patients received BoNTA 225 U, 150 U and 75 U, respectively, and 72 patients received placebo. Within the PR stratum, 44, 43 and 40 patients received BoNTA 225 U, 150 U and 75 U, respectively, and 46 patients received placebo. Table 2 illustrates baseline characteristics of all enrolled patients stratified by treatment group
Baseline characteristics (all enrolled patients)

SD, Standard deviation; MIDAS, Migraine Disability Assessment Scale.
There were no statistically significant differences between treatment groups in their demographic characteristics. All patients had a history of moderate to severe migraine headaches. The mean time from onset of migraines was 23.1 years and the mean age at onset was 19.7 years. The mean baseline MIDAS score of 38.7 indicated severe disability associated with their migraine episodes. During baseline, >97% of each strata and treatment group were using acute headache pain medications [e.g. ergotamines, triptans, simple analgesics, opioids and combination medications (ICHD-II sections 8.2.1–8.2.5)]. The mean number of days in the baseline period patients used acute headache pain medication was 6.5, 6.4, 6.3 and 6.4 days in the PNR group and 6.2, 5.2, 5.6 and 5.9 days in the PR group for the BoNTA 225 U, 150 U, 75 U and placebo groups, respectively.
Patient disposition
A total of 81% of patients (401/495) completed the study and 19% (94/495) discontinued: 6.1% (30/495) for administrative reasons (lost to follow-up and personal reasons); 5.9% (29/495) for lack of efficacy (BoNTA 225 U n = 6; BoNTA 150 U n = 5; BoNTA 75 U n = 5; placebo n = 13); 2.8% (14/495) for adverse events (BoNTA 225 U n = 4; BoNTA 150 U n = 4; BoNTA 75 U n = 4; placebo n = 2); 0.4% (2/495) for protocol violations (one for improper entry and one for inability to follow study instructions); and 3.8% (19/495) for other reasons. The percentage of patients discontinuing the study was similar for both BoNTA and placebo groups in the PNR (18.4% BoNTA; 19.4% placebo) and PR strata (17.3% BoNTA; 26.1% placebo).
Analyses of efficacy
Mean change from baseline in the frequency of migraine episodes per 30-day period
At day 180, the primary end-point, the mean change from baseline in the frequency of migraine episodes in the PNR stratum per 30-day period was −1.6, −1.7, −1.5 and −1.4 in the BoNTA 225 U, 150 U, 75 U and placebo groups, respectively (Table 3a). The differences between the groups were not statistically significant (P = 0.817). There were no statistically significant between-group differences observed at any time points. Pairwise comparisons between the groups showed a statistically significant difference at day 60 between the BoNTA 150 U group compared with placebo (−1.5 vs. −0.7, P = 0.042). In the PR stratum, although there were no statistically significant between-group differences observed at any time point, from day 90 to the end of the study (day 270), all treatment groups experienced a mean change from baseline of greater than two migraine episodes per 30-day period (Table 3b). From day 240 to day 270, pooled data of the two strata showed decreases from baseline ranging from 2.2 to 2.5 in the BoNTA treatment groups and 2.4 in the placebo group.
Mean change from baseline in the mean number of migraine headaches per 30-day period (placebo responder stratum) [Mean (SD)]
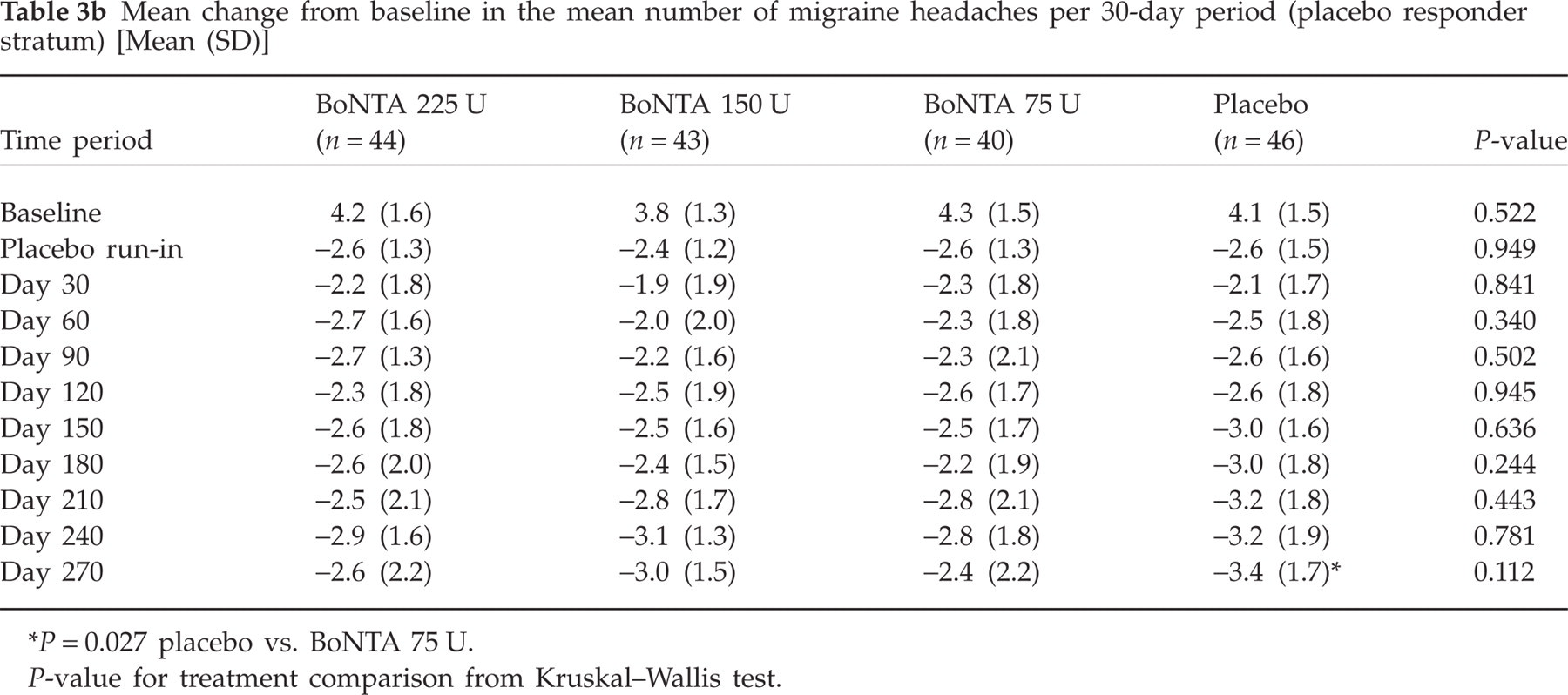
P = 0.027 placebo vs. BoNTA 75 U.
P-value for treatment comparison from Kruskal–Wallis test.
Mean change from baseline in the number of migraine headaches per 30-day period (placebo non-responder stratum) [Mean (SD)]

P = 0.042 BoNTA 150 U vs. placebo.
P-value for treatment comparison from Kruskal–Wallis test.
Percentage of patients with a decrease from baseline of ≥50% migraine episodes per 30-day period
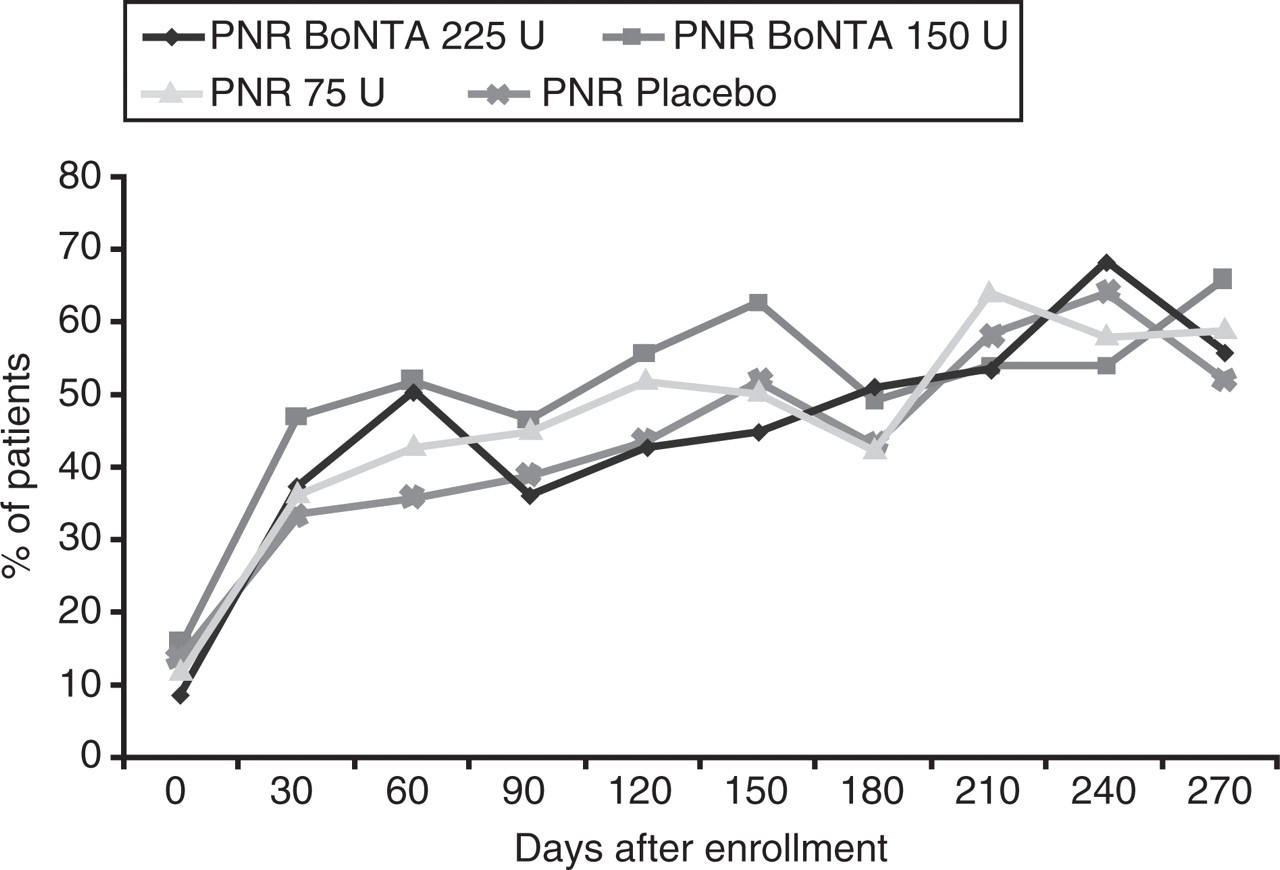
For the PNR stratum, the number of patients with at least a 50% decrease from baseline in the frequency of migraine episodes per 30-day period generally increased throughout the study, to >50% of patients in the BoNTA treatment arms by day 270 and 44% in the placebo arm (Fig. 2). However, no statistically significant between-group differences were observed at any time point. At day 60, pairwise comparisons between the groups showed a statistically significant difference favouring the BoNTA 150 U group; 39.5% of patients in the BoNTA 150 U group had at least a ≥50% decrease from baseline in the frequency of migraine episodes per 30-day period compared with 24.3% in the placebo group (P = 0.046).

Percentage of patients with a decrease of at least 50% in migraine attacks per 30-day period.
Pooled data of the two strata (PNR and PR) showed that from day 150 onward, the percentage of patients who experienced at least a 50% decrease from baseline in the frequency of migraine episodes per 30-day period was >50% in all treatment groups. No statistically significant differences were found between the treatment groups.
Percentage of patients with a decrease from baseline of two or more migraine episodes per 30-day period
For the PNR stratum, by day 60, 35.7–51.9% of patients in all treatment groups had a decrease from baseline of two or more migraine episodes per 30-day period, with this percentage generally being maintained or increasing through day 270 (Fig. 3). From day 210, >50% of patients in all treatment groups experienced a decrease from baseline of two or more migraine episodes per month. No statistically significant between-group differences were observed at any time point. Pairwise comparisons between the groups showed statistically significant differences at day 60 between the BoNTA 150 U and placebo groups (51.9% vs. 35.7%, P = 0.047) and at day 150 between the BoNTA 225 U and 150 U groups (44.8% vs. 62.5%, P = 0.036).
Percentage of patients with a decrease of two or more migraine attacks per 30-day period.
Pooled data of the two strata (PNR and PR) show that from day 60 onward, approximately 50% of patients in all treatment groups experienced a decrease of two or more migraine episodes per 30-day period, and from day 210 onward, this percentage increased to ≥60% of patients in all treatment groups. Pairwise comparison between the groups showed statistically significant differences at day 180 between the BoNTA 225 U and 75 U groups (61.5% vs. 46.9%; P = 0.037).
Other efficacy measures
In the analyses of other efficacy measures, there were only occasional statistically significant differences between treatment groups favouring either BoNTA or placebo, including the number of headache days, the number of days with migraine, the frequency of moderate to severe migraine episodes, the number of days with use of acute headache medication during the study, and the patients' global assessment of response to treatment. No trend favouring either BoNTA or placebo was observed on any of these measures.
Acute medication use during the course of treatment
During the course of the study, >50% of all patients continued to use acute headache medications and >70% of all patients continued to use concomitant migraine medications, which included not only acute headache medications but also ancillary agents such as anxiolytic, antiemetic and propulsive agents.
Safety and tolerability
One or more adverse events, regardless of causality, were reported for 76.7%, 77.6% and 77.2% of patients treated with BoNTA 225 U, 150 U and 75 U compared with 54.2% of patients treated with placebo. The overall incidence of adverse events was greater in each BoNTA group than in the placebo group (P < 0.001). The majority of adverse events were transient and mild or moderate in severity.
The most frequently reported treatment-related adverse events in the BoNTA groups were muscular weakness (in areas of injection sites), neck pain, neck rigidity, blepharoptosis, myalgia, skin tightness and injection site pain (Table 4). The most frequently reported treatment-related adverse events in the placebo group were neck pain, neck rigidity and headache. The majority of treatment-related adverse events were mild or moderate in severity.
Number (%) of Patients With Treatment-Related Adverse Events, Reported by = 3 Patients in Any Treatment Group

Adverse events are ordered in descending order according to the percentage in any treatment group.
P-values for among-group comparisons are from Pearson's χ2 or Fisher's exact tests.
Only 14/495 patients (2.8%) discontinued the study due to adverse events (four patients in each of the BoNTA groups and two patients in the placebo group). In eight of 12 BoNTA-treated patients who discontinued due to adverse events, the reason was related to neck pain/rigidity, and in six of 12 patients, eye problems and/or blepharoptosis was a factor.
A total of seven patients experienced serious adverse events (two in the BoNTA 225 U group, two in the BoNTA 150 U group, one in the BoNTA 75 U group and two patients in the placebo group), all of which the investigators considered to be unrelated to study treatment. There were no clinically relevant differences between the BoNTA and placebo groups in changes from baseline to follow-up for laboratory variables or vital signs.
Discussion
In this exploratory trial, both BoNTA and placebo treatment resulted in sustained mean reductions from baseline in the frequency of migraine episodes per month throughout the course of the study. However, the primary end-point was not met, and the BoNTA groups failed to demonstrate a superior treatment effect compared with placebo.
The mean reduction from baseline in the frequency of migraine episodes per month at the predetermined primary efficacy time point (day 180) was 1.5–1.7 episodes in the three BoNTA treatment groups and 1.4 episodes in the placebo group for the PNR group. Pooled results of the two treatment strata (PNR and PR) indicate that from day 240 to day 270, there were mean decreases from baseline ranging from 2.2 to 2.5 migraine episodes per month in all treatment groups. These results are similar, or larger in magnitude, to those observed in randomized, double-blind, placebo-controlled migraine trials evaluating topiramate (decreases from baseline in active groups of 2.1 and 2.3 migraine episodes) (1, 16), gabapentin (decrease of 2.0) (36) and extended-release divalproex sodium (decrease of 1.2) (37). However, comparisons of data from different clinical trials are limited and should be evaluated cautiously given variabilities in study design, patient population (i.e. size and characteristics) and duration.
A strong placebo response was evident in this trial. Analyses involving 50 placebo-controlled migraine clinical trials of oral treatments found average placebo-response rates ranging from 27% to 30% (38–40). Couch, in an analysis of eight trials that evaluated various oral prophylactic migraine treatments, found that approximately 27% of patients given placebo improved by at least 50% (38). However, in this trial, the pooled data of the two strata showed that, from day 150 onward, the percentage of patients who experienced at least a 50% decrease from baseline in the frequency of migraine headache episodes per 30-day period was >50% in the placebo treatment group.
Placebo response in headache trials is well documented and may be due in part to the neurological correlate of a patient's expectation of a clinical effect (known as expectancy) of treatment (41). Patient expectancy in addition to the perceived potency of a particular intervention may interact with neuronal systems to mediate an antinociceptive response (42–45). In a study using saline injections as placebo, Zubieta and colleagues found a strong positive correlation between activation of the endogenous opioid system and expectations of analgesia in addition to the patient's estimate of a treatment's analgesic efficacy (44). Patients may perceive an injectable placebo as having greater analgesic potency than an oral placebo, which theoretically could result in a stronger antinociceptive response. This may have been an underlying causative factor in the strong placebo response observed in this trial.
Additionally, the design of this trial included three BoNTA randomization groups and one placebo group. Therefore, patients entered the trial conscious that they had a 75% probability of being randomized to active treatment. Linde et al. have demonstrated that an uneven randomization scheme with a greater number of active treatment arms may result in higher patient expectations of efficacy and a more robust response to placebo than an even 1 : 1 randomization (46). This may also have contributed to the strong placebo response observed in this trial.
BoNTA prophylactic treatment of migraine was safe and well tolerated in this trial. The discontinuation rate due to adverse events in this study was only 2.8%, which compares favourably with the discontinuation rates due to adverse events observed in other migraine prophylaxis trials. In the clinical trials evaluating topiramate, gabapentin and divalproex sodium as migraine prophylaxis, the discontinuation rates due to adverse events in patients receiving active medication were approximately 19%, 16% and 8%, respectively (36, 37, 47).
Although BoNTA treatment was not superior to placebo in this trial, data from several randomized, double-blind, placebo-controlled clinical trials have demonstrated that BoNTA significantly reduces the mean frequency of headache/migraine episodes per month from baseline compared with placebo in episodic migraine patients. In a trial evaluating BoNTA as prophylactic migraine treatment, BoNTA 25 U (but not 75 U) significantly reduced the frequency of moderate-to-severe migraine episodes from baseline compared with placebo at months 2 (1.6 vs. 0.4, P = 0.008) and 3 (1.9 vs. 1.0, P = 0.042) and the frequency of migraine episodes of any severity at month 3 (2.1 vs. 0.9, P = 0.014) (29). In migraineurs with two to eight attacks per month, BoNTA 100 U administered with a fixed-site protocol did not result in superior results compared with placebo (30). However, a post hoc analysis of an episodic migraine trial, conducted by Aurora and colleagues, showed that BoNTA treatment (mean, 188–192 U) resulted in a significant mean reduction from baseline of 4.0 migraine episodes at day 180 (vs. placebo: 1.9 migraine episodes; P = 0.048) in patients who experienced ≥12 headache days per month (31).
This trial is part of a series of large, placebo-controlled exploratory trials designed to investigate the therapeutic effect of BoNTA as prophylactic treatment of migraine and headache, identify a responsive patient population and establish a treatment protocol. Further study is needed to improve the understanding of the mechanism of the interaction of BoNTA with the nociceptive pathway and the types of headache patients that may optimally respond to BoNTA treatment.
Dosing and results reported in this study are specific to the formulation of botulinum toxin type A manufactured by Allergan, Inc. (Irvine, CA, USA). The Allergan, Inc. formulation is not interchangeable with other botulinum toxin products and cannot be converted by using a dose ratio (48).
Acknowledgements
This study was sponsored by Allergan, Inc., Irvine, CA, USA. X.L. and C.T. are employed by Allergan, Inc. and own stock in the company. The European BoNTA Headache Study Group: Reto Agosti (Zürich, Switzerland); Guy Arnold (Berlin, Germany); Fayyaz Ahmed (Hull, UK); Gérard Besson (Grenoble Cedex 9, France); Mitchell F. Brin (Irvine, USA); Victor Chan (Valence Cedex 9, France); Pierre Clavelou (Clermont-Ferrand, France); Vida Demarin (Zagreb, Croatia); Hans-Christoph Diener (Essen, Germany); Andrew Dowson (Surrey, UK); Rozalina Dimitrova (Irvine, USA); Leslie Findley (Essex, UK); Jean Fraser (Wigan, UK); Claire Harding (Cardiff, UK); Hartmut Göbel (Kiel, Germany); Morag Horne (Berkshire, UK); Matti Ilmavirta (Jyvaskyla, Finland); Davor Janculjak (Osijek, Croatia); Rigmor Jensen (Copenhagen, Denmark); Gareth John (Manchester, UK); Tapani Jolma (Pori, Finland); Thorbjorn Kjaernli (Alesund, Germany); Ksenija Willheim (Rijecka, Croatia); Jacqueline Maroni (Glasgow, UK); Martin Marziniak (Homburg Saarland, Germany); Nadine Memran (Nice, France); Julio Pascual (Santander, Spain); Anne Christine Poole (Oslo, Norway); Maja Relja (Zagreb, Croatia); John Robinson (Liverpool, UK); Erkki Sako (Turku, Finland); Mansur Salman (Birmingham, UK); Mikaël Saulay, Diploma of ENSAI (Irvine, USA); Jean Schoenen (Liege, Belgium); Jean Michel Senard (Toulouse Cedex, France); Claudia Sommer (Wurzburg, Germany); Susan Taylor (Chorley, UK); Catherine Thompson (High Wycombe, UK); Catherine Turkel (Irvine, USA); Jean-Marc Visy (Reims, France); Anton Vladic (Zagreb, Croatia); Andelko Vrca (Zagreb, Croatia).
