Abstract
Background
Previous studies have shown an analgesic effect of ginger in the acute treatment of migraine, and there is anecdotal evidence of its efficacy in migraine prophylaxis.
Objective
This study aimed to evaluate the potential of ginger to prevent migraine attacks.
Methods
This double-blind, placebo-controlled randomized clinical trial took place at the Headache Clinic, Universidade Federal de Minas Gerais (Belo Horizonte, Minas Gerais, Brazil), involving 107 patients. Only subjects diagnosed with episodic migraine, aged between 18 and 60 years old, and who were not taking any prophylactic medication, were enrolled in the study. After one month of observation, subjects selected for the study were randomized 1:1 into placebo and treatment groups. Patients received capsules three times per day of 200 mg of dry extract of ginger (5% active ingredient) or placebo (cellulose) for three months. Visits were performed monthly and the patients were asked to fill in a migraine diary. The adherence to treatment was evaluated by counting capsules.
Results
The percentage of patients who responded to treatment (i.e. a reduction of 50% in the number of migraine attacks at the end of treatment) did not differ between the groups. There was a decrease in the number of days with severe pain, analgesic use for acute migraine and duration of migraine attacks in both groups, without significant difference between ginger and placebo groups.
Conclusions
Ginger provides no greater benefit in the prophylactic treatment of migraine when compared to placebo. This trial is registered at ClinicalTrials.gov (NCT02570633).
Introduction
Migraine can decrease functional capacity and quality of life, being one of the main causes of disability worldwide. In the Global Burden of Disease Study 2016, it is estimated that migraine has been responsible for 45 million years of life lived with disability worldwide (1).
Prophylactic treatment, when effective, can decrease the frequency and severity of migraine attacks, reducing the personal and societal burden related to migraine. Approximately 38% of patients with migraine are candidates for prophylactic treatment; however, only 3 to 13% currently undergo regular prophylaxis (2). A large proportion of patients with migraine do not adhere to treatment because of dissatisfaction with drug efficacy and/or the occurrence of side effects, among other factors (3). Adherence to medication prophylaxis varies between 41 and 95% in the first two months and declines significantly to 7–55% with one year of treatment (4). Besides, lower levels of adherence are associated with depression (5).
A common cause of non-adherence to migraine treatment is the occurrence of side effects related to prophylactic drugs, such as weight gain, mood symptoms, sleep and memory disorders (3,6). Therefore, the use of complementary and/or alternative strategies with theoretically fewer side effects has been on the rise among patients with migraine (7).
Behavioral, physical therapies and the use of nutraceuticals have been shown to be effective in preventing migraine attacks (7). The analgesic effect of foods and nutritional supplements has been investigated in different conditions. In this line, ginger has shown an analgesic effect in the treatment of osteoarthritis, dysmenorrhea and muscle pain (8–10). For instance, Rahnama et al. showed that the use of 500 mg of ginger powder three times a day two days before the onset of the menstrual period and continued for the first three days of the menstrual period was more effective than placebo in decreasing the severity of dysmenorrhea-related pain (11). Previous studies also showed that using ginger extract two or three times a day for at least 1 week decreases pain in osteoarthritis (12–16).
Recently, our group showed that the addition of ginger extract (400 mg) to ketoprofen in the treatment of acute migraine promotes a further reduction in pain and an improvement in functional status (17). The analgesic effect of ginger has been attributed to the capacity of its active components (gingerols and shogaols) to inhibit prostaglandin biosynthesis by decreasing the expression of cyclooxygenase-2 enzyme (COX-2) as non-steroidal anti-inflammatory drugs (18). Ginger also has antiemetic properties when administered in the treatment of nausea and vomiting induced by chemotherapy or during pregnancy (19,20). A case study of a patient who received 500 to 600 mg of ginger powder mixed with water three times a day suggested the potential of ginger as a preventive measure against migraine without any reported side-effects (21). However, the effect of ginger in migraine prophylactic treatment has not been evaluated in a clinical trial. Therefore, this study describes the prophylactic use of ginger extract in migraine through a double-blind placebo-controlled randomized clinical trial. This trial is registered at ClinicalTrials.gov (NCT02570633).
Methods
Selection of patients
Migraine patients of both sexes, aged between 18 and 60 years, who agreed to participate by signing the informed consent form, were enrolled in the study. The inclusion criteria were: (i) Migraine diagnosis according to The International Classification of Headache Disorders 2nd Edition (22) for at least one year and age at onset below 50 years, with or without aura (22); (ii) fewer than 15 headache days per month; (iii) no prophylactic treatment for migraine in the last three months.
The exclusion criteria were: (i) Headaches not characterized as migraine; (ii) analgesic, alcohol or illicit drug abuse; (iii) individuals using any prophylactic treatment for migraine; (iv) hypersensitivity to ginger; (v) other neurological diseases (e.g. epilepsy, stroke); (vi) use of anticoagulant drugs; (vii) pregnant or lactating women.
Trial design
This was a double-blind, placebo-controlled randomized clinical trial adapted from the Guidelines for Controlled Trials of Drugs in Migraine (23), which took place at the Headache Clinic, University Hospital, Universidade Federal de Minas Gerais (UFMG) (Belo Horizonte, MG, Brazil). This study was registered at the Trials Registry Platform (NCT02570633) and was approved by the UFMG human ethics research committee (CAAE: 28236814.3.0000.5149), Brazil. All participants gave written consent to participate in the study.
The study period lasted four months. An observation period of one month preceded the treatment phase. During this observation period, patients filled in a headache diary containing information about headache frequency, duration and intensity, and the use of analgesics.
The treatment or intervention lasted three months and the visits occurred monthly (T30: 30 days of treatment; T60: 60 days of treatment, and T90: 90 days of treatment). In the interval between each visit, the patients were also asked to fill in a headache diary comprising information about headache attack frequency (number of attacks per month), duration in hours of each attack, intensity of pain (using a 4-point scale), analgesic use for symptomatic treatment, and potential side effects. Patients were advised to not change their lifestyle (diet and physical activity habits) throughout the intervention period.
Patients were instructed to take 200 mg of ginger extract (5% of gingerol) or placebo (cellulose) three times a day, totaling 600 mg of extract, comprising 30 mg of gingerol, in the intervention/treatment group. The capsules of both placebo and ginger extract were made of gelatin, were odorless, and had the same color and format. Clinicians and patients could not discriminate between placebo and ginger capsules. Ginger extract was obtained at Amphora Pharmacy (Belo Horizonte, Brazil), and the amount of gingerol in the extract was confirmed by an independent laboratory (Ideal Pharma, São Paulo). It is worth mentioning that doses below 2 g of ginger extract containing 5% gingerols are highly tolerable and not toxic to humans (24).
The allocation of patients to each group was randomized and double-blind (both participants and care providers were blinded). The randomization sequence was created by the Randperm function of the Matlab Mathworks software (Matrix Laboratory) with the ratio of 1: 1. The enrollment of patients and the intervention assignment were performed by different researchers.
No change to trial outcomes occurred after its onset.
Clinical and demographic measures
Demographic data (age, sex and marital status), headache features (migraine diagnosis, length of disease, age of disease onset and headache impact) and anthropometric data (weight, height and calculated body mass index (BMI)) were acquired. Headache impact was evaluated by the Headache Impact Test, version 6 (HIT-6) (25) and the Migraine Disability Test (MIDAS) (26,27) questionnaires. Depression and anxiety symptoms were evaluated, respectively, by the Beck Anxiety and Depression Inventories (BDI and BAI) (28,29).
Evaluation of treatment efficacy
The primary endpoint was the percentage of patients who responded to treatment; that is, a reduction of 50% in the number of migraine attacks at the end of treatment.
For the analysis of the primary outcome of the study, only those individuals who adhered to more than 80% of the treatment, evaluated by counting capsules and the equation: [(Number of capsules remaining − Number of capsules that should have been used) × 100] were considered (4,5).
Secondary endpoints for treatment efficacy were also considered, including: i) Changes in the number of days with severe pain, number of days that required the use of analgesic drugs, maximum and minimum duration of migraine attacks; ii) changes in the HIT-6 and MIDAS.
Side-effects and treatment adherence were also evaluated.
Statistical analyses
Statistical analyses were performed using SPSS, version 20.0 (IBM Corp., Armonk, NY, USA). The Kolmogorov–Smirnov test was used to test normality. To evaluate the differences between the two groups, Simple t-test and Mann–Whitney test were performed for the mean and median comparisons, respectively. To evaluate the differences between different times of treatment, Paired samples t-test and Wilcoxon test were used for the mean and median comparisons, respectively. Categorical variables were compared using chi-square analyses. Continuous variables taken at different time intervals were compared using a Generalized Estimating Equation (GEE) model to evaluate the effect of group allocation (placebo or ginger), adjusting for time effect (group*time). The Bonferroni post-test identified the presence of significant effects.
Sample size was estimated in at least 104 participants (52 in the intervention group and 52 in the placebo group) considering the following: 25% pain relief with ginger treatment, 22% confidence interval range (the difference between the highest and lowest confidence interval range) (8,9,30) and 30% dropout rate. Calculation of sample size was based on 5% alpha error, and 80% statistical power (31).
Results
Demographic and clinical characteristics
Three hundred and nineteen (319) individuals were initially assessed, of whom 107 were included in the study: There were 54 in the placebo group and 53 in the group that received ginger extract. Figure 1 shows the study flow diagram.
Flow diagram of the study.
Demographic and clinical characteristics in individuals with migraine.

BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; HIT-6: Headache Impact Test – version 6; MIDAS: Migraine Disability Test.
Chi-square test.
Mann-Whitney test.
Simple t-test.
Treatment efficacy
At the end of three months of treatment, 42% of patients in the ginger group and 39% in the placebo group responded to treatment; that is, had a reduction of 50% or more in the number of migraine attacks per month. Accordingly, there was no difference between groups in the primary end point (p > 0.05).
Secondary end points including days with pain (Figure 2(a)), days with severe pain (Figure 2(b)), days requiring the use of analgesics (Figure 2(c)), number of migraine attacks (Figure 2(d)) and maximum duration of migraine attacks (Figure 2(f)) also decreased on the follow-up (time) in both groups, without significant difference between them (p > 0.05) and without interaction time*group (p > 0.05).
Data collected in migraine diary (secondary end-points). (a) Number of days with pain; (b) number of days with severe pain; (c) number of days when drugs were used for acute migraine treatment; (d) migraine attack frequency; (e) minimum duration and (f) maximum duration of migraine attacks.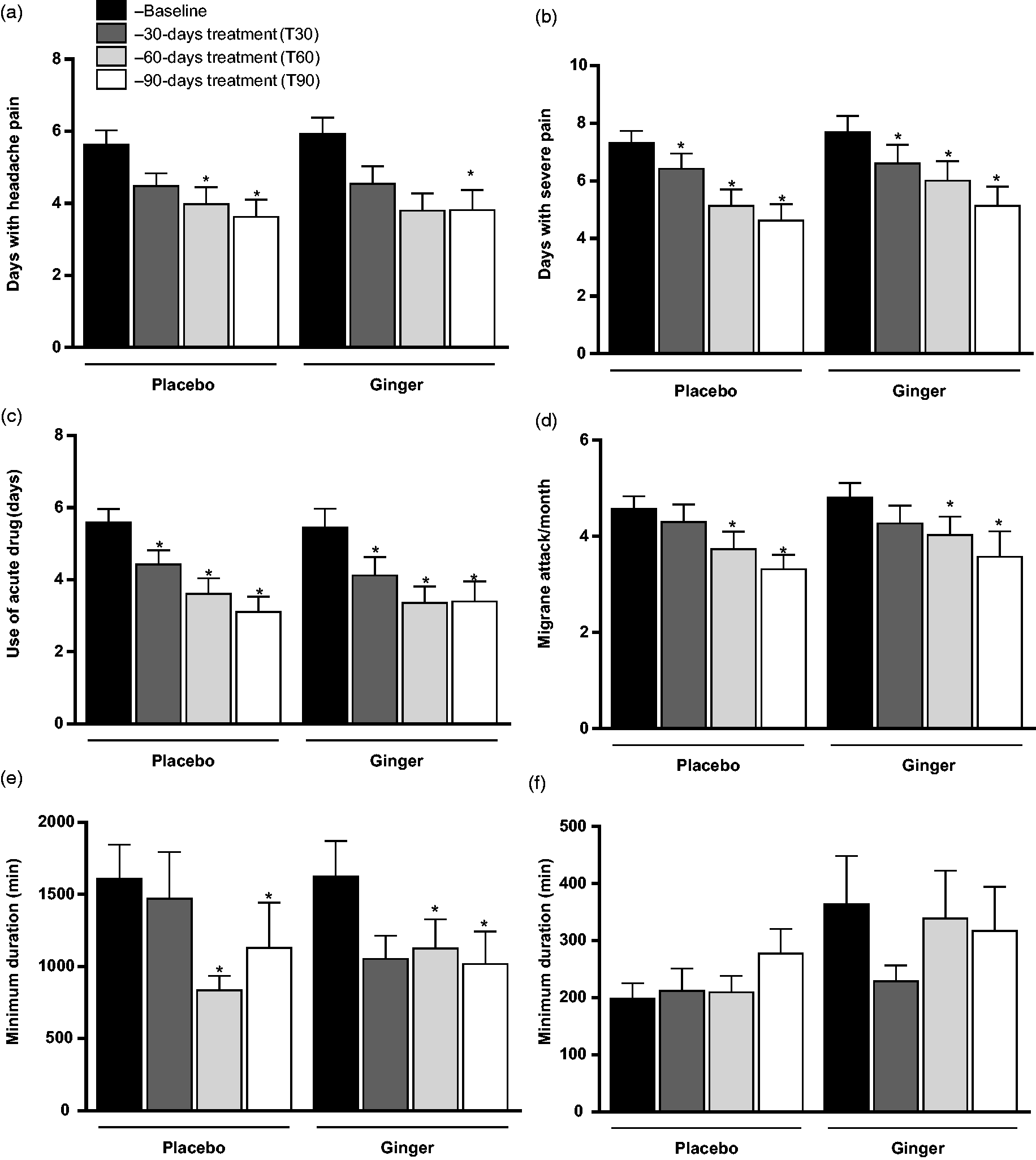
Scales of migraine impact, depression and anxiety throughout the treatment.

T30: 30-days treatment; T60: 60-days treatment; T90: 90-days treatment; BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; HIT-6: Headache Impact Test – version 6; MIDAS: Migraine Disability Test.
Mann-Whitney test.
Test: Generalized Estimating Equation (GEE) model followed by Bonferroni post-test.
Different from baseline, p < 0.05.
Patients in both groups also decreased the scores in the BDI and BAI (Table 2). Interestingly, after 60 days of treatment, BDI and BAI correlated positively with total days with migraine pain (R = 0.304, p < 0.01 and R = 0.252, p = 0.02, respectively) and after 90 days of treatment, BAI correlated positively with total days with migraine pain (R = 0.309, p < 0.01). At the end of the treatment, both scales correlated positively with frequency of migraine attacks per month (R = 0.215; p = 0.05 and R = 0.213, p = 0.05, BDI and BAI, respectively).
Treatment tolerability
Sixteen individuals in the ginger group reported side effects, and four of them chose to leave the study because of that. In the placebo group, eight patients reported side effects, and one of them chose to leave the study. The side effects reported in the ginger group were: Heartburn (n = 12), constipation (n = 1), reduced appetite (n = 1) and nausea (n = 3), while in the placebo group: Nausea (n = 4) and heartburn (n = 4). The frequency of side effects was significantly higher in the ginger group only in the first 30 days of treatment (p < 0.01).
Adherence to treatment, as evaluated by the number of capsules remaining, was 95.5% in the ginger group and 95.2% in the placebo group (p = 0.84).
Discussion
Previous studies have shown the efficacy of ginger, in extract or powder, in reducing and preventing pain associated with medical conditions (8–10,17). In this study, we showed that the use of ginger extract in the prophylactic treatment of migraine was not effective in decreasing the frequency of migraine attacks. People who received ginger extract responded similarly to people under placebo.
The placebo response rate in our study is in accordance with the literature on preventive trials of migraine, ranging from 14–50% depending on the duration and design of the study (32). Previous studies have suggested that placebo may change biochemical and cellular responses in the brain toward an analgesic effect. Besides, some factors contribute to this response, including the individual’s tendency to change behavior when they are involved in a study and the influence of cognitive factors on pain perception, such as attention, anxiety, and previous experiences (32). In this study, factors such as patient care, monthly visits and the patient’s expectation regarding a new migraine approach may have contributed to the high placebo response. This is corroborated by the observation that depression scores decreased over the course of the treatment and correlated with the number of migraine attacks. Migraine has a strong comorbidity with depression and previous studies have shown that patients with migraine are more likely to have depression than the general population (33–35).
We and others showed that ginger is effective in decreasing the intensity of migraine pain when administered as an acute strategy (17,36). In a double-blind, randomized and controlled clinical trial, Maghbooli showed that the use of 250 mg of ginger powder during migraine attacks was as effective as 50 mg of sumatriptan in relieving pain intensity 2 h after ginger intake (36). We also showed that oral intake of 400 mg of ginger extract in addition to an intravenous non-steroidal anti-inflammatory drug (ketoprofen) in treatment of acute migraine promotes reduction in pain and improvement in functional status (17). Furthermore, pre-clinical studies have suggested a neuroprotective role for ginger. Ginger components decrease the expression of inflammatory mediators in microglia and astrocytes and upregulate the levels of neurotrophic factors (37,38). Accordingly, as migraine has been associated with neurogenic inflammation with subsequent activation of trigeminal nociceptors (39), ginger might have a modulatory effect on it. However, our data do not support a therapeutic advantage to the use of ginger in migraine prophylaxis compared to placebo.
One limitation of the study includes the lack of proper pharmacological assessment of the ginger capsules prior to the trial. The administration of ginger extract occurred three times a day (i.e. with an average interval of 4–6 hours). According to a pharmacokinetic study, after the consumption of ginger extracts, the peak plasma concentration (Tmax) of serum gingerols occurs between 45 and 120 minutes, with elimination half-lives of two hours (24). The posology chosen for this study was based on the rationale of providing a reasonable amount of gingerols in the least number of times to prevent adherence problems (40). The consumption of ginger capsules more frequently could decrease adherence, and higher doses could lead to gastrointestinal side effects. Conversely, previous studies have observed that taking ginger extract two or three times a day was enough to improve pain (11,12,15). As strengths of the study, the careful enrollment process and its design, with blinded randomization and follow-up, must be mentioned. In sum, this study stands out as the first double-blind placebo-controlled randomized clinical trial developed to specifically evaluate the prophylactic use of ginger in migraine prophylaxis. Despite its analgesic and potential neuromodulatory effects, ginger does not have a beneficial prophylactic effect for the treatment of migraine compared to placebo.
Clinical implications
Ginger does not promote a therapeutic advantage in migraine prophylaxis compared to placebo.
Footnotes
Acknowledgements
The authors would like to give thanks for the support of the headache outpatient clinic medical team and nutrition students Isabel Myrian Guimarães Campos, Tamara Carneiro Medeiros de Souza and Luciana Alves de Andrade for their help during data collection and care with the participants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Brazilian government funding agency Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
