Abstract
Objectives: This is the second of a pair of studies designed to evaluate the efficacy and safety of onabotulinumtoxinA (BOTOX®) for prophylaxis of headaches in adults with chronic migraine.
Methods: PREEMPT 2 was a phase 3 study, with a 24-week, double-blind, placebo-controlled phase, followed by a 32-week, open-label phase. Subjects were randomized (1:1) to injections of onabotulinumtoxinA (155U–195U; n = 347) or placebo (n = 358) every 12 weeks for two cycles. The primary efficacy endpoint was mean change in headache days per 28 days from baseline to weeks 21–24 post-treatment.
Results: OnabotulinumtoxinA was statistically significantly superior to placebo for the primary endpoint, frequency of headache days per 28 days relative to baseline (−9.0 onabotulinumtoxinA/−6.7 placebo, p < .001). OnabotulinumtoxinA was significantly favoured in all secondary endpoint comparisons. OnabotulinumtoxinA was safe and well tolerated, with few treatment-related adverse events. Few patients (3.5% onabotulinumtoxinA/1.4% placebo) discontinued due to adverse events.
Conclusions: The results of PREEMPT 2 demonstrate that onabotulinumtoxinA is effective for prophylaxis of headache in adults with chronic migraine. Repeated onabotulinumtoxinA treatments were safe and well tolerated.
Introduction
Chronic migraine (CM) is a disabling complex neurological disorder recognized as a complication of migraine in the the International Classification of Headache Disorders, second edition (ICHD-II) (1). Patients with CM experience headache on ≥15 days per month, of which at least eight headache days per month meet criteria for migraine without aura or respond to migraine-specific treatment (2). CM is associated with significant disability and reduced health-related quality of life (HRQoL) (3,4). Approximately 1.3% to 2.4% of the general population suffers from CM (5–7), and one in five CM sufferers cannot work because this condition impacts their ability to lead productive lives (8). CM is frequently complicated by overuse of acute pain medications (9–11). Some drugs have regulatory approval for migraine prophylaxis; none are approved specifically for CM prophylaxis. Only a few trials have investigated preventive therapies for CM (12–15).
OnabotulinumtoxinA (BOTOX®, Allergan, Inc., Irvine, CA) has been reported to relieve pain associated with a variety of conditions, including migraine headache (12,13,16–26). The presumed mechanism for headache prophylaxis is blockade of peripheral signals to the central nervous system, which inhibits central sensitization (27,28). A systematic series of exploratory controlled trials was conducted to assess the efficacy and safety of onabotulinumtoxinA in patients with episodic migraine (EM) (<15 headache days per month) (29,30) and chronic daily headache (CDH) (≥15 headache days per month) (12,13,31). In the two largest EM exploratory studies, no significant between-group differences were observed in the frequency of headache episodes (29,30). In one CDH study that evaluated patients with chronic tension-type headache (CTTH), no significant between-group difference favouring onabotulinumtoxinA was observed in the number of tension-type headache-free days per month (31). Efficacy of onabotulinumtoxinA in EM and CTTH has therefore not been established, although it is also possible that patient selection criteria, dosage and injection paradigm of these exploratory studies were not optimal (29–32).
The post hoc analysis of one of the phase 2 onabotulinumtoxinA CDH studies led to key design features of the onabotulinumtoxinA CM phase 3 studies (13,33). We report the double-blind phase results of the second of two large, randomized, placebo-controlled, parallel-group, phase 3 studies that evaluated the safety and efficacy of onabotulinumtoxinA in adults suffering with CM. Results from PREEMPT 1 are reported in a companion manuscript.
Methods
Study design
The Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) clinical program consisted of 2 studies: PREEMPT 1, conducted from 23 January 2006 to 16 July 2008 at 56 North American sites; and PREEMPT 2, conducted from 7 February 2006 to 11 August 2008 at 66 global sites (50 North America and 16 European). PREEMPT 2 was conducted to evaluate the efficacy and safety of onabotulinumtoxinA for prophylaxis of headaches in adults with CM. PREEMPT 2 consisted of a 28-day baseline screening period (hereafter referred to as baseline), a 24-week, double-blind, parallel group, placebo-controlled phase featuring two injection cycles every 12 weeks, followed by a 32-week, open-label phase with three onabotulinumtoxinA injection cycles (Figure 1). Patients used an interactive voice response system (IVRS) telephone diary to record their headache symptoms and acute treatments. Patient diary-day compliance was defined as the percentage of days for which a patient entered diary information into the IVRS system. A maximum three-day recall was allowed, although patients were encouraged to call daily.
PREEMPT study design.
This study was in compliance with the Declaration of Helsinki ethical principles, Good Clinical Practices, principles of informed consent, and requirements of public registration of clinical trials in the United States (ClinicalTrials.gov identifier NCT00168428). PREEMPT 2 was approved at each site by an independent ethics committee or a local institutional review board. Prior to administration of the study procedure, written informed consent was obtained from each randomized participant.
Study participants
Men or women aged 18–65 years with a history of migraine meeting the diagnostic criteria listed in ICHD-II (2004) section 1, migraine (1)—with the exception of “complicated migraine” (i.e., hemiplegic migraine, basilar-type migraine, ophthalmoplegic migraine, migrainous infarction)—and with headache occurring on ≥15 days/four weeks were eligible.
Patients were excluded if they had any medical condition that might put them at increased risk if exposed to onabotulinumtoxinA (e.g., myasthenia gravis, Eaton-Lambert syndrome, amyotrophic lateral sclerosis, any other significant disease that could interfere with neuromuscular function). Other exclusion criteria were diagnosis of other primary/secondary headache disorder, use of any headache prophylactic medication within 28 days of day 1 of baseline, Beck Depression Inventory score of >24 at day 1 of baseline, temporomandibular disorder, fibromyalgia, psychiatric disorders that could interfere with study participation, or previous exposure at any time to any botulinum toxin serotype. Prior to administration of study treatment, women of childbearing potential were required to have a negative urine pregnancy test and have been using a reliable means of contraception.
To be randomized at the end of the baseline screening phase, patients had to provide diary data on ≥20 of 28 days and have had ≥15 headache days, of which each consisted of ≥4 hours of continuous headache and of which ≥50% were migraine or probable migraine days (referred to hereafter as migraine days); they also had to have had ≥4 distinct headache episodes, each lasting ≥4 hours.
Randomization, stratification and study treatment
Qualified subjects were randomized (1:1) in a double-blind fashion to onabotulinumtoxinA or placebo. Randomization was stratified based on the frequency of acute headache pain medication use during baseline (designated as “medication overuse–yes” or “medication overuse–no”), with treatments balanced in blocks of four within each medication-overuse stratum for each investigator site. Patients in the medication overuse–yes stratum had overused acute headache pain medications during baseline, with intake of simple analgesics on ≥15 days, or other medication types or combination of types for ≥10 days, with intake ≥2 days/week from the category of overuse. Investigators were trained not to enroll patients who frequently used opioids as their acute headache pain medication. The randomization sequence was generated using SAS programming language (SAS Institute, Cary, NC, USA) and was stored in a central server with access granted to the randomization programmers. The programmers then released the information to personnel who packed the medication kits, and to the vendor who managed the patient electronic diary (Perceptive Informatics, Waltham, MA, USA) for purposes of central implementation of the randomization and treatment-kit assignment. Throughout the double-blind phase of the study, the patients, the investigators who administered the study treatment and assessed safety and outcomes and the sponsor study management personnel were all masked to the treatment-group assignment. At the end of the baseline screening phase, when the investigator attempted randomization of a subject, the central implementation computer program determined if the subject met the quantitative inclusion/exclusion criteria as per the patient reported diary data. If qualifications were met, the subject number was linked to the next randomization number grouped within strata for that site, and the site was notified of the medication kit assigned to that randomization number.
The PREEMPT injection and dose paradigm was based on the experiences from phase 2 studies (12,13) and will be detailed elsewhere. Study injections were administered as 31, fixed-site, fixed-dose, intramuscular (IM) injections (minimum dose 155 U) across seven specific head/neck muscle areas (corrugator, procerus, frontalis, temporalis, occipitalis, cervical paraspinal and trapezius) every 12 weeks over 24 weeks (two cycles). Depending on the location(s) of the patient’s predominant pain and severity of palpable muscle tenderness, a follow-the-pain strategy, with additional dosing was allowed per protocol. At the investigator’s discretion, up to 40 U additional onabotulinumtoxinA (maximum dose 195 U) or placebo injections could be given to one or both sides in up to three muscle groups (occipitalis, temporalis, and trapezius). Injections were administered IM using a sterile 30-gauge, 0.5 inch needle and 0.1 mL of onabotulinumtoxinA (5 U) or placebo injected at each site. Dosing and results of this study are specific to the formulation of onabotulinumtoxinA manufactured by Allergan, Inc. (Irvine, CA, USA).
Efficacy and safety measures
The primary efficacy endpoint was mean change from baseline in frequency of headache days for the 28-day period ending with week 24. A headache day was defined as a calendar day (00:00 to 23:59) when the patient reported four or more continuous hours of a headache, per the patient diary. Subsequent to study initiation, but prior to study completion and treatment unmasking, the protocol and statistical analysis plan for PREEMPT 2 was amended to change the primary and secondary endpoints, making frequency of headache days the PREEMPT 2 primary endpoint. This change was made based on several factors: availability of PREEMPT 1 data, guidance provided in newly issued International Headache Society clinical trial guidelines for evaluating headache prophylaxis in CM (34) and the earlier expressed preference of the US Food and Drug Administration (FDA), all of which supported using headache day frequency as a primary outcome measure for CM.
All other efficacy analyses primarily examined the mean change from baseline to the 28-day period ending with week 24. Secondary efficacy variables were frequency of migraine days (defined as a calendar day with ≥4 continuous hours of headache meeting ICHD-II criteria for migraine 1.1, 1.2 or 1.6), frequency of moderate/severe headache days (defined as a calendar day with ≥4 continuous hours of headache and a maximum severity of moderate or severe, per the patient diary, among all headache episodes reported on that day regardless of duration), monthly cumulative headache hours on headache days, proportion of patients with severe (≥60) Headache Impact Test (HIT)-6 score, and frequency of headache episodes (defined as patient-reported headache with a start and stop time indicating that the pain lasted ≥4 continuous hours). Other prespecified efficacy assessments evaluated at week 24 included acute headache pain medication intakes (all categories; referred to hereafter as acute pain medication intakes) and three assessments of disability to reflect changes in functioning, vitality, psychological distress and health-related quality of life: the mean change in total HIT-6 (35), the Migraine-Specific Quality of Life Questionnaire (MSQ v2.1) (36,37) and the daily average of the Headache Impact score (HIS).
Statistical analyses
Enrollment of 650 patients was planned. For headache day frequency, a week 24 minimum sample size of n = 325 per group, with standard deviation of 6.7, would have >90% power to detect ≥1.75 between-group difference in mean change from baseline, using a two-sided α = 0.05. A large sample size was planned because of the long study duration (56 weeks per patient). This would allow sufficient sample size for the long-term safety evaluations (>150 patients with five active treatment cycles).
All efficacy analyses used the intent-to-treat population, which included all randomized patients. For each primary and secondary variable, prespecified comparisons between treatment groups were done by analysis of covariance of the change from baseline, with the same variable’s baseline value as a covariate, with main effects of treatment group and medication overuse strata. The baseline covariate adjustment was prespecified as the primary analysis; sensitivity analyses (e.g., rank-sum test on changes from baseline without a baseline covariate) were also performed. Scores for months with at least 20 days of diary data were prorated to 28-day equivalents. Scores for months with less than 10 days of diary data were estimated using a modified last observation carried forward (mLOCF) methodology. This involved the substitution of the patient’s previous 28-day period score multiplied by the ratio of the mean across all patients in the 28-day period of interest divided by the mean across all patients in the previous 28-day period. Scores for months with 10–19 days of diary data were estimated using an average of the prorated and the mLOCF estimates. The mLOCF method of imputation of missing data was prespecified, but sensitivity analyses were also done (e.g., using observed data, without imputation). For binomial variables, the between-group comparisons were done with Pearson’s Chi-square or Fisher’s exact tests, except that logistic regression, with the same variable’s baseline as covariate, was used for variables with baseline imbalance. A two-sided test with p ≤ .05 was considered to be statistically significant.
To control the type 1 error rate for multiple secondary endpoints in the amended PREEMPT 2 protocol and analysis plan, a fixed-sequence gate-keeping approach was used for the five ranked secondary variables at the week 24 primary visit. If the p value of a secondary endpoint was not ≤.05, the tests of any lower-ranked secondary endpoints were not considered statistically significant, regardless of individual p value.
Safety analyses were performed on all randomized patients who received at least one dose of study medication at day 0.
Results
Demographic and baseline headache characteristics
Baseline demographics and characteristics
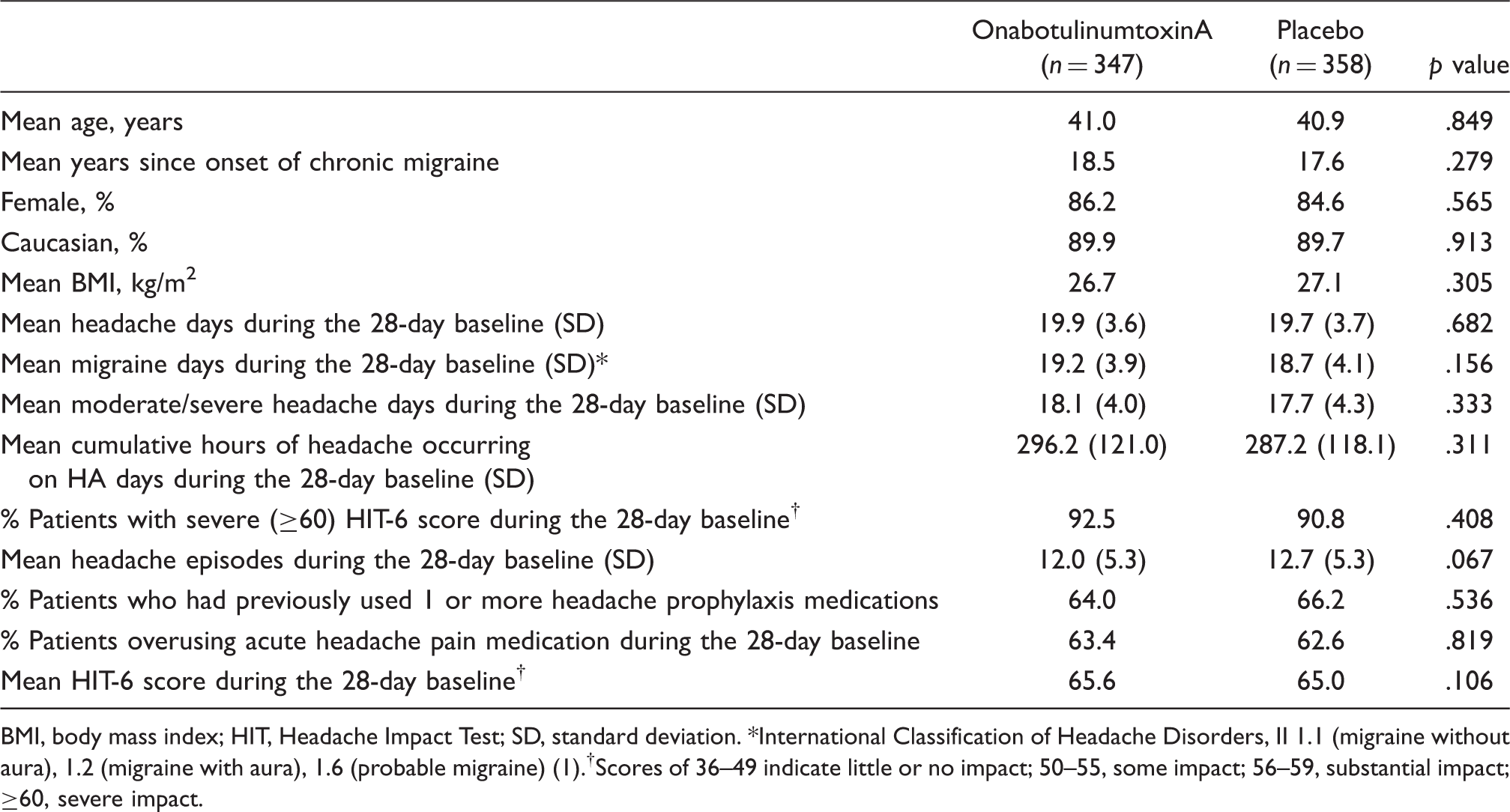
BMI, body mass index; HIT, Headache Impact Test; SD, standard deviation. *International Classification of Headache Disorders, II 1.1 (migraine without aura), 1.2 (migraine with aura), 1.6 (probable migraine) (1).†Scores of 36–49 indicate little or no impact; 50–55, some impact; 56–59, substantial impact; ≥60, severe impact.
Efficacy results
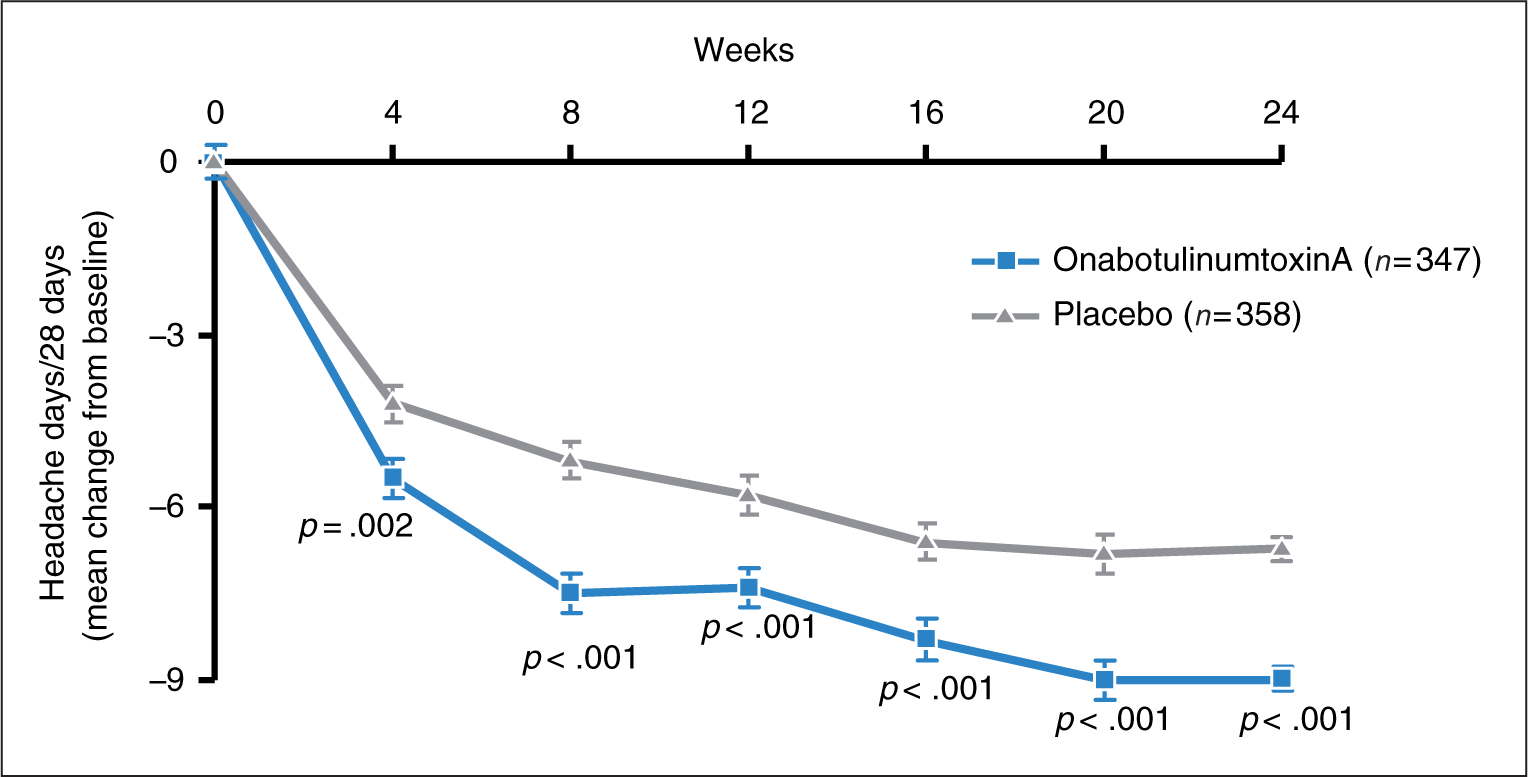
Primary endpoint: headache days. Statistically significant decreases from baseline with a between-group difference favouring onabotulinumtoxinA were observed for the primary endpoint, mean change from baseline in frequency of headache days from the first post-treatment study visit (week 4) through week 24 (−9.0 days onabotulinumtoxinA vs. −6.7 placebo, p < .001; 95% confidence interval [CI] [−3.25, −1.31]) (Figure 3; Table 2).
Patient disposition. PREEMPT 2 primary endpoint: mean change from baseline in headache day frequency. Headache day frequency at baseline: 19.9 ± 0.2 onabotulinumtoxinA vs. 19.7 ± 0.2 placebo, p = .682. All data presented as mean ± standard error. Efficacy of onabotulinumtoxinA at week 24 HIT, Headache Impact Test. *Primary efficacy endpoint. †Significant between-group differences favouring onabotulinumtoxinA. ‡International Classification of Headache Disorders, II 1.1 (migraine without aura), 1.2 (migraine with aura), 1.6 (probable migraine) (1). §Scores of 36–49 indicate little or no impact; 50–55, some impact; 56–59, substantial impact; ≥60, severe impact. 

Secondary efficacy variables. Large mean decreases from baseline with significant between-group differences favouring onabotulinumtoxinA were observed for all of the five rank-ordered secondary variables, starting at either the first or the second post-treatment study visit and continuing through all subsequent visits, including the week 24 primary time point (Table 2), in rank order: frequency of migraine days (p < .001), frequency of moderate/severe headache days (p < .001), total monthly cumulative hours of headache occurring on headache days (p < .001), proportion of patients with severe HIT-6 scores (p = .003) and frequency of headache episodes (p = .003). Large within-group improvements for mean change from baseline in frequencies of acute pain medication intake (all categories) were observed, although there were no significant between-group differences (p = .132). A post hoc analysis was conducted to identify potential patterns of intake by medication class (i.e., ergotamines, triptans, simple analgesics, opioids, combination analgesics, multiple analgesics). The result of this exploratory analysis was that the frequency of triptan intakes was significantly reduced from baseline in the onabotulinumtoxinA compared to the placebo group at week 24 (−3.0 onabotulinumtoxinA vs. −1.7 placebo, p < .001).
Headache impact on functioning and health-related quality of life. OnabotulinumtoxinA-treated patients in PREEMPT 2 had improved functioning compared with placebo-treated patients, as demonstrated by a significant decline in disability (mean change from baseline in total HIT-6 score) at all time points (p < .001). OnabotulinumtoxinA treatment significantly improved quality of life (p < .001) at weeks 12 and 24 compared to placebo as measured by each of the three MSQ role function domains—restrictive, preventive, and emotional—and as also measured by mean change in HIS at all time points (p < .001).
Safety and tolerability
Summary of overall adverse events reported in the 24-week, double-blind phase

AEs, adverse events. *Discontinuations during double-blind or open-label phases due to AEs witht onset during the double-blind phase.
Discussion
Previous clinical trials examining the prophylaxis of various headache disorders have generally excluded subjects with CM (14,32). Patients with CM are often considered treatment refractory and prone to overuse of acute pain medication to alleviate their symptoms (11). Within the PREEMPT patient population, two-thirds overused acute pain medication during the 28-day baseline period and approximately two-thirds had also tried previous headache prophylaxis medication without success due to ineffectiveness and/or intolerability. Even with these significant challenges faced by CM patients, treatment with onabotulinumtoxinA in PREEMPT 2 compared to placebo significantly improved multiple headache symptom measures in the primary and secondary efficacy variables, including headache-related disability.
There are few randomized controlled trials of CM treatment to support evidence-based approaches to the care of these patients (2). Available studies are limited by their diagnostic and eligibility criteria, sample size and failure to account for the use of prior and/or concomitant acute and preventive headache medications (2,3,14,15,38–41). The lack of large controlled trials, coupled with, until recently, the absence of operational diagnostic criteria and clinical trial guidelines, posed a significant challenge to efforts to identify endpoints and outcome measures that could accurately reflect the clinical benefit of any intervention in CM (1,2,34).
The analyses from PREEMPT 1 were considered along with other factors when it was decided, prior to patient unmasking in PREEMPT 2, to amend the PREEMPT 2 primary and secondary endpoints and analysis plan. With the results of PREEMPT 1 known prior to analysis of PREEMPT 2, there may be a concern regarding the independence of the findings of PREEMPT 2, thus inflating the overall type-1 error rate. To better control for such potential type-1 error for PREEMPT 2, a highly conservative Bonferroni adjustment was examined at the week 24 primary time point that modified the critical levels from .05 to .01 for p value comparisons still applied to each variable in the gate-keeping rank order. This adjustment was based on the change in the selection of the primary endpoint from among all five variables that were primary or secondary in PREEMPT 1, even though the high correlation among these variables means that the inflation of type-1 error was not as extreme as accounted for by such a correction. The week 24 efficacy results for the final selection of primary variable (frequency of headache days) and all five ranked secondary efficacy variables in PREEMPT 2, including headache episodes, the primary variable in PREEMPT 1, remained significant for onabotulinumtoxinA versus placebo. This result differs from the PREEMPT 1 trial. The week 24 mean change from baseline in frequency of headache episodes in the onabotulinumtoxinA group in both PREEMPT 1 and PREEMPT 2 was similar (i.e., −5.2 and −5.3, respectively). However, the mean change from baseline for the placebo groups in these trials was different (i.e., −5.3 and −4.6). It is possible that the significant baseline imbalance (p = .023) seen in PREEMPT 1 (higher mean number of headache episodes in the placebo group [13.4] than the onabotulinumtoxinA group [12.3]), which was not observed in PREEMPT 2 (placebo 12.0; onabotulinumtoxinA 12.7), confounded the post-treatment effect. When this baseline imbalance seen in PREEMPT 1 was treated as an anomaly, a post hoc analysis of headache episode counts (not change from baseline) found that onabotulinumtoxinA significantly reduced headache episode counts compared to placebo at weeks 4, 8, 20 and 24 (42).
Results from the analysis of acute pain medication intakes (all categories) demonstrated a mean reduction from baseline in intake across both placebo and onabotulinumtoxinA-treated groups. Since the intake of acute pain medication (all categories) was similar between the treatment groups, the significance of onabotulinumtoxinA treatment across multiple efficacy endpoints cannot be attributed to detoxification from acute pain medication during the study. Although this presents an apparent discrepancy between a reduction in frequency of headache days and acute pain medication intake, we do not believe this is a failure to report. Patient compliance rate for reporting daily symptoms using the electronic diary was high (>93%) and included a report on their acute pain medication intake. This discrepancy may have also been due to the complexity of analyzing all categories of acute pain medication intake together, as post hoc analyses showed significant reduction compared to placebo in use of triptans as a result of onabotulinumtoxinA treatment. This trial has another important finding. Clinical experience claimed in the past that patients overusing acute pain medication would not respond to preventive therapy. This was clearly not the case in the present study.
The headache-related burden and disability in individual patients with CM is multifaceted, encompassing headache frequency, duration and severity. At week 24, the onabotulinumtoxinA group experienced significant improvements over the placebo group across multiple symptom dimensions, including frequencies of headache days, migraine days and moderate/severe headache days; total cumulative hours of headache; headache episodes; and headache-related disability (HIT-6). The mean change from baseline in HIT-6 disability scores exceeded the established clinically meaningful between-group minimum difference of 2.3 (43). These differences in headache burden and disability resulted in significant improvements in functional performance and quality of life. As shown by these trials in this complex and disabled population, multiple outcome measures are useful to portray the multifaceted aspects of the disorder that contribute to a reduction in headache-related disability and improvement in headache-specific quality of life.
The majority of patients (≥89%) completed the 24-week, double-blind, phase of PREEMPT 2, indicating a favourable safety and tolerability profile. There were more AEs and treatment-emergent AEs reported in the onabotulinumtoxinA treatment arm than in the placebo treatment arm; however, these events were considered to be mild to moderate in severity and were short-lived, resulting in few discontinuations due to AEs. There was one serious AE reported that was considered treatment-emergent and that required patient hospitalization. These safety results are consistent with the known safety and tolerability profile of onabotulinumtoxinA, and no new safety events were observed.
There are several limitations to this study. Currently, there are no approved agents (34) for the prophylaxis of CM; therefore, the PREEMPT 2 study design did not include an active comparator. A large placebo response has been observed in migraine prophylaxis studies (34), which is consistent with our observations. In general, the placebo effect in pain trials such as this one can be attributed to expectation, Pavlovian conditioning and reduction of anxiety (44). Prophylactic migraine studies have indicated a high variability in rates of placebo response in comparison to acute migraine treatment studies (44). Additionally, parenteral procedures in pain trials are associated with an increased placebo response (44), perhaps due to high patient expectations when undergoing an injection treatment. Despite rigorous blinding practices, it is possible that patients could have become effectively unblinded in the course of this study. Injections of onabotulinumtoxinA into the forehead are associated with muscle relaxation. This physical response, possibly observed by some patients in the onabotulinumtoxinA treatment arm, may have inflated the reporting of the active response. Conversely, there was a lack of a nocebo response, in which patients in the placebo arm could have observed a parallel lack of physical response and became equally unblinded to the placebo. However, the presence of a placebo response suggests that this did not happen and that the blind was maintained. The placebo response could also be explained partly by regression to the mean or spontaneous improvement.
Population-based epidemiological studies have demonstrated that the study population in PREEMPT 2 is representative of the typical patient with CM seen in clinical practice (45). Therefore, physicians could expect similar results when treating patients with CM in the community. It should be noted that the efficacy of onabotulinumtoxinA demonstrated in the PREEMPT 2 study may not extrapolate to other patient populations who were specifically excluded from these trials, such as patients with episodic migraine, CTTH and secondary headache disorders.
The PREEMPT 2 study is the largest well-designed, placebo-controlled clinical trial in this severely disabled patient population. The results of PREEMPT 2 are consistent across multiple headache symptom measures, and demonstrate the superiority of onabotulinumtoxinA in efficacy, in reducing disability and in improving functioning, vitality and overall health-related quality of life. PREEMPT 2 confirms onabotulinumtoxinA (155–195 U) as a safe and effective treatment for adults with CM.
Disclosure
This study was sponsored by Allergan, Inc., Irvine, CA.
Conflict of interest statement
HCD has received honoraria for participation in clinical trials, contribution to advisory boards and/or oral presentations from Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, CoLucid, Bohringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grunenthal, Janssen-Cilag, Lilly, La Roche, 3M Medica, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brummer, Sanofi-Aventis and Weber & Weber. He has also received financial support for research projects from Allergan, Almirall, AstraZeneca, Bayer, GlaxoSmithKline, Janssen-Cilag and Pfizer. Headache research at the Department of Neurology in Essen, where HCD is professor, is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union. DWD has received honoraria from Allergan, Merck, Neuralieve, Coherex, Kowa, Minster, NeurAxon, H Lundbeck, Endo, Pfizer, Nupathe and MAP Pharmaceuticals, in addition to being a consultant to and on the advisory board of these pharmaceutical companies. He has also received funding from Advanced Neurostimulation Systems, St. Jude Medical Center and Medtronic. SKA received grants and research support from Advanced Bionics, Alexza, Allergan, Capnia, GlaxoSmithKline, MAP Pharmaceuticals, Merck, Ortho-McNeil, Neuralieve, NuPathe and Takeda. She is a consultant for Ortho-McNeil, Merck, GlaxoSmithKline, Allergan, Neuralieve, NuPathe and MAP Pharmaceuticals. She has also received honoraria from Merck, GlaxoSmithKline, Kowa, NuPathe and Ortho-McNeil. CCT, RED and MFB are employees of Allergan, and own stock in the company. SDS and RBL have received honoraria and research funding from Allergan, in addition to being consultants to and on the advisory board of Allergan.
Footnotes
Acknowledgements
The authors thank the patients who participated in the studies and their families. We also thank the regional lead coordinating investigators and Allergan Global Clinical Operations for their support, principal investigators and their staff for conducting the trial and current and past BOTOX® Headache Development Advisory Board Members for their input and intellectual contributions to the development and advancement of the BOTOX® clinical development program. We would like to thank Allergan, Inc., for funding IntraMed Educational Group, New York, NY, USA, to provide editorial support in the preparation and styling of this manuscript.
Werner J. Becker, Andrew Blumenfeld, F. Michael Cutrer, Astrid Gendolla, Christine Lay, Vince Martin, Maja Relja, Jack D. Schim, Timothy R. Smith, Thomas W. Ward and Paul Winner.
Jacqueline Abbas, Salim Ali, Billy Bahia, Steve Balyakin, Anne Blanco, Terry Boodhoo, Antoinette Brown, Roger Chan, Raymona Chin, George Demos, Christopher Dick, Dawn Drake, Ilana Fainaru, Laurie Fitch, Melissa Gilbert, Rozanno Gonzales, Amy Ho, Diane Hook, Courtney Huynh, Xiaofang Lei, Kimberlee Marruffo, Mary Ann Miller-Messana, David Nguyen, Dolly Nguyen, Shobhal Patel, Mimi Phomma, Stacey Quimpo, Anna Rask, Carminna Reyes, Mary Ann Robinson, Julie Roth, Regina Shin, Mai Sirimanne, Beatrix Taylor, Anisse Tse, Amanda VanDenburgh, Kathy Vu and Caroline Writer.
Reto Agosti, Werner J. Becker, Tanya Bilchik, Eugene Blonsky, Steven Bowman, Paul Brownstone, Guy P. Boudreau, Roger Cady, Albert Cook, Hans-Christoph Diener, Keith Edwards, Arthur Elkind, Michael Elliott, Giles Maxwell Erlington, Stefan Evers, Gerald Ferencz, Julia Files, Alan Finkel, Ian Finkelstein, Benjamin Frishberg, Nestor Galvez-Jimenez, Marek Gawel, Peter Goadsby, Hartmut Gobel, Edward Goldstein, Brian Grosberg, John Hinton Jr, Patrick Hogan, Davor Janculjak, Constance Johnson, Milind Kothari, John Claude Krusz, Klaus Langler, Sylvia Lucas, Paul Mazzeo, Donald McCarren, Tamara Ann Miller, Michael Morse, Joseph Nicolas, Pari Nikpey, C. Phillip O’Carroll, Volker Pfaffenrath, Neil Pugach, Maja Relja, Uwe Reuter, Marcia Ribeiro, Perry Richardson, Michael Rivner, Sara Sacco, Peter Sandor, Joel Saper, Lucian Sitwell, Timothy Smith, Claudia Sommer, Egilius Spierings, Michael Stein, Andreas Straube, Frederick Taylor, Martin Taylor, Stewart Tepper, Ira Turner, Martin Veilleux, Ranka Vurdelja, Jerome Walker, Joseph Weissman, Stuart Weatherby, Maria-Carmen Wilson and James Wymer.
Sheena K. Aurora, Werner J. Becker, Andrew Blumenfeld, Rami Burstein, Carl Dahlof, Hans-Christoph Diener, David M. Dodick, Anthony W. Fox, Fred G. Freitag, Richard B. Lipton, Ninan T. Mathew, Jane Osterhaus, Julio Pascual, Joel R. Saper, Stephen D. Silberstein, K.M.A. Welch and Paul Winner.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
