Abstract
Clinical trials of migraine therapy often require treatment when migraine pain intensity is moderate or severe, but many physicians find this practice artificial and patients often prefer to treat while pain is mild. This randomized, placebo-controlled study assessed the efficacy of zolmitriptan 2.5 mg in treating migraine while pain is mild, in patients who typically experience migraine attacks that are initially mild, but progress to moderate or severe. The intent-to-treat population comprised 280 patients (138 zolmitriptan; 148 placebo), with mean MIDAS grades of 29.6 (zolmitriptan) and 27.6 (placebo). Zolmitriptan 2.5 mg provided a significantly higher pain-free rate at 2 h (43.4% vs. 18.4% placebo; P <0.0001). Significantly fewer zolmitriptan patients reported progression of headache pain to moderate or severe intensity 2 h postdose (53.7% vs. 70.4% placebo; P < 0.01), or required further medication within 24 h (46.4% vs. 71.1% placebo; P <0.0001). The efficacy of zolmitriptan was more pronounced in patients treating during the first 15 min following pain onset. Adverse events were reported in 31.2% of patients treated with zolmitriptan (vs. 11.3% for placebo), and the incidence was lower in patients who treated early after attack onset. Zolmitriptan provides high efficacy when treating migraine while pain is mild, with the clinical benefits being more pronounced when treating early after migraine onset.
Introduction
Development of the 5-HT1B/1D receptor agonists (‘triptans’) for treatment of migraine over the past 10 years has revolutionized acute migraine therapy. Triptans are now considered to be the mainstay of therapy in many migraineurs. An extensive clinical trial program has proven zolmitriptan to be an effective and well-tolerated second generation triptan (1, 2).
Migraine clinical trials have traditionally assessed headache response (defined as a reduction in pain intensity from moderate or severe intensity at baseline to mild or no pain) (1, 3). This endpoint was chosen primarily to maximize the statistical power of the studies by incorporating 2-point improvements in headache intensity and to avoid treatment of nonmigraine headache such as tension-type headaches (4), and has become the basis for regulatory approval for new triptan drugs. However, in the clinical setting many physicians find this practice artificial and patients often prefer to treat attacks while the pain is mild, rather than waiting for pain to intensify. Indeed, International Headache Society (IHS) guidelines state that in principle, treatment in migraine clinical trials should be started as early as possible, to mimic clinical practice (5).
One open-label zolmitriptan study (6) and retrospective analyses of sumatriptan data (7, 8) showed that treating migraine in the mild phase was associated with higher pain-free rates than treating when pain intensity was moderate or severe. In two prospective, placebo-controlled multicentre studies with sumatriptan, pain-free efficacy rate was increased by treating while pain was still mild (9). However, to date, data from these studies have only been published in abstract format and thus details of the methodology are not known. The trial described here was prospective, rigorously designed solely to address the treatment of migraine in the mild phase, and subsequently had strict inclusion criteria to maximize its scientific value.
Until now, the efficacy of zolmitriptan in the treatment of migraine while pain is mild has not been investigated in a controlled, prospective study. This randomized, placebo-controlled study assessed the efficacy of oral zolmitriptan 2.5 mg compared with placebo in treatment of disabling migraine while pain is mild. The study also assessed other benefits of early intervention, such as whether treatment with zolmitriptan could prevent or reduce progression to more severe migraine and the impact of migraine on patients’ normal activities. Importantly, the current study also recorded the time to treatment from migraine onset, in order to study the influence of treating early.
Methods
Patients
Patient inclusion criteria were:
aged 18–65 years;
an established diagnosis of migraine as defined by IHS criteria (10) with an age of onset <50 years;
ability to recognize a migraine headache while intensity is mild;
migraine attacks that are initially mild but progress to become moderate or severe;
experienced at least 1 migraine headache per month in the last 3 months;
ability to differentiate between migraine and nonmigraine headache;
moderate or severe migraine-associated disability, as assessed using the Migraine Disability Assessment Scale (MIDAS) questionnaire (11).
The MIDAS questionnaire records time lost due to migraine from employment or school, household work, or family and social activities. Moderate or severe disability corresponds to a MIDAS disability grade of III (MIDAS score 11–20) or IV (MIDAS score 21+).
Exclusion criteria included:
history of basilar, opthalmoplegic or hemiplegic migraine headache;
frequent nonmigraine headaches;
history of, or symptoms suggestive of, ischaemic heart disease or other vascular disease including Prinzmetal angina, Wolff–Parkinson–White syndrome or other cardiac accessory conduction pathways or arrhythmias;
systolic BP ≥ 150 mmHg or diastolic BP ≥ 95 mmHg;
use of monoamine oxidase type A inhibitors, methysergide or methylergonovine;
breast-feeding or pregnancy;
hypersensitivity to zolmitriptan or commonly used pharmaceutical excipients;
severe hepatic impairment; participation in another clinical trial.
Study design and treatments
This multicentre, randomized, double-blind, placebo-controlled, parallel-group study was conducted in accordance with the Declaration of Helsinki (12). The protocol was approved by a recognized ethics committee pertinent to each study location and patients provided written informed consent before participating in the trial.
Eligible patients were randomized on a 1 : 1 basis to receive oral zolmitriptan 2.5 mg or matching placebo. Patients were allocated to treatment in balanced blocks of 4 within each trial centre.
Patients were instructed to treat a single migraine headache during the mild intensity phase, within 4 h of onset; the time to treatment was recorded. For persistent or recurrent headache of moderate to severe pain intensity, participants could take a second dose of the same trial treatment or an approved escape medication > 2 h after the first trial treatment. Patients returned to the clinic within 2 weeks of treating a migraine or 6 weeks after trial medication was dispensed, whichever was the earlier.
Efficacy assessment
Data for efficacy assessments were recorded on diary cards. Headache intensity was measured on a standard 4-point rating scale (0 = none; 1 = mild; 2 = moderate; 3 = severe). The primary efficacy measure was pain-free rate (i.e. ‘no pain’) at 2 h after the first dose of zolmitriptan 2.5 mg or placebo. Secondary endpoints included:
pain-free rate at 30 min, 1 h and 1.5 h;
nonprogression of headache within 2 h postdose and 12 h postdose;
use of further medication within 24 h;
patients (%) with moderate or severe headache pain at 30 min, 1, 1.5 and 2 h;
use of additional medication within 24 h;
percentage of patients with moderate or severe headache pain at 30 min, and 1, 1.5 and 2 h;
ability to perform normal activities at 30 min and 1, 1.5 and 2 h postdose;
reduction in the impact of migraine headache on usual activities at 2 h postdose;
associated symptoms (nausea, photophobia and phonophobia) recorded in diary cards.
Non-progression of headache at 2 h was defined as headache intensity that remained mild (using the 4-point rating scale above) or improved to ‘no pain’ at 2 h after dosing, and had not progressed to moderate or severe pain at any of the previous timepoints. Non-progression at 12 h was defined as headache intensity remaining mild or improving to ‘no pain’ at 2 h, and not worsening in the 12 h after treatment, and the patient not taking further medication (escape medication or a second dose of the study medication).
Safety assessment
Safety assessments comprised standard screening assessments (medical history, physical examination, blood pressure, height and bodyweight) and reporting of adverse events. A detrimental change in condition was recorded as an adverse event only if occurring within 24 h after taking the study treatment. However, all serious adverse events that occurred during the study were recorded, irrespective of timing. The study population included all randomized patients who took study treatment.
Statistical analyses
Binary endpoints were those that required a yes/no response (e.g. pain-free response at 2 h), and were analysed using logistic regression. All time-to-event endpoints were analysed using Cox's regression model (proportional hazards model). All tests of significance were 2-sided and used the 5% level of significance. Endpoints were analysed for the intent-to-treat (ITT) population, which included all patients who treated a migraine attack with study treatment and completed at least one efficacy assessment. In addition, a per-protocol population was assessed for the primary endpoint only.
Sample size
The study was sized to detect a clinically meaningful difference between treatments in the primary endpoint (pain-free response at 2 h). Sample size was estimated using a χ2 test of equal proportions, with a 5% two-tailed significance level and 90% power. In order to detect a 20% difference between zolmitriptan and placebo, 128 patients per group (256 in total) were required to treat one migraine attack with the study treatment.
Results
Study participants
A total of 302 patients from 24 centres in 3 countries (USA, France and Norway) were randomized to one of two treatment groups: zolmitriptan 2.5 mg, n = 150; placebo, n = 152. Twenty-two patients did not treat a migraine attack during the study period. Hence, 280 patients received study medication and were included in the safety population (zolmitriptan 2.5 mg, n = 138; placebo, n = 142). No patients were excluded from the ITT population therefore this was identical to the safety population. The two treatment groups were well balanced with respect to demographic and baseline migraine characteristics (Table 1). In particular, both groups showed considerable headache-related disability, with high mean MIDAS scores (29.6 for zolmitriptan 2.5 mg and 27.6 for placebo).
Patient demographic and migraine characteristics (ITT population)‡
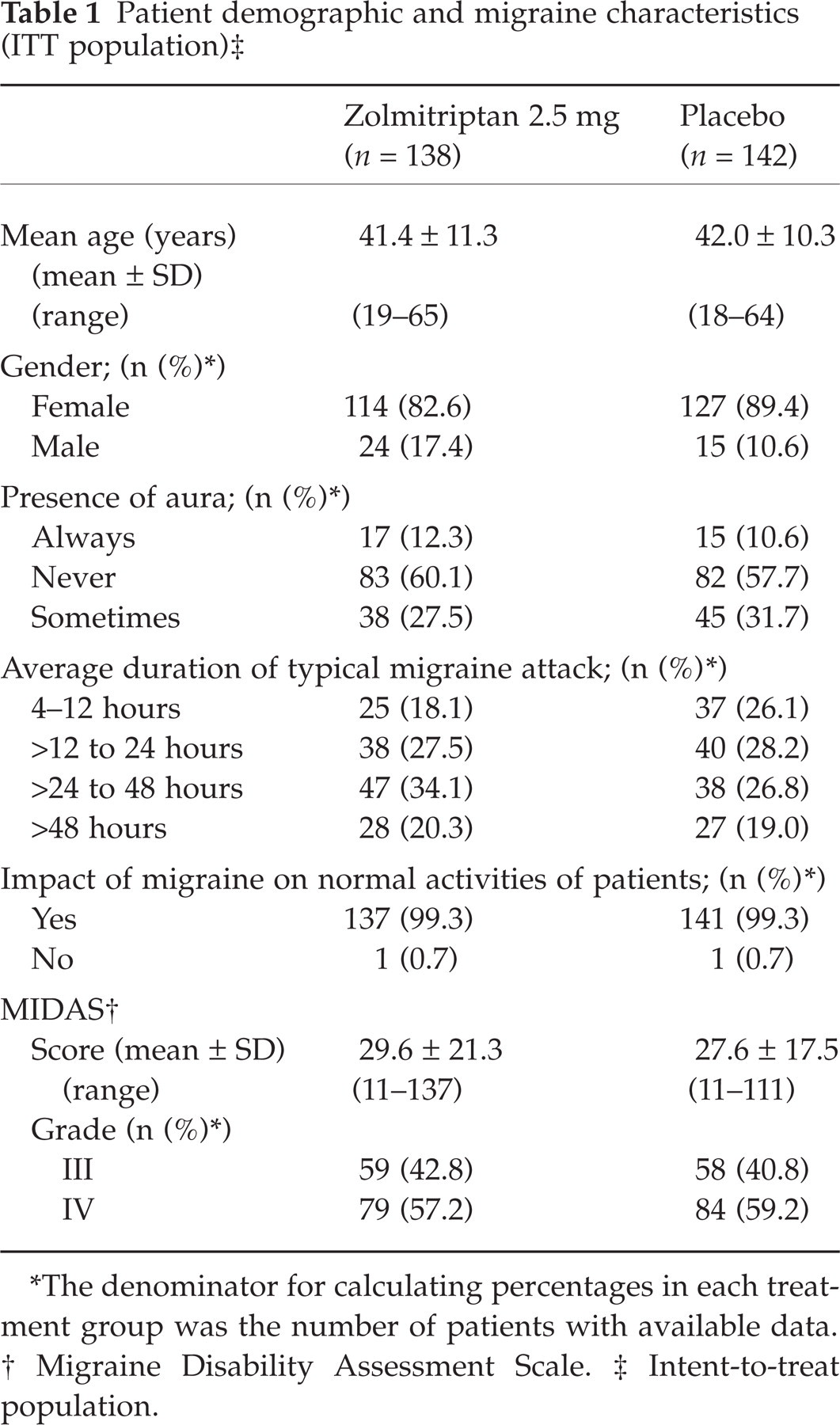
∗The denominator for calculating percentages in each treatment group was the number of patients with available data.
†Migraine Disability Assessment Scale.
‡Intent-to-treat population.
The proportion of patients withdrawn from the study was similar in the two treatment groups; 12 (8%) in the zolmitriptan group and 14 (9.2%) in the placebo group. The most common reason for withdrawal from the study was ‘protocol noncompliance’ (8 and 12 patients in the zolmitriptan and placebo groups, respectively; in most cases, this was due to patients not having a migraine attack within 6 weeks of randomization). Other specific reasons for withdrawal included ‘patient was lost to follow-up’ and pregnancy. Four patients in the placebo group were withdrawn after treatment and were therefore included in the ITT and safety populations. The per-protocol population comprised 126 patients in each treatment group; 12 and 16 patients from the zolmitriptan and placebo groups, respectively, were excluded from the per-protocol population. The most common reasons for exclusion were failure to treat a migraine while of mild intensity (n = 11) and use of analgesics less than 6 h prior to treatment (n = 9).
Time to dosing
Most patients in the study treated their migraine attack early. In the ITT population, 38.4% and 39.4% of patients in the zolmitriptan 2.5 mg and placebo groups, respectively, treated within 15 min and 54.3% and 56.3%, respectively, treated within 30 min
Pain-free rate
Significantly more patients in the zolmitriptan 2.5 mg group were pain-free at 2 h compared with the placebo group (43.4% vs. 18.4%; odds ratio (OR) 3.28, 95% CI 1.90–5.66, P < 0.0001) (Fig. 1). The corresponding values for the per-protocol population were similar (42.7% vs. 20.0%; OR 2.87, 95% CI 1.63–5.05, P < 0.001). Hence, the per-protocol population was not investigated in further detail.

Pain-free response rates at 2 h for all patients and for those patients who treated within 15 min of migraine onset. □ Zolmitriptan 2.5mg;  Placebo. ∗P < 0.001 vs. placebo; †P < 0.001 vs. placebo.
Placebo. ∗P < 0.001 vs. placebo; †P < 0.001 vs. placebo.
Further analysis showed that the difference in pain-free rate at 2 h between zolmitriptan 2.5 mg and placebo was even greater in patients who treated an attack within 15 min (57% vs. 20%, P < 0.001; Fig. 1). At the earlier timepoint of 90 min, significantly more patients in the zolmitriptan 2.5 mg group were pain-free compared with the placebo group (29.4% vs. 14.2%; OR 2.48, 95% CI 1.36–4.52, P < 0.01).
Patients taking zolmitriptan 2.5 mg within 15 min showed even higher pain-free rates compared with placebo at all time points measured up to 2 h: 43.4% vs. 16.4% (P < 0.01) at 90 min, 18.9% vs. 10.9% at 1 h and 5.7% vs. 1.8% at 30 min.
Headache progression
There was a significant difference in favour of zolmitriptan in the proportion of patients whose mild pain progressed to more severe intensity within 2 h after treatment (53.7% vs. 70.4%, P < 0.01) (Fig. 2). Within 12 h, the proportion of patients with mild pain that had progressed was higher in both groups, but a significant difference remained (59.0% vs. 77.0%, P < 0.01) (Fig. 2).

Progression of migraine within 2 and 12 h. □ Zolmitriptan 2.5mg;  Placebo. ∗∗P < 0.01 vs. placebo.
Placebo. ∗∗P < 0.01 vs. placebo.
This difference in favour of zolmitriptan in the proportion of patients with progression of mild pain within 2 h was also more pronounced in patients who treated within 15 min (48.1% vs. 71.2%, P < 0.05)
The proportion of patients requiring further medication within 24 h from initial treatment was significantly lower in the zolmitriptan group than in the placebo group (46.4% vs. 71.1%, P < 0.0001). Most patients using further medications took either escape medication (non steroidal anti-inflammatory drugs (NSAIDs), antiemetics, analgesics and sedatives were permitted) or a second dose of study medication. Again, additional benefits of treating within 15 min were observed; 41.5% of zolmitriptan recipients who treated early required additional medication compared with 69.6% of placebo recipients (P < 0.01).
Impact on normal activities
At 2 h after dosing, significantly more patients in the zolmitriptan group were able to perform normal activities compared with placebo recipients (68.4% vs. 50.7%, P < 0.01) (Fig. 3a). The impact of migraine on usual activities was reduced in 54.3% of the zolmitriptan recipients at 2 h after dosing. Patients who took zolmitriptan within 15 min of headache onset reported less impact of the migraine on usual activities than those treating later (Fig. 3b).

Reduction in the impact of migraine on usual daily activities at 2 h. (a) Ability to perform normal activities; (b) Comparing the reduction in impact on usual activities for all patients and those treated within 15 mins. □ Zolmitriptan 2.5mg; ░ Placebo. †P < 0.01 vs. placebo.
Tolerability profile
Adverse events were reported by 31.2% (43/138) of patients in the zolmitriptan group and 11.3% (16/142) of patients in the placebo group. The majority of these adverse events were transient, self-limiting and mild to moderate in intensity. Table 2 shows the adverse events that occurred in ≥ 2% of study participants.
Adverse events occurring in ≥ 2% of patients

Only one serious adverse event was reported. This patient experienced a hypersensitivity reaction nearly 10 h after taking zolmitriptan; however, the symptoms resolved after treatment with epinephrine.
Patients who treated early with zolmitriptan had lower rates of adverse events than those who treated later after onset of the attack (26%, 23%, 44%, 46% and 34% for time to treatment of 0–15, 15–30, 30–45, 45–60 and >60 min, respectively, after onset of attack).
Discussion
This is the first prospective, placebo-controlled study to investigate the efficacy of zolmitriptan therapy in the treatment of disabling migraine while pain is mild. This study recruited patients with migraine pain that typically began as mild, could be recognized as migraine, and subsequently increased in intensity to cause severe disability. The results of this trial demonstrate that in patients with headaches that are likely to progress to more severe and disabling attacks, treating with zolmitriptan during the mild phase, especially within the first 15 min following onset of pain, offers significant clinical benefits. Zolmitriptan produced significantly higher pain-free rates, and was shown to significantly reduce the impact of migraine on daily activities, compared with placebo. Treating early was also associated with a reduction in disability as measured by impairment of activity. Furthermore, zolmitriptan 2.5 mg significantly reduced progression of migraine pain from mild to more severe intensity compared with placebo.
Results of this study showed zolmitriptan 2.5 mg to provide significantly higher 2-h pain-free rates compared with placebo, with more than 40% of patients being pain-free. The therapeutic gain between placebo and zolmitriptan 2.5 mg was 25% for 2-h pain-free rates in this study, which is similar to that shown in previous studies of zolmitriptan 2.5 mg in the treatment of moderate or severe migraine attacks (29% and 20%) (13, 14). Achieving a pain-free state is one of the most challenging endpoints that clinicians and patients seek. This study was in line with IHS guidelines for migraine clinical trials, which encourages pain-free response as a primary endpoint in clinical trials (5).
While pain relief is clearly of most importance to migraineurs, there is also a need to investigate additional outcomes that are meaningful and relevant to patients. The disabling effects of migraine and its associated symptoms are significant and cause patients to miss time from paid employment, social activities and household work (15, 16). The high level of migraine-related disability in the patients in the current study was evident by high MIDAS scores. Zolmitriptan significantly reduced the impact of migraine on daily activities and progression of migraine pain compared with placebo. Contrary to what might be expected, i.e. that early treatment results in recurrence and a need for further medication, zolmitriptan recipients experienced a reduced need for further medication within 24 h compared with placebo.
Previous retrospective analyses of sumatriptan studies have shown that treating migraine while pain is mild confers higher pain-free response rates than treating when pain is moderate to severe in intensity (7, 8). The researchers suggested that early intervention while migraine pain is mild might lead to improved treatment outcomes for patients. However, these studies did not specifically assess the time to treatment in patients with migraine in the mild phase. This study suggests that the terminologies for ‘early treatment’ and ‘treatment while pain is mild’ are not completely interchangeable. Rather, it is necessary to clarify whether the improved efficacy rates associated with treating migraine while pain is mild are a factor of baseline pain intensity alone, or whether the time to treatment is implicated.
The current study assessed not only the treatment of migraine while baseline pain is mild, but also the time to treatment. Interestingly, the majority of patients chose to treat an attack early; over half of patients treated within 30 min and over one third treated within 15 min. An analysis of patients who chose to treat very early (within 15 min) showed that the difference in 2-h pain-free rates in favour of zolmitriptan was even more pronounced (56.6% vs. 20.0%). Similarly, the difference in favour of zolmitriptan in the proportion of patients with nonprogression of headache was more marked in patients who treated very early, as was the difference in favour of zolmitriptan in the proportion of patients who reported reduced impact on their normal activities, and reduced need for additional medication. Furthermore, early intervention with zolmitriptan led not only to better efficacy, but also to a low incidence of adverse events. The tolerability profile for zolmitriptan seen in this study is consistent with previous studies (17–19).
In conclusion, zolmitriptan provides high pain-free response rates in the treatment of migraine while pain is mild, and can provide other significant patient benefits, such as reducing the progression to more severe migraine, reducing the impact that migraine has on normal activities, and reducing the need for additional medication. Furthermore, the clinical benefits of treating migraine while pain is mild are even more pronounced in those patients who choose to treat mild migraine early, highlighting the need for formulations that give patients the flexibility to treat as soon as migraine pain starts.
Footnotes
Acknowledgements
This study was supported by AstraZeneca Pharmaceuticals LP.
Study participants: Pauline Boulan-Predseil, Issy les Moulinhaux, France; Daniel H. Brune, Peoria, IL, USA; James M. Ferguson, Salt Lake City, UT, USA; Alvin E. Goldansky, Phoenix, AZ, USA; Anne Poole Jørgensen, Oslo, Norway; T. Kjærnli, Alesund, Norway; Jack A. Klapper, Denver, CO, USA; John Claude Krusz, Dallas, TX, USA; David Kudrow, Encino, CA, USA; Michel Lantéri-Minet, Nice, France; Christian Lucas, Lille, France; Dennis C. MKey, Mogadore, OH, USA; Ninan T. Mathew, Houston, TX, USA; Gary P. Plundo, Delmont, PA, USA; Sharon E. Prohaska, Kansas City, MO, USA; Sid Rosenblatt, Irvine, CA, USA; Øyvind Røsjø, Oslo, Norway; Thierry Soisson, Orleans, France; S. Bror Strandquist, Tonsberg, Norway; O. Bjorn Tysnes, Bergen, Norway; Jean Marc Visy, Reims, France; Kerri Wilks, Towson, MD, and Atlanta, GA, USA; Devin D. Zimmerman, South Bend, IN, USA; Carlos A. Zubillaga, Holiday, FL, USA.
The authors thank Andrée Rose and Marianne Wells for their assistance in preparing the manuscript.
