Abstract
In this international, multicentre, double-blind, placebo-controlled, single attack study, ‘triptan naive’ migraine patients were randomized in an 8:8:1 ratio to receive zolmitriptan 5 mg, sumatriptan 100 mg or placebo. The all-treated analysis included 1058 patients who took study medication. The primary endpoint, complete headache response, was reported by 39%, 38% and 32% of patients treated with zolmitriptan, sumatriptan and placebo, respectively, with no significant difference between treatment groups. In patients with moderate headache at baseline, complete response was significantly greater following zolmitriptan than after placebo (48% vs. 27%; P = 0.01); there was no significant difference between sumatriptan and placebo groups (40% vs. 27%). In patients with severe baseline headache (where a greater reduction in headache intensity is required for a headache response), there was no significant difference between any groups in complete headache response rates. For secondary endpoints, active treatment groups were significantly superior to placebo for: 1-, 2- and 4-h headache response (e.g. 2-h headache response rates: zolmitriptan 59%; sumatriptan 61%; placebo 44%; P < 0.01 vs. placebo); pain-free response rates at 2 and 4 h; alleviation of nausea and vomiting; use of escape medication and restoration of normal activity. The incidence of adverse events was similar between zolmitriptan and sumatriptan groups but was slightly lower in the placebo group. The lack of difference between active treatments and placebo for complete response probably reflects the high placebo response obtained, which is probably a result of deficiencies in trial design. For example, the randomization ratio may result in high expectation of active treatment. Thus, while ethically patient exposure to placebo should be minimized, this must be balanced against the scientific rationale underpinning study design.
Keywords
Migraine is a chronic episodic disorder with a prevalence of about 15–20% in women and 6% in men (1–4). A major advance in the treatment of migraine came with the development of agonists selective for the 5-hydroxytryptamine(5-HT) receptor subtype 1B/1D, which have also contributed to the understanding of migraine pathophysiology (5). 5-HT1B/1D agonists able to penetrate the intact blood–brain barrier, such as zolmitriptan are likely to act at several sites within the trigeminovascular system, including constriction of cerebral vasculature, both central and peripheral inhibitory effects on afferent transmissions within trigeminal pathways (6–8) and modification of serotonin-dependent central nervous system activity (9).
At the time that this study was conducted, sumatriptan was the only available 5-HT1B/1D agonist for migraine therapy. This study compared the efficacy and tolerability of zolmitriptan 5 mg with that of sumatriptan 100 mg (the recommended dose at the time of the study) in the acute treatment of migraine. The primary objective was a comparison between sumatriptan and zolmitriptan and the study was powered to show a 10% difference in efficacy between these active treatment groups. However, to confirm the efficacy of sumatriptan and zolmitriptan, a placebo group was also included. The size of the placebo group was small (8:8:1 randomization to zolmitriptan, sumatriptan and placebo) because of a reluctance of ethics committees in several European countries (where sumatriptan efficacy had been established) to approve an active comparator clinical trial with a high exposure of patients to placebo, for the acute treatment of migraine.
An effective migraine therapy must provide rapid relief of migraine headache and associated symptoms. To assess the clinical efficacy of migraine treatments, the International Headache Society (IHS) recommended the primary endpoint to be the number of migraine attacks resolved within 2 h of treatment and not recurring nor requiring escape medication within 24 h (10). However, during the clinical evaluation of sumatriptan, Glaxo formulated a primary endpoint which is still used routinely (11). This primary endpoint assessed headache response rate defined as a reduction in headache intensity from severe or moderate at baseline to mild or none. Although pain-free response and headache recurrence were evaluated separately they were not a component of the primary endpoint or combined as a composite endpoint as recommended by the IHS for attacks resolved (migraine attacks resolved within 2 h with no relapse within 24 h). Using the established headache response definition, a reduction in headache pain of only 1 point (from moderate to mild pain) is regarded as treatment success although such an effect may have little clinical relevance (12, 13). It is probable that a 2-point change in headache severity will be significantly more relevant to the patient than a 1-point change (12). Therefore, with increased knowledge of how to assess migraine it appears there is potential to improve the parameters measured. Indeed, the IHS is currently revising its guidelines for evaluation of migraine therapies. To address this issue, an attempt was made in this study to identify an efficacy parameter which gave a clinically meaningful outcome for the patient. Complete response within 2–24 h was therefore chosen as the primary efficacy variable for the present study. Part of this study has previously been reported (14).
Methods
Patients
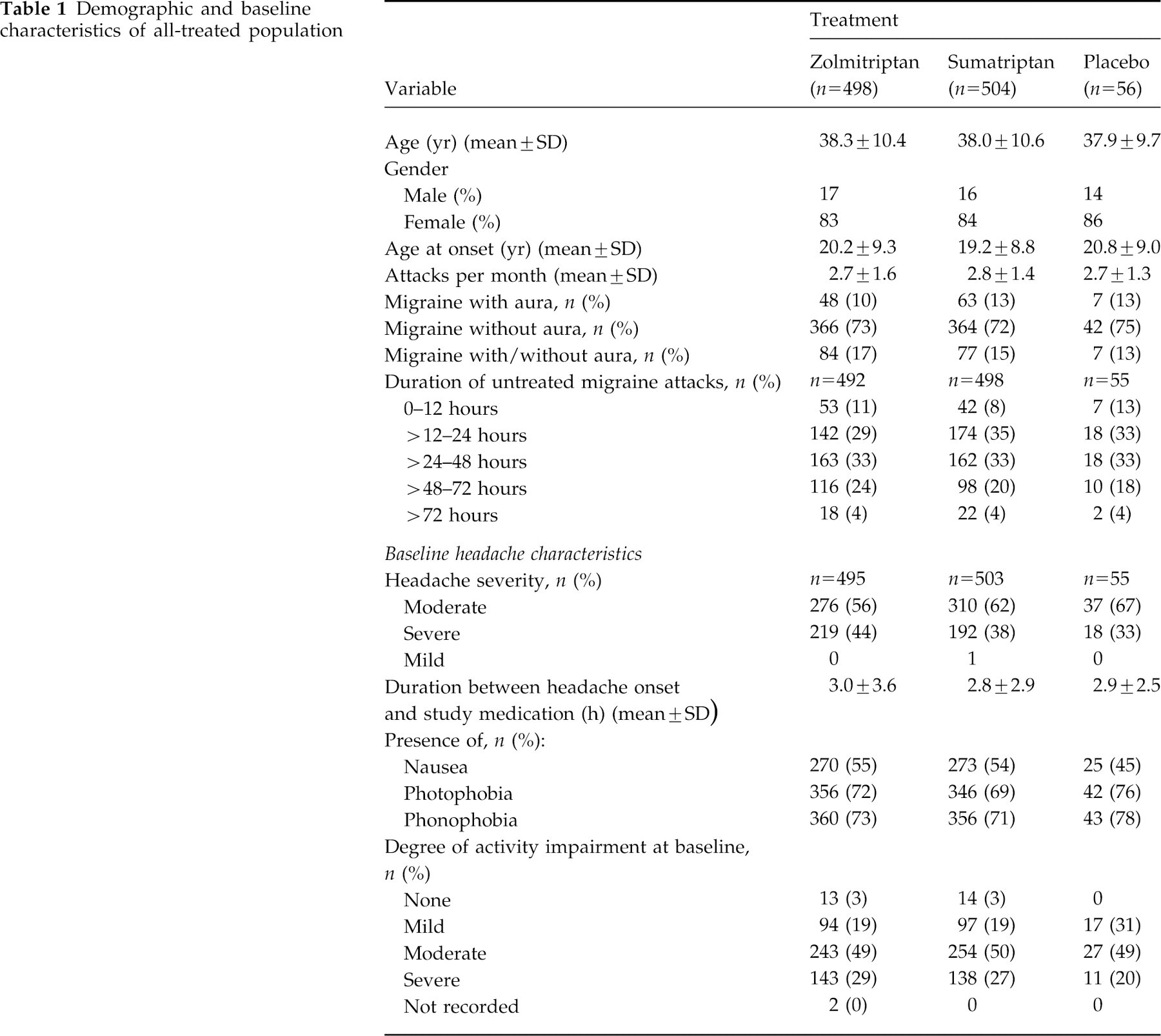
Male or female outpatients (18–65 yr) with an established diagnosis of migraine with or without aura (15) were recruited from 106 centres in 20 countries. Patients were eligible for inclusion if they had a history of migraine of at least 1 year's duration, an age at migraine onset of <50 yr and had experienced an average of 1–6 attacks per month for the 6 months preceding the study. In addition, patients were required to be able to distinguish migraine from other types of headache and to comply with study procedures including completion of a patient diary. Only patients who had never taken sumatriptan or zolmitriptan were eligible for inclusion. The rationale for this was to alleviate the potential for bias by patients having preconceived ideas/experience regarding the efficacy of the active treatments. Patients were excluded if they had received regular treatment during the month preceding the study with psychoactive drugs (e.g. hypnotics, benzodiazepines, neuroleptics, antidepressants) or drugs with a clinically important action at a 5-HT receptor. However, patients were permitted to use medications such as β-blockers, calcium channel blockers (excluding flunarizine), clonidine and valproic acid for migraine prophylaxis. Patients were also excluded from the study for the following reasons: history of drug or alcohol abuse, diagnosis of basilar, ophthalmoplegic or hemiplegic migraine, history or symptoms of ischaemic heart disease or other vascular disease, angina pectoris, Wolff-Parkinson–White syndrome or other cardiac accessory conduction pathways or dysrhythmias, uncontrolled hypertension, any medical or psychiatric condition which may have put the patient at risk or interfered with efficacy assessments, experience of non-migraine headaches on more than 6 days per month in the past 6 months, and participation in a clinical trial within 30 days of the study. In addition, women who were pregnant or lactating or who were not using adequate contraception were excluded from the study. All patients were required to provide written informed consent.
Study design
In this international, multicentre, double-blind, placebo-controlled study, patients were randomized in an 8:8:1 ratio to receive a single oral dose of zolmitriptan 5 mg, sumatriptan 100 mg or placebo, respectively. Each patient was supplied with two tablets to be taken simultaneously and blinding was maintained using a double-dummy technique. Patients were to treat a single migraine headache of moderate or severe intensity with or without aura and were required to take medication within 6 h of headache onset or of waking with headache. Patients were not permitted to take study medication if they had used an anti-emetic for treatment of the current migraine headache, an analgesic within the preceding 6 h or an ergot derivative within the previous 48 h. Escape medications could be taken 2 h after the study medication if migraine symptoms persisted. However, ergot derivatives were not permitted until 12 h after study medication. Sumatriptan could not be used as an escape medication.
Patients returned to the clinic within 1–2 weeks of treating a migraine headache, at which time patient diaries and medication wallets were collected.
The study received the approval of local Ethics Committees and was conducted according to Good Clinical Practice and in accordance with the Declaration of Helsinki.
Assessments
At screening, details of patients' medical history, headache history and concomitant medications were taken. Patients underwent a physical examination and 12-lead electrocardiogram (ECG). In addition, laboratory assessments including haematology, clinical chemistry and urinalysis were performed. Laboratory assessments and ECGs were repeated on return to the clinic. ECGs were reviewed by the investigator and then by an independent cardiologist who was blinded to the treatment assignment. The independent cardiologist's aim was to ensure uniformity of interpretation, determine whether ECGs showed any significant changes at follow-up and whether the change represented an ischaemic event.
Patients were withdrawn from the study if they withdrew consent, did not return for the follow-up visit, failed to treat a migraine headache within 10 weeks of randomization or failed to meet the admission criteria after randomization.
Efficacy assessments
Patients recorded migraine characteristics (e.g. severity, presence or absence of aura, nausea, photophobia, phonophobia) prior to and at 1,2 and 4 h after medication. The primary objective was to compare the complete headache response rates of zolmitriptan, sumatriptan and placebo in the acute treatment of migraine headache. Complete headache response was defined as a reduction in headache pain from moderate/severe at baseline to mild or no pain 2 h after taking study medication with no moderate or severe recurrence within 24 h. Use of escape medication was permitted 2 h post-dose if migraine symptoms were still present. Complete response was therefore additionally evaluated by counting patients who took escape medication as non-responders. Complete response was also analysed separately for patients with moderate and severe baseline pain.
Secondary objectives were to compare headache and pain-free response rates at 1, 2 and 4 h post-dose. A headache response was defined as the reduction of headache intensity from moderate or severe pain at baseline to mild or no pain (11). Pain-free response was a reduction of headache intensity to no pain. In addition, other secondary objectives were to compare: the proportion of patients whose migraine-associated symptoms were effectively treated, use of escape medication after 2 h, incidence of recurrence (a headache of moderate or severe intensity occurring within 24 h of treatment in a patient whose initial moderate/severe headache had reduced to mild/no pain 2 h post-treatment), meaningful migraine relief (a subjective global assessment of treatment response incorporating all migraine symptoms), time to meaningful migraine relief, and degree of activity impairment at 1, 2,4 and 24 h (rated as severe, moderate, mild or no impairment).
Tolerability
All adverse events were recorded in patient diaries and were assessed by the investigators for intensity, seriousness and relationship to study medication. In addition, any action taken as a result of the adverse event was reported.
Statistical analysis
For each efficacy endpoint, the treatments were compared to placebo using Mantel-Haenszel chi-squared tests, adjusted for country (country was used instead of centres since there were many small centres). Country by treatment interactions were assessed and were not statistically significant. Confidence intervals were calculated using Pratt's approximation. The all-treated and protocol-preferred populations gave similar results, and therefore results are given only for the all-treated population.
The trial was designed to detect a difference of 10% in complete response between patients receiving zolmitriptan and those receiving sumatriptan and a difference of 30% between patients receiving sumatriptan and those receiving placebo. Assuming that 30% of patients recruited do not complete the study, it was estimated that approximately 1170 patients would need to be recruited.
Results
A total of 1311 patients were enrolled, of whom 1058 took study medication and were included in the safety analysis. Of the 253 patients excluded from all analyses, 225 did not take study medication and 28 were lost to follow-up. The demographic and baseline characteristics of the patients who received study medication (all-treated population) are shown in Table 1. In the all-treated patient population, 59% patients reported headache of moderate intensity and 41% of patients reported severe headache. The mean duration between onset of headache and the first dose of study medication was 2.9 h.
Demographic and baseline characteristics of all-treated population
Efficacy
One thousand and fifty-eight patients were included in the all-treated patient population for the efficacy analysis. The primary efficacy parameter, complete response (2–24 h), was compared between all groups. Results are shown in Fig. 1. There was no statistically significant difference in complete response between groups. Analysis of complete response by baseline headache severity showed that of those patients in the all-treated population with moderate baseline headache, 48% in the zolmitriptan group, 40% in the sumatriptan group and 27% of the placebo group had a complete response (P = 0.01 for zolmitriptan vs. placebo) (Fig. 2). There was no significant difference in complete response of moderate baseline headache between sumatriptan and placebo. For patients with severe baseline headache, there was no significant difference in complete response rates between placebo (44%) and either active treatment (27% zolmitriptan, 35% sumatriptan, Fig. 2).

Complete response rates for the all-treated patient population by treatment group. Values are presented as mean and 95% confidence intervals.

Complete response rates for the all-treated patient population by headache severity and treatment group. Values are presented as mean and 95% confidence intervals+. □ = moderate baseline pain; ■ = severe baseline pain. *P = 0.01 vs placebo. +Baseline severity not recorded by 3 zolmitriptan-treated patients, 1 sumatriptan-treated patient and 1 patient treated with placebo.
Complete response was also analysed with respect to the use of escape medication. A total of 406 patients in the all-treated population used escape medication. A significantly greater proportion of patients in the placebo group (57%) required escape medication than in the zolmitriptan or sumatriptan groups (both 38%; P < 0.05). When those patients taking escape medication were classified as treatment failures, complete response rate was 37% (180/491), 39% (195/498) and 25% (14/55) for zolmitriptan, sumatriptan and placebo groups, respectively. Whether the use of escape medication was taken into account or not, the relative efficacy of the active treatment groups remained unaltered.
For the secondary efficacy parameters, headache and pain-free response rates and improvements in normal activities were recorded at 1-, 2- and 4-h time-points. Results are summarized in Table 2. Headache response rates with zolmitriptan and sumatriptan were significantly superior to those with placebo at all time-points assessed (P < 0.05 vs. placebo). The 2-h headache response rate was 59%, 61% and 44% for zolmitriptan, sumatriptan and placebo, respectively. Pain-free response rates were also significantly greater for both active treatments at 2 and 4 h post-dose. There was no significant difference between active treatments for either headache or pain-free response rates. The percentage of patients with no activity impairment increased with time for each treatment group. When those patients taking escape medication were counted as treatment failures, headache response rate at 4 h was 73% (322/441), 75% (329/436) and 38% (17/45) for zolmitriptan, sumatriptan and placebo groups, respectively. Corresponding pain-free response rates at 4 h were 51% (227/441), 51% (224/437) and 20% (9/45).
*P < 0.05 versus placebo.
+1- and 2-hour data not influenced by use of escape medication.
++One patient with mild baseline headache was included for the three time points in the sumatriptan group.
Headache recurrence was reported in a similar percentage of patients in all treatment groups. Thus, for patients reporting a headache response at 2 h, 26% (74/288), 28% (84/304) and 25% (6/24) of those in the zolmitriptan, sumatriptan and placebo groups, respectively, reported recurrence. The time to recurrence, however, differed markedly between active treatment groups and placebo. The median time to recurrence for zolmitriptan and sumatriptan groups was 15.4 and 15.3 h, respectively, and in the placebo group was 3.6 h.
Effects on non-headache symptoms were also recorded and are summarized in Table 3. The incidence of nausea at 4 h was significantly lower in the active treatment groups than in the placebo group (both P < 0.01), although this could have been influenced by the use of escape medication between 2 and 4 h. However, in patients who experienced nausea at 4 h and who did not take escape medication, the incidence was 10%, 13% and 38% for zolmitriptan, sumatriptan and placebo, respectively, and consistent with the data presented in Table 3. The incidence of vomiting was low at each assessment point but at 4 h was also significantly lower in the zolmitriptan (4%) and sumatriptan (3%) treatment groups than with placebo (18%; both P < 0.01 vs. placebo).
Effect of treatment with zolmitriptan 5 mg, sumatriptan 100 mg or placebo on non-headache symptoms of migraine +
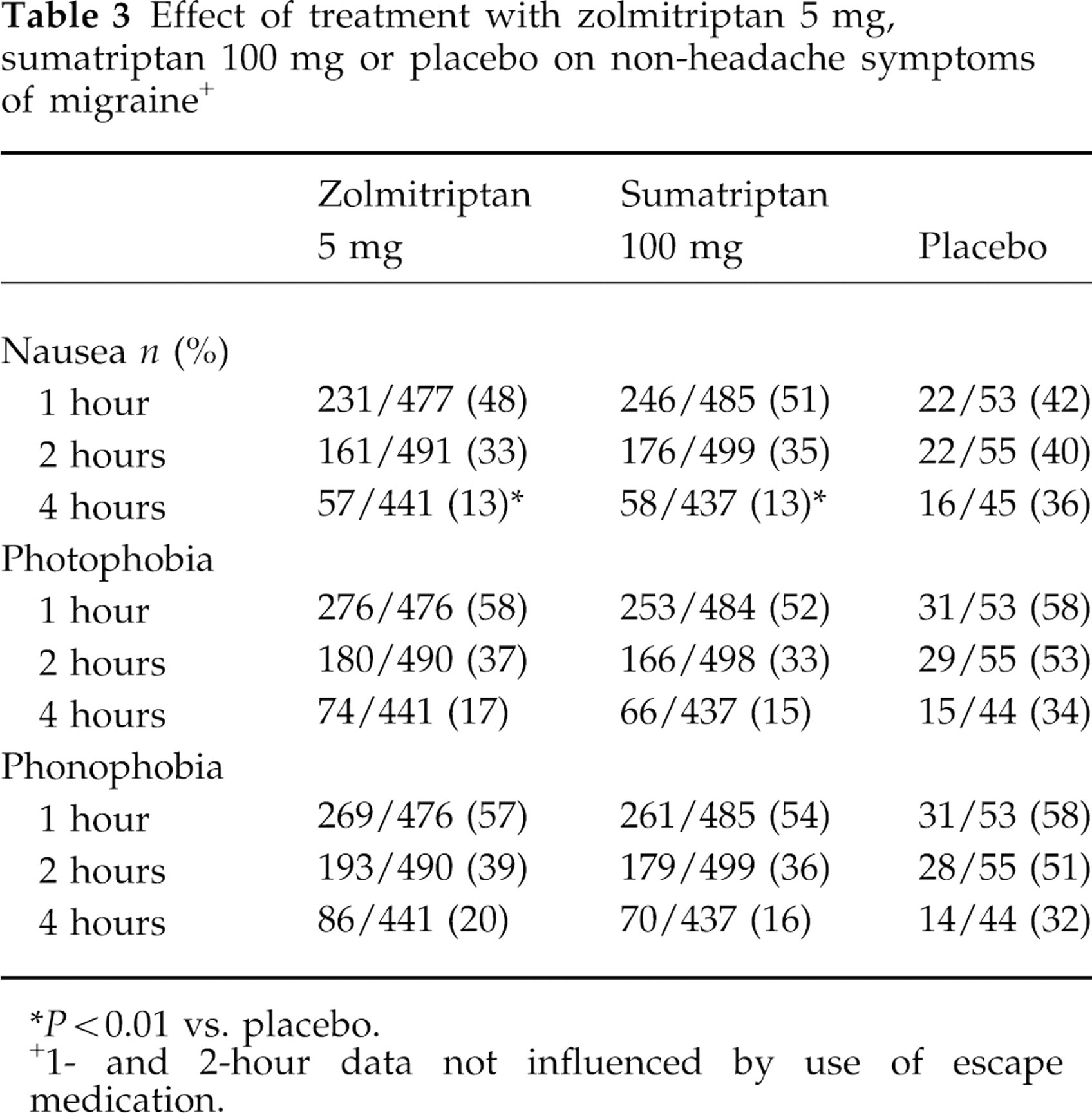
*P < 0.01 vs. placebo.
+1- and 2-hour data not influenced by use of escape medication.
Meaningful migraine relief was reported by 78% of zolmitriptan-treated patients, 81% of sumatriptan-treated patients and 71% of placebo-treated patients. The median time to meaningful migraine relief in the active treatment groups (2.0 h for both zolmitriptan and sumatriptan groups) was half that of the placebo group (4.0 h). Within 2 h of study drug administration (no escape medication used), meaningful migraine relief was reported by 52% (235/453) of zolmitriptan-treated patients, 53% (239/452) of sumatriptan-treated patients and 26% (14/53) of placebo recipients.
Tolerability
All safety evaluations were based on the available data from the 1058 patients in the all-treated population. For adverse events, 1039 patients had data available. At least one adverse event was reported by 58% (287/491) of patients treated with zolmitriptan, by 57% (279/492) of patients treated with sumatriptan and by 23% (13/56) of those treated with placebo. The most frequently reported adverse events are summarized in Table 4. The profile of adverse events was similar across treatment groups. Adverse events were predominantly of mild or moderate intensity. The incidence of severe adverse events was similar across all three groups (5–7%). The incidence of CNS-related adverse events was also similar between active treatment groups but was slightly lower in the placebo group. The most commonly reported CNS-related events were somnolence (zolmitriptan 8%, sumatriptan 6%, placebo 4%) and dizziness/vertigo (zolmitriptan 9%, sumatriptan 9%, placebo 2%).
Number of patients treated with zolmitriptan 5 mg, sumatriptan 100 mg or placebo experiencing adverse events* with an incidence of at least 5%

*Adverse events reported on 1039 patients with available data; patients can experience more than one adverse event.
Atypical symptoms thought to be possibly of cardiac origin occurred with an incidence of 0–2% across the three treatment groups. The most frequent of these events was palpitations which occurred with 2% incidence in each treatment group. Only one patient was considered by the independent cardiologist to have significant treatment-emergent electrical changes in ECG. This patient, who had received sumatriptan, showed ST depression considered indicative of an ischaemic episode.
Chest-related symptoms such as chest pain, tightness and heaviness ranged in frequency from 0 to 2%. Chest pressure was the most frequently reported of these symptoms, occurring with an incidence of 1%, 2% and 2% in the zolmitriptan, sumatriptan and placebo groups, respectively.
Of the adverse events reported in ≥5% of any treatment group, greater than 50% were experienced within 1 h of dose administration. Thus, 36% of zolmitriptan-treated patients and 35% of sumatriptan-treated patients experienced these adverse events within 1 h and this increased by a further 15% and 14% of patients, respectively, at 2 h and by a further 7% and 6% of patients, respectively, at 24 h post-dose. In the placebo group, the overall incidence of the most frequently reported adverse events increased from 11% at 1 h to 16% at 2 h and to 21% at 24 h post-dose.
There were no drug-related serious adverse events. Clinically significant changes in laboratory parameters occurred in two patients treated with zolmitriptan. In one patient, several biochemical parameters were outside the normal range at follow-up (8 days post-zolmitriptan administration). However, these were considered to be possibly associated with the use of a dextropropoxyphene/paracetamol/caffeine combination. In a second patient, serum aspartate aminotrans-aminase (AST) was elevated above the normal reference range at follow-up, although other biochemistry parameters including serum alanine aminotransferase, total bilirubin and alkaline phosphatase were all within normal ranges. A subsequent sample showed AST levels only slightly above the normal range.
Discussion
In this trial complete response rate (2–24 h) was chosen as the primary efficacy parameter for between-group comparisons of efficacy. Although this parameter did not show any significant difference between zolmitriptan and sumatriptan and between the two active treatments and placebo, several secondary parameters indicated benefit of zolmitriptan and sumatriptan over placebo. Furthermore, comparison of complete response rates for a subgroup of patients with headache of moderate intensity at baseline did show zolmitriptan to be significantly superior to placebo in the all-treated population. Secondary parameters that showed a significantly more favourable effect of active treatments compared with placebo were: headache response at 1, 2 and 4 h, the proportion of patients either pain free or experiencing nausea and vomiting, and the proportion of patients who reported improvements in their normal activities.
Although complete response is a composite efficacy measure of headache response without headache recurrence, it does not capture those patients who have obtained partial relief/benefit from treatment and the time to headache recurrence. Deferring headache symptoms for >8 h could have health and economic benefits for patients in work as well as benefits to other aspects of normal family life, for example less disruption of parental roles and less postponement of household duties or social activities. Median times from treatment to recurrence for both active treatments was approximately 15 h, considerably longer than placebo.
The discriminatory validity of complete response as a primary endpoint had not been evaluated before it was used in this study. As this endpoint does not take into account the clinical value of a response for those patients who experience treatment failure between 2 and 24 hours, it may fail to identify clinically relevant differences between treatments. Thus, in association with the small placebo group and the extremely wide confidence interval for complete headache response (95% CI 24.5, 53.6) for placebo, meaningful comparison between the active treatment groups and placebo in this study is difficult. As results from the secondary endpoints show statistically significant differences between active treatments and placebo, these may be more appropriate parameters for the differentiation of acute treatments.
Although there are experimental studies in animals and electrophysiological studies in man to suggest that zolmitriptan has a central mechanism of action (7, 9, 19), while animal studies suggest sumatriptan does not (20), the incidence of adverse events was similar between both active treatment groups. Importantly, there was no increase in the incidence of CNS adverse events in the zolmitriptan group relative to the sumatriptan group. The profile of adverse events was similar between active treatments and placebo; both active treatments were well tolerated.
It is likely that the unusually high placebo response observed in the present study is related to the design of the trial. The size of the placebo group was small resulting in large confidence intervals for efficacy parameters. The small size of the placebo population was based on the reluctance of the ethics committees of some countries (where efficacy of sumatriptan had been established) to include placebo groups in trials for migraine. However, these results suggest that if a placebo group is to be included it must be of sufficient size to allow meaningful between-group comparison.
This study was initiated during the early stages of clinical development of zolmitriptan when only one placebo-controlled dose-range finding study with 88 evaluable placebo patients was available (16). International Headache Society Guidelines recommended that drugs used for the acute treatment of migraine should be compared with placebo and when two presumably active drugs are compared then a placebo control should be included to test the sensitivity of the patient sample (10). The objective of this study was to compare the efficacy and safety of sumatriptan, zolmitriptan and placebo. Although the IHS guidelines recommend that when a new drug is being tested to show a difference from standard treatment (sumatriptan) there is no need to include a placebo group, given the limited zolmitriptan vs placebo data available, it was felt, despite the views of a number of ethics committees, that the value of a placebo group, albeit small, was justified. Furthermore, the study was a single attack study with escape medication available to patients 2 h after the study medication, if migraine symptoms persisted. Therefore the potential impact on the patients was deemed to be acceptable by the study designers. Had placebo and zolmitriptan performed as in the previous dose-range finding study (7% Complete Headache response vs 49% for placebo and zolmitriptan 5 mg respectively, 2:1 randomization) (16) then the power of the study would have been adequate. The results of this present study are also in contrast to those of another subsequent dose-range finding study (17) which demonstrated a significantly superior complete response rate for zolmitriptan 5 mg (45%) over placebo (7%). While zolmitriptan complete response in the present study (39%) is similar to that seen in the two reported dose-range finding studies (16, 17) the placebo response (32%) is approximately five times greater than that observed elsewhere.
Patients were informed of the randomization ratio and were therefore aware that there was a 16 : 1 chance that they would receive active treatment. Consequently, it is probable that they would have a high expectation of receiving active treatment, which is likely to produce a larger placebo response (particularly when only one attack is being treated), thereby reducing the difference between placebo and the two active treatments. In addition, although patients were naive to sumatriptan, it is likely that they were aware of the efficacy of sumatriptan compared with conventional treatments which may have further increased their expectation of a good response.
In conclusion, the lack of a significantly superior effect for the two study drugs, zolmitriptan and sumatriptan, compared with placebo for the primary endpoint is most likely due to the very small placebo group, which should be avoided in future trials. In this study, zolmitriptan 5 mg was shown to be at least as effective as sumatriptan 100 mg and as well tolerated, in the acute treatment of migraine.
Footnotes
Acknowledgements
We would like to thank the investigators who took part in this study (* manuscript review panel): A Black; G Donnan; A Zagami; G Herkes; G Lord (Australia); J Rainer; G Ladurner; U Baumhackl; W Amberger; D Klinger; E Deisenhammer; T Broussalis (Austria); J Schoenen; J Jacquy; P Louis (Belgium); J Gawel (Canada); J Olesen*; D Daugaard; A Korsgaard; K Kjaersgaard Pedersen; P Tfelt-Hansen*; K Jensen; PE Hansen; A Ladermann (Denmark); AE Kaasik; R Zupping* (Estonia); M Reunanen; E Säkö; M Haanpaä; H Havanka; I Rautakorpi; C Hedman; J Liukkonen; M Partinen; J-M Seppä; M Färkkilä; E Kinnunen; M Ilmavirta (Finland); H Massiou; H Petit; G Chazot; G Geraud*; P Henry; B Mihout; C Meyrignac; M Dujardin; A Laporte; A Giacomino; JF Wilmann; H Vilarem (France); HC Diener*; S Evers; W Paulus; W Kuhl; H Göbel; A Beckmann-Reinholdt; E Scholz; I Neu; A Peikert; A Straube; V Pfaffenrath*; H Klepel (Germany); C Karageorgiou; S Harmousi; I Milonas (Greece); JWM ter Berg; EACM Sanders; LJMM Mulder; JG Kok; J Van der Zwan; J Haan; M Ferrari* (Holland); G Nappi*; GC Manzoni; A Carolei; M Giacovazzo (Italy); G Arthur; R Frith (New Zealand); T Haldorsen; B Engelsen; A Edland; R Solhoff; O Rösjö; JP Larsen (Norway); A Prusinski; T Domzal; A Szczudlik (Poland); T Pavia; P Monteiro, L Cunha (Portugal); JM Lainez; H Liano; J Pascual Gomez; M Noya Garcia, R Leira; AN Granell; F Aranda, M Gracia Naya (Spain); C Dahlöf; J Boivie; C-G Cederberg; H Nilsson; L Edvinsson (Sweden); HR Isler (Switzerland); M Gross; A Dowson*; C Hawkes, RN Corston; RJ Abbott; M Johnson; P Rolan; TJ Steiner; L Findley; J Jestico; M Wilkinson (UK).
We would also like to thank Dr Gaelen Wagner (Duke University Medical Center, Core ECG Laboratory, Durham, North Carolina, USA) for providing independent assessment of all ECG recordings, and Esther Kito, Jane Bryttan and Luigi Giorgi (Glaxo Wellcome) for their contribution in study design, study implementation and data analysis of the study.

