Abstract
This phase II study investigated the efficacy, tolerability and dose-response relationship of oral zolmitriptan in the treatment of a single migraine attack in Japanese patients. A bridging analysis then assessed the validity of extrapolating western clinical data to these Japanese patients. In this multicentre, randomized, double-blind, placebo-controlled study, patients received a single dose of placebo or zolmitriptan 1, 2.5 or 5 mg. The primary endpoints were 2-h headache response and the tolerability of zolmitriptan. A statistically significant dose-response relationship was observed for the 2-h headache response (P = 0.003). The 2.5 mg group had significantly greater 2-h headache response than the placebo group (P = 0.032). The adverse event profile was similar to that reported in western patients, and no adverse events unique to the Japanese population were observed. The bridging analysis report confirmed similar efficacy and tolerability of zolmitriptan in Japanese and western populations. In the Japanese patients, the estimated response rates were 34.3%, 45.2%, 57.7% and 66.2% for placebo, and zolmitriptan 1, 2.5 and 5 mg, respectively, while in the western population the corresponding rates were 39.9%, 49.6%, 61.2% and 71.7%. Zolmitriptan is effective and well tolerated in the acute treatment of migraine in Japanese patients. The optimal dose was 2.5 mg, although the 5 mg dose may provide further benefit for some patients. The bridging analysis supports extrapolation of data from western to Japanese patients.
Introduction
Zolmitriptan is an orally active 5-HT1B/1D receptor agonist widely used in the acute treatment of migraine. An extensive clinical trial programme conducted primarily in Caucasian patients in the USA and Europe has established that zolmitriptan is consistently effective and well tolerated across varying patient subgroups and migraine types (1–4). The optimal dosage of zolmitriptan in these studies was found to be 2.5 mg, with 2-h headache response rates of 62–65% and a good safety profile.
The results from the above studies were considered in the preparation of the clinical study design and plan for evaluating the efficacy and tolerability of zolmitriptan in Japanese subjects.
Data from a concurrent study demonstrated that the pharmacokinetics of zolmitriptan in Caucasian and Japanese healthy volunteers were comparable (5). Furthermore, and consistent with previous pharmacokinetic and clinical efficacy studies in Caucasian subjects, this study demonstrated that zolmitriptan 2.5 mg was well tolerated in Japanese subjects. Therefore, as no clinically relevant differences between Japanese and Caucasian subjects have been identified to date, the possibility of using the present phase II dose–response trial as a bridging study, supporting the extrapolation of overseas clinical data to Japanese subjects, was considered.
Therefore, the objectives of this study were twofold. First, to investigate the efficacy, tolerability and dose–response of oral zolmitriptan in the acute treatment of a single migraine attack in Japanese patients. Secondly, to present the results of the bridging analysis report, directly comparing results from the Japanese population with those obtained in a similar phase II study previously conducted in a western population, and to determine whether extrapolation of western data to Japanese patients is valid.
Methods
Patients
Male and female patients aged 18–64 years, with an established diagnosis of migraine with or without aura (according to International Headache Society criteria), were included in the study. Eligible patients had an age at migraine onset of <50 years, a history of migraine symptoms for at least 1 year and experienced one to six migraine attacks per month in the 3 months prior to the study.
Exclusion criteria were: a history of basilar, ophthalmoplegic or hemiplegic migraine; non-migraine headaches reported on >10 days per month during the previous 6 months; ischaemic heart disease, dysrhythmias or cardiac accessory pathway disorders (e.g. Wolff-Parkinson–White syndrome); severe liver or renal impairment; uncontrolled hypertension; pregnancy or lactation; severe allergies or hypersensitivity to drugs; participation in a clinical study during the past 3 months; or required use of ergotamine preparations.
This study was designed and conducted in compliance with the ethical principles of good clinical practice and the Declaration of Helsinki, and all patients gave written informed consent before any study procedures were begun.
Study design and treatments
This was a randomized, placebo-controlled, double-blind, parallel-group study conducted in 81 centres throughout Japan. Participating investigators were neurologists specializing in the treatment of headache throughout Japan and all recruited a small number of patients (maximum 10), thus decreasing the potential for any centre-to-centre bias. After a screening visit to determine eligibility for the study, patients were randomized into a 1 : 1 : 1 : 1 ratio to receive zolmitriptan 1, 2.5 or 5 mg or placebo for the acute treatment of a single migraine attack.
Patients were requested to treat only moderate or severe migraine headaches with study medication. Before administration of the study treatment, use of other medication was restricted: acute treatment with ergotamine was not permitted within the previous 48 h, while analgesics, steroids, antidepressants, antiemetics, anticonvulsants or sedatives were not allowed in the previous 8 h. Patients could not take approved escape medication until completion of the 4-h post-dose assessment.
Assessments
Patient diary cards were used to record data on the treated migraine headache immediately before and at 0.5, 1, 2 and 4 h after administration of the study medication. The primary endpoints were the headache response rate at 2 h, defined as a reduction in migraine intensity from moderate or severe at baseline to mild or no pain, and an overall safety rating for zolmitriptan (the proportion of patients with ‘no problem’). Secondary outcome measures included headache response at 0.5, 1 and 4 h, pain-free response rate (reduction from moderate or severe at baseline to no pain) at 2 h, complete response (a headache response at 2 h and then no recurrence or use of escape medication within 24 h), improvement of associated headache symptoms, the incidence of headache recurrence, use of escape medication, the patients' global impression of treatment and the tolerability profile of zolmitriptan.
Enrolment, baseline and follow-up safety evaluations included laboratory assessments, vital signs and a 12-lead electrocardiogram. All adverse events occurring between dosing and follow-up were recorded on patient diary cards and converted to WHOART adverse event classifications.
Statistical analysis
The dose–response relationship for all endpoints was tested statistically using the Cochran-Armitage trend test at the one-sided 5% significance level. Headache response rates at 2 h and use of escape medication in the zolmitriptan 2.5 mg and placebo groups were also compared directly using the chi-square test at the two-sided 5% significance level. Treatment by centre interactions had been fully tested for and not detected in several previous studies, so this aspect was not explored in the current multicentre study. Efficacy analysis was performed on the evaluable case population (common expression in Japanese studies, equivalent to per protocol population), whereas the safety analysis was performed on the all-treated population. However, for the primary efficacy endpoint of 2-h headache response, the all-treated population was also analysed.
From the overseas data, it was assumed that 2-h headache responses of 30%, 45%, 60% and 65% could be expected in this study with placebo and zolmitriptan 1, 2.5 and 5 mg, respectively. A total of 200 evaluable patients (50 per group) would give over 90% power to detect a dose response across the four groups, using the Cochran-Armitage test (5% significance level). Moreover, at a significance level of 5%, 50 evaluable patients per group would give over 80% power to detect a significant difference between placebo and zolmitriptan 2.5 mg (the optimal dose in western populations), using a two-sided chi-squared test.
Bridging analysis
The methods for the bridging analysis report were defined prior to Key code break meeting for the Japanese dose–response study. Data re-analysis was then performed using the same statistical methods and population definitions as were used in the chosen phase II study in the Caucasian population, which had a close similarity in design (1). That study also used a randomized, double-blind, placebo-controlled design, and investigated the efficacy and tolerability of zolmitriptan 1, 2.5, 5 and 10 mg in the treatment of a single moderate or severe migraine. The primary endpoint was 2-h headache response, while secondary endpoints included safety, pain-free response, headache response at 0.5, 1 and 4 h, use of escape medication, and improvement of associated symptoms. The main differences between the two studies were that the western study also included a 10-mg dose and the optional use of a second dose. The primary endpoint for the bridging analysis was 2-h headache response.
To directly compare the two studies, two statistical approaches were used. First, a logistic regression model was fitted using dose as a categorical covariate and baseline headache intensity as a factor, allowing formal comparisons of the response rate to each dose of zolmitriptan with that of placebo, at a significance level of 5%. Secondly, a logistic regression model was fitted using dose as a continuous covariate and baseline intensity as a factor, allowing the dose–response curve across the range of doses to be estimated, at a significance level of 5%. The western study was one of the previously mentioned studies where existence of centre by treatment interactions was explored and not found, so such analyses were not repeated for the bridging analysis. Centre effects were also tested for, but found to not significantly contribute to the logistic regression models. Analyses were performed on the protocol-preferred populations of both studies. The adverse events of both studies were described in the bridging analysis report using modified COSTART terms.
Results
Patient characteristics
A total of 289 patients were randomized to treatment; of these, 58 patients were excluded because they did not take trial medication (Table 1). A further two patients were excluded because of improper acquisition of informed consent or incorrect administration of study medication. Therefore, the all-treated population consisted of 229 patients. An additional 27 patients were excluded from this group because of protocol deviations or violation of inclusion/exclusion criteria, resulting in an evaluable case population of 202 patients in which the assessment of efficacy was performed.
Patient accountability

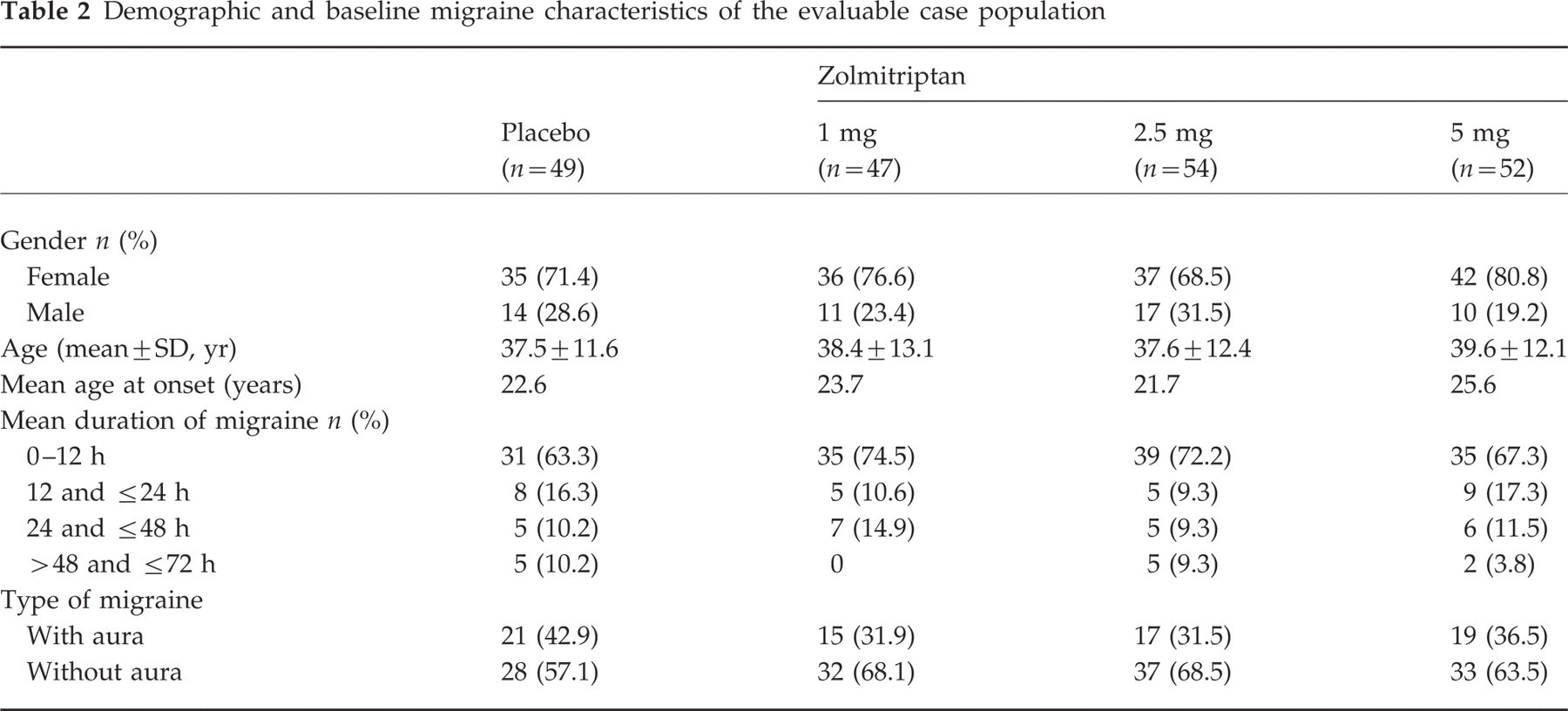
The demographic characteristics were similar across the four treatment groups (Table 2). Overall, 74% of patients were female and the mean age of the patient population was 38 years. The mean age at migraine onset was 23 years and the mean number of attacks per month was 3.0. The majority of patients typically experienced migraine without aura (64%) and associated symptoms of nausea (90%), vomiting (54%) and photophobia (56%). Phonophobia was experienced by 45% of patients. The majority of treated attacks were of moderate intensity (73%). The median time between onset of migraine and treatment with study medication was 2.8 h.
Demographic and baseline migraine characteristics of the evaluable case population
Efficacy
A statistically significant dose–response relationship was observed for the primary endpoint of 2-h headache response. Response rates in the evaluable case population were 53.3% (95% CI 37.9% to 68.3%), 55.6% (41.4% to 69.1%) and 65.4% (50.9% to 78.0%) for zolmitriptan 1.0, 2.5 and 5.0 mg, respectively, compared with 37.5% (23.9% to 52.7%) for placebo (Cochran-Armitage test P=0.003; Fig. 1). Similar response rates occurred in the all-treated population: 52.0%, 54.2% and 66.7% for zolmitriptan 1.0, 2.5 and 5.0 mg, respectively, compared with 34.5% for placebo (Cochran-Armitage test P<0.001). Comparison of response rates for zolmitriptan 2.5 mg and placebo in the all-treated population found a statistically significant difference (P=0.032); in the evaluable case population this comparison did not reach significance (P=0.068). Comparable results were observed when 2-h headache response rates were analysed by baseline migraine characteristics (Table 3). Zolmitriptan was also similarly effective regardless of the pre-treatment migraine duration (Table 3).
Number (%) of patients with a 2-h headache response according to demographic or baseline migraine characteristics


Headache response rates at 2 h after acute treatment of migraine with placebo or zolmitriptan 1, 2.5 or 5 mg in Japanese patients.
Secondary endpoints
For the secondary endpoints of headache response at 0.5 and 1 h, no statistically significant dose–response relationship was found (P=0.423 and P=0.283, respectively). At 4 h, the dose–response relationship was significant (P=0.032), with response rates of 75.6%, 76.5% and 78.3% for zolmitriptan 1.0, 2.5 and 5.0 mg, respectively, and 61.0% for placebo (Table 4).
Percentage of patients responding on secondary endpoints

∗ P=0.041 vs. placebo (chi-square test).
There was no significant dose–response relationship for the percentage of patients pain free at 2 h, although a numeric trend was evident. In addition, there was no consistent dose–response relationship for the improvement of associated symptoms, although at 4 h a significant dose–response relationship was observed for improvement in vomiting.
A significant dose–response relationship was observed for the percentage of patients experiencing a complete response at 24 h: 37.8%, 46.3% and 46.2% of patients receiving zolmitriptan 1, 2.5 or 5 mg, respectively, reported a complete response compared with 22.9% of placebo recipients (P=0.004; Table 4). There was a significant difference between the zolmitriptan 2.5 mg group and placebo in the use of escape medication (P=0.041). No significant dose–response relationship was observed for recurrence rates or for patients' impression of treatment, although a higher proportion of patients in the zolmitriptan 2.5 mg and 5 mg groups considered treatment to be ‘useful’ or ‘very useful’ compared with placebo or zolmitriptan 1 mg (Fig. 2).

Patient impression of treatment after acute treatment of migraine with placebo or zolmitriptan 1, 2.5 or 5 mg in Japanese patients. ▪ very useful,  useful,
useful,  no opinion,
no opinion,  not useful, never use again.
not useful, never use again.
Tolerability
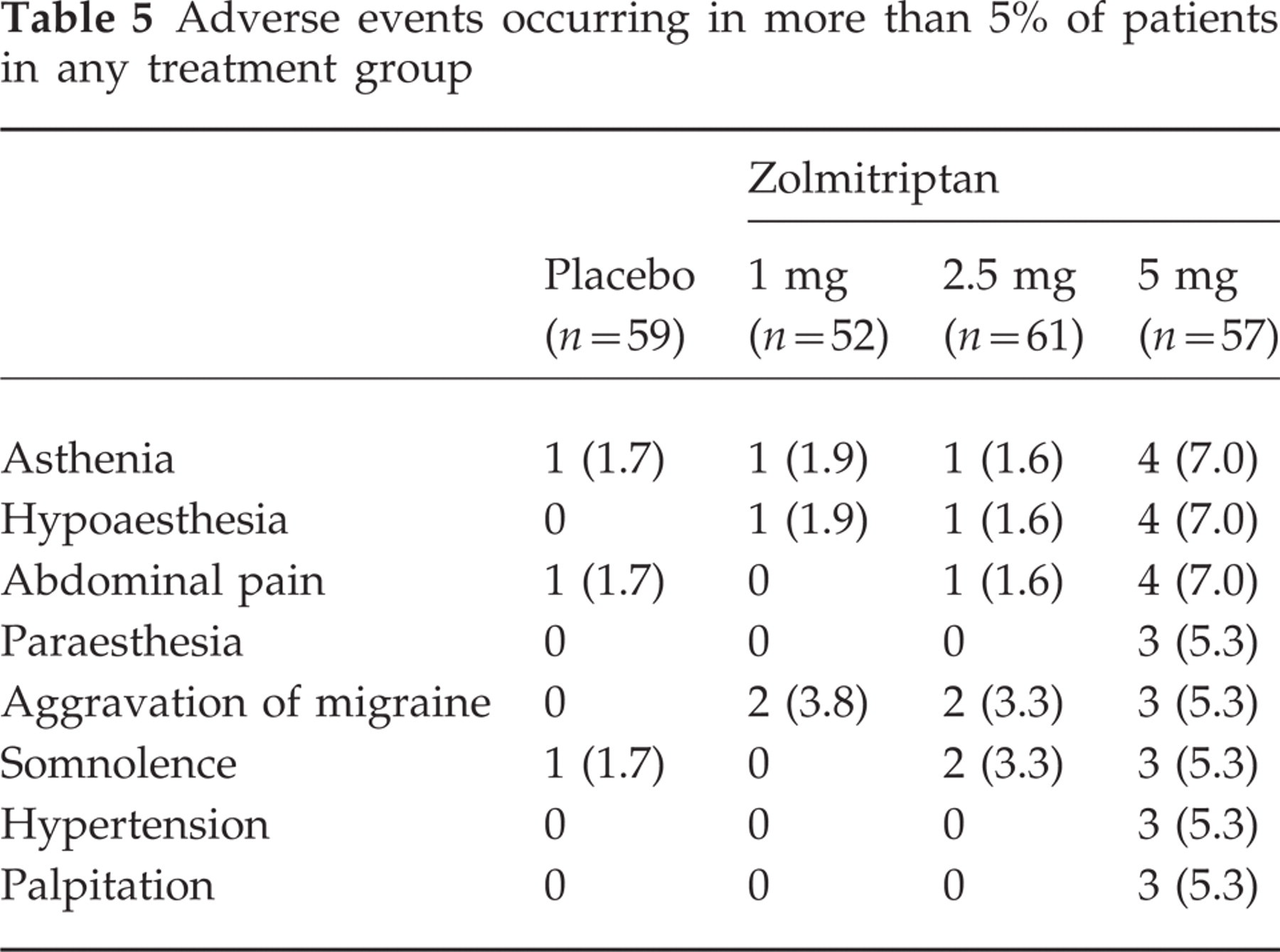
Zolmitriptan was well tolerated, with the majority of patients reporting ‘no problem’ with respect to tolerability (79.7%, 80.8%, 68.9% and 59.6% of patients in the placebo, zolmitriptan 1, 2.5 and 5 mg groups, respectively). Adverse events were predominantly mild or moderate in intensity, and all resolved without intervention. No adverse events unique to the Japanese population were observed. The most frequently reported adverse events overall were asthenia, hyperaesthesia, aggravation of migraine, somnolence, paraesthesia and abdominal pain (Table 5). A significant dose-dependent increase in the incidence of adverse events was observed (P<0.001), with 13.6%, 15.4%, 32.8% and 43.9% of patients who received placebo or zolmitriptan 1, 2.5 or 5 mg, respectively, reporting at least one adverse event. No serious adverse events were reported.
Adverse events occurring in more than 5% of patients in any treatment group
An analysis of the relationship between the severity of adverse events and patient impression of treatment showed that the presence of adverse events did not affect patient ratings of the acceptability of treatment. In the zolmitriptan 5 mg group, 46.7% of patients who did not experience any adverse events assessed the treatment as ‘useful’ or ‘very useful’, compared with 56.0% of those who experienced mild or moderate events. Similarly, in the 2.5 mg dose group, 43.6% of patients with no adverse events found the treatment ‘useful’ or ‘very useful’ compared with 38.9% of those who experienced mild or moderate adverse events.
There were no clinically significant changes in laboratory values or vital signs.
Bridging analysis
Study populations for the bridging analysis were redefined according to the inclusion criteria of the phase II study in the western population, and therefore include a slightly different number of patients. The protocol-preferred populations included 191 patients from the Japanese study and 751 patients from the western study. The all-treated safety populations were 231 and 855 patients, respectively. The demographics of the two study populations were similar: in the Japanese study, 74.3% of patients were female and the mean age was 38.6 years, while in the western study, 88.1% of patients were female and the mean age was 41.2 years. The migraine histories of the two groups were also comparable in terms of attack frequency, age at onset, and the proportion of patients who did not normally experience aura. However, the average duration of untreated migraines was shorter in Japanese patients, with 70.7% reporting migraines of less than 12-h duration. In comparison, the majority of migraines (60.1%) in western patients lasted between 24 and 72 h.
Comparison of the 2-h headache response rates in the Japanese study population with those of the previously studied western population showed similar response rates and confirmed the dose–response relationship (Table 6).
Estimated response rates using logistic regression models with quadratic trends and adjusting for baseline intensity

Logistic regression analysis of 2-h headache response, with dose as a categorical variable and baseline intensity as a factor, found a statistically significant difference in the Japanese study between zolmitriptan 2.5 mg vs. placebo (P=0.017) and zolmitriptan 5 mg vs. placebo (P=0.001), but not for 1 mg vs. placebo (P=0.102). A headache response at 2 h was 2.6 (95% confidence interval: 1.2–5.8) and 4.2 (95% CI: 1.8–10.0) times more likely in patients treated with zolmitriptan 2.5 and 5 mg, respectively, than in those treated with placebo. In western patients, all three doses were significantly more effective than placebo, and patients were 2.5 (95% CI: 1.5–4.2), 4.0 (95% CI: 2.5–6.4) and 4.4 (95% CI: 2.8–7.1) times more likely to have a headache response with zolmitriptan 1, 2.5 and 5 mg, respectively, than with placebo.
Further logistic regression analyses were performed using dose as a continuous variable and baseline intensity as a factor. The derived parameter estimates are shown in Table 7. Dose–response curves were defined from the parameter estimates of these models, allowing comparisons of the dose–response curves for the two study populations. The estimated response rates showed a clear similarity between the Japanese and western populations. In the Japanese study group, estimated response rates were 34.3%, 45.2%, 57.7% and 66.2% for placebo and zolmitriptan 1, 2.5 and 5 mg, respectively, while in the western population the corresponding rates were 39.9%, 49.6%, 61.2% and 71.7% (Fig. 3). Taken together, these analyses of the primary endpoint data show that the dose–response curves in these two studies were remarkably similar; as already recognized from western data, the dose–response curve in the Japanese population demonstrated a flattening of the curve at the 2.5 mg dose. These findings related to the primary endpoint were supported by the analyses of secondary endpoints. Headache response rates at 1 and 4 h were higher in all zolmitriptan treatment groups than in the placebo group for both study populations. A clear dose–response relationship was also observed for complete response rates. In the Japanese study population, complete response rates were 19%, 41%, 47% and 54% for placebo and zolmitriptan 1, 2.5 and 5 mg, respectively, compared with rates of 17%, 30%, 40% and 45% for the western study population.
Parameter estimates of the 2-h headache response in the Japanese and western study populations using a continuous model with linear and quadratic trends


Estimated 2-h headache response rates (using logistic regression models with quadratric trends adjusted for baseline intensity) with 95% confidence intervals in Japanese (▪) and western (□) patients.
Recurrence rates were lower with all doses of zolmitriptan than with placebo, and this was consistent for both study populations. In the Japanese study, the incidence of recurrence within 24 h in patients with a response at the 2-h time point was 41%, 16%, 13% and 17% for placebo and zolmitriptan 1, 2.5 and 5 mg, respectively. Comparative rates in the western study population were 46%, 36%, 37% and 32%.
Tolerability
The profile of adverse events in the Japanese population was similar to that reported in western patients, although the overall incidences were somewhat lower in the Japanese patients (24% vs. 39%). This difference was observed for all doses, including placebo, suggesting that this is more likely to be a cultural effect rather than being treatment related. The type of adverse events reported was similar in the two study populations, with the most common events in Japanese patients being asthenia (4.1%), paraesthesia (3.5%), aggravation reaction (3.5%), somnolence (2.9%), and vomiting (2.9%), while in the western study the most frequent events were dizziness (8.1%), paraesthesia (7.0%), somnolence (6.1%), nausea (5.6%), warm sensation (4.3%) and asthenia (2.9%). In both studies, the majority of adverse events were mild or moderate in intensity.
Discussion
This phase II dose–response study demonstrates that oral zolmitriptan is effective in the acute treatment of migraine in Japanese patients. A significant dose–response relationship was observed for the primary endpoint of 2-h headache response, and the results indicated that 2.5 mg is the minimally effective dose. Although the comparison of the 2-h headache response between zolmitriptan 2.5 mg and placebo showed no statistically significant difference in the evaluable case population, a significant difference was observed in the all-treated population. One possible reason for the lack of significance observed in the evaluable case population was that more patients in the zolmitriptan group treated a migraine of severe intensity compared with the placebo group (29.6% vs. 20.4%). Baseline migraine intensity is a well-known prognostic factor for headache response, and therefore this imbalance in the distribution of baseline intensity may have prevented the detection of a statistically significant difference. This is supported by the finding of the bridging analysis report, where analyses of these study results adjusted for baseline intensity and demonstrated a significant difference between zolmitriptan 2.5 mg and placebo.
The bridging analysis was performed to directly compare the efficacy and tolerability data obtained in the Japanese patients with those from a previous phase II study conducted in a western population, and thereby to determine whether extrapolation of western data into Japanese patients is valid. The bridging analysis of the efficacy data from the two studies shows that zolmitriptan is similarly effective in Japanese and western populations. As highlighted above, the bridging analysis provided baseline-adjusted estimated response rates and these showed that 2-h response rates to zolmitriptan 2.5 mg were 57.7% and 61.2% in the Japanese and western populations, and with zolmitriptan 5 mg were 66.2% and 71.7%, respectively. In both populations, the 2.5 mg dose was clearly more effective in the treatment of migraine and associated symptoms compared with placebo and the 1 mg dose, while the 5.0 mg dose provided small but consistent improvements over the 2.5 mg dose.
Zolmitriptan was well tolerated in Japanese patients, and the profile of reported adverse events was similar to those previously reported with zolmitriptan in mainly Caucasian populations (6). No adverse events unique to the Japanese population were observed. Direct comparison with a western population in the bridging analysis confirmed that the nature of adverse events was similar in the two studies, although a smaller proportion of patients in the Japanese study experienced adverse events (24% vs. 39%). Interestingly, this lower incidence of adverse events was observed even though it has previously been shown that the AUC and Cmax of zolmitriptan are approximately 20% higher in Japanese subjects (5). Therefore, the data obtained in the present study confirm assumptions that although Japanese subjects have a somewhat higher exposure to zolmitriptan, this will not be associated with clinically relevant differences in efficacy or tolerability. The incidence of adverse events was dose-dependent, with a marginal increase in the frequency of adverse events in the 5 mg dose group.
In conclusion, the results from this dose–response study and from the bridging analysis show that oral zolmitriptan is effective and well tolerated in the Japanese population. Balancing both efficacy and tolerability, 2.5 mg was considered to be the optimal dose, as is also recommended for western populations, although the 5 mg dose may provide further benefit for some patients. The similar efficacy and tolerability of zolmitriptan observed in the bridging analysis of Japanese and western study data support the extrapolation of western clinical data to Japanese patients.
