Abstract
Understanding factors influencing patients' preference will improve guidance to make rational choices in expanded symptomatic migraine treatment. The objective of this open-label, cross-over study was to explore patients' preferences for sumatriptan 50 mg vs. zolmitriptan 2.5 mg tablets, focusing on factors influencing this preference. One hundred consecutive migraine patients attending our clinics were asked to treat three attacks with each medication and then fill out a preference questionnaire. Ninety-four migraineurs completed the trial and 42 (44%, 95% CI 34–58%) reported that they preferred zolmitriptan 2.5 mg over sumatriptan 50 mg tablets and 27 (29%, 20–38%) preferred sumatriptan 50 mg. The remaining 25 (27%, 18–36%) did not show any preference. For the initial treatment of the attacks, there were more patients needing just one tablet of zolmitriptan 2.5 mg compared with sumatriptan 50 mg (67 vs. 39%). The reasons for preference among those 69 patients who had shown preference for either of the two triptans were: a faster onset of action (speed of onset) (73%), a longer duration of the effects (39%), fewer adverse events (35%) and lower price (13%). Only one-quarter of the studied migraine population thought that sumatriptan 50 mg and zolmitriptan 2.5 mg were equivalent, which suggests that most migraine patients differentiate between triptans. A faster onset of action (speed of onset) was the most important reason for preference.
Introduction
The launching of new anti-migraine 5-HT1B/1D agonists (triptans) is expanding the treatment options for the symptomatic management of migraine attacks. Even though these drugs can be considered as very similar, as they belong to the same pharmacological group, their pharmacodynamic and pharmacokinetic properties are somewhat different. These heterogeneous properties within the class of triptans could account for some of the differences in efficacy that have already been demonstrated in clinical trials. The results obtained in direct comparative controlled clinical trials are, however, of limited value since, in most cases, only two triptans have been compared. In addition, the information from these trials can be misleading for several reasons, i.e. selection of responders to one of the compared triptans, a biased definition or selection of end-points. Furthermore, the results obtained in different controlled clinical trials cannot be used for a valid and reliable comparison between triptans. The design of these clinical trials may be comparable but the studies have been conducted under different conditions at different times and may perhaps have not used the same inclusion and/or exclusion criteria (1, 2)
Which triptan to choose for a given patient is a frequently appearing problem among those doctors who are treating migraine patients today. Generally speaking, data from non-comparative and comparative clinical trials do not demonstrate dramatic differences between triptans in the majority of the efficacy and safety parameters and the statement ‘For a difference to be a difference it has to make a difference’ seems warranted. In addition, the characteristics of the migraineur and his/her headache attacks do not allow a prediction as to which triptan will be more appreciated. In spite of this, it is quite common in clinical practice that migraine patients express greater satisfaction with one triptan over another, although results obtained in controlled clinical trials were very comparable.
Further understanding of the factors that influence patients' preferences will undoubtedly provide doctors with relevant guidance to make rational choices for their symptomatic treatment of migraine. The aim of this study was to explore patients' subjective preference for sumatriptan 50 mg vs. zolmitriptan 2.5 mg in the acute treatment of migraine, especially focusing on which factors influence their preference.
Patients and methods
Patients with migraine with and without aura, diagnosed according to IHS criteria (3), were recruited consecutively from patients attending our two neurology clinics. Eligible patients were those who either currently were using triptans for their migraine attacks or those who, after their visit, were recommended to use a triptan for symptomatic treatment of their migraine attacks. The two only available oral formulations of sumatriptan, 50 mg tablets, and zolmitriptan, 2.5 mg tablets, were prescribed to them. In our country the price is the same for these two triptans, where the patient has to pay 40% of the total cost of the medication. Patients were informed that sumatriptan 50 mg and zolmitriptan 2.5 mg could be very similar with respect to efficacy and tolerability, but that they, in spite of this, may have preference for one over the other. In addition, that they could use up to two tablets in order to relieve the symptoms of their migraine attack.
We used an open-label, non-randomized, cross-over study design. Patients were asked to treat at least three consecutive attacks with one medication before switching over to the other. They were asked to record how many tablets they used per attack. Finally, the patients were asked to return to our clinics in order to fill in a preference questionnaire. The preference questionnaire included simple questions on: the number of tablets of each triptan used as the initial treatment for their migraine attacks; whether they preferred sumatriptan, zolmitriptan or both; and, if they had expressed a preference, their reasons for this. A number of possible reasons for preference: faster onset of action (speed of onset), longer duration of the effect, better tolerability and lower cost were given in the questionnaire but they could also fill in other reasons.
This study has not received any direct or indirect financial support from any pharmaceutical company. Questionnaire data were personally analysed by the authors once all the patients had finished the study. Neither GlaxoWellcome nor Astra-Zeneca were informed when this study was taking place.
Student's and chi-square tests were used when statistical tests were performed. Results are given as mean ±
Results
Demographics
One hundred migraine patients entered into this preference study. Six did not complete the trial (they were lost to follow-up before treating six attacks) and thus were not included in the analysis. The age of the 94 patients who completed the study was 40 ± 9 years. Eighty-four (89%) were women. Seven (8%) were suffering from migraine with aura. Regarding average frequency of migraine attacks in the previous 3 months, 42 had less than three episodes per month, 44 between three and six episodes per month and the remaining eight more than six per month.
Fifty (53%) of these 94 migraine patients were currently using sumatriptan 50 mg tablets for acute treatment of their migraine attacks; 31 (33%) were using zolmitriptan 2.5 mg tablets. Both groups of patients were naïve for the other triptan. The remaining 13 (14%) had no experience of any of the triptans before entering this study. Thirty-two patients were using prophylactic medications during the study. These medications were not modified during the study period.
Preference questionnaire
Forty-two migraine patients (44%, 95% CI 34–54%) preferred zolmitriptan 2.5 mg tablets over sumatriptan 50 mg tablets, 27 (29%, 95% CI 20–38%) preferred sumatriptan over zolmitriptan and the remaining 25 (27%, 95% CI 18–35%) did not show any subjective preference (Fig. 1). Demographic analysis of these patients showed that the age and sex distributions were not significantly different for the three groups.

Percentage of patients preferring zolmitriptan 2.5 mg, sumatriptan 50 mg or either of the two medications in this study.
Sixty-three out of 94 patients (67%) used one tablet only of zolmitriptan 2.5 mg with respect to number of tablets used for the initial treatment of the migraine attack (not considering the treatment of a possible recurrence, Fig. 2). The corresponding number when using sumatriptan 50 mg was 36 out of 94 patients (39%) and this difference is statistically significant (P = 0.001). When this item was analysed in the two groups preferring one medication to the other, this difference became even more evident (Figs 3 and 4). Among those 42 patients who expressed preference for zolmitriptan 2.5 mg, 81% and 31% took only one tablet of zolmitriptan and sumatriptan, respectively (P = 0.000004) (Fig. 4). By contrast, among those 27 patients who expressed preference for sumatriptan 50 mg tablets, no difference between the two medications could be demonstrated with respect to number of tablets used for the initial treatment of the migraine attack (Fig. 3).

Percentages of patients needing either just one or more than one (either sometimes one and sometimes two or always two tablets) as the initial (not including recurrence) treatment of migraine attacks. Notice that fewer patients needed just one tablet of zolmitriptan 2.5 (67%) compared with sumatriptan 50 mg (39%). □ sumatriptan 50 mg, ▪ zolmitriptan 2.5 mg.

Percentage of patients needing just one tablet or more than one tablet (that is always two tablets or sometimes one and sometimes two tablets) in the group of 29 patients expressing preference for sumatriptan 50 mg. As compared with the percentages in Fig. 2 for the whole group, patients preferring sumatriptan 50 mg tended in a higher proportion to need just one tablet.
The reasons for preference among those 69 patients who had shown preference for either of the two triptans were: a faster onset of action (speed of onset) (73%), a longer duration of the effects (39%), fewer adverse events (35%) and lower price (13%) (Fig. 5). With the exception of price, these reasons for preference in percentages did not differ among those patients who preferred sumatriptan and those who preferred zolmitriptan (Fig. 6). Regarding the number of reasons for preference, 29 patients (42%) gave only one single reason, 32 (46%) gave two reasons and the remaining 8 (12%) gave three or four reasons.

Percentage of patients needing one or more than one tablet in the 42 cases expressing preference for zolmitriptan 2.5 mg.
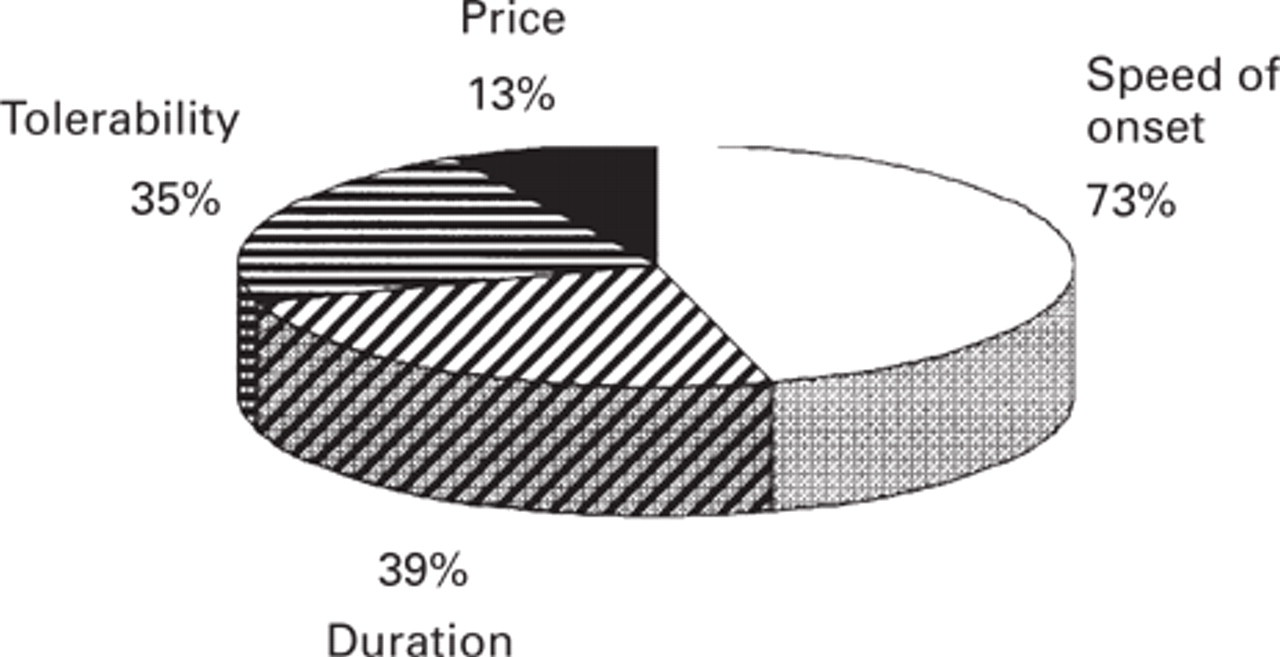
Distribution of reasons for preference in the 69 patients expressing preference. Patients could give one or more reasons to explain their preference. Speed of onset was the main reason for preference.

Distribution of reasons for preference in percentage in the 27 patients preferring sumatriptan 50 mg and in the group of 42 patients preferring zolmitriptan 2.5 mg. Speed of onset was the main reason for preference for the two triptans, followed by longer duration of their effects and better tolerability. Price, meaning fewer number of tablets, was chosen as a reason for preference more frequently by patients preferring zolmitriptan. □ sumatriptan 50 mg, ▪ zolmitriptan 2.5 mg.
There was no significant difference, either in preference or in the reasons for preference, in patients taking prophylactic treatment compared with those who were not on this treatment.
Discussion
To the best of our knowledge, this is the first large preference study involving the two most experienced triptans. Before commenting on the results, it is important to consider the potential drawbacks and confounding factors in this particular study. Ideally, these studies should be blind and include only triptan-naive patients. Otherwise, theoretically, there might be a trend in an open design for negative feelings about one medication to spill over into the evaluation of the other drug (1). In any case, multiple attack, cross-over designs, like the one in our study, can also offer some practical advantages as they seem to reflect more reliably what happens in daily clinical practice as compared with parallel, one-attack protocols. In addition, most of our patients had experience with both triptans prior to this study and they were not very selected as about half were routinely taking zolmitriptan and half sumatriptan in the months before this study was undertaken.
Taking these considerations into account, in our opinion the most important finding of this study is that almost three-quarters of migraine patients preferred one drug to the other, that is, only one-quarter of our migraine patients thought that sumatriptan 50 mg and zolmitriptan 2.5 mg tablets were equivalent. This finding has practical consequences in clinical practice as it means that poor satisfaction with one triptan does not necessarily implicate poor satisfaction with another triptan. Thus, in migraine patients who are not satisfied with one triptan we should try further triptans, ideally in three attacks, before labelling them as poor responders.
In this study 15% of patients preferred zolmitriptan 2.5 mg tablets to sumatriptan 50 mg. This rather small difference between the two drugs should be interpreted with caution, as 95% CI overlap and as there could be an ordering effect. In terms of percentages, there was no clear difference why patients preferred one medication to another, except for price. Our results confirm that the main reason to prefer a symptomatic anti-migraine treatment is faster onset of action (speed of onset), which was pointed out by more than seven out of 10 patients expressing preference for one of the two medications (4, 5). Longer duration of the effects, implicitly implying less recurrence, and better tolerability were the following reasons for preference given by around one-third of patients.
Price was identified by 4% of patients preferring sumatriptan and 19% of patients preferring zolmitriptan as one of the reasons for preference. It could be simplistically argued that this 15% difference could account for the 15% difference in preference between the two medications. We think this is only partly true. On the one hand, it is clear that there is in practice a difference in price for the two medications as more patients needed two tablets of sumatriptan 50 mg than of zolmitriptan 2.5 mg as the initial treatment for their migraine attacks and the price for the two tablets is identical in Spain. This could mean that, in a neurological setting, zolmitriptan 2.5 mg is nearer to the ideal dose for a higher proportion of migraine patients/attacks than sumatriptan 50 mg. It seems logical that a patient prefers zolmitriptan 2.5 mg to sumatriptan 50 mg, both in terms of price and convenience, if frequently one tablet vs. two tablets is needed. On the other hand, however, price and convenience do not seem to totally explain this difference in preference. Analysing our data, it is striking that only one patient preferred zolmitriptan over sumatriptan exclusively due to price. In addition, even though the percentages for reasons to prefer one drug over the other were very similar, the numbers of patients in this series showing preference due to faster onset of action (speed of onset), longer duration or fewer adverse events were always higher for patients preferring zolmitriptan.
The results of this open preference trial indicate that most patients differentiate between the two available triptans (sumatriptan and zolmitriptan) in daily clinical practice, obliging the clinicians to look for the triptan that best suits the needs of each migraine patient. Regardless of the triptan that was preferred, a faster onset of action (speed of onset) was the most important factor in choosing one drug over the other.
Footnotes
Acknowledgements
The authors are indebted to Dr Carl Dahlöf for his critical reading of the manuscript.
