Abstract
The efficacy and tolerability of rizatriptan (MAXALT™) and zolmitriptan (ZOMIG™) were compared in a randomized, double–blind, double–dummy, stratified (on prior use of rizatriptan and/or zolmitriptan), placebo–controlled, single attack study in 766 patients. Rizatriptan tended to provide freedom from pain sooner than zolmitriptan (hazard ratio 1.26, P = 0.075), acting within 60 min following dosing. More patients were pain free at 2 h on rizatriptan than on zolmitriptan (43.2% vs. 35.6%, P = 0.041), while headache relief at 2 h was similar (70.5% vs. 66.8%). At 2 h, fewer patients on rizatriptan had symptoms of photophobia (35.6% vs. 43.5%, P = 0.029) and nausea (25.2% vs. 32.5%, P = 0.046), and more patients on rizatriptan had normal function (45.4% vs. 37.0%, P = 0.025) than zolmitriptan. Headache recurred in 28% of patients taking rizatriptan, 29% taking zolmitriptan and 26% taking placebo. Both active treatments were effective compared to placebo and were well tolerated. The most common side–effects with rizatriptan were asthenia/fatigue, somnolence and dizziness, while the most common side–effects with zolmitriptan were asthenia/fatigue and dizziness.
Rizatriptan (MAXALT™) is a 5–HT1B/1D receptor agonist which is used for the acute treatment of migraine at oral doses of 5 mg and 10 mg. In clinical trials, both doses were shown to be highly effective and well–tolerated compared to placebo (1, 2), with an onset of action as early as 30 min after dosing (3). Rizatriptan 10 mg was more effective than rizatriptan 5 mg with respect to headache relief, freedom from pain and associated migraine symptoms, as well as return to normal function and need for additional medication (1, 4). Rizatriptan 10 mg is the recommended dose in most countries, while 5 mg should be used in patients taking propranolol.
In placebo–controlled studies conducted vs. oral sumatriptan, rizatriptan 10 mg acted sooner than sumatriptan 50 mg (3) and 100 mg (5), while the 5 mg dose of rizatriptan acted sooner than sumatriptan 25 mg through 2 h post–dosing (3). All active treatments were well tolerated.
Zolmitriptan (ZOMIG™) is also a 5–HT1B/1D receptor agonist used for the acute treatment of migraine. The 2.5 mg dose has demonstrated efficacy in the treatment of migraine (6) and 2.5 mg is the starting dose in the United States and the recommended dose in the European Union. Direct comparator studies are useful in helping physicians to select the most appropriate therapy for a patient. The study described below was thus conducted to compare the efficacy and safety of rizatriptan 10 mg with zolmitriptan 2.5 mg.
Methods
Patients
A total of 882 women and men who met IHS criteria for migraine (7) with or without aura were enrolled. Patients had to have at least a 6–month history of migraine and usually experienced 1–8 attacks per month. Patients were excluded if they had clinical evidence of cerebrovascular or cardiovascular disease including significant ECG abnormality, or if they had a history within 1 year or current evidence of drug or alcohol abuse. Patients with any contraindication or sensitivity to 5–HT1B/1D agonists, or those who had received treatment with any other investigational compound or device within the past 30 days were also excluded, as were pregnant women or nursing mothers.
Approvals were obtained from local regulatory authorities and Ethical Review Committees, and each patient who participated in the trial gave written informed consent. The study conformed to Good Clinical Research Practice.
Study design and procedure
This randomized, double blind, placebo–controlled outpatient study compared the clinical profiles of rizatriptan and zolmitriptan tablets for the acute treatment of a single migraine attack. The study was conducted at 66 international sites. A planned total of approximately 800 male or female patients were to be enrolled to yield approximately 650 evaluable patients in a rizatriptan: zolmitriptan:placebo ratio of 2:2:1. At randomization patients were stratified based on prior use of rizatriptan and/or zolmitriptan to reduce potential bias from previous use of these agents.
Patients took a single dose of study medication for a migraine headache that was not resolving spontaneously and was of moderate or severe intensity on a 4–point headache severity scale (0 = none, 1 = mild, 2 = moderate, 3 = severe), provided that they had not taken any prohibited medications (monoamine oxidase inhibitors or methysergide within the past 2 weeks; propranolol within the last 3 days; any triptan, ergot derivatives, or opiates within the past 24 h; and any other analgesic or antiemetic within the past 6 h). Other migraine prophylaxis was permitted. Non–responders, whose headaches failed to improve to grade 0 or 1 (absent or mild) at 2 h post–dose, were permitted optional additional analgesia (e.g. NSAIDs or opiates) and antiemetics. Patients with headache relief at 2 h (reduction in severity from grade 2 or 3 at baseline to grade 0 or 1) who then experienced recurrent headache (return to grade 2 or 3) within 24 h of initial dosing were allowed to take additional analgesics or anti–emetics.
Outcome measures
At baseline, and at 0.5, 1, 1.5, 2, 3 and 4 h after dosing, patients rated their headache severity in a diary using the 4–grade scale described above. A number of parameters were derived from this scale for analysis:
Pain free: The reduction of headache severity from grade 2 or 3 at baseline to grade 0 (complete abolition of headache pain).
Time to pain free within 2 h (primary efficacy endpoint): The first time (0.5, 1, 1.5 or 2 h) that a patient reported the complete abolition of headache pain in the 2 h after dosing.
Headache relief: The reduction of headache severity from grade 2 or 3 at baseline to grade 0 or 1.
Time to headache relief within 2 h: The first time (0.5, 1, 1.5 or 2 h) that a patient reported headache relief in the 2 h after dosing.
Patients also recorded their functional disability (0 = normal, 1 = mildly impaired, 2 = severely impaired, 3 = unable to do activities, requires bed–rest), and the presence or absence of associated symptoms (nausea, vomiting, photophobia and phonophobia) at the same time points as the ratings of headache severity. At 2 h post–dose, patients also recorded their satisfaction with the study medication (1 = completely satisfied, couldn't be better; 2 = very satisfied; 3 = somewhat satisfied; 4 = neither satisfied, nor dissatisfied; 5 = somewhat dissatisfied; 6 = very dissatisfied; 7 = completely dissatisfied, couldn't be worse). The use of additional analgesia/anti–emetics taken after 2 h was recorded, as well as the time and maximum severity of any headache recurrence. At 24 h post–dose, each patient completed the validated 24–h Migraine–specific Quality–of–Life Questionnaire© (8, 9), which assesses quality of life over five domains (work/social/energy/symptoms/feelings) and was translated for country use.
Statistical analyses and power calculations
The study was powered to test the primary hypothesis; namely that rizatriptan 10 mg would be superior to zolmitriptan 2.5 mg in time to pain–free up to 2 h after drug administration. The power calculation for the time to pain–free analysis was based on the log–rank test. With a planned sample size of 260 patients receiving rizatriptan and 260 patients receiving zolmitriptan, the study had approximately 95% power to detect a hazard ratio of 1.8 based on a 2–sided test to compare time to pain–free distributions, at a type I error rate of α = 0.05 and assuming proportional hazards. The power of the test for comparing time to pain–free between active treatments and placebo (n = 130) was at least 95% based on a hazard ratio of 2.7.
The efficacy analysis was based on an ‘all–patients–treated’ approach that included all patients who recorded at least one efficacy rating after dosing (n = 727). Missing values were replaced by carrying forward the preceding value after baseline, except for the time to event analysis in which a patient was censored at the time of the last available data.
The time to pain–free analysis was performed using binary regression models for grouped survival data (10, 11). Covariates included in the model were treatment effect, time interval, baseline headache severity, stratum and region. Percentages of patients with headache relief, pain–free and with associated symptoms were analysed using logistic regression models. Cox regression was used for the analysis of use of additional medication data. The satisfaction with medication and 24–h quality of life individual domain data were analysed using analysis of variance methods. The functional disability data were analysed using a cumulative logistic regression model. Treatment effects were quantified by means, percentages or hazard ratios as appropriate.
As there was only one primary endpoint and one primary comparison, there was no need for multiplicity adjustment for the primary endpoint. No adjustments were made for secondary endpoints except for quality of life, where Hochberg's procedure (12) was used as these data included five domains. The overall type I error rate was set at α = 0.05.
All patients who took study medication were included in the safety analysis, the primary endpoint of which was the overall incidence of adverse events. Fisher's Exact Test was used for pairwise treatment comparisons.
Results
Study population
A total of 882 patients were randomized to treatment. Of those randomized, 116 did not take study medication, the primary reason being lack of migraine headache during the study period (9 months). A total of 766 patients were treated with study medication and completed the study. Thirty–nine patients completed the diary several days after dosing and therefore were excluded from the efficacy analysis.
The baseline characteristics of the 766 patients are displayed in Table 1. The groups were similar with regard to age, sex, race, baseline headache severity, presence of aura and use of migraine prophylaxis. Thirty per cent of patients had previous experience of any triptan in the past 30 days; less than 1% had taken rizatriptan and 8% had used zolmitriptan. Six per cent had used ergots.
Patient baseline characteristics
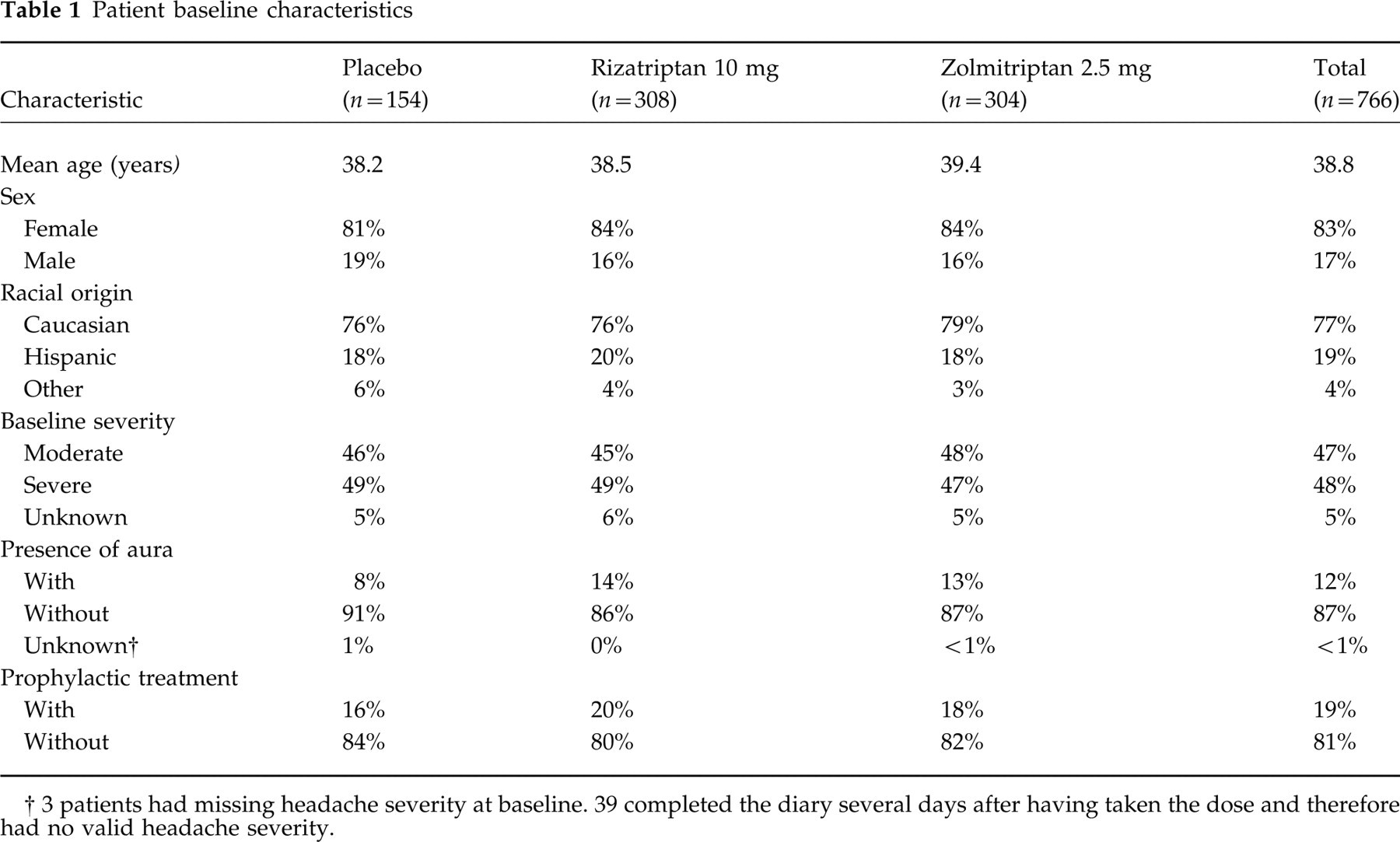
3 patients had missing headache severity at baseline. 39 completed the diary several days after having taken the dose and therefore had no valid headache severity.
Efficacy
Time to pain free within 2 h
On the primary efficacy measure of time to pain–free within 2 h, rizatriptan 10 mg was marginally superior to zolmitriptan 2.5 mg. The hazard ratio of rizatriptan vs. zolmitriptan was 1.26 (95% CI = 0.98–1.64, P = 0.075). This means that, at any time in the 2–h period, headache in a patient on rizatriptan was 26% more likely to be eliminated in the next few minutes than in a patient taking zolmitriptan. A pre–planned analysis also examined pain freedom sustained through the 2–h period by censoring patients who did not have pain freedom sustained through to 2 h (initially reported pain–free within the 2–h period but then relapsed). The analysis censored nine patients (two in the rizatriptan group, six in the zolmitriptan group and one in the placebo group). In this analysis rizatriptan was superior to zolmitriptan with a hazard ratio of 1.31 (95% C.I. = 1.01–1.71, P = 0.041).
Headache relief, time to headache relief and pain–free up to 2 h
The proportions of patients who reported headache relief and pain–free at time points up to 2 h are shown in Table 2. In terms of headache relief, both rizatriptan and zolmitriptan were superior to placebo from 1 h onwards (P < 0.001), with rizatriptan being superior to zolmitriptan at the 1 h time–point (42.5% vs. 35.3%, P = 0.03). Headache relief rates at 2 h were 70.5% with rizatriptan and 66.8% with zolmitriptan (P = 0.23). The hazard ratio for time to headache relief was 1.22 in favour of rizatriptan (95% C.I. = 1.00–1.48, P = 0.053), with the ratio for sustained relief being 1.23 (P = 0.047).
Percentages of patients reporting headache relief and pain–free up to 2 h

n: Number of patients with headache assessment at 2 h.
P < 0.05 vs. zolmitriptan.
P < 0.05, P < 0.001 vs. placebo.
Regarding the pain–free measurement, rizatriptan was superior to placebo from 1 h onwards and was superior to zolmitriptan at 2 h, with pain–free rates at 2 h of 43.2% with rizatriptan and 35.6% with zolmitriptan (P = 0.041) (Fig. 1). Zolmitriptan was superior to placebo only from 90 min following dosing.

Percentages of patients who reported pain–free in the 2-hour interval following dosing. ▪=rizatriptan 10 mg (n=292); ♦=zolmitriptan 2.5 mg (n=289); •=placebo (n=146). ∗P < 0.05 vs. placebo. ‡P < 0.05 vs. zolmitriptan.
Associated symptoms
The proportions of patients without associated migraine symptoms of nausea, vomiting, photophobia and phonophobia at 0 and 2 h are summarized in Table 3. There were no differences between treatments with respect to vomiting. Both active treatments were superior to placebo in relieving nausea, photophobia and phonophobia from 1 hour onwards. Rizatriptan was superior to zolmitriptan in the proportion of patients with no nausea (75% vs. 67%, P = 0.046) and no photophobia at 2 h (64% vs. 56%, P = 0.029).
Secondary efficacy measures

@: Treatment comparisons were based on a comparison of normal function vs. functionally disabled.
P < 0.05 vs. zolmitriptan.
:P < 0.05, P < 0.001 vs. placebo.
Functional disability
Rizatriptan was superior to zolmitriptan in the proportion of patients with normal function from 1.5 h onwards; 45.4% of patients on rizatriptan had normal function at 2 h vs. 37.0% of patients taking zolmitriptan (P = 0.025) (Table 3). Rizatriptan was superior to placebo from 1 h onwards, while zolmitriptan was superior to placebo from 90 min after dosing.
Satisfaction with medication at 2 h
Patients on rizatriptan were more satisfied with their medication than those on zolmitriptan at 2 h (mean score ± standard error 3.38 ± 0.10 vs. 3.67 ± 0.10, P = 0.038). Patients in both active treatment groups were more satisfied than those taking placebo (mean score 4.97 ± 0.13, P < 0.001); 62.7% of patients on rizatriptan reported being completely, very or somewhat satisfied at 2 h compared with 54.6% of patients taking zolmitriptan (P = 0.045) and 23.1% of patients taking placebo (P < 0.001).
Need for additional medication from 2 to 24 h
The cumulative percentages of patients taking additional medication from 2 to 24 h are shown in Table 3. Fewer patients in both active treatment groups needed additional medication from 2 to 24 h than those taking placebo (P < 0.001) while there was no difference between active agents. The need for further treatment between 2 and 4 h post–dose was smaller for patients receiving rizatriptan (18.5%) than for patients receiving either zolmitriptan (24.9%, P = 0.045) or placebo (53.4%, P < 0.001).
Headache recurrence within 24 h
Approximately 28% of patients on rizatriptan, 29% of patients on zolmitriptan and 26% of patients taking placebo reported recurrence. The mean duration of relief (time of first recorded pain relief to time of recurrence) was 11.4, 11.9 and 9.7 h for the rizatriptan, zolmitriptan and placebo groups, respectively. Interpretation of recurrence rates is difficult because recurrence is both conditional on initial headache relief at 2 h and confounded by the use of additional medication. A post hoc analysis, which excluded patients who took additional medication before the recurrence, gave recurrence rates of 26% for rizatriptan, 26% for zolmitriptan and 16% for placebo.
24–h quality of life
Rizatriptan and zolmitriptan were superior to placebo on all five quality of life domains (work/social/energy/symptoms/feelings) (P < 0.01) and there were no differences between the active treatments. The mean domain score ranged from 12.80 to 13.62 for rizatriptan, from 12.43 to 13.39 with zolmitriptan and from 10.65 to 11.14 for placebo.
Pain–free at 2 h without recurrence or need for additional medication
A post hoc analysis examined the proportion of patients who were pain–free at 2 h, then did not report a recurrence or take any additional medication from 2 to 24 h. In this analysis rizatriptan (32.2%) was superior to zolmitriptan (23.5%, P = 0.013) and placebo (6.8%, P < 0.001).
Effect of stratification
There was no interaction between treatment and stratum for any endpoint. This means that the efficacy and tolerability differences between the two compounds were unaffected by previous experience with rizatriptan and/or zolmitriptan.
Safety
Both active treatments were generally well tolerated and the adverse events that did occur were mostly rated as mild or moderate in intensity and transient. There were two serious clinical adverse experiences following study drugs. One patient was hospitalized for appendicitis 13 days after taking rizatriptan, while another was hospitalized for abdominal pain 4 days after taking zolmitriptan. No patient was withdrawn from the study because of a clinical adverse experience.
Table 4 summarizes the clinical adverse event profile of rizatriptan and zolmitriptan vs. placebo. All 766 patients who took study medication were included in the safety analysis. The overall incidence of any clinical adverse events was significantly higher in the rizatriptan and zolmitriptan groups than in the placebo group (31.2% vs. 38.8% vs. 22.1%, P < 0.05 vs. placebo, P = 0.051 rizatriptan vs. zolmitriptan). Only three types of adverse experiences occurred in ≥ 5% of the patients in at least one treatment group: asthenia/fatigue, dizziness and somnolence. The percentages of patients reporting asthenia/fatigue were 6%, 5% and 3% in the rizatriptan, zolmitriptan and placebo groups, respectively. The percentages having dizziness were 5%, 6% and 3% in the rizatriptan, zolmitriptan and placebo groups, respectively, while the incidence of somnolence was 6% for rizatriptan, 4% in the zolmitriptan group and 3% for the placebo group. Chest pain (also including chest pressure, heaviness, tightness, cramp, retrosternal pain or oppression and thoracic oppression) was reported by 2% of patients taking rizatriptan, 4% of patients taking zolmitriptan and 1% of placebo–treated patients.
Summary of overall incidence of clinical adverse events and the most common clinical adverse events (incidence ≥ 5% in any one treatment group)

P < 0.05 vs. placebo.
Chest pain also includes chest pressure, retrosternal pain or oppression, chest tightness, chest cramp, thoracic oppression and chest heaviness.
Comment
The results of this active comparator study in 766 migraine patients indicate that rizatriptan 10 mg has advantages over zolmitriptan 2.5 mg in the treatment of migraine attacks. On the primary outcome measure of time to pain–free (chosen because pain–free is a more clinically relevant endpoint than headache relief), a patient on rizatriptan was 26% more likely to experience freedom from pain in the next few minutes than a patient taking zolmitriptan at any time in the 2–h period following dosing (P = 0.075). This measure, however, refers to the efficacy of both agents over the full 2–h period and assumes that patients who report pain–free prior to 2 h do not relapse. It is thus also important to consider the population who has pain–free sustained throughout the entire period. In this case rizatriptan was superior to zolmitriptan, with a patient being 31% more likely to have freedom from pain sustained through the 2–h period following dosing.
Rizatriptan was also better than zolmitriptan on a wide range of other outcome measures assessed within 2 h of dosing. It was superior concerning the percentage of patients who were pain–free at 2 h (the recommended IHS method to assess efficacy in clinical trials in migraine (13)), associated migraine symptoms (nausea and photophobia), functional disability and satisfaction with medication at 2 hours. The use of additional medication from 2 to 4 h (a useful measure of early efficacy) was also lower with rizatriptan than with zolmitriptan. In addition, more patients demonstrated sustained efficacy with rizatriptan than zolmitriptan, defined as becoming pain–free by 2 h, with no subsequent recurrence or need for additional medication. These effects were shown to be independent of previous exposure to rizatriptan or zolmitriptan. Both agents had similar effect on pain relief and phonophobia at 2 h and quality of life over 24 h.
Both active treatments were generally well tolerated in comparison to placebo. Adverse events were mostly mild or moderate and transient in nature. The commonest adverse events following rizatriptan and zolmitriptan were similar to those reported in previous studies (1–6) and consisted of asthenia/fatigue, dizziness and somnolence.
