Abstract
The goal of this study was to evaluate the coronary vasoconstrictive effects of high doses of eletriptan compared with a standard dose of sumatriptan. Patients with no clinically significant coronary artery disease were randomized to receive high-dose intravenous eletriptan (n = 24) vs a standard dose of sumatriptan (n = 18; 6 mg subcutaneously) vs placebo (n = 18). Serial angiograms were obtained. The primary non-inferiority analysis found equivalence between the mean maximum change in left anterior descending coronary artery diameter for eletriptan, -22% [95% confidence interval (CI) -26, -19], and sumatriptan, -19% (95% CI -22, -16). The change due to placebo was -16% (95% CI -20, -12). No individual cases of clinically significant vasoconstriction were observed. The results confirm that eletriptan has a broad cardiovascular safety margin, with plasma concentrations comparable to three to five times the Cmax of an oral 80-mg dose associated with modest vasoconstriction equivalent to standard therapeutic doses of sumatriptan.
Introduction
Triptans are potent and selective 5-HT1B/1D agonists that have demonstrated significant efficacy in the acute treatment of migraine (1, 2). As a class, triptans are contraindicated in patients with ischaemic heart disease due to their vasoconstrictor effects, with case reports suggesting that treatment in this subgroup may precipitate anginal episodes and, on rare occasions, acute myocardial infarction (3). Pathological changes in diseased coronary arteries appear to alter their basal tone and reactivity (4). In contrast, in humans with normal coronary arteries, triptan challenge is associated with only mild vasoconstriction (5). Coronary tone in healthy arteries is predominantly modulated by endothelial factors and adrenergic inputs (4). Serotonergic inputs, primarily mediated by 5-HT2 receptors (6), may also alter vasomotor tone. While ergotamine binds to 5-HT2 receptors, triptans do not do so to a significant degree (1, 5). Thus, triptans represent an important advance over ergotamine in terms of the cardiovascular safety margin.
The effect of 5-HT1B/1D receptor activity on vasomotor tone is modest since coronary arteries are not richly endowed with the 5-HT1B/1D-receptor populations that are targeted by triptans. Consistent with this, in vitro studies of isolated human arteries indicate that eletriptan has an 86-fold greater selective activity at the middle meningeal artery compared with the coronary artery, while sumatriptan exhibits a 30-fold selectivity (7). The results of in vitro studies, though, may only be very cautiously extrapolated to the clinical setting.
No previous study has systematically measured the vasoconstrictive effect of a triptan in vivo across a wide range of doses based on a double-blind placebo-controlled methodology that included use of an active control. The goal of the current study was to assess, in normal volunteers, the vasoconstrictive effects of eletriptan at plasma concentrations of at least three times the C max for the 80-mg oral dose. The prespecified hypothesis was that supramaximal doses of eletriptan would have no greater coronary vasoconstrictive effects than therapeutic doses of sumatriptan.
Methods
Patients without prior documented coronary disease undergoing diagnostic coronary angiography for clinical indications consented to participate in the study. Patients confirmed with angiography as having normal coronary arteries (<20% focal stenosis and without diffuse irregularities) were evaluated during the same catheterization procedure after the randomized double-blind administration of either intravenous (i.v.) eletriptan, subcutaneous (s.c.) sumatriptan, or placebo.
The study was approved by an Institutional Review Board (IRB) and all subjects provided written informed consent. A screening evaluation included a medical history and physical examination. The patients were men and women aged 18–60 years. Subjects were excluded for any of the following reasons: women who were pregnant or nursing; known hypersensitivity to triptans; concomitant use of CYP3A4 inhibitors, serotonin selective reuptake inhibitors, or monoamine oxidase inhibitors; current cardiovascular illness, including systolic blood pressure ≥ 180 mmHg or diastolic blood pressure ≥ 100 mmHg, Prinzmetal's angina, chest pain symptoms, or ECG findings likely to be due to coronary artery spasm; history of prior stroke, or myocardial infarction (or pathological Q-waves or a current ECG consistent with infarction); any history of valvular or congenital heart disease, cardiomyopathy, heart failure, or any clinically significant cardiac arrhythmia or conduction abnormality; or any acute or unstable medical condition that might compromise the potential subject's safety.
Study treatment
Patients underwent diagnostic coronary angiography according to standard procedures; the angiographic films were immediately reviewed and patients meeting entry criteria were randomized, double-blind, to receive either i.v. eletriptan (n = 24), s.c. sumatriptan (n = 18), or placebo (n = 18). Intravenous eletriptan infusion was adjusted to achieve a mean plasma concentration three or more times the C max of 80 mg of eletriptan in volunteers, with a minimum target concentration of 299 ng/ml of eletriptan. There was a preplanned replacement of all subjects whose eletriptan plasma concentration at 40 min postinfusion was < 299 ng/ml. If no 40-min measurement was available, a minimum concentration of 121 ng/ml at 15 min was used as a proxy criterion. Interim analyses of eletriptan plasma concentrations were performed, and the infusion protocol adjusted to ensure that all patients met the minimum target concentration. Sumatriptan was administered subcutaneously employing a single standard therapeutic dose of 6 mg.
The study drug was administered using a double-dummy design to maintain the blind across two routes of administration, i.v. (eletriptan/placebo), and s.c. (sumatriptan). An unblinded third party (hospital research pharmacist) not otherwise involved with study subjects or procedures prepared all study treatments. Eletriptran-HBr was supplied in an aqueous solution of 2 mg/ml with matching placebo for infusion consisting of 0.9% NaCl solution. A two-step infusion was used to permit assessment of plasma concentration, which would approximate three concurrent 80-mg doses of eletriptan. This design also enabled safety and tolerability to be evaluated between the two infusion steps. The first 26-mg dose was infused over 20 min at a rate of 0.650 ml/min (1.30 mg/min), followed by administration of the remaining 46 mg, infused over 20 min at a rate of 1.150 ml/min (2.30 mg/min). In the actual study, patients were initially exposed using the same two-step infusion procedure to a 36-mg dose of eletriptan (n = 12), and then a 52-mg dose of eletriptan (n = 6). The total infused dose was revised upward twice, based on interim pharmacokinetic data to ensure that sufficient patients achieved the targeted C max.
Sumatriptan succinate for s.c. administration was supplied as the Imitrex®, STATdose® SYSTEM Kit, which consisted of disposable syringe cartridges containing 6 mg of sumatriptan in 0.5 ml of saline solution (GlaxoSmithKline, Research Triangle Park, NC, USA). Matching placebo (0.5 ml of 0.9% NaCl solution) for s.c. administration was similarly prepared by an unblinded third party.
Study procedures
At baseline, heart rate, aortic and femoral artery blood pressures, blood sample, and a 12-lead ECG were obtained. Continuous ECG and blood pressure monitoring were performed throughout the procedure. Serial angiograms were obtained at the preinfusion baseline, at 5, 15, and 40 min after the start of infusion, and at 10 min postinfusion, using a 6-F coronary catheter and non-ionic contrast medium; a single angiogram was performed at each time point at the same angle determined to provide optimal visualization of the mid left anterior descending artery (mid-LAD) and proximal circumflex. Blood samples were collected (either from the femoral artery catheter or a femoral venous catheter, if present) at baseline, at 5, 15, and 40 min after the start of the infusion, and at 10 min postinfusion. Mean plasma levels were calculated for the time point at which maximum change in the coronary artery diameter occurred for each patient.
Safety assessments included monitoring of adverse events such as chest pain as well as changes in heart rate and aortic pressure throughout the study procedure. As predefined in the protocol, the study infusion was to be stopped or intracoronary vasodilators administered if subjects had a > 50% focal or diffuse vasoconstriction that was asymptomatic or > 25% focal or diffuse vasoconstriction associated with either chest symptoms or ischaemic changes on the ECG. Following completion of the study protocol, patients were managed as with any standard diagnostic catheterization, with vascular sheath removal and haemostasis, ECG and blood pressure monitoring, and administration of i.v. fluids. Patients were discharged home in 4–8 h; investigators contacted them by telephone 3–7 days later to determine if any poststudy adverse events had occurred.
Quantitative coronary angiography
Quantitative coronary angiographic (QCA) analysis was performed by an independent laboratory (PERFUSE, Harvard Clinical Research Institute, Boston, MA, USA), blinded to study drug allocation. For every patient, three consecutive-end diastolic images were used at each time point to calculate the mean segment diameter (MSD) in the mid-LAD and proximal circumflex arteries. In addition to the protocol-defined regions, the remainder of the coronary tree was evaluated at each time point for evidence of any global or focal vasoconstriction.
Eletriptan/sumatriptan assays
Samples were centrifuged at 3 °C for 10 min at 1500
Statistical analyses
The prespecified primary outcome parameter was the log of the ratio of minimum post-baseline MSD to baseline MSD for the mid-LAD. The same parameter was also calculated for the proximal circumflex coronary artery. The mean of the logged ratio of the minimum post-baseline vs baseline coronary artery diameter (CAD) was calculated and compared for eletriptan vs sumatriptan. The difference in logged means, and corresponding 95% confidence interval limits, were antilogged to give a ratio of geometric means (of ratios) and corresponding 95% confidence interval limits. The primary, a priori non-inferiority analysis compared the effect of eletriptan vs sumatriptan on the mid-LAD.
Results
Overall, 162 subjects presenting for diagnostic angiographic evaluation of chest pain or other symptoms or non-invasive findings suspicious for ischaemia consented to and were screened for the study. After completion of the diagnostic angiogram, 60 subjects were qualified to enter into the treatment phase of the study. Excluded were 102 subjects with evidence of significant coronary artery disease or cardiomyopathy documented by the diagnostic study. Eletriptan was administered to 24 patients (15 men, nine women; mean age 48 years; age range 33–58); sumatriptan was administered to 18 patients (12 men, six women; mean age 46 years; age range 36–59); placebo was administered to 18 patients (11 men, seven women; mean age 46 years; age range 30–60). There were no differences between the groups with respect to age, gender, or concomitant medications. A prior history of migraine was obtained in three patients in the eletriptan group and one in the placebo group. Prior to catheterization, all patients were receiving at least one concomitant medication, with the following classes of medications being the most common among subjects in the eletriptan, sumatriptan, and placebo groups, respectively: non-steroidal anti-inflammatory drugs, n = 21, 16, 15; β-blockers, n = 8, 12, 14; vasodilators, n = 14, 7, 8; drugs for gastrointestinal ulcers, n = 9, 4, 5; statins, n = 7, 4, 4; antihistamines, n = 5, 6, 4; and ACE inhibitors, n = 3, 5, 2. For this study, no medication was allowed in the 8 h before catheterization, during which time subjects were allowed only water. All patients were treated with sedative-hypnotic medications as part of the standard catheterization protocol.
Clinical, haemodynamic, and ECG responses
Of the 24 subjects who received eletriptan, 23 completed the study and one discontinued due to a protocol violation not related to study drug. The protocol violation became evident during the course of study angiography; it consisted of a congenital myocardial bridge associated with coronary systolic narrowing at the flexion point where the coronary artery entered the myocardium. The 18 subjects who received sumatriptan and the 18 who received placebo completed the study. All patients completed the study without serious adverse events, were discharged home following the catheterization, and were physically well as ascertained by telephone follow-up 3–7 days later. During the study itself, no patient developed clinical ischaemia or any of the prespecified end points (any coronary vasoconstriction > 50%, or > 25% with ECG changes) which would have necessitated premature termination of the study. There were no serious treatment-related adverse events. No patients receiving eletriptan or sumatriptan experienced chest pain, whereas two patients receiving placebo did (11%); no patients had clinically significant ECG changes that would have necessitated drug discontinuation. Minor ECG changes noted included the following: in the eletriptan group, one subject experienced non-specific transient ST-T wave changes and one subject a non-specific transient ST elevation without evidence of ischaemia; in the sumatriptan group, one subject experienced a transient T-wave abnormality; in those receiving placebo, no subjects experienced ECG abnormalities.
Rapid i.v. infusion of eletriptan, subcutaneously administered sumatriptan, and placebo resulted in the following mean (±SE) changes in heart rate: for eletriptan, + 2.2 ± 1.6 beats/min; for sumatriptan, − 6.7 ± 1.5 beats/min; and for placebo, − 2.7 ± 1.2 beats/min. Study treatment resulted in the following mean (±SE) changes in systolic/diastolic femoral artery blood pressure: for eletriptan, +16.4 ± 3.2/+ 10.0 ± 1.2 mmHg; for sumatriptan, +10.3 ± 4.0/+2.8 ± 1.9 mmHg; and for placebo, − 2.5 ± 2.4/+3.8 ± 1.6 mmHg. Study treatment resulted in the following mean (±SE) changes in systolic/diastolic aortic blood pressure: for eletriptan, + 21.2 ± 2.7/+11.2 ± 2.0 mmHg; for sumatriptan, + 9.5 ± 4.1/+4.1 ± 1.6 mmHg; and for placebo, + 2.8 ± 3.3/−1.7 ± 1.4 mmHg.
Eletriptan and sumatriptan plasma concentrations
Twenty of the 24 patients achieved plasma concentrations that were higher than the targeted minimum concentration of 299 ng/ml at 40 min (or 121 ng/ml at 15 min if 40-min data were unavailable). The eletriptan infusion protocol achieved the desired targets, with a mean plasma concentration of 660 ng/ml (range 299–1310) at 40 min after the start of the infusion.
The mean concentration for sumatriptan at 15 min after the start of the infusion, the expected time of maximum concentration, was 68 ng/ml (range 41–107 ng/ml). This was a concentration similar to the C max of 71 ng/ml (range 49–110 ng/ml) for 6-mg s.c. sumatriptan reported in the US package insert (USPI).
Quantitative coronary angiographic findings
The primary outcome measure of this study was the maximal per cent change in CAD as determined by QCA (expressed as the per cent of baseline diameter). In the eletriptan group, despite higher plasma concentrations than those observed at therapeutic doses, the magnitude of change in CAD for eletriptan was similar to that observed on sumatriptan (Fig. 1a,b). The maximum mean change in left anterior descending CAD [±95% confidence interval (CI), based on geometric mean ratio] for eletriptan was − 22% (95% CI − 26, − 19), for sumatriptan was − 19% (95% CI − 22, − 16), and for placebo was −16% (95% CI −20, − 12). One patient experienced reduction in LAD diameter of 39% at the flexion point of a myocardial bridge; this congenital anomaly was technically a protocol violation that should have excluded study entry.

(a) Maximal per cent change in left anterior descending coronary artery: comparison of sumatriptan 6 mg vs supramaximal eletriptan i.v. (b) Maximal per cent change in proximal circumflex coronary artery: comparison of sumatriptan 6 mg s.c. vs supramaximal eletriptan i.v.
The maximum mean change in proximal circumflex CAD for eletriptan was − 19% (95% CI − 22, − 17); for sumatriptan it was − 17% (95% CI − 21, − 14); and for placebo it was − 13% (95% CI − 17, − 10). Furthermore, the time course of change in CAD was similar across all three treatments both during infusion and immediately postinfusion (Fig. 2).

Mean per cent change in left anterior descending coronary artery diameter at 5 min, 15 min, and 40 min after the start of infusion, and at 10 min after the completion of infusion.
No patient in any group developed clinically meaningful vasoconstriction (>30% reduction from baseline diameter). The magnitude of vasoconstriction observed in the eletriptan and sumatriptan groups was no different from that seen in patients receiving placebo and was not associated with ECG changes or chest symptoms; in fact, there were only two patients who reported chest pain and they were on placebo.
A supra-therapeutic dose of eletriptan given intravenously was equivalent to sumatriptan 6-mg s.c. in its effect on CAD (Fig. 1a,b); this observation was further demonstrated by the non-inferiority analysis (Table 1). The criterion for passing non-inferiority required that the lower limit of the antilogged 95% CI be > 0.90, a level achieved for both the left anterior descending and the proximal circumflex arteries. There was extensive overlap in maximal coronary artery constriction after eletriptan challenge compared with placebo, suggesting that the small changes in CAD observed on supra-therapeutic doses of i.v. eletriptan were within the normal physiological range. There was no correlation between eletriptan C max and changes in CAD (Fig. 3).

Scatter plot showing lack of correlation between maximum eletriptan plasma concentration and maximum reduction in coronary artery diameter (mid-LAD). Circled value is protocol violator with congenital coronary anatomic anomaly.
Comparison of effect of eletriptan and sumatriptan on coronary artery diameter: results of a non-inferiority analysis
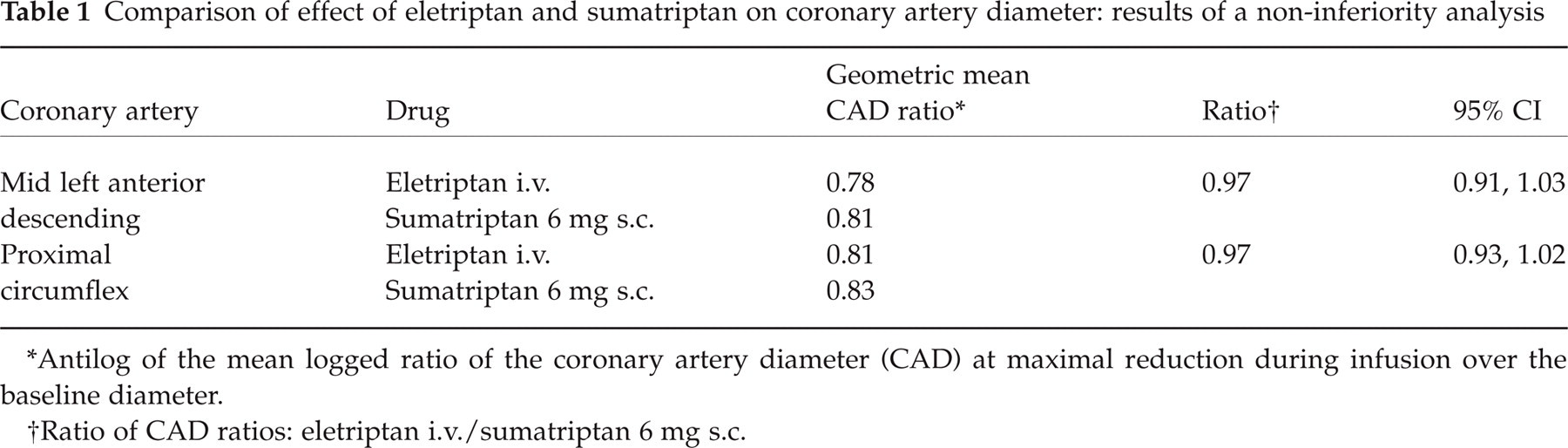
Antilog of the mean logged ratio of the coronary artery diameter (CAD) at maximal reduction during infusion over the baseline diameter.
Ratio of CAD ratios: eletriptan i.v./sumatriptan 6 mg s.c.
Discussion
This study demonstrates that in patients with normal coronary arteries, the antimigraine drug eletriptan, administered at plasma concentrations in excess of three times the C max of an oral 80-mg dose, results in only a mild and clinically insignificant degree of coronary vasoconstriction, similar to that seen in patients receiving a standard dose of sumatriptan or placebo (Fig. 1a,b). Furthermore, there were no individual cases of clinically meaningful vasoconstriction (>50% with associated ECG changes, or > 25% without ECG changes). Overall, high-dose eletriptan was well tolerated, and was not associated with any clinically significant ECG changes, haemodynamic changes, or serious adverse events.
Clinical studies of patients receiving triptans document that up to 10% develop chest discomfort, but this symptom is rarely associated with ECG changes or any cardiovascular-related effects, and thus appears to be predominantly non-cardiac in nature (8–10). The results of this study provide further evidence that chest symptoms associated with triptan treatment are likely to be non-cardiac in origin. It is important to qualify the current results by noting that the lack of clinically meaningful triptan effects on healthy coronary arteries cannot be extrapolated to patients with significant coronary atherosclerosis, whether clinically silent or symptomatic. For this reason, it is essential that patients be medically evaluated for coronary artery disease prior to prescribing a triptan. For patients who are asymptomatic, especially those 60 years or older, this should include an assessment of known cardiovascular risk factors such as hypertension, smoking, glucose intolerance, and hypercholesterolaemia (11).
Most previous studies evaluating the potential effects of triptans on coronary arteries have been based on in vitro studies of isolated vessels (5, 7, 12–15). Previous human studies of the vasoconstrictive effect of triptans on coronary arteries have been limited by small sample sizes, and typically do not include placebo and/or active comparator controls, and are not designed to evaluate dose–response effects (15–20). Previous studies have documented a modest degree of vasoconstriction, in the same 20% range observed in the current study (16–21).
A preclinical study (7) found that both eletriptan and sumatriptan exhibit selectivity in their contractile effects on isolated human middle meningeal artery compared with the coronary artery (86-fold for eletriptan and 30-fold for sumatriptan). Eletriptan's approximately three-fold greater selectivity for cranial vs coronary arteries may help to explain why use of high doses of eletriptan in the current study were associated with coronary vasoconstriction equivalent to standard doses of sumatriptan. Alternatively, it is possible that the concentration of eletriptan achieved in the current study was above the level at which maximum 5-HT1B-mediated coronary vasoconstriction occurs (5). The lack of a notable vasoconstrictive trend at higher eletriptan plasma concentrations (Fig. 3) appears to support the latter hypothesis.
Interestingly, treatment with placebo alone resulted in a progressive pattern of coronary vasoconstriction during the catheterization procedure (mean maximum reduction in LAD: − 16% (95% CI − 20, − 12) (Fig. 2). This non-pharmacological vasoconstrictive effect is consistent with literature reporting a fair degree of variability in CAD in response to daily activities, as well as in response to a variety of psychological stressors, many of which are much less stressful than angiography (22–24). These findings indicate that use of a randomized, double-blind, placebo-controlled methodology is important for studies evaluating the effect of drugs on cardiovascular physiology. It has been common practice in catheterization studies for a patient's baseline physiological parameters to serve as the ‘control’, but this study found reason to question this practice.
The main limitation of the current study was the use of a normal volunteer sample. It is possible that some susceptible migraineurs (even with normal coronary arteries) might be more likely to respond to triptan administration with coronary vasospasm. While this is a concern, postmarketing surveillance data help us to assess the risk in context, with less than one serious cardiovascular event reported per million patients treated with a triptan (11). Data from two large prescription database studies provide further evidence, with both reporting relative risk ratios of < 1 for serious cardiac events, and for all-cause mortality, for migraineurs treated with triptans compared with those not receiving triptan therapy (25, 26).
Conclusions
Intravenous doses of eletriptan, with plasma concentrations equivalent to three to five times the C max of an oral 80-mg dose of eletriptan, resulted in mild and clinically insignificant reductions in coronary diameter, reductions no different from those observed on either a standard therapeutic dose of 6 mg sumatriptan administered subcutaneously or placebo. Overall, high-dose eletriptan was well tolerated by the cardiovascular system, and was not associated with any significant ECG or haemodynamic changes, nor was it linked with any serious adverse events.
Footnotes
Acknowledgements
Funding for this study was provided by Pfizer Inc.
