Abstract
Aim
To explore a possible differential effect of sumatriptan on extracerebral versus cerebral arteries, we examined the superficial temporal (STA), middle meningeal (MMA), extracranial internal carotid (ICAextra), intracranial internal carotid (ICAintra), middle cerebral (MCA) and basilar arteries (BA).
Methods
The arterial circumferences were recorded blindly using high-resolution magnetic resonance angiography before and after subcutaneous sumatriptan injection (6 mg) in 18 healthy volunteers.
Results
We found significant constrictions of MMA (16.5%), STA (16.4%) and ICAextra (15.2%) (p ≤ 0.001). Smaller, but statistically significant, constrictions were seen in MCA (5.5%) and BA (2.1%) (p ≤ 0.012). ICAintra change 1.8% was not significant (p = 0.179). The constriction of cerebral arteries was significantly smaller than the constriction of extracerebral arteries (p < 0.000001).
Conclusion
Sumatriptan constricts extracerebral arteries more than cerebral arteries. We suggest that sumatriptan may exert its anti-migraine action outside of the blood–brain barrier.
Introduction
The triptans, serotonin receptor B/D agonists, are the mainstay in acute treatment of migraine (1). Almost 20 years have passed since the first triptan, sumatriptan, was launched (2), but the mode-of-action of triptans is still a subject of intense debate (3,4). Sumatriptan was originally developed as a cranial vasoconstrictor acting on the 5-HT1B/1D receptors in cephalic vessels (5). However, several modes-of-action as well as multiple sites-of-action have been proposed in animal experiments (6). Recently it has been reported that sumatriptan has a blocking effect on the nociceptive transmission between peripheral and central trigeminovascular neurons (7). Using high-resolution magnetic resonance angiography (MRA) in a study of migraine without aura patients (8), we recently showed that sumatriptan constricted the middle meningeal artery (MMA), but not the middle cerebral artery (MCA), during migraine attacks. In contrast, we have previously reported that sumatriptan constricted both the MMA and the MCA in healthy volunteers (9), but suggested that it mainly constricted the MMA. To further explore a possible differential effect of sumatriptan on extracerebral versus cerebral arteries, we examined the effect of sumatriptan on the following additional arteries: the superficial temporal artery (STA), the extracranial part of the internal carotid artery (ICAextra), the basilar artery (BA) and the intracranial part of the internal carotid artery (ICAintra). To the best of our knowledge this has not previously been studied. We hypothesized that sumatriptan would constrict the extracerebral arteries significantly more than the cerebral arteries. To test this hypothesis, we used high-resolution MR-angiography in 18 healthy volunteers with a 3T MR scanner before and after subcutaneous injection of 6 mg sumatriptan.
Methods
Participants and study design
We recruited 18 healthy subjects (11 women and 7 men, mean age 25 years (range 22–28 years), mean weight 65 kg (range 53–77 kg)). Exclusion criteria were: a history of migraine or any other type of headache (except episodic tension-type headache more than once a month), history of serious somatic disease, daily intake of any medication (except oral contraceptives) and general contraindications for MRI scan. All female participants used safe contraceptive methods. All participants gave written informed consent to participate. The Ethical Committee of Copenhagen approved the study (H-K-20060083), which was conducted in accordance with the Helsinki II Declaration of 1964 as revised in Edinburgh in 2000. Briefly, the original study was a randomized double-blind placebo controlled cross-over study, where the participants received CGRP and placebo (isotonic saline) over 20 minutes. On both days 6 mg sumatriptan (Imigran® injection, Glaxo Wellcome Operations, Bernard Castle, UK) was injected subcutaneously 40 minutes after start of infusion. MR-angiography was recorded at baseline, 30 minutes after start of infusion and 15 minutes after sumatriptan injection (Figure 1). Parts of the study focusing on the effect of CGRP have been published before (9). To examine the effect of sumatriptan in arteries not exposed to CGRP, here we performed analyses of the acquired MR-angiography images for the placebo day only. Values for MCA and MMA were reported previously (9). Here, we analyse STA, BA and the extracranial as well as the intracranial part of ICA to study the effect of sumatriptan. The data analysis was done blindly with respect to time sequence using automated vessel wall analysis software (10).
The experimental design. Baseline MR-angiography (MRA) was recorded in 18 healthy volunteers followed by an infusion of placebo (isotonic saline) for 20 minutes. Injection of 6 mg sumatriptan subcutaneously was given 45 minutes after the start of placebo infusion. MRA were recorded 15 minutes before and after sumatriptan injection.
Experimental procedures
All participants reported to the laboratory headache-free. Tobacco, coffee, tea, cocoa or other methylxanthine-containing foods or beverages were not allowed 12 hours before study start. Participants were placed in the supine position and a venous catheter (Venflon®, Becton Dickinson, Franklin Lakes, NJ, USA) was inserted for infusion into the left antecubital vein. Blood samples were then collected to determine baseline levels of haematocrit, potassium and sodium. The subjects were monitored with ECG (by ECG monitoring function attached to the MR scanner), end-tidal partial pressure of carbon dioxide (CO2) (capnograph, Datex, Helsinki, Finland), blood oxygen saturation, blood pressure and heart rate (Veris® monitor, Medrad, Warrendale, PA, USA). MR images were recorded from a 3.0-Tesla Philips Achieva Scanner (Philips Medical Systems, Best, The Netherlands) using an eight-element phased-array receiver head coil.
Data acquisition and analysis
Details concerning data acquisition and imaging protocols have been reported previously elsewhere (9). The MR images were transferred from the scanner to a remote workstation in DICOM format and then analyzed by LKEB-MRA vessel wall analysis software (10). The software provides automated contour detection and quantification of the luminal boundaries in vessel segments. There is a slight overestimation of the vessel size as measured by the software, but since we are measuring a change in the circumference this systematic error will be eliminated in the final measurements. We identified the MCA, the BA and an intracranial part of the ICA (ICAintra) on the MCA scan images, whereas the MMA, the STA and the extracranial part of the ICA (ICAextra) were identified on the MMA scan images (Figure 2). In general, the larger a vessel, the better the analysis. The ICAintra and BA are larger arteries than MCA, and the ICAextra and STA are larger than MMA. The analyses of all four additional arteries were even more precise than MCA and MMA (narrower confidence intervals) (Figure 3). The MCA was identified by marking the branch from the main trunk of the ICA. The ICAintra was identified just before the ICA becomes MCA. The BA segment was marked using the point where it divides into the posterior cerebral arteries as reference. The MMA segment was identified by marking the branch from the main trunk of the maxillary artery and ICAextra and STA segments were identified using the ipsilateral start of the MMA as a reference. In each scan, a vessel segment with the exact same start and end point was identified. The software automatically calculated a path-line and measured the circumference of the vessel every 0.2 mm perpendicular to the centreline, from which the average of 26 values (5-mm-long vessel segment) was finally obtained. The software uses a local gradient for contour detection. The location of the maximum gradient (which is where the contour is located) is not influenced by signal-to-noise ratio (SNR) until the noise becomes very large (but in such conditions even human observers have difficulty drawing the contours). All images had to pass a full quality assurance (QA). The image quality of each artery was assessed by measuring SNR by dividing the mean signal intensity from a region of interest with the standard deviation of the mean signal intensity from the air surrounding the subject. The SNR level was acceptable for all arteries (ICAin: 187, BA: 185, MCA: 171, ICAex: 104, STA: 77, MMA: 72).
Positions of the measured vessel segments are marked with black lines: (a) superficial temporal artery, (b) extracranial part of the internal carotid artery, (c) middle meningeal artery, (d) middle cerebral artery, (e) basilar artery and (f) intracranial part of the internal carotid artery. The superficial temporal artery (STA) (open circle) changed −16.4% (95% CI −20.7 to −12.2), the extracranial internal carotid artery (ICAextra) (open square) change was −15.2% (95% CI −16.9 to −13.5) and the middle meningeal artery (MMA) (open triangle) changed −16.5% (95% CI −21.8 to −11.2). The middle cerebral artery (MCA) (closed circle) changed by −5.5% (95% CI −8.69 to −2.38). The basilar artery (BA) (closed triangle) showed a small but significant change by −2.1% (95% CI −3.1 to −1.1), whereas the intracranial internal carotid artery (ICAintra) (closed square) change was not significant −1.8% (95% CI −4.3 to 0.65). Error bars represent 95% CI.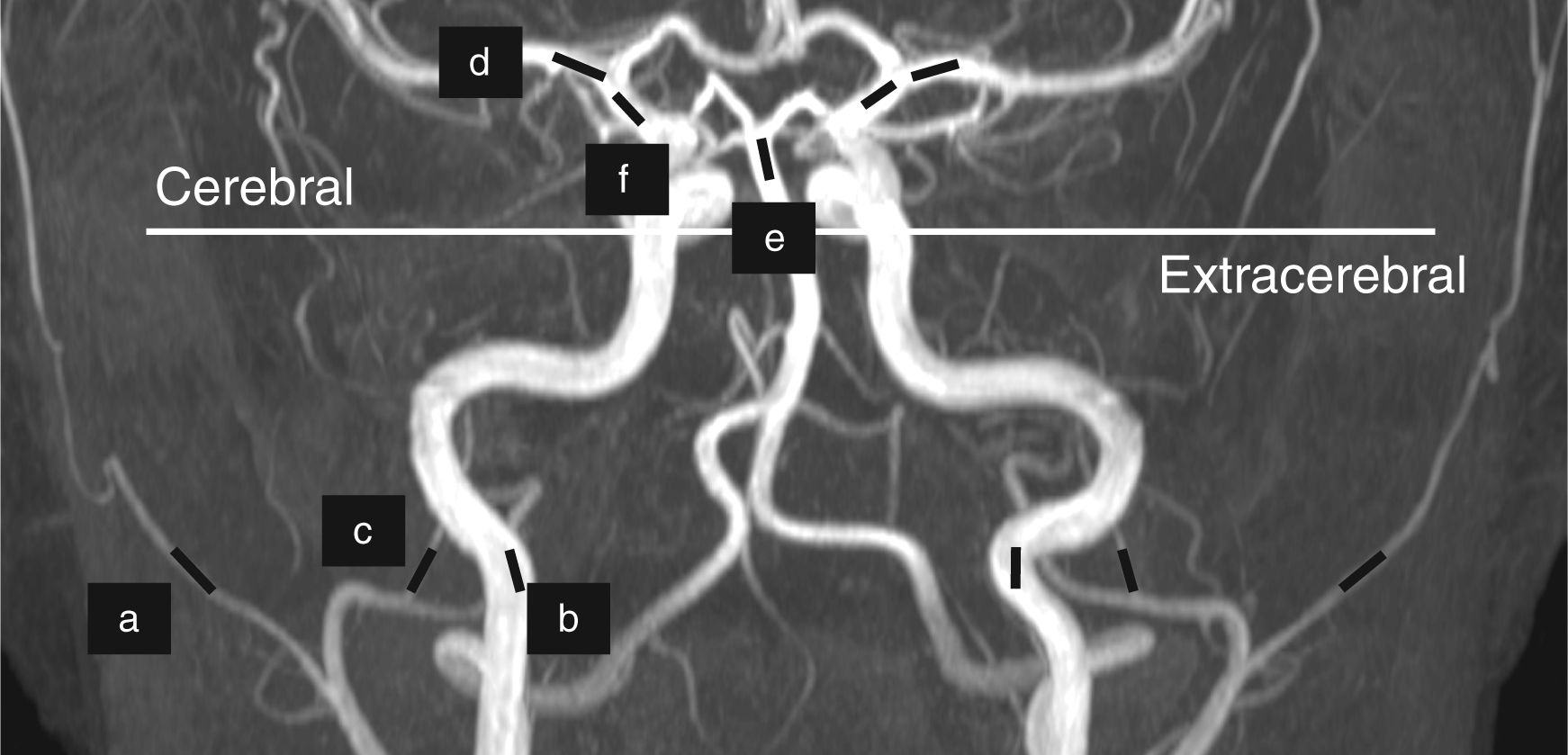

Statistical analysis
All absolute values are presented as median and quartiles in parenthesis. Percent changes are reported as mean with 95% CI.
The primary endpoints were differences in the BA, ICAextra, ICAintra, MCA, MMA and STA circumferences between pre- and post-sumatriptan recordings. To explore a possible selective effect of sumatriptan, we compared the relative changes ((post-sumatriptan – pre-sumatriptan)/pre-sumatriptan) between the cerebral and extracerebral arteries. The Wilcoxon signed rank test was used to test all endpoints. The analyses were performed using IBM® SPSS® Statistics 20.0 for MAC. Level of significance at 0.05 was accepted.
Results
Median absolute change of circumferences (quartiles) of right- and left-sided arteries between before, and 15 minutes after 6 mg subcutaneous sumatriptan injection in healthy volunteers.
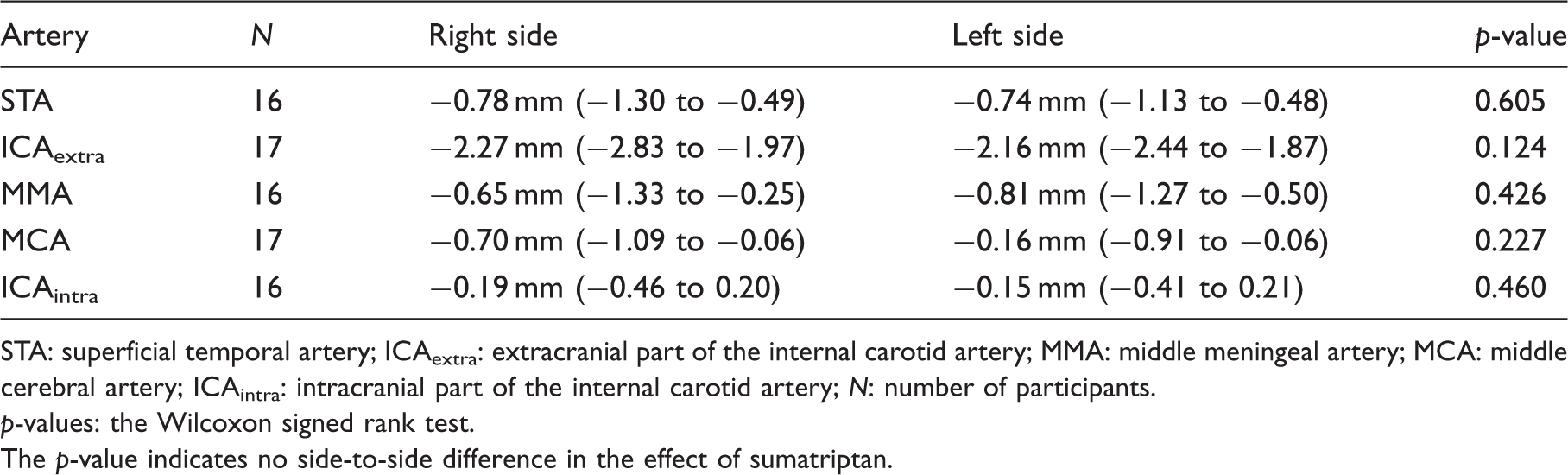
STA: superficial temporal artery; ICAextra: extracranial part of the internal carotid artery; MMA: middle meningeal artery; MCA: middle cerebral artery; ICAintra: intracranial part of the internal carotid artery; N: number of participants.
p-values: the Wilcoxon signed rank test.
The p-value indicates no side-to-side difference in the effect of sumatriptan.
The effect of sumatriptan on extracerebral arteries
Median circumferences (quartiles) of selected intra- and extracerebral arteries before, and 15 minutes after 6 mg subcutaneous sumatriptan injection in healthy volunteers.

STA: superficial temporal artery; ICAextra: extracranial part of the internal carotid artery; MMA: middle meningeal artery; MCA: middle cerebral artery; BA: basilar artery; ICAintra: intracranial part of the internal carotid artery; N: number of participants.
p-values: the Wilcoxon signed rank test.
The effect of sumatriptan on cerebral arteries
We found a constriction of the MCA (p = 0.003) and the BA (p = 0.012), but not of the ICAintra (p = 0.179) after sumatriptan (Table 2). Sumatriptan changed the circumference of MCA by −5.5% (95% CI −8.69 to −2.38). The BA circumference showed a small but significant change by −2.1% (95% CI −3.1 to −1.1), whereas the ICAintra change was not significant −1.8% (95% CI −4.3 to 0.65) (Figure 3).
The effect of sumatriptan on cerebral versus extracerebral arteries
The mean change of the extracerebral arterial segments was −16.0% (95% CI −18.1 to −13.9) compared with a −3.4% (95% CI −5.0 to −1.8) change of the cerebral arterial segments. Sumatriptan induced significantly larger constriction of the extracerebral (STA, ICAextra and MMA) than of the cerebral (MCA, BA and ICAintra) arteries (p < 0.000001) (Figure 4).
Mean per cent circumference change of the cerebral (n = 41) and extracerebral (n = 49) arterial segments after sumatriptan. Mean change of the cerebral arteries (open square) was −3.4% (95% CI −5.0 to −1.8) and 16.0% (95% CI −18.1 to −13.9) of the extracerebral arteries (closed square). Error bars represent 95% CI.
The effect of sumatriptan on the heart rate, blood pressure and respiration
Mean (±SEM) end-tidal partial pressure of CO2 (PetCO2), blood oxygen saturation (SatO2), heart rate (HR), systolic (SBT) and diastolic blood pressures (DBT).

Discussion
The major finding of the present study was that sumatriptan constricted both the extracerebral and cerebral arteries in healthy volunteers. The explorative analysis revealed that sumatriptan exerts a stronger vasoconstrictive effect on the extracerebral (mean circumference reduction by 16.0%) than on the cerebral arteries (mean circumference reduction by 3.4%).
Human in vitro studies of the sumatriptan effect on cephalic arteries
Jansen et al. (11) obtained human arteries from patients undergoing neurosurgery and characterized the 5-HT receptor subtypes in the temporal, middle meningeal and pial arteries in vitro. The authors reported no significant difference in the contractile effect (-log concentration of agonist eliciting half maximum constriction (pD2)) of sumatriptan between the middle meningeal (pD2: 6.5) and pial arteries (pD2: 6.2). Furthermore, there was no difference between the pial and temporal arteries (pD2: 5.9). The sumatriptan effect was tested on arteries that were suspended in tissue baths, hence it was not possible to distinguish possible differences in effect between intra- and periluminal applications of sumatriptan. No difference in the contractile effect between the pial and temporal arteries is in contrast to our findings. Theoretically, there could be a difference in receptor distribution between the proximal MCA (recorded by MRA) and distal pial branches, but, to date, no study has reported such a difference.
The effect of sumatriptan has also been examined in post-mortem human arteries. Parsons et al. (12) investigated the effect of GR43175 (sumatriptan) on endothelium denuded human basilar artery in vitro and reported a potent contractile effect of GR43175 on the basilar artery (pD2: 6.45). This has been confirmed in two more studies of the basilar artery (13,14). Interestingly, it seems that the potencies (pD2 values) found in post-mortem and in vitro vessels from patients undergoing neurosurgery are comparable. The effect of sumatriptan on the internal carotid artery (ICA) has been investigated in two studies (15,16). One post-mortem study reported the lowest effect in the ICA compared with the meningeal, temporal and basilar arteries (15). However, the exact localization of the used ICA was not described. Hardebo (16) reported the presence of potent contractile sumatriptan receptors in post-mortem human intracranial ICA (i.e. proximal extracavernous and the proximal, intermediate and distal intracavernous). No difference in receptor potency between segments of the vessel was detected. The in vitro studies cannot, therefore, explain the discrepancy between the sumatriptan response in the ICAintra and ICAextra in our study.
Collectively, human in vitro studies suggest the following rank order of sumatriptans constrictive effect on cephalic arteries: the middle meningeal > the basilar > the pial > the temporal > internal carotid artery.
The discrepancy between human in vivo and in vitro studies of sumatriptan
The rank order of sumatriptan’s constrictive effect in the present study is in contrast to the human in vitro studies. We found the following rank order: MMA > STA > ICAextra > MCA > BA > ICAintra. Although it is assumed that there are local variations in the receptor occurrences along the arteries in favour of the most distal branches, it does not explain the response of the BA. It has been proposed that the BBB is more permeable in the circumventricular areas (17), wherein the rostral part of the BA is located, which is the area we have examined in the study (Figure 2). Furthermore, according to the rank order gained from in vitro studies, a more pronounced constriction of the BA should be expected. However, we found that BA constricted eight times less than the MMA and the STA (i.e. ∼2% vs ∼16%), when we compared the proposed most permeable part of the BA with the STA. One could criticize the small number of BA investigated in our study (n = 8). However, we calculated a narrow confidence interval (95% CI −3.1 to −1.1), indicating that the finding was relatively precise. Another important difference between in vivo and in vitro conditions could be cerebrovascular autoregulation, which is most sensitive to changes in blood CO2 levels and the blood pressure. We saw no change in the end-tidal partial pressure of CO2 after sumatriptan (Table 3). Furthermore, the mean arterial blood pressure was increased (8–10%), which theoretically should cause a secondary vasoconstriction to maintain constant cerebral blood flow. However, larger arteries play only a minor role in the autoregulation compared with the smaller arterioles. It is therefore unlikely that these systemic parameters caused the different arterial responses between cerebral and extracerebral arteries in the present study.
Given that there seems to be no difference in receptor potency between extracerebral and cerebral arteries, and that sumatriptan receptors have been identified in the smooth muscle cells of extracerebral as well as cerebral arteries (including MCA) (18), the smaller effect of sumatriptan on cerebral arteries in the present study suggests a limited ability of sumatriptan to cross the blood–brain barrier (BBB). This is supported by imaging studies of the effect of sumatriptan during migraine attacks. Sumatriptan was not able to affect neuronal activity in the brain stem during migraine attacks (19). It has been debated for more than a decade whether drugs must reach the central nervous system to induce anti-migraine effects (3,20). The present data suggest that sumatriptan may exert its main anti-migraine action outside of the BBB.
Is the effect of sumatriptan on cerebral arteries real or a time effect?
The slight cerebral arterial circumference reductions found in this study could be a result of a time effect, as the sumatriptan response was not placebo controlled. Before sumatriptan administration, the subjects were scanned at baseline and after a placebo infusion (isotonic saline) (Figure 1). In a previous publication (9), we presented the changes 30 minutes after placebo infusion: MCA dilated 2.9% (95% CI −0.80 to 4.63) and MMA dilated 4.8% (95% CI −0.51 to 10.12) (p > 0.05). In addition, there was no circumference change of any artery after placebo (ICAin −0.8%, ICAex 0.3%, STA −1.3% and BA 1.6%) (p ≤ 0.368) (data not shown). In another study using the same MRA method as the present study, we scanned healthy volunteers 20 minutes after placebo and found MMA dilated by 1.2% and MCA constricted by −0.8% (p > 0.05). These participants were scanned again after 2 hours and the MMA increase was 6.4%, whereas the MCA decrease was −2.0% (p > 0.05) (21). These results suggest that the changes found in the present study, although statistically significant in two out of three cerebral arteries, are close to the variation seen in repetitive arterial measurements.
Peripheral site of action of sumatriptan in migraine: vascular or neuronal?
The present study, consistent with several previous human in vivo studies (8,21,22), confirms a potent peripheral vasoconstrictive effect of sumatriptan. Similar constrictions (i.e. MMA −19.8% vs MCA −5.3%) have also been reported in migraine patients without aura (8). However, the role of mechanical vasoconstriction in the anti-migraine effect of sumatriptan has been disputed (7,23). In particular, the efficacy of CGRP antagonists (24) and of the 5-HT1F-receptor agonist lasmitidan (25), underlines the importance of non-vascular actions. Levy et al. (7) reported that sumatriptan caused a disruption of communication between the peripheral (sensory terminals) and central trigeminovascular neurons, and Jakubowski et al. (26) found that sumatriptan was not effective once allodynia (correlates to sensitization of second order neurons) was established during a migraine attack. We previously found a unilateral dilatation of the MMA and MCA during migraine attack (8). Interestingly, all patients reported amelioration of migraine after sumatriptan, but the MCA dilatation was still present.
Conclusion
In contrast to previous in vitro studies, the present paper demonstrates that the extracerebral arteries (MMA, STA, ICAextra) constricts significantly more than the cerebral arteries (MCA, BA, ICAintra) after subcutaneous sumatriptan administration. We therefore suggest that sumatriptan may not cross the blood–brain barrier to an extent that is enough to cause constriction of cerebral arteries comparable with the constrictive levels in vitro.
Clinical implications
The present paper demonstrates that the extracerebral arteries (MMA, STA, ICAextra) constrict significantly more than the cerebral arteries (MCA, BA, ICAintra) after subcutaneous sumatriptan administration. We therefore suggest that sumatriptan may not cross the blood–brain barrier and primarily exerts its effect outside of the blood–brain barrier.
Footnotes
Funding
The study was supported by grants from the University of Copenhagen, the Lundbeck Foundation through the Center for Neurovascular Signaling (LUCENS), The Research Foundation of the Capital Region of Denmark; Danish Council for Independent Research-Medical Sciences (FSS) (grant 271-08-0446); The Novo Nordisk Foundation (R172-A14333).
Acknowledgements
The authors thank all participating subjects; Dr Anders Hougaard, radiographers Bente S Møller, Marjut Lindhardt and Helle J Simonsen; physicist Dr Adam E Hansen.
Conflict of interest
JO has received grants and/or research support from, has been a consultant and/or scientific adviser for, and has been in the speakers’ bureau of Allergan Inc, AstraZeneca Pharmaceuticals LP, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Janssen Pharmaceutical Products, Lundbeck, Merck and Pfizer. MA has received grant support and honoraria for lecturing from Merck, and honoraria for lecturing from Pfizer, GlaxoSmithKline, Norpharma and AstraZeneca, and is a consultant and/or scientific adviser for Allergan, Amgen and Alder. FMA has received honoraria for lecturing from Allergan. There are no conflicts of interest for the remaining authors.
