Abstract
A novel model-based meta-analysis was used to quantify the dose-response relationship of sumatriptan and eletriptan for the proportion of patients that achieve migraine pain relief up to 4h after treatment. The proportion of patients that became pain free was also evaluated. This analysis includes some unique features, allowing comparison of sumatriptan and eletriptan doses that have not been directly compared in a head to head study and also permitting comparison between the two drugs at multiple time points up to 4 h after treatment. Because the analysis allows comparison of response to blinded sumatriptan with that to marketed sumatriptan and contains timepoints as early as 0.5 h, it is especially suited to detection of possible effects of encapsulation on sumatriptan's therapeutic effectiveness and thus was employed to assess this also. Data from 19 randomized placebo controlled clinical trials were jointly analysed using a random-effects logistic regression model. The results of this analysis show a significant clinical benefit of eletriptan 40 mg compared to sumatriptan 100 mg at any point in time up to 4 h after treatment. The benefit of eletriptan 40 mg is greatest around 1.5-2 h after treatment with an absolute difference at 2 h of 9.1% (7.4-11.5%) more patients achieving pain relief and 7.3% (5.8-8.6%) more patient achieving pain free when compared to sumatriptan 100 mg. An absolute benefit of more than 5% of patients is maintained from 45 min up to 4 h after treatment for pain relief and from 1.5 h up to 4 h for pain free. Eletriptan 20 mg was superior to sumatriptan 50 mg and similar to sumatriptan 100 mg for pain relief while it was similar to sumatriptan 50 mg for pain free. The benefit of eletriptan 20 mg when compared to sumatriptan 50 mg is greatest around 1.5-2 h after treatment with an absolute difference at 2 h of 5.0% (2.9-8.1%) more patients achieving pain relief. An absolute benefit of more than 3% of patients was maintained from 1 h up to 3 h after treatment. No significant difference was found between eletriptan 20 mg and sumatriptan 50 mg for the fraction of patients that became pain free. No significant effect of encapsulation of sumatriptan was found on the time course of response up to 4 h after treatment when compared to commercial sumatriptan.
Introduction
The introduction of selective 5-HT1B/1D agonists, or triptans, has provided a safe and effective treatment option for acute migraine. Eletriptan is a recent addition to the treatment arsenal that has been shown to be more efficacious than its predecessor sumatriptan in head-to-head comparative studies (1–3). Given the numerous triptans that are available, important differentiators between them might include speed of onset, efficacy, duration of action, tolerability and safety. In a large comparative trial, eletriptan 40 mg demonstrated significantly greater efficacy for pain relief at 2 h than sumatriptan 100 mg (67% vs. 59%) (1). Significant benefits of eletriptan 40 mg were also seen for other endpoints such as pain free at 2 h, treatment acceptability, and sustained pain relief. Similar results were obtained in two other comparative trials (2, 3).
The objective of this paper is to characterize the dose–response relationship of sumatriptan and eletriptan for patients that achieve migraine pain relief up to 4 h after treatment on basis of the collective clinical knowledge. The dose–response relationship for the fraction of patients that become pain free was also analysed. We combined the results from 19 randomized controlled clinical trials representing about 11 400 patients into a meta-analysis using a random-effects logistic regression model. An integrated analysis of all relevant and available clinical trials provides a meaningful way to assess potential differences in treatment success on the basis of current clinical knowledge (4). One of the benefits of such an integrated analysis is to augment the results of comparative clinical trials with trials that evaluated either sumatriptan alone or eletriptan alone. In addition to augmenting the comparison of doses that have been compared in head-to-head trials, the current analysis allows a comparison of eletriptan and sumatriptan doses that have not been compared directly in controlled studies. The analysis also allows a comparison between the two drugs at any time point up to 4 h after treatment.
Methods
Studies included
Using Medline, we performed a comprehensive search for clinical trials that included sumatriptan for the treatment of migraine. The cut-off date for the retrieval of publications was August 30, 2002. Pfizer provided study reports of clinical trials that evaluated eletriptan. The results from all of these trials have since been published (1–3,5–8). For inclusion in the analysis, each trial had to meet the following criteria:
randomized double-blind placebo controlled trial;
treatment of moderate or severe migraine in adults within 8 h of onset;
measurement of relief from migraine pain on a four point categorical scale of none, mild, moderate, severe;
includes efficacy results for the first attack;
no remedication or rescue before 2 h.
We specifically searched for studies that included multiple doses of sumatriptan or eletriptan, or studies that assessed pain relief at multiple time points after treatment. Data at 4 h after dosing were included only if no rescue or remedication was allowed until this point in time. Dose–response studies available in the summary basis for FDA approval of sumatriptan were also evaluated (9). An overview of the 19 clinical trials that were included in the analysis is provided in Table 1.
Summary of trials included in the analysis: number of patients in each dose group based on the 2- h headache response, and the time points that were evaluated. P = placebo; Sn = Sumatriptan dose (mg); En = Eletriptan dose (mg)

Endpoints
Pain relief was defined as an improvement in headache pain score from a baseline of moderate or severe intensity to mild or no pain. Pain free was defined as an improvement in headache pain score from a baseline of moderate or severe intensity to no pain. Pain free data was available for 17 out of the 19 trials (no pain free data in [12, 14]).
Statistical analysis
A trial specific random-effects nonlinear logistic regression analysis was used to quantify the time course of the dose–response relationship of sumatriptan and eletriptan for the proportion of patients that achieve pain relief or that become pain free. The following general model structure was used to capture the dose–response relationship for the probability of having pain relief or pain free (P) for each drug:

In this equation, E 0 is the intercept, reflecting placebo response; E max is the maximal drug effect, reflecting the maximal difference in response between placebo and active treatment; Dose is the dose of the drug; ED50 is the dose of the drug to achieve 50% of Emax; η is a trial specific random effect with variance ω2; and g(x} is the inverse logit-transformation to constrain the probabilities between 0 and 1. An important aspect in performing an analysis that incorporates data from different trials is to account for potential heterogeneity among the different studies. The trial specific random-effects model accounts for potential heterogeneity due to, for example, random trial-to-trial difference in patient populations, so that a realistic comparison can be made across all trials of the effect of sumatriptan and eletriptan relative to placebo as well as each other.
The Laplacian approximation to the log-likelihood as implemented in the NONMEM program (NONMEM version V) was used to provide maximum likelihood estimates of the model parameters (23). The dependency of the model parameters (E 0 , E max , ED 50) on drug, time since dosing, and encapsulation was evaluated. Final model selection was done on the basis of the Log Likelihood criterion (P < 0.05). The difference in − 2 times the Log of the Likelihood (−2LL) between a full and reduced model is approximately asymptotically χ2 distributed with degrees of freedom equal to the difference in number of parameters between the two models. A decrease of more than 3.84 in −2LL is significant at the P < 0.05 level for 1 additional parameter. Standard errors of the parameter estimates were approximated using the asymptotic variance-covariance matrix.
Results
Model
The following models best described the intercept (E 0), maximal difference between placebo and active treatment (E max), and dose at which 50% of the maximal effect is attained (ED 50):
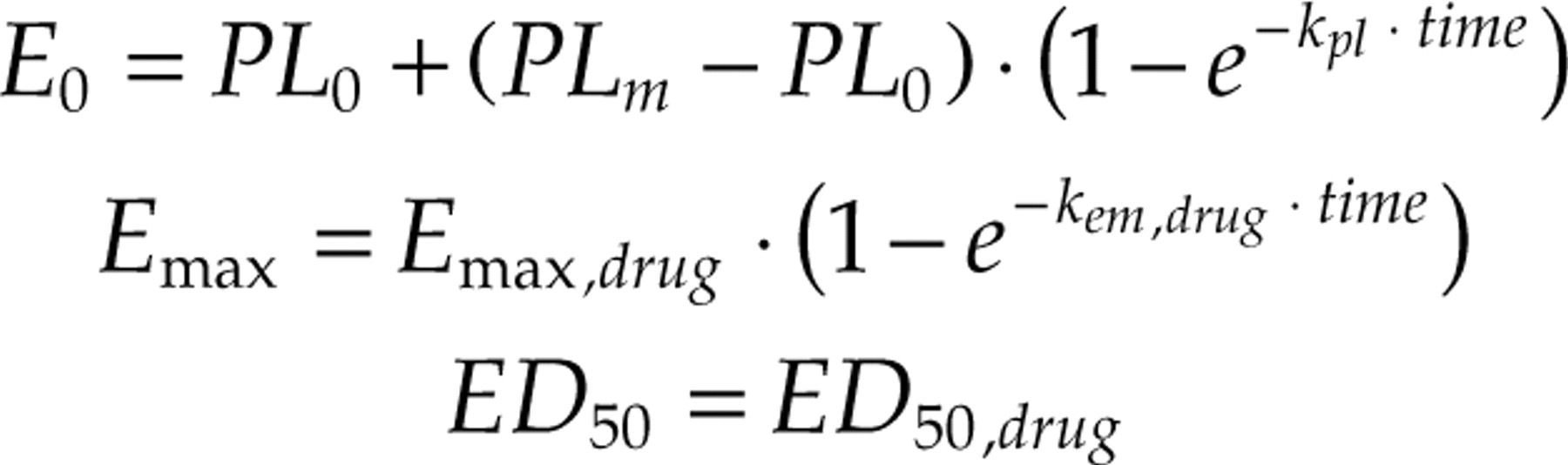
The model parameters are presented in Table 2.
Estimated model parameters and their 90% confidence interval
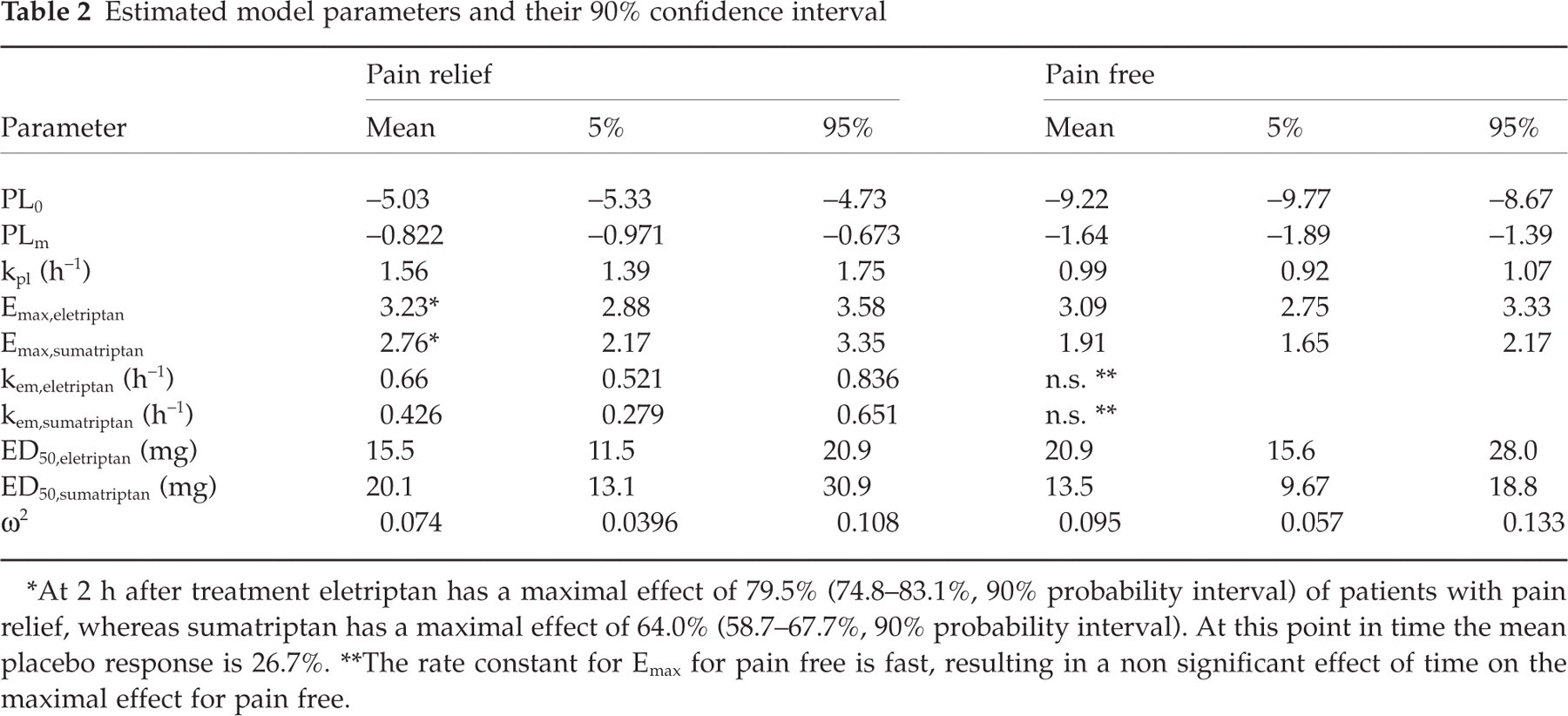
At 2 h after treatment eletriptan has a maximal effect of 79.5% (74.8–83.1%, 90% probability interval) of patients with pain relief, whereas sumatriptan has a maximal effect of 64.0% (58.7–67.7%, 90% probability interval). At this point in time the mean placebo response is 26.7%.
The rate constant for Emax for pain free is fast, resulting in a non significant effect of time on the maximal effect for pain free.
Figure 1 shows the fit of the dose–response model for eletriptan and sumatriptan to the combined data from all trials stratified by time since dosing. The figure indicates an adequate summarization of the data by the proposed dose–response model. This is confirmed by the fact that a model that assumes an independent effect for each dose (i.e. an

Fit of the dose-response model for (a,c) eletriptan and (b,d) sumatriptan to the combined data from all trials, stratified by time since dosing ○ 4 h; ▴ 2 h;□ 1 h; × 0.5 h. (a,b) show pain relief, whereas (c,d) shows the results for pain free. The solid lines represent the model-predicted dose response relationships at specific time points after dosing in a typical trial. The symbols reflect the mean (across all trials) observed fraction of patients with pain relief or pain free at each evaluated dose and time point after adjusting for the random trial-to-trial differences. The error bar around each of the symbols reflects a 95% confidence interval on the observed fraction of patients.
The intercept or placebo response of the model was found to be an exponential function of time, starting at PL
0 at time 0 and increasing to PL
m at infinite time with a rate constant of k
pl. The data at 0 mg (placebo) in Fig. 1 shows that the exponential model captures the placebo response well. This is confirmed by the fact that a model for E
0 that assumes an independent effect for each time point (i.e. an
The maximal drug effect for pain relief was found to be an exponential function of time with a statistically significant different E max and rate constant (k em) for eletriptan and sumatriptan, indicating differences in efficacy and rate of onset between the two compounds (P < 0.001).
Figure 1 shows that this simple dose–response model describes the data of both eletriptan and sumatriptan well. The rate constant with which E max is being achieved is significantly larger for eletriptan when compared to sumatriptan, reflecting a quicker onset of effect for eletriptan vs. sumatriptan. Together with a larger E max of eletriptan this results in a greater maximal attainable effect of eletriptan compared to sumatriptan at any time point up to 4 h after dosing. The maximal drug effect for pain free was found to be constant with time, but significantly different between eletriptan and sumatriptan (P < 0.001). The fact that no significant time effect was found for pain free means that the rate of onset of effect is similar between placebo, sumatriptan and eletriptan.
Heterogeneity
Figure 1 shows how well the model characterizes the aggregate data from all trials. A statistically significant random trial effect was found for pain relief as well as pain free (P < 0.001), indicating heterogeneity between the studies on the overall mean. The random effect influences all dose groups in a specific study and shift all responses up or down on the logit scale to a similar extent. An important question is whether, after accounting for these study effects, the responses observed in each trial are well predicted by the model, and no further heterogeneity is present. An example of this is shown for the pain relief data at 2 h in Fig. 2. Figure 2 shows that after we account for those trial specific random effects, the observed response for each treatment group and each trial is well predicted. Figure 2 is similar to Fig. 1, however, in this figure each point represents the outcome of a specific trial, instead of the mean response across all trials. As in the previous figure, the symbols represent the observed fraction of patients that have pain relief, adjusted for the trial-specific random effect so that they can be easily compared to the model mean prediction. The figure shows that response for each treatment group in each trial is well described by the model, especially for the treatments of particular interest, 50 and 100 mg sumatriptan and 20 and 40 mg eletriptan. For clarity only the two-hour data is shown, but similar homogeneity is observed for the other time points and the pain free data. This homogeneity suggests that the headache responses in each trial are quite similar after accounting for a difference in the intercept for each trial. We can interpret this to mean that the random differences in patient populations from trial-to-trial affect the response to placebo and active treatment to a similar extent. This implies homogeneity in the difference in response between placebo and active treatment, similar to what was found in the meta-analysis by Ferrari et al. (4). The only difference is that this analysis considers the difference on the logit scale to be consistent, whereas Ferrari et al. used the absolute difference.
Homogeneity of treatment outcome across trials. The symbols reflect the mean and 95% confidence interval for the fraction of patients with pain relief at 2 hours for all treatment options in each of the evaluated trials after adjusting for the random trial-to-trial differences. Each symbol reflects the results of one trial. The solid vertical lines represent the model-predicted response for a typical trial.
Encapsulation
The comparative studies between eletriptan and sumatriptan encapsulated the commercial sumatriptan tablets to ensure adequate medication blinding. The issue has been raised that the encapsulation could potentially impact the response by significantly delaying absorption. In addition to the comparative studies between eletriptan and sumatriptan, one other study reported the use of an encapsulated form of sumatriptan (20). The benefit of the current analysis is that we can compare the time course of effect of the encapsulated form of sumatriptan to the time course of response to commercial (unencapsulated) sumatriptan. Figure 3 shows this comparison for 100 mg sumatriptan for the pain relief data. The different symbols reflect the observed mean response of 100 mg encapsulated sumatriptan and 100 mg commercial sumatriptan after accounting for random trial-to-trial differences. The solid line is the model-predicted time course of response of 100 mg sumatriptan in a typical trial. The figure clearly shows that there are no differences in headache response at any point in time between 100 mg encapsulated sumatriptan and 100 mg commercial sumatriptan. If anything, at two hours the response to encapsulated sumatriptan is slightly larger than the response to commercial sumatriptan. At two hours this difference is estimated to be 3.4% (90% confidence interval: − 3.2 to 6.4). Similar results are found for 50 mg Sumatriptan. The absence of an effect of encapsulation on the time course of response of sumatriptan is confirmed by a detailed statistical analysis. There was no significant difference in E max (P = 0.14), the rate constant for the maximal effect, k em (P = 0.15), or the ED 50 (P = 0.19) between encapsulated sumatriptan compared to commercial sumatriptan for the pain relief data. Because the encapsulated form of sumatriptan used in the eletriptan comparative studies is likely to be different from the encapsulation used in the other studies we also tested for the effect of encapsulation of sumatriptan in the eletriptan comparative trials vs. sumatriptan in all other trials. No significant effects of encapsulation were found, confirming the absence of an effect of encapsulation in general and in the eletriptan comparative studies in particular on the time course of headache response to sumatriptan.

Lack of impact of encapsulation on the time course of response to 100 mg Sumatriptan. The solid line represents the model-predicted time course of response to 100 mg Sumatriptan in a typical trial. The symbols reflect the observed mean response to 100 mg encapsulated sumatriptan (▵) and commercial (unencapsulated) sumatriptan (○) after accounting for random trial-to-trial differences. The error bar reflects a 95% confidence interval on the observed fraction of patients with pain relief.
Predictions
The model can be used to simulate the expected time course of the dose–response relationship for eletriptan and sumatriptan and it's associated uncertainty. The predictive distribution of the headache pain free and pain relief response was derived by sampling 5000 sets of model parameters from the variance matrix of the parameter estimates. For each set of parameters the dose–response relationship of eletriptan and sumatriptan was calculated for a typical trial (i.e. the trial specific random effect is zero, yielding the dose–response relationship for a representative patient population in a typical trial). The 90% uncertainty interval is taken between the 5th and 95th percentile of the predictive distribution.
Pain relief
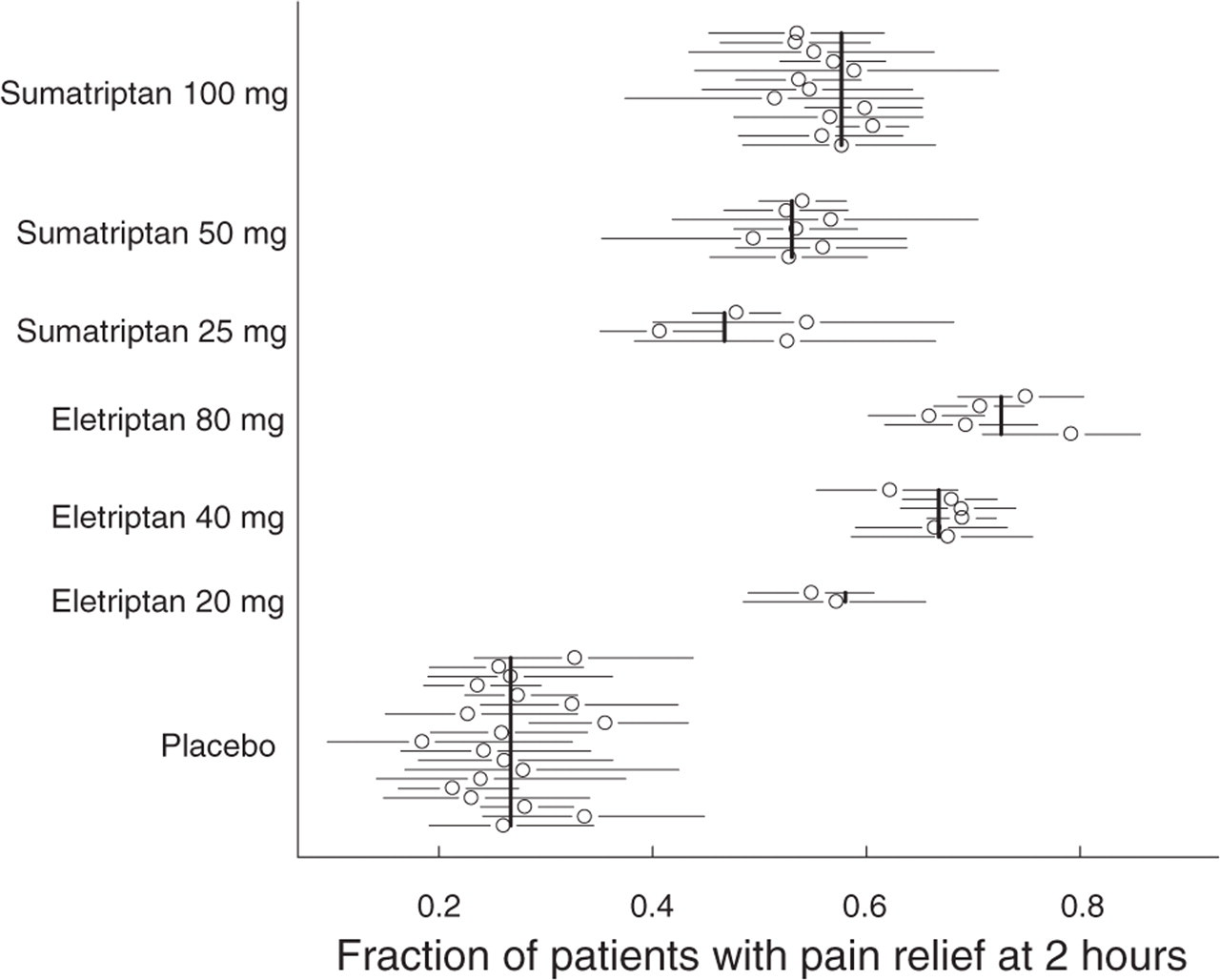
Table 3 shows the expected difference from placebo in the percentage of patients that have migraine pain relief (therapeutic gain) for sumatriptan 50 and 100 mg, and eletriptan 20 and 40 mg. Even though eletriptan 80 mg was included in the dose–response analysis, this dose is not included in the predictions. Because eletriptan 20 and 40 mg are the approved doses in the US, these doses were chosen for detailed comparison with sumatriptan. The therapeutic gain for sumatriptan 100 mg at 2 h is 31% (27–33%, 90% uncertainty interval). This value is similar to the 29% reported in the meta-analysis by Ferrari et al. (4), confirming the predictive validity of the model. Sumatriptan 100 mg has a small benefit compared to sumatriptan 50 mg. The difference however, is less than 5% of patients with headache relief at any point in time. The table shows the benefit of eletriptan 40 mg over 100 mg sumatriptan at all time points up to 4 h after treatment, whereas eletriptan 20 mg seems to be equivalent to 100 mg sumatriptan. The 2-hour therapeutic gain for both sumatriptan 100 mg and eletriptan 20 mg is about 31%.
Difference from placebo in the percentage of patients that have pain relief (therapeutic gain) for sumatriptan 50 and 100 mg, and eletriptan 20 and 40 mg as a function of time. Mean and 90% probability interval are shown
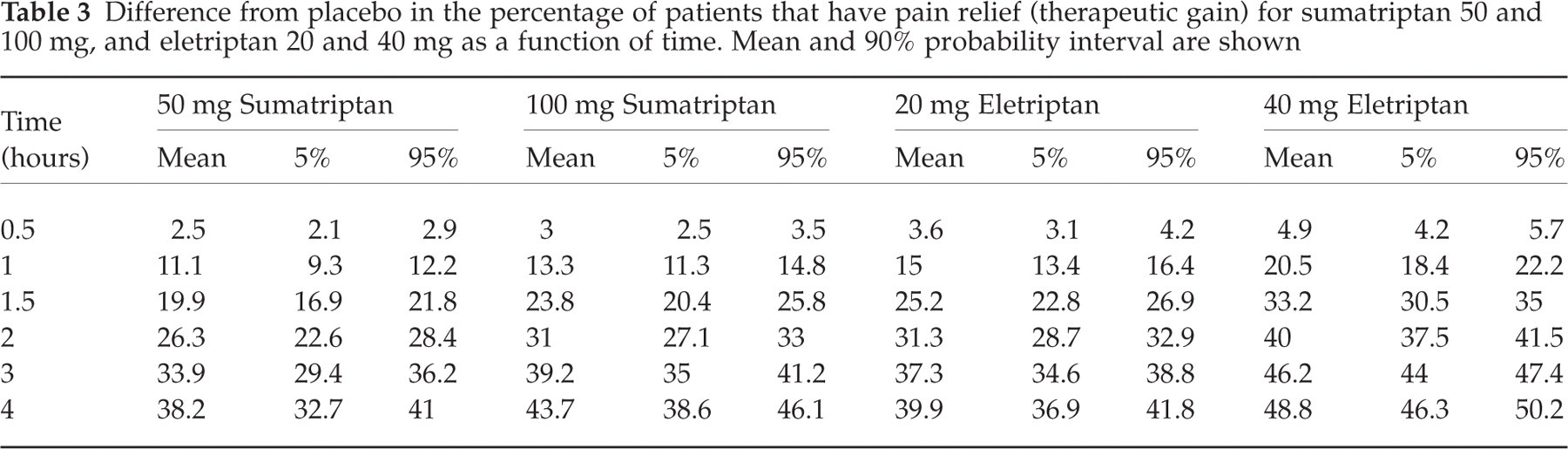
Because we have analysed the dose–response relationship of eletriptan and sumatriptan jointly, we can make a direct comparison between the two drugs. Figure 4 shows the therapeutic gain of eletriptan 40 mg over sumatriptan 100 mg and of eletriptan 20 mg over sumatriptan 50 mg. The solid line is the expected difference in per cent patients with pain relief in a typical trial, whereas the dashed lines span a 90% uncertainty interval. The scale of the figure is based on the therapeutic gain of sumatriptan over placebo at 2 h so that the magnitude of the benefit of eletriptan over sumatriptan can be interpreted relative to the benefit of sumatriptan over placebo. Table 4 summarizes the difference in headache response between sumatriptan 50 mg and eletriptan 20 and 40 mg as well as the difference between sumatriptan 100 mg and eletriptan 20 and 40 mg. The data shows that eletriptan 20 mg is similar to sumatriptan 100 mg. The expected difference is less than 5%, and the 90% uncertainty interval includes 0 up to 4 h after treatment. Eletriptan 40 mg, on the other hand, is superior to sumatriptan 100 mg. The expected therapeutic benefit is greater than 5% between 1 and 4 h after dosing. The benefit peaks at about 1.5 h after dosing with an absolute benefit of 9.4% (7.6–11.6, 90% probability interval). This is a relative benefit in therapeutic gain of 39% over the response of 100 mg sumatriptan at this point in time. Both eletriptan 20 and 40 mg are superior to sumatriptan 50 mg. The expected therapeutic benefit of eletriptan 20 mg compared to sumatriptan 50 mg is greater than 3% between 1 and 3 h after dosing. This is a statistically significant benefit because the lower bound of the uncertainty interval excludes 0 up to 3 h after treatment.
Difference from sumatriptan 50 and 100 mg in the percentage of patients that have pain relief (therapeutic gain) for eletriptan 20 and 40 mg as a function of time. Mean and 90% probability interval are shown
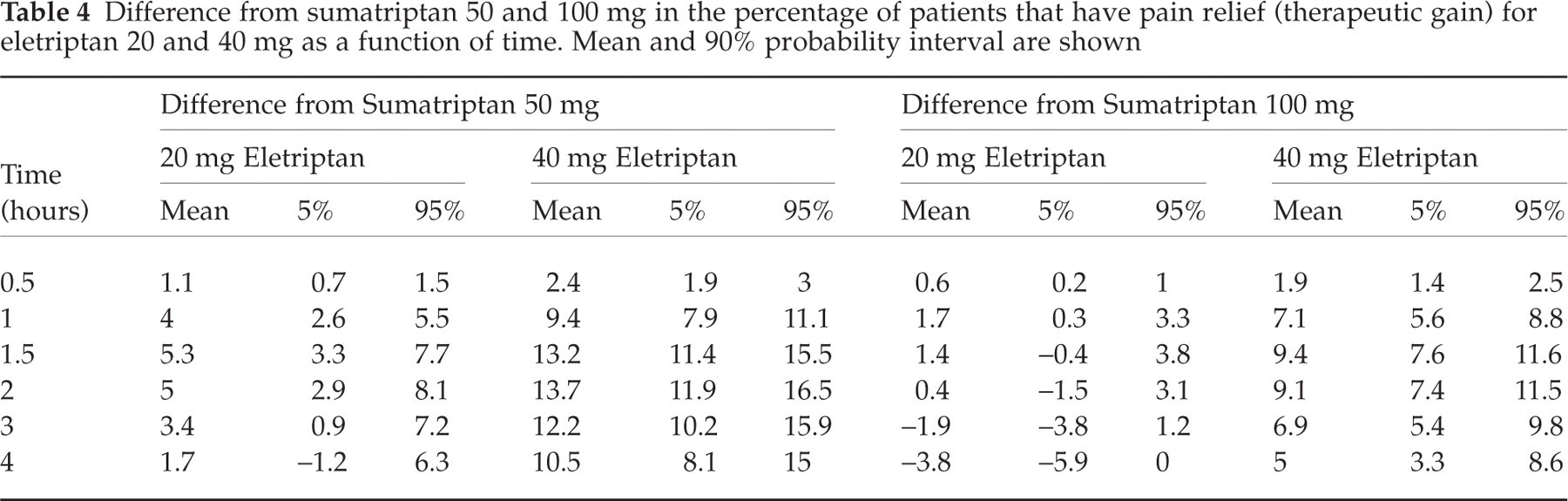
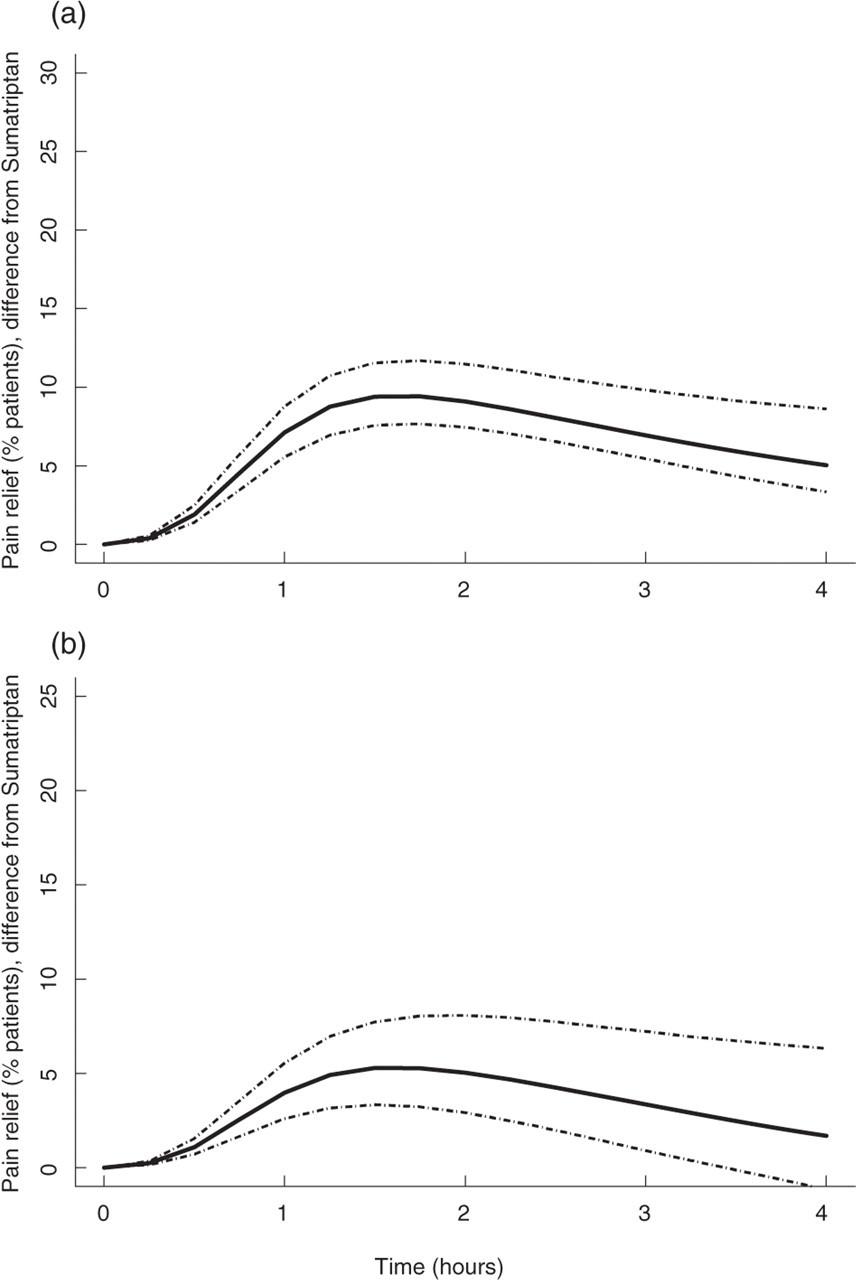
Time course of the absolute difference in the per cent of patients with pain relief (a) between eletriptan 40 mg and sumatriptan 100 mg, and (b) between eletriptan 20 mg and sumatriptan 50 mg. The predictions are shown for a typical patient population in a typical trial. The solid lines represent the model predictions; the dashed lines span a 90% probability interval for those predictions.
Another way of looking at the difference between sumatriptan 100 mg and eletriptan is to consider the probability that eletriptan is superior or equivalent to sumatriptan given the joint data from the 19 clinical trials. We have chosen an absolute difference of 5% in the per cent of patients that have pain relief as a significant clinical difference (for comparison this would be a 16% relative increase in therapeutic gain of 100 mg sumatriptan over placebo at 2 h). Equivalence is then defined as being within + 5% or −5% of the sumatriptan 100 mg headache response. Superior would be a greater than 5% benefit, and inferior, a greater than 5% reduction in response. On basis of these definitions, 20 mg eletriptan has a greater than 99% probability of being equivalent to 100 mg sumatriptan up to 3 h post treatment. At 4 h the probability of being equivalent is 86%. Conversely, eletriptan 40 mg has a greater than 98% chance of being superior to sumatriptan 100 mg up to 3 h post treatment. At 4 h the probability of being superior, i.e. more than 5% absolute difference in patients having headache relief, is 64%.
Pain free
Table 5 shows the expected difference from placebo in the percentage of patients that are pain free (therapeutic gain) for sumatriptan 50 and 100 mg, and eletriptan 20 and 40 mg. The therapeutic gain for pain free for sumatriptan 100 mg at 2 h is 20.5% (18%-23%, 90% uncertainty interval). This value is similar to the 19% reported in the meta-analysis by Ferrari et al. (4), confirming the predictive validity of the model. Sumatriptan 100 mg has a small benefit compared to sumatriptan 50 mg. The difference however, is less than 5% of patients that are pain free at any point in time. The table shows the benefit of eletriptan 40 mg over 100 mg sumatriptan at all time points up to 4 h after treatment, whereas eletriptan 20 mg seems to be equivalent to 50 mg sumatriptan. The 2-hour therapeutic gain for both sumatriptan 50 mg and eletriptan 20 mg is about 17%.
Difference from placebo in the percentage of patients that become pain free for sumatriptan 50 and 100 mg, and eletriptan 20 and 40 mg as a function of time. Mean and 90% probability interval are shown

Figure 5 shows the therapeutic gain of eletriptan 40 mg over sumatriptan 100 mg and of eletriptan 20 mg over sumatriptan 50 mg. The solid line is the expected difference in per cent patients that are pain free in a typical trial, whereas the dashed lines span a 90% uncertainty interval. The scale of the figure is based on the therapeutic gain of sumatriptan over placebo at 2 h so that the magnitude of the benefit of eletriptan over sumatriptan can be interpreted relative to the benefit of sumatriptan over placebo. The data shows that eletriptan 20 mg is similar to sumatriptan 50 mg. The expected difference is very close to zero up to 4 h after treatment. Eletriptan 40 mg, on the other hand, is superior to sumatriptan 100 mg. The expected therapeutic benefit is greater than 5% between 1.5 and 4 h after dosing. The absolute benefit at 2 h after dosing is 7.3% (5.8–8.6%, 90% probability interval). This is a relative benefit in therapeutic gain of 35% over the response of 100 mg sumatriptan at this point in time.

Time course of the absolute difference in the per cent of patients that are pain free (a) between eletriptan 40 mg and sumatriptan 100 mg, and (b) between eletriptan 20 mg and sumatriptan 50 mg. The predictions are shown for a typical patient population. The solid lines represent the model predictions; the dashed lines span a 90% probability interval for those predictions.
Discussion
This paper presents an integrated analysis comparing the therapeutic benefit of sumatriptan and eletriptan for the per cent of patients that achieve migraine pain relief as a function of the dose administered and time since dosing up to 4 h after treatment. The benefit of such an integrated analysis is that it summarizes the clinical performance of the two compounds in a large number of patients, in this case more than 11000, from a large number of trials. Whereas the gold standard for comparing two treatment options is a comparative clinical trial, the large sample sizes required to show potential clinical meaningful differences often limit the comparison of a large number of treatment options. The results of such comparative studies can be greatly enhanced by adding information from other trials, thereby allowing for the comparison of treatments that have not been compared directly. Ferrari et al. (4) recently published an example of the benefit of such a meta-analysis in the field of migraine. The authors compared the relative benefits of seven triptans for pain relief at 2 h, pain free at 2 h, recurrence, intrapatient consistency of response, and tolerability. In the analysis presented in this paper we highlight in more detail the clinical benefit of eletriptan, one of the newest triptans, vs. sumatriptan, the oldest but still widely prescribed triptan. Our analysis differs from Ferrari et al. (4) in that we explore the full dose–response relationship of sumatriptan and eletriptan for headache pain measured as relief and pain free as well as the full time course of response up to 4 h after treatment. To accomplish this, a model-based approach is taken, which allows the direct comparison of the therapeutic benefit of any dose of sumatriptan and eletriptan from start up to 4 h after dosing. The model-based approach augments the lack of information for certain dose strengths and time points by using information from neighbouring doses and time points, increasing the power to detect potential differences.
An additional benefit of the integrated analysis is that we can make a comparison between the trials that use an encapsulated form of sumatriptan and all other trials in which commercial sumatriptan was used. One of the issues raised about the comparative studies is that an encapsulated form of commercial sumatriptan was sometimes used to accomplish blinding and it has been claimed that this introduced a clinically significant delay in absorption (24). One way to address the encapsulation issue is to look for pharmacokinetic differences through bio-equivalence studies. However, those studies are generally conducted in healthy volunteers and not during a migraine attack, factors that can influence the absorption of triptans. Even if pharmacokinetic data in migraine patients were available, the comparison on the basis of plasma concentrations is made difficult by a potential time delay between plasma concentrations and migraine pain relief due to transport of the drug to the site of action. This makes the clinical relevance of potential differences in the pharmacokinetic profile hard to predict. Another way to explore the possible clinical impact of encapsulation is to compare the actual clinical response in patients receiving encapsulated and unencapsulated formulations of sumatriptan. This was done in the current analysis, the results of which show no impact of encapsulation on the headache pain relief response of sumatriptan at any time point up to 4 h after dosing. Figure 3 clearly shows that the response to encapsulated sumatriptan was similar to the response of commercial, unencapsulated sumatriptan. Because of the large sample size and the multiple time points at which this comparison is made, the analysis is quite sensitive to detect potential differences. No statistical significant impact of encapsulation was found on the dose–response relationship of sumatriptan (Emax and ED50) and the rate of onset of effect (kem).
When combining results from different trials, careful attention must be given to the consistency or homogeneity of the outcomes across the trials. Because of potential random, or known, trial-to-trial differences in the patient populations one cannot simply take the mean of the results across all trials. The random effects analysis that we used accounts for such trial-to-trial differences so that an accurate comparison across trials can be made. The analysis showed that the outcomes are very similar from trial-to-trial after accounting for a random effect on the intercept of the model. Since the intercept impacts both placebo and active treatment to a similar extent, this implies that the difference in response (or therapeutic gain) between treatment options is quite consistent from trial-to-trial. This is similar to the results reported by Ferrari et al. (4).
The results of the analysis clearly show the significant clinical benefit of eletriptan 40 mg compared to sumatriptan 100 mg for the per cent of patients that have headache pain relief as well as pain free. As soon as 45 min after treatment the therapeutic gain for pain relief of eletriptan 40 mg compared to sumatriptan 100 mg is more than 5% of patients, which is maintained up to 4 h post treatment (Fig. 4). The benefit of eletriptan is greatest around 1.5–2 h after treatment with a therapeutic gain of more than 9% additional patients having pain relief. Because of the large number of patients included in the analysis and homogeneity across the trials the superiority of eletriptan is well defined by a narrow range of uncertainty (Figs 4 and 4). For example, if we consider an absolute difference of more than 5% of patients clinically superior, there is a greater than 99% chance that eletriptan 40 mg is clinically superior to sumatriptan 100 mg for pain relief between 1 and 3 h after dosing. Otherwise said, there is a greater than 99% chance that the absolute difference in response between these two treatment options is greater than 5% of patients. Starting at 1.5 h after treatment and up to 4 h the therapeutic benefit for pain free is more than 5% of patients for eletriptan 40 mg compared to sumatriptan 100 mg. The therapeutic benefit of eletriptan 40 mg over sumatriptan 100 mg is not associated with less tolerability or more adverse events (1–3). The meta-analysis of Ferrari et al. (4) supports the equivalent tolerability profile of eletriptan 40 mg compared to sumatriptan 100 mg for any adverse event, CNS related adverse events, and chest symptoms. This study also showed the superior performance of eletriptan 40 mg with respect to recurrence of headache within 24 h. Interestingly, the meta-analysis by Ferrari et al. (4) showed a numerical but not statistically significant benefit of eletriptan 40 mg over sumatriptan 100 mg for pain relief at 2 h after treatment, despite the fact that all comparative trials between these two treatment options have shown an effect difference of between 8 and 11% in the benefit of eletriptan, consistent with the results of our analysis. This difference may be due to the fact that we have included an additional large comparative trial that was not included in the analysis of Ferrari et al. (4) thereby significantly increasing the sample size as well as to the fact that we included the complete time course, both of which increase the ability to detect the (statistical) significance of treatment differences. The model-based analysis is further validated by the fact that the predicted benefit of sumatriptan 100 mg vs. placebo is similar to the results presented by Ferrari et al. (4).
The current analysis allows for a comparison between eletriptan 20 mg and sumatriptan 100 mg or 50 mg; combinations that have not been compared extensively in comparative trials. Eletriptan 20 mg is equivalent to sumatriptan 100 mg for pain relief if equivalence is defined as being within +5% or −5% of patients of the sumatriptan 100 mg response. According to this definition of equivalence, there is a greater than 99% probability that eletriptan 20 mg is equivalent to sumatriptan 100 mg up to 3 h after dosing. At 4 h, the probability of being equivalent is still 86%. For pain free, eletriptan 20 mg is equivalent to sumatriptan 50 mg up to 4 h after dosing. Conversely, eletriptan 20 mg has a small, but statistically significant benefit over sumatriptan 50 mg for pain relief, with a maximum therapeutic gain of about 5% of patients between 1.5 and 2 h. It is interesting to note that the benefit of increasing the dose from 20 mg eletriptan to 40 mg eletriptan is much larger than the benefit of increasing the sumatriptan dose from 50 mg to 100 mg. The absolute therapeutic gain for pain relief of eletriptan 40 mg over eletriptan 20 mg at 2 h after dosing is 8.7% (90% probability interval: 7.1–10.2%). With a total response rate of 58% of patients achieving pain relief after eletriptan 20 mg at 2 h, this therapeutic benefit implies that 21% of the patients that do not respond to eletriptan 20 mg are expected to respond to eletriptan 40 mg.
In conclusion, the meta-analysis confirms the superior efficacy for pain relief and pain free of eletriptan 40 mg vs. sumatriptan 50 mg and 100 mg up to 4 h after treatment. Eletriptan 20 mg was found to be superior to sumatriptan 50 mg for pain relief and equivalent to sumatriptan 50 mg for pain free. Encapsulation did not impact the efficacy of sumatriptan during this time frame.
Footnotes
Acknowledgements
This study was supported by a grant from Pfizer Inc.
