Abstract
We examined pericranial muscle tenderness and abnormalities in the second exteroceptive suppression period (ES2) of the temporalis muscle in chronic tension-type headache (CTTH; n = 245) utilizing a blind design and methods to standardize the elicitation and scoring of these variables. No ES2 variable differed significantly between CTTH sufferers and controls (all tests, P > 0.05). We found no evidence that CTTH sufferers with daily or near daily headaches, a mood or an anxiety disorder, or high levels of disability exhibit abnormal ES2 responses (all tests, P > 0.05). CTTH sufferers were significantly more likely than controls to exhibit pervasive tenderness in pericranial muscles examined with standardized (500 g force) manual palpation (P < 0.005). Female CTTH sufferers exhibited higher levels of pericranial muscle tenderness than male CTTH sufferers at the same level of headache activity (P < 0.0001). Elevated pericranial muscle tenderness was associated with a comorbid anxiety disorder. These findings provide further evidence of pericranial hyperalgesia in CTTH and suggest this phenomenon deserves further study. Basic research that better elucidates the biological significance of the ES2 response and the factors that influence ES2 assessments appears necessary before this measure can be of use in clinical research.
Keywords
Introduction
The pathogenesis of chronic tension-type headache (CTTH) is largely unknown. Nociception from pericranial muscles, sensitization of pain transmission circuits at the trigeminal nucleus/dorsal horn, and dysregulation of central pain modulation have each been postulated to play a significant role in the pathophysiology of chronic tension-type headache (e.g. (1–5)).
Elevated pericranial muscle tenderness in CTTH probably reflects a sensitization of pain transmission circuits at the trigeminal nucleus and dorsal horn (6), though sensitization of peripheral nociceptors in pericranial muscles has not been ruled out. Elevations in pericranial muscle tenderness in CTTH have been observed in a number of studies (6–13), although stringent methodological precautions, such as electronic monitoring of palpation pressure by blind assessors, has been rare (6, 7)
The finding of a shortened or abolished second exteroceptive suppression period (ES2), or silent period, in CTTH argues for a dysfunction in supraspinal pain modulation systems in CTTH (14). Subsequent to the original report, abnormal ES2 responses have been reported in CTTH in some studies (15–17), but not in others (11, 12, 18, 19), leaving the status of this important observation unclear. Differences in subject samples or in assessment procedures across studies may have produced these discrepant ES2 findings. Two of the four studies reporting negative findings examined young adults (18–20 years old) (11, 12); in contrast, studies reporting positive findings appear to have assessed older adults (mean age 35–45 years) (14–17). Age alone cannot account for the differences in results, because two other studies of older adults (mean age 35–45 years) also reported negative results (18, 19). However, it has been argued that a dysfunction in supraspinal pain modulation systems (and thus abnormal ES2 responses) is most likely to be present when CTTHs are continuous, nearly continuous or severe (4), or when symptoms of depression are present (20). Differences in the composition of patient samples with regard to headache frequency or severity, or the presence of comorbid affective symptoms thus may have contributed to discrepant findings across studies. The small samples in previous studies increase the likelihood of such sampling variability. To date no study has included a sufficient number of subjects to address this issue by comparing ES responses in subsamples of patients who differ in these characteristics.
Methodological differences in the elicitation of ES2 also may have contributed to variability in results across studies, because no two studies have utilized the same procedure (stimulus intensity, stimulus duration, and the number of recordings) for obtaining an ES2 response. Comparable results might not be expected unless all recording parameters are consistent. Consequently, a study group of the European Headache Federation (21) proposed a standard methodology for assessing ES2 responses that recommended averaging 10 rectified electromyographic (EMG) recordings at 20 mA intensity of 0.2 ms duration. Only one study conducted since the publication of these recommendations has adopted this methodology, and this study (12) yielded negative findings.
The scoring of ES2 responses also is problematic because ES2 does not begin and end abruptly. Thus, differences in scoring procedures are probably also a source of variability in results across studies. The manual scoring of ES2 periods used in all of the studies reporting positive findings to date may yield spurious findings, particularly when not conducted blindly, because judging the onset and offset of ES2 responses can be difficult and unreliable across scorers (22, 23). It is thus noteworthy that the three studies in which assessments were conducted blindly (12, 18, 19), one of which employed completely automated scoring with a computer program (12), all reported negative results.
The purpose of the present study was two-fold. First, we investigated the presence of pericranial muscle tenderness and attenuated ES2 responses in a large (n = 245) carefully diagnosed adult patient sample of CTTH sufferers and in matched controls utilizing a blind design as well as methods for standardizing both the elicitation and scoring of these psychophysiological variables. Second, we determined whether ES2 or pericranial muscle tenderness (PMT) abnormalities were more likely to occur in the following subgroups of CTTH sufferers: (i) individuals with continuous or nearly continuous headaches, (ii) individuals with comorbid mood or anxiety disorders, or (iii) individuals who reported significant disability associated with their headaches.
Subjects and methods
Subjects
Individuals with frequent tension-type headaches were recruited from the general population in both urban (Columbus, Ohio and surrounding suburban areas) and rural (southern Ohio and western West Virginia) areas to participate in a clinical trial comparing drug and non-drug treatments for CTTH (24). Headache sufferers (n = 245) were required to meet International Headache Society (IHS) (25) diagnostic criteria for CTTH (IHS 2.2), and could not meet diagnostic criteria for headache induced by chronic substance use or exposure (IHS 8.2) at each of two assessments: a neurological evaluation by a project neurologist and an independent structured diagnostic interview conducted by a second staff member. In order to obtain an assessment of psychophysiological variables in the absence of prophylactic or psychotropic therapies, only participants who were not taking prophylactic headache medications, antidepressants, mood stabilizing medications, or regularly using anxiolytics were evaluated. Finally, participants were required to record at least 15 headache days during 1 month of daily headache recordings prior to the psychophysiological evaluation to confirm the diagnosis of CTTH.
The CTTH sample was predominately female (78%) and Caucasian (95%; 3% African-American, 2% other ethnic background), with a mean age of 37 years (range 17–65 years). Males were slightly older (mean 41 years) than females (mean 36 years), t (243) = 2.62, P = 0.009. Although all patients were required to identify tension-type headaches as their primary headache problem, 28% of patients also received a concurrent migraine diagnosis. However, we limited inclusion of subjects with concurrent migraine to well-controlled migraine occurring no more than 1 day per month. CTTH sufferers reported an average of a 12-year (mean 148 months) history of problem tension-type headaches with headaches occurring at the current frequency for more than 7 years (mean 86 months). Approximately two-thirds (67%) of patients reported a family history of problem headaches. Most patients (79%) reported having consulted a physician for their headache problems.
Healthy controls (n = 47) were identified by CTTH sufferers from among their peers or responded to advertisements for persons who did not experience problem headaches. Controls were paid $30 for completing a structured diagnostic interview, medical history, psychophysiological assessment, and psychosocial evaluation. Controls were required to report fewer than 10 headaches per year, could not receive a diagnosis of migraine or suffer from a chronic pain disorder. Controls did not differ from CTTH sufferers in gender (74.5% female), age (mean 37.3 years), or ethnic background (87.2% Caucasian; 6.4% African American; 6.3% other ethnic background).
All participants gave written informed consent to participate in the study, which was approved by the Ohio University Institutional Review Board.
Psychophysiological evaluation
Pericranial muscle tenderness
Prior to beginning the study, the examination techniques of the two neurologists who were to evaluate PMT were standardized in training sessions. Healthy controls were randomly scheduled among headache sufferers and no information about a participant's headache or psychosocial history was available to the examiners of PMT. Participants were instructed to refrain from discussing their headaches (or lack thereof) with the examiner until the completion of the assessment. During the examination, the neurologist palpated 10 pericranial muscle sites with the tip of the right index finger with the subject in a seated position. Finger pressure was standardized (500 g force) using a specially adapted palpometer from Dolorimeter Systems Inc. (Victoria, BC) that attaches to the index finger. The palpometer consists of a thin-film pressure transducer and connects to an electronic device that displays the pressure exerted on the transducer by the examiner's finger as each muscle is palpated. Detailed information on the palpometer has been published previously (26). Tenderness was recorded according to the Total Tenderness System (27), which has previously proved to be reliable (7). Five pairs of muscles (suboccipital, posterior cervical, middle trapezius, masseter, and temporalis) were each palpated bilaterally at predefined points. The assessor rated tenderness for each of the 10 palpation sites on a four-point scale (0–3) and the scores were summed from the 10 sites to a Total Tenderness Score (maximum possible score = 30).
Exteroceptive suppression periods
Exteroceptive suppression was recorded according to a previously described method (12, 28) in accordance with the standards recommended by the European Headache Federation (21). Briefly, temporalis EMG was recorded on the subject's right side and square wave electrical stimulation (0.2 ms duration, 20 mA intensity) was applied to the ipsilateral labial commissure during 2–3 s of maximal molar clenching. Prior to the assessment, electrical stimulation of 0.2 ms duration was administered at 0 mA and increased to 20 mA in increments of 2 mA to familiarize the subject with the sensation. Sensory and discomfort thresholds were assessed at this time. During the assessment 10 20-mA stimuli were hand triggered by the examiner at 10-s intervals during each ES2 assessment. EMG signals were bandpass filtered from 20 Hz to 500 Hz and sampled at a rate of 2 kHz for 75 ms prior to and 175 ms after the delivery of each stimulus.
Data reduction
Individual 250-ms sweeps of EMG activity associated with each of the 10 stimulation trials were rectified and averaged online. ES suppression periods were automatically scored and ES parameters were calculated using a computer program (29). Briefly, the program first expressed the rectified and averaged signal as a percentage of the prestimulus baseline (defined as the mean level of EMG activity between −75.0 ms and −25.0 ms before electrical stimulation). The signal was then subjected to 10 point smoothing, which consists of a running average of 5 ms of data. Figure 1 provides a graphic display of smoothed EMG activity where ES responses can be seen. The computer software program then automatically identified ES1 and ES2 suppression periods from the smoothed EMG waveform using computer algorithms and generated scores for all major ES parameters. See French et al. (29) for details.
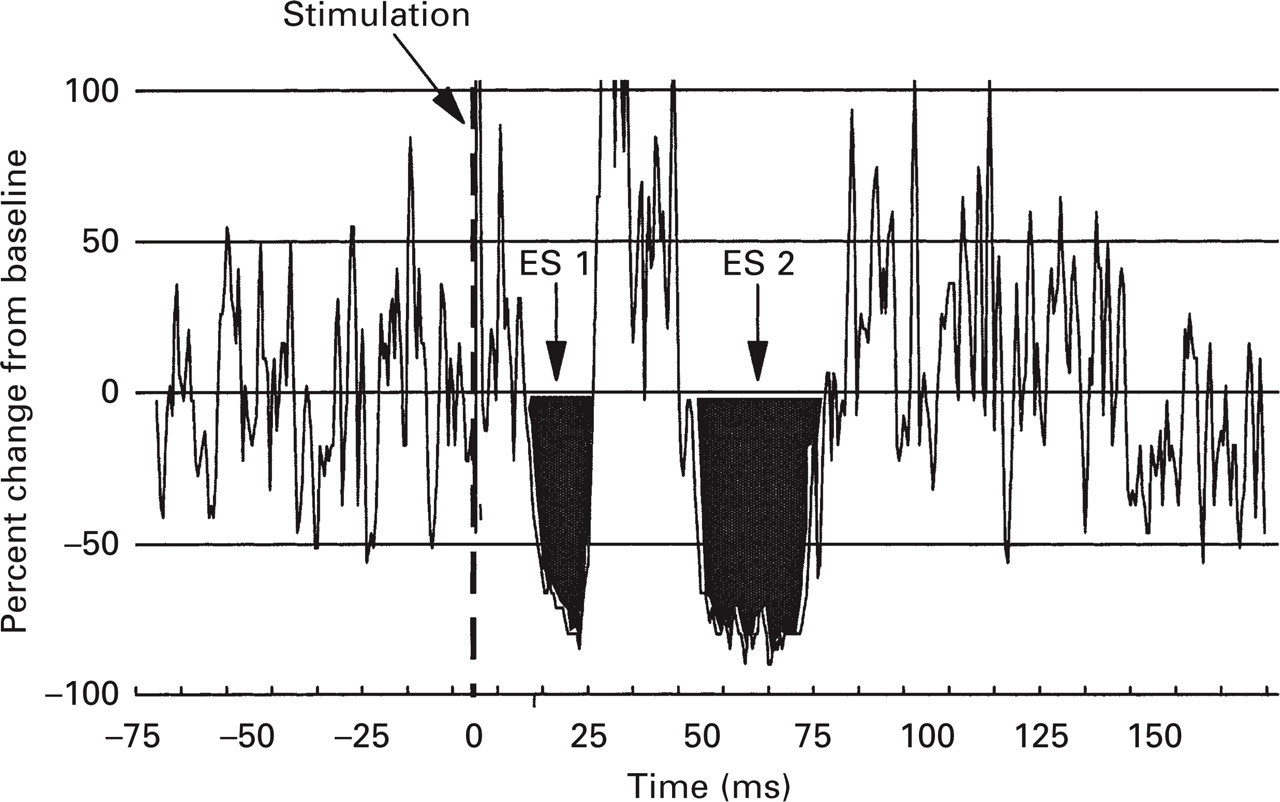
Unsmoothed waveform of 10 averaged and rectified electromyographic signals.
Headache recordings
CTTH sufferers recorded headache activity four times daily for 1 month following the neurological and psychophysiological evaluation using a 10-point rating scale with five anchors that ranged from ‘No pain’ to ‘Extremely painful—I can't do anything when I have a headache' (30). The Headache Index (average of 28 weekly ratings including 0 s), the number of headache days (pain rating > 1), and the number of days with a headache of at least moderate severity (pain rating > 5) provided measures of headache activity. Peak headache intensity ratings (the peak headache intensity rating for each week) were averaged to provide a measure of peak pain intensity.
Psychological evaluation
Psychiatric diagnosis
The Primary Care Evaluation of Mental Disorders (PRIME-MD) (31) is designed to facilitate the diagnosis of psychiatric disorders commonly seen in medical settings. It includes a patient-completed questionnaire of key symptoms and a clinician-administered structured interview (Clinician Evaluation Guide) that yields a subset of diagnoses included in the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) (32). Psychologists made diagnoses of the most commonly encountered mood (major depressive disorder, dysthymia and minor depressive disorder) and anxiety disorders (panic disorder, generalized anxiety disorder and anxiety disorder not otherwise specified) using the PRIME-MD Mood and Anxiety Modules.
The Headache Disability Inventory
The Headache Disability Inventory (HDI) (33) was designed to assess ‘the burden of chronic headaches’ using 25 items that inquire about the perceived impact of headaches on emotional functioning (e.g. ‘I feel desperate because of my headaches’) and daily activities (e.g. ‘Because of my headaches I am less likely to socialize’). Items were designed specifically to assess the concerns of individuals with recurrent headache disorders. The HDI appears to exhibit reasonable short-term (1 week, r = 0.93–0.95) and longer-term (2 month, r = 0.76–0.83) stability, and patient reports on the HDI appear to be reasonably congruent with spouse reports (33, 34). The HDI has the advantage of assessing both the affective and disability components of headache impact (35).
Results
Headache recordings
Daily diary data revealed that on average CTTH sufferers recorded 26 headache days per month. About two-thirds (65%) of patients recorded daily or near daily headaches (25 days or more per month) and only a small number (12%) of patients recorded fewer than 20 headache days per month. On average, pain levels were at least moderate (> 5 on a 10-point scale) on 14 days per month with an average peak intensity of 6.5. Females and males did not differ in number of headache days or number of days of at least moderate pain.
Exteroceptive suppression periods
Baseline temporalis muscle activity during clenching did not differ significantly
in CTTH (males' mean = 188.1 µV/s,
Electrophysiological data are presented in Table 1. None of the ES parameters differed in females and males (all P > 0.10), so males and females were combined for subsequent analyses. ES parameters calculated using a 50% suppression criterion are reported in Table 1. Conclusions were unchanged when 0% or 80% suppression criteria were used to calculate ES parameters.
Data from the electrophysiological assessment

Data from absent suppression periods excluded.
ES1 latencies, maximum suppression, duration, and area of suppression period did not differ significantly in CTTH sufferers and controls. Similar percentages of CTTH sufferers (15%) and controls (21%) had absent ES2 suppression periods. Regardless of whether subjects with absent suppression periods were included in the analysis, ES2 latencies, maximum suppression, duration and area of suppression periods did not differ significantly between CTTH sufferers and controls (all tests, P > 0.05).
Several subgroups of CTTH sufferers were hypothesized to be especially likely to exhibit abnormal ES2 responses. These included CTTH sufferers with daily or nearly daily (25 or greater headache days per month; n = 159) headaches, CTTH sufferers with a PRIME-MD mood disorder (n = 70) or anxiety disorder (n = 128) diagnosis, CTTH sufferers with a long history of CTTHs, defined as chronicity above the median (median = 53 months), and CTTH sufferers with high levels of disability, defined by scores above the median (median = 36) on the HDI. In no case did the ES2 parameters for the designated subgroup of patients differ significantly from the ES2 parameters for the remaining CTTH sufferers, or from the ES2 parameters of controls (all tests, P > 0.05). ES2 parameters (duration, area and maximum suppression) were uncorrelated with PMT total tenderness scores (r = 0.02 females, −0.37 males for ES2 duration).
ES2 parameters did show significant but small correlations with three of six headache measures: a comorbid migraine diagnosis (r = 0.16, P < 0.05) and a longer chronicity of CTTHs (r = 0.15, P < 0.05) were associated with slightly longer ES2 suppression periods, and higher peak pain intensities were associated with slightly shorter suppression periods (r = −0.16, P < 0.05). These latter findings are not significant when the α level is adjusted for the number of correlations conducted; in any event the observed correlations are too small to be of clinical significance.
Pericranial muscle tenderness
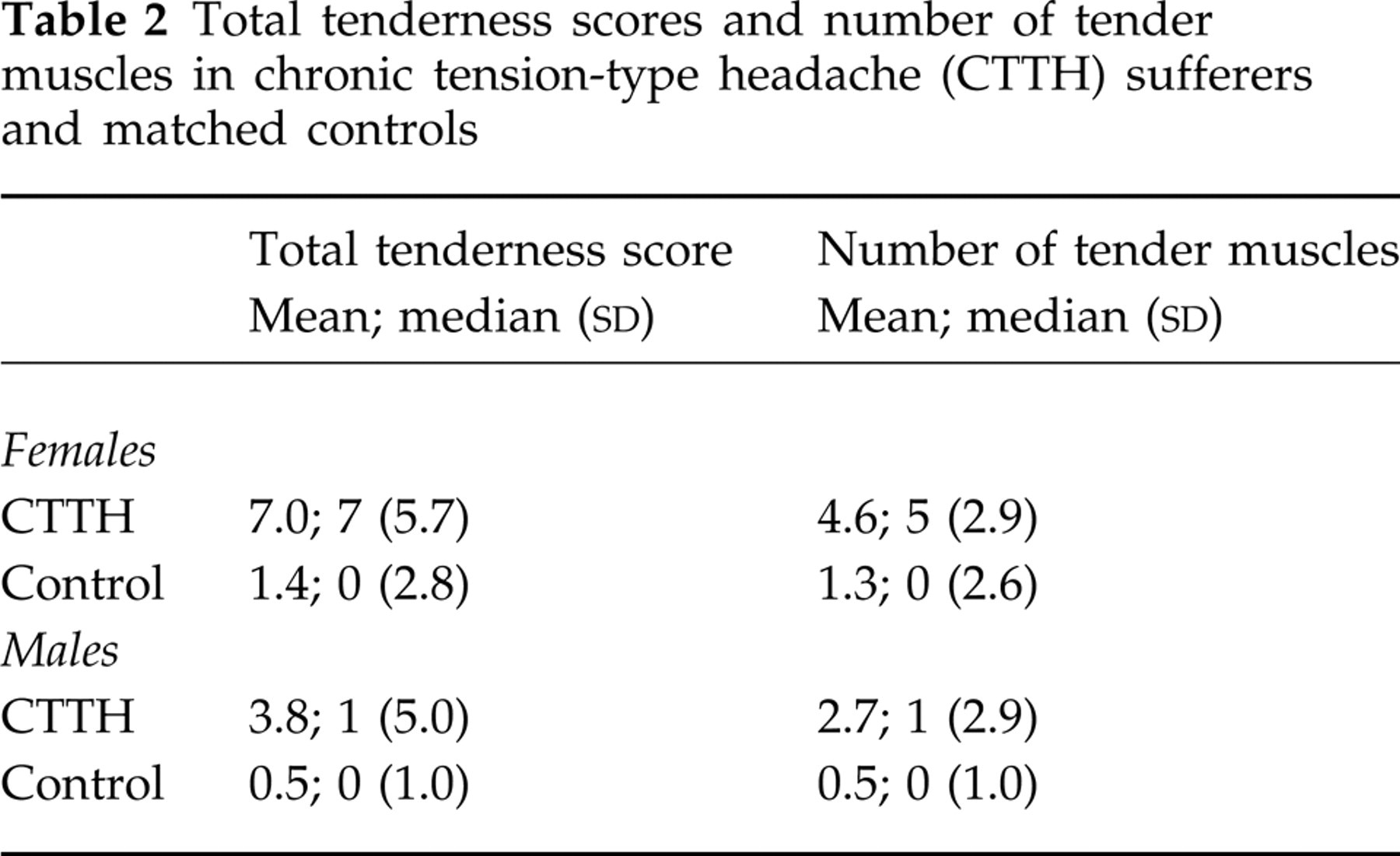
CTTH sufferers, especially females, exhibited pervasive PMT, while similar levels of tenderness were infrequently observed in healthy controls. In Fig. 2 it can be seen that female CTTH sufferers were two to seven times more likely than female controls to exhibit tenderness in each of the 10 muscles that were examined (all at least P < 0.02). Female CTTH sufferers thus obtained significantly higher total tenderness scores (Z = −6.5, P < 0.0001) and exhibited tenderness in a greater number of muscles (Z = −6.09, P < 0.0001) than did female controls (see Table 2). Tenderness associated with CTTH was also widespread, with female CTTH sufferers five times more likely than female controls to exhibit pervasive tenderness (11, 12) defined as tenderness in at least two of the five muscle groups examined (76% CTTH vs. 14% controls; P < 0.0001).

Percentage of female chronic tension-type headache (CTTH) patients (▪) and female controls (□) exhibiting tenderness to moderate pressure (500 g force) in each of 10 muscles. All differences between CTTH and controls were significant (P ≤ 0.02).
Total tenderness scores and number of tender muscles in chronic tension-type headache (CTTH) sufferers and matched controls
Among males, CTTH sufferers also obtained higher total tenderness scores (Z = −2.76, P = 0.0058) and exhibited a greater number of tender muscles than did controls (Z = −2.74, P = 0.006). Male CTTH sufferers also were more likely than male controls to exhibit pervasive tenderness (52% CTTH vs. 8% controls; P = 0.006).
Tenderness levels observed in female CTTH sufferers were significantly higher than tenderness levels observed in male CTTH sufferers (Table 2; P < 0.0001). Because levels of headache activity did not differ in males and females, this difference was not simply a result of higher levels of headache activity in females than males. Among healthy controls, males and females did not differ significantly in tenderness.
CTTH sufferers who had experienced a headache in the previous 24 h and CTTH sufferers who had been headache-free for at least 24 h did not differ significantly in total tenderness scores or number of tender muscles (medians = 5 and 4, respectively, for both groups). Thus, differences in tenderness levels between CTTH sufferers and healthy controls were not restricted to CTTH sufferers who had experienced a headache in the last 24 h. In fact, CTTH sufferers who had been headache-free during the 24 h prior to the assessment exhibited significantly higher total tenderness scores and a greater number of tender muscles than did healthy controls (both P < 0.0001).
Pericranial muscle tenderness (total tenderness scores) was examined in the same subgroups of CTTH sufferers examined in the analysis of ES2 data above. It can be seen in Table 3 that tenderness was significantly elevated in CTTH sufferers with an anxiety disorder diagnosis, although this difference only reached statistical significance in females, and in males who also reported high levels of disability on the HDI.
Pericranial muscle tenderness (PMT) total tenderness scores in subgroups of chronic tension-type headache (CTTH) sufferers

Discussion
Exteroceptive suppression
In our large sample of carefully diagnosed CTTH patients we did not observe the abnormalities in ES2 responses observed by some (14–16), but not by other investigators (11, 12, 18, 19). This is despite the fact that our methodology for the assessment of ES2 parameters closely followed the recommendations of a consensus panel of the European Headache Federation and utilized a computerized scoring system to eliminate the possibility of scoring bias (29).
It has been suggested that anxiety or depression might reduce ES2 duration (20, 36). No prior investigation has been large enough to test this hypothesis. In the present study, the ES2 parameters did not differ in CTTH patients diagnosed (by the PRIME-MD) with a mood or an anxiety disorder or in CTTH patients with no sign of psychiatric disorder. If a shortened ES2 response is associated with even subclinical levels of depression or anxiety as has been hypothesized (20), this abnormality should have been evident in subgroups of patients in our sample with diagnosed mood or anxiety disorders.
It also would be reasonable to hypothesize that ES abnormalities would most likely be observed when tension-type headaches are continuous or nearly continuous or severe (4). However, we found no evidence that ES2 responses differed in CTTH sufferers with near daily headaches and CTTH sufferers with less frequent headaches. Moreover, headache parameters showed no consistent relationship to ES2 parameters. Our findings provide no indication that CTTHs are associated with an abnormal ES2 response and suggest that additional basic research that better elucidates the biological significance of the ES2 response and the factors that might influence or confound ES2 assessments may be necessary before this measure can be used effectively in clinical research.
Other investigators have employed different, albeit equally indirect, methods of assessing the role of supraspinal pain modulation in musculoskeletal pain disorders (37–40). For example, Maixner and colleagues (39) found that the submaximal effort tourniquet procedure for invoking diffuse noxious inhibitory control systems (DNIC) fails to suppress chronic temporomandibular disorder (TMD) pain in the same manner as it suppresses acute pain in healthy controls (41). Similarly, Kosek & Hansson (38) found that the tourniquet procedure for inducing DNIC systems failed to suppress pain responses to pressure stimuli in patients with fibromyalgia in the same manner as it does in healthy controls. These findings led both research groups to conclude that the patients with the pain disorders they studied were less able to activate endogenous supraspinal pain modulation than were healthy controls. Examination of DNIC systems in CTTH sufferers and controls using similar methodologies may thus provide useful information. These and other (28) findings further suggest that the dynamic assessment of anti-nociceptive responses to challenge may yield different results than the static assessment procedures more typically employed.
Pericranial muscle tenderness
CTTH sufferers exhibited greater tenderness than controls in all 10 muscles examined when a digital palpometer was used to standardize applied pressures. Using the criterion of tenderness in at least two of the pericranial muscle groups evaluated (11, 12), 76% of female and 52% of male CTTH sufferers exhibited pervasive tenderness. Female CTTH sufferers were about five times as likely as female controls to exhibit pervasive muscle tenderness, and male CTTH sufferers were about seven times as likely as male controls to exhibit pervasive tenderness. Pericranial muscle tenderness was thus quite successful in distinguishing CTTH sufferers from controls. However, pericranial muscle tenderness is less effective in differentiating CTTH sufferers from migraine sufferers who also are likely to exhibit elevated levels of tenderness (e.g. (11, 12, 42)), possibly reflecting sensitization of pain transmission pathways in both CTTH and migraine.
Females exhibited significantly higher levels of tenderness than males, despite the fact that the level of headache activity recorded by males and females did not differ. Tenderness might thus reflect important gender differences in pain mechanisms, or merely the greater readiness of females to display pain behaviours (43). Signal detection procedures (44) might provide one method for evaluating these two explanations. If sex differences in tenderness reflect sex differences in pain mechanisms, an understanding of the physiological mechanisms underlying these sex differences may shed light on pain mechanisms in a number of disorders where chronic muscular pain occurs more frequently in women than men.
An anxiety disorder diagnosis was associated with elevations in pericranial muscle tenderness. Anxiety disorders may be associated with high levels of muscle tension, or with increased awareness of and increased sensitivity to bodily sensations (e.g. (45, 46)), either of which might elevate PMT levels. In addition, an anxiety disorder might aggravate the sensitization of trigiminal nucleus pain transmission circuits, or dysfunction in supraspinal pain modulation systems that have been hypothesized to underlie muscle tenderness. However, the association between anxiety disorders and PMT levels in CTTH requires replication before underlying mechanisms are explored.
The pathophysiology of pericranial muscle tenderness is poorly understood, but in CTTH may result from sensitization of second-order neurones and plasticity in trigeminal nucleus caudalis pain transmission circuits (2). Additional research is necessary to further our understanding of the cellular and molecular basis of this sensitization of pain pathways (47). This would not only improve our understanding of CTTH, but would probably lead to the development of improved strategies for the management of CTTH and other chronic myofascial pain disorders.
Footnotes
Acknowledgements
Support for this research was provided in part by a grant from The National Institutes of Health (NINDS no. NS32374). Thanks to Lars Bendtsen (University of Copenhagen) for informing us about the availability of a device for standardizing finger pressure during palpation. Appreciation is also expressed to Kimberly Hill, Peter Malinoski, Adriana Meade, Angela Nicolosi, Carol Nogrady, Cornelia Pinnell, France Talbot and Michael Stensland. G.L.L. is now at St. Vincent Health Center, Erie, Pennsylvania and D.J.F. is now at the University of Moncton, Moncton, NB, Canada.
