Abstract
Increased pain sensitivity in the central nervous system may play an important role in the pathophysiology of chronic tension-type headache (CTTH). Previous studies using pain thresholds as a measure of central pain sensitivity have yielded inconsistent results and only a few studies have examined perception of muscle pain without involvement of adjacent tissues. It has been suggested that suprathreshold testing might be more sensitive than threshold measurements in evaluation of central hyperexcitability in CTTH. The aim of the study was to compare pain ratings to suprathreshold single and repetitive (2 Hz) electrical stimulation of muscle and skin in cephalic (temporal and trapezius) and extracephalic (anterior tibial) regions between patients with CTTH and healthy subjects. In addition, we aimed to examine gender differences in pain ratings to suprathreshold stimulation and degree of temporal summation of pain between patients and controls. Pain ratings to both single and repetitive suprathreshold stimulation were higher in patients than in controls in both skin and muscle in all examined cephalic and extracephalic regions (P < 0.04). Pain ratings to both single and repetitive suprathreshold electrical stimulation were significantly higher in female compared with male patients (P < 0.001) and in female compared with male controls (P ≤ 0.001). The degree of temporal summation of muscular and cutaneous pain tended to be higher in patients than in controls but the differences were not statistically different. This study provides evidence for generalized increased pain sensitivity in CTTH and strongly suggests that pain processing in the central nervous system is abnormal in this disorder. Furthermore, it indicates that suprathreshold stimulation is more sensitive than recording of pain thresholds for evaluation of generalized pain perception.
Keywords
Introduction
Sensitization of the central nervous system (CNS) may play an important part in the pathophysiology of chronic tension-type headache (CTTH) and other myofascial pain disorders (1, 2). Pain thresholds to mechanical, heat and electrical stimulation have been extensively studied in CTTH (3–11). Overall these studies have revealed lower pain thresholds in CTTH than in healthy controls, but the differences have been subtle and studies examining only small numbers of subjects have not detected any difference (5, 6). Stimulus–response functions of pressure pain in patients with CTTH and fibromyalgia exhibited a marked upward shift in rated pain intensity at suprathreshold stimulus intensities, while the pain threshold was shifted only marginally (12, 13). Thus, suprathreshold testing might be more sensitive than threshold measurements in evaluation of central hyperexcitability in CTTH (2, 14). Intramuscular suprathreshold pain perception has never been studied in patients with CTTH.
The experimental models used to investigate central hyperexcitability of nociceptive pathways include the study of wind-up in animals (15) and temporal summation of pain in humans (16). In humans, temporal summation has been described as a gradual increase in pain sensitivity to repetitive stimuli (16). These studies have suggested that temporal summation of pain might be a perceptual correlate of wind-up in humans and one of several initiators of central sensitization (17). Temporal summation has been studied in patients with fibromyalgia and found to be more pronounced than in controls (18).
The aim of the present study was to compare pain ratings to suprathreshold single and repetitive electrical stimulation of muscle and skin in cephalic and extracephalic regions between patients with CTTH and healthy subjects. The second aim was to examine gender differences in pain ratings to suprathreshold single and repetitive electrical stimulation of muscle and skin in cephalic and extracephalic regions. Moreover, we aimed to examine whether temporal summation of muscular and cutaneous pain differed between patients and healthy subjects.
Methods
Subjects
We recruited 21 patients with a diagnosis of CTTH according to the criteria of the first edition of International Headache Classification of the International Headache Society (IHCD-1) (19), from advertisements in local newspapers and from the out-patient headache clinic at the Danish Headache Centre at Glostrup University Hospital. All patients completed a diagnostic headache diary (20) during a 4-week run-in period to confirm the diagnosis. At screening, we undertook a full physical and neurological examination. The inclusion criteria were a diagnosis of CTTH and age between 18 and 65 years. Exclusion criteria were: a history of more than 1 day with migraine per month; use of any kind of daily medication including prophylactic headache therapy but not oral contraceptives; excessive alcohol use; and serious somatic or psychiatric disorders including depression (Hamilton Depression Score ≥17) (21). Twenty-one healthy volunteers served as controls (Table 1). They had never had migraine and had less than 12 days per year with tension-type headache. The study was approved by the local ethics committee and was undertaken in accordance with the Helsinki Declaration of 1975, as revised in 1983. All subjects gave informed consent to participate.
Clinical data on patients with chronic tension-type headache and healthy controls

Values are means (ranges).
Experimental setting and test paradigms
All subjects were examined for pain sensitivity at the temporal, trapezius and anterior tibial regions sitting in a dental chair with headrest. Pericranial tenderness was recorded according to the Total Tenderness Score [TTS; total score of eight pairs of pericranial muscle and tendon insertion points, each scored on a 4-point scale (0–3) at each location with a maximum possible score = 48] (22). Quantitative sensory testing was performed to measure local tenderness, electrical pain thresholds and pain ratings to suprathreshold single and repetitive electrical stimulation at standard anatomical points in the temporal, trapezius and anterior tibial regions, at the non-dominant side. In the temporal region, the standard point was located 3 cm rostral to the centre of the midway between the lateral orbital edge and the anterior part of the external auditory meatus. In the trapezius region, the standard point was located in the centre of the descending part of the trapezius muscle midway between the processus spinosus of the 7th cervical vertebra and the acromion. In the anterior tibial region, the standard point was located between 1/3 and 2/3 of the distance between the lower tip of the kneecap (apex patellae) and the lateral malleolus.
Local tenderness scores
The local tenderness score (LTS) was assessed by pressure-controlled palpation. A palpometer (23) was used to control palpation pressure. Pressure intensity used in the study was 160 arbitrary units. Palpation was performed with small rotating movements of the observer's second and third fingers. Pressure was sustained for 4–5 s. Tenderness was scored on a 0–100-mm visual analogue scale (VAS) blinded for the observer (Hanne Andresen (H.A.)). The method has previously been proven to be reliable (22). Measurement of total and local tenderness was performed by the same observer (H.A.).
Electrical stimulation
Intramuscular electrical stimulation was induced by two needle electrodes with uninsulated tips (3 mm) (13R24; Dantec Measurement Technology A/S, Copenhagen, Denmark). Cutaneous electrical stimulation was induced by a bipolar saline-soaked surface stimulation electrode, which was mounted to the skin with tape (Dantec Measurement Technology A/S). Electrical stimuli of 1 ms stimulus duration were generated by a constant current stimulator (Counterpoint; Dantec Measurement Technology A/S). Thus, a single stimulus lasted 1 ms. Repetitive stimuli consisted of single stimuli delivered at 2 Hz for 2.5 s (train of five stimuli) (16). The needle electrodes were inserted 15 mm into the muscle with the cathode 5 mm proximal and the anode 5 mm distal to the standard points in the trapezius and the anterior tibial muscles. In the temporal muscle, the needle electrodes were inserted slantingly 10 mm into the muscle with the cathode 5 mm rostral and the anode 5 mm caudal to the standard points. The correct intramuscular position of electrodes was verified by demonstration of electromyographic (EMG) activity in response to voluntary muscle contraction. The EMG signals from the trapezius and anterior tibial muscles were recorded by an electromyograph (Counterpoint; Dantec Measurement Technology A/S). Assignment to measurement of electrical pain thresholds and pain ratings to suprathreshold stimulation (site of stimulation, muscle vs. skin) was done in a randomized and balanced design. A flow chart of electrical stimulation paradigm is shown in Fig. 1. All measurements in electrical stimulation paradigm were performed by the same observer (S.A.).

Flow chart of intramuscular and cutaneous electrical stimulation.
Intramuscular and cutaneous electrical pain thresholds
The electrical pain threshold was defined as the weakest stimulus perceived as painful by the subject after single electrical stimulation. Pain threshold to single electrical stimulation was assessed by the method of limits (3, 24). The stimulus strength was gradually increased up to the first report of painful sensation with notation of value in mA. The procedure was performed three times with a minimum interval of 30 s between successive determinations and the threshold was calculated as the mean of the three values.
Suprathreshold electrical stimulation
Subjective pain ratings (VAS) to both single and repetitive electrical stimulation with intensity of 1.5 times the individual pain threshold were assessed (25). The subjects rated the fifth stimulus in the train of repetitive stimuli. The procedure was performed three times with a minimum interval of 30 s between successive determinations and the pain rating was calculated as the mean of the three values.
Temporal summation
Temporal summation of muscular and cutaneous pain was induced by delivering a train of five 1-ms pulses repeated at 2 Hz with an intensity of 1.5 times the individual pain threshold (suprathreshold repetitive stimulation as described above) (16, 26).
Data analysis
The primary endpoint was the difference in subjective pain ratings to suprathreshold single and repetitive stimulation in both muscle and skin in temporal, trapezius and anterior tibial regions between patients and controls. The quantitative measure of temporal summation, the degree of temporal summation, was the difference between pain rating to fifth stimulus in the train of electrical stimulation and pain rating to a single stimulus at intensity of 1.5 times the individual pain threshold (26). The Shapiro–Wilk test for normal distribution was used to analyse distribution of the data. Results that were not normally distributed were presented as median with quartiles unless marked otherwise. The Mann–Whitney U-test was used to test recording for unpaired observations. The Wilcoxon signed ranks test was used to test recordings for paired observations (gender differences). Statistical analyses were done using SPSS®, version 12.0 software (SPSS Inc., Chicago, IL, USA). Five percent was accepted as level of significance.
Results
All patients and healthy subjects completed the study and their clinical characteristics are presented in Table 1. In patients with tension-type headache the TTS, 15 (9–25), was significantly higher than the TTS in control subjects, 1 (0–5) (P < 0.0001).
Local tenderness score by manual palpation
The LTS was significantly higher in patients than in control subjects in the temporal muscle [15.0 ± 4.0 mm (mean ± SEM) vs. 0.4 ± 0.3 mm of VAS (P = 0.001)], in the trapezius muscle [12.5 ± 4.1 mm vs. 0.6 ± 0.4 mm of VAS (P = 0.001)] and in the anterior tibial muscle [3.2 ± 2.0 mm vs. 0.0 ± 0.0 mm of VAS (P = 0.004)].
Intramuscular and cutaneous electrical pain thresholds
Pain thresholds to single electrical stimuli measured in the temporal, trapezius and anterior tibial muscles and in overlying skin were not different between patients and controls (Table 2).
Pain thresholds (mA) to single electrical stimulation
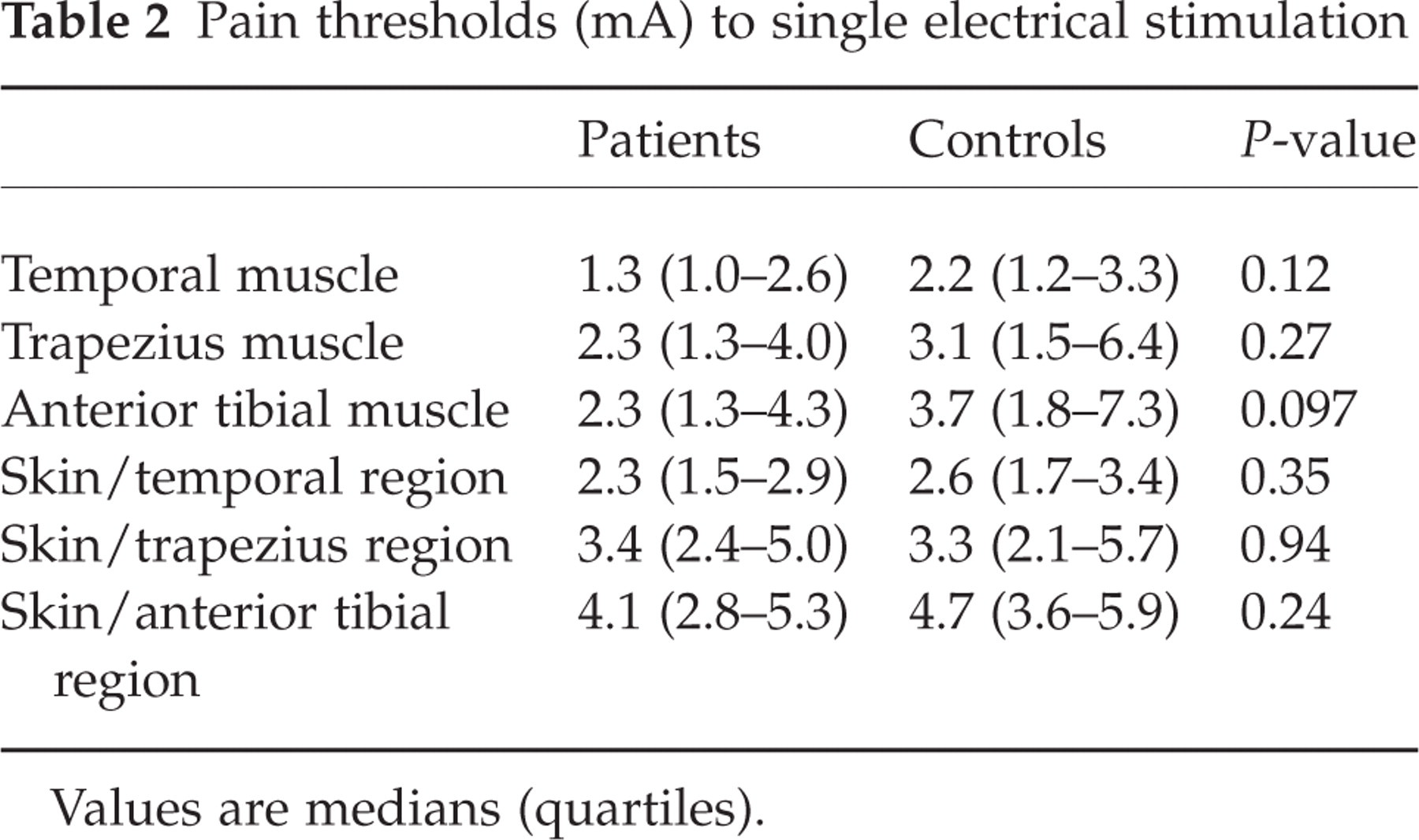
Values are medians (quartiles).
Suprathreshold pain ratings
Pain ratings to both single and repetitive suprathreshold electrical stimulation were significantly higher in patients than in controls in the temporal, trapezius and anterior tibial muscles (Table 3). In skin, there were also significantly higher pain ratings to both single and repetitive suprathreshold electrical stimulation in patients than in controls in the temporal, trapezius and anterior tibial regions (Table 3).
Pain ratings (visual analogue scale) to single and repetitive (2-Hz) suprathreshold electrical stimulation

Values are medians (quartiles).
Pain ratings to both single and repetitive suprathreshold electrical stimulation were significantly higher in female compared with male patients (Table 4a) and in female compared with male controls (Table 4b), except for pain ratings to repetitive stimulation in the anterior tibial muscle.
Pain ratings (visual analogue scale) to single and repetitive (2-Hz) suprathreshold electrical stimulation in male and female healthy controls

Values are medians (quartiles).
Pain ratings (visual analogue scale) to single and repetitive (2-Hz) suprathreshold electrical stimulation in female and male patients with chronic tension-type headache

Values are medians (quartiles).
Temporal summation
The degree of temporal summation of muscular and cutaneous pain was higher in patients than in controls at all examined locations, but the differences did not reach statistical significance (Table 5).
Degree of temporal summation of muscular and cutaneous pain in temporal, trapezius and anterior tibial regions

Values are medians (quartiles).
Discussion
Present findings and pathophysiological considerations
The major outcome of the present study was the demonstration of higher pain ratings to all tested suprathreshold stimuli in patients with CTTH than in healthy controls. This was true for both single and repetitive suprathreshold electrical stimulation at all the examined locations, i.e. temporal, trapezius and anterior tibial regions and for both muscle and skin. Another important finding was increased local tenderness for manual palpation with standardized suprathreshold pressure in the temporal, trapezius and anterior tibial regions in patients with CTTH. Thus, our data clearly demonstrate generalized increased pain sensitivity in patients with CTTH. It could be argued that generalized hyperalgesia in our patients may have been due to a greater pain-specific hypervigilance at the cortical level. However, this is less probable, because pain thresholds did not differ between patients and controls. Janke et al. (27) demonstrated that depression increased vulnerability to TTH in patients with frequent headaches during and following the laboratory stress test and was associated with elevated pericranial muscle tenderness. It has been suggested that anxiety and depression may contribute to increased excitability in central nociceptive pathways in patients with CTTH (27, 28). Both patients and control subjects in our study had a Hamilton Depression Score <17, which rules out influence of depression on our results.
The degree of temporal summation of muscular and cutaneous pain tended to be higher in patients than in controls but the differences were not statistically different. The optimal stimulus paradigm for induction of temporal summation in patients with chronic pain is unknown. It must be kept in mind that temporal summation reflects only part of the process of central sensitization (15, 29). In addition, it has been demonstrated that wind-up in humans is not facilitated by central sensitization induced by injection of capsaicin in skin, suggesting that central sensitization and wind-up are independent phenomena (30).
The hyperalgesia in CTTH demonstrated in the present study could be due to disturbance of the nociceptive system with peripheral or central sensitization, or both (31, 32). The fact that the hyperalgesia is generalized, i.e. found in all the examined tissues, and of comparable degree in all examined locations, together with the fact that the locus of pain in patients with CTTH is in the cephalic region and not in the extremities, makes it highly unlikely that the hyperalgesia is caused by peripheral sensitization of nociceptors in muscle and skin. The hyperalgesia is best explained by abnormal pain modulation in the CNS. This could be caused by prolonged nociceptive input from tender pericranial myofascial tissues resulting in segmental central sensitization at the level of the upper cervical spinal dorsal horn/trigeminal nucleus with secondary sensitization of supraspinal neurons, e.g. in the thalamus or somatosensory cortex (2). This hypothesis is further supported by a recent study demonstrating a decrease in grey matter brain structures involved in pain processing in patients with CTTH (33). This decrease was positively correlated with duration of headache and the authors interpreted the data as being the consequence of central sensitization generated by prolonged input from pericranial myofascial structures. Decreased antinociceptive activity from supraspinal structures, i.e. deficient descending inhibition, may also contribute to the increased pain sensitivity in CTTH. The nociceptive flexion reflex threshold has been found to be decreased in patients with CTTH compared with controls, indicating dysfunction of antinociceptive systems in CTTH (9, 34).
Gender differences
We found that females in both the patient and control groups had a higher pain rating than males to single and repetitive suprathreshold stimulation in 23 of 24 comparisons, the only exception being pain rating to repetitive in anterior tibial muscle in patients. The latter finding may be due to a type 1 error. Our results are in agreement with previous findings of increased pain sensitivity in female subjects to experimental painful stimuli over males (35, 36). The increased pain sensitivity in females may be due to gender differences in various psychological and physiological factors, internal hormonal factors, nociceptive input integration in the CNS and modulation of the afferent input by antinociceptive activity from supraspinal structures. Interestingly, the diffuse noxious inhibitory control (DNIC), which is part of a central pain modulatory system that relies on spinal and supraspinal mechanisms, has been shown to be unable to attenuate temporal summation of second pain in female patients with fibromyalgia and female controls compared with male controls (37). This may also be the case for female patients with CTTH and needs to be addressed.
Mechanisms of generalized hyperalgesia in chronic headache
Both animal and human studies emphasize the role of the convergence mechanism in the neuroplastic changes in nociceptive neurons in pathological pain conditions, including headaches (38, 39). The convergent afferent input to spinal cord wide-dynamic-range (WDR) neurons from skin, viscera and muscles has been implicated in referred pain patterns in superficial and deep pain localization (40, 41). Sessle et al. (39) have demonstrated convergence of cutaneous, tooth pulp, visceral, neck and muscle afferents onto nociceptive and non-nociceptive neurons in trigeminal subnucleus caudalis (medullary dorsal horn) and suggested a role for these neurons in mediating deep pain and in spread and referral of pain in the craniofacial region. In addition, it has been shown that trigeminal activation leads to a sensitization of second-order neurons receiving cervical input (42). It has been suggested that this mechanism might be involved in the referral of pain from trigeminal to cervical structures (42). Convergence and interaction between cutaneous and deep afferent inputs also seems to be a characteristic of somatosensory neurons in the thalamus and somatosensory cerebral cortex (43–45). The pathophysiological basis for the increased susceptibility to central sensitization is unknown. Possible mechanisms include an impaired supraspinal inhibition of nociceptive transmission in the spinal dorsal horn due to a serotonergic dysfunction. In the sensitized state, the afferent Aβ-fibres that normally inhibit Aδ- and C-fibres by presynaptic mechanisms in the dorsal horn will, in contrast, stimulate the nociceptive second- and third-order neurons. In addition, the effect of Aδ- and C-fibre stimulation of the nociceptive dorsal horn neurons will be potentiated and the receptive fields of the dorsal horn neurons will be expanded (46). The nociceptive input to supraspinal structures will therefore be considerably increased, which may result in increased excitability of supraspinal neurons (47) as well as decreased inhibition or increased facilitation of nociceptive transmission in the spinal dorsal horn (48), i.e. in generalized pain hypersensitivity. Thus, we hypothesize that frequent nociceptive input from muscles in the cephalic region in patients with infrequent episodic headache induces sensitization of second-order neurons in the trigeminal nucleus caudalis and dorsal horn of the cervical spinal cord. Subsequently, patients develop frequent episodic tension-type headache. Continuous nociceptive input in these patients sensitizes third-order neurons in the thalamus and neurons in the somatosensory cortex. Thus, patients develop CTTH and present with generalized hyperalgesia as demonstrated in our study.
CTTH and fibromyalgia
Central sensitization of the nociceptive pathway is also believed to play a major role in the pathophysiology of fibromyalgia. Generalized hypersensitivity to various nociceptive stimuli has also been demonstrated in fibromyalgia (18, 49, 50). As in CTTH, the stimulus–response function for manual pressure over a tender muscle in fibromyalgia is linear and thus qualitatively different from that of normal muscle (13). Despite the similarities in the pathophysiology, there are also differences between CTTH and fibromyalgia. Monopolar needle EMG recordings in the trapezius muscle comparing tender and non-tender points on palpation in fibromyalgia and CTTH demonstrated a higher ratio between tender point EMG and non-tender point EMG in patients with fibromyalgia than in patients with CTTH (51). In fibromyalgia, but not in CTTH, sustained muscle activation might thus represent a peripheral trigger.
Methodological considerations
Previous pain perception studies have indicated that patients with CTTH are hypersensitive to stimuli applied in both the cephalic and extracephalic regions. The pressure, thermal and electrical pain thresholds in the cephalic region have been reported to be lower in patients than in controls (4, 7, 8, 11, 52). Lower extracephalic pressure-pain thresholds were demonstrated in the Achilles tendon (11) and finger (4) in patients with CTTH. In addition, Langemark et al. (9) found that patients with CTTH scored higher on the VAS scale to electrical stimulation of the sura nerve. The above-mentioned studies that found a difference in pain thresholds between patients and healthy subjects all investigated a relatively large number of patients, i.e. from 32 to 50 patients (4, 8, 9), while studies with a small sample size, i.e. 12–14 patients (5, 6), detected no significant difference. In agreement with this, pain thresholds did not differ between patients and controls in the present study. It has been calculated that large groups are required for group comparisons of pressure-pain thresholds due to considerable interindividual variation in this parameter (53). Our results confirm the previous assumption that suprathreshold stimulation is more sensitive than pain threshold measurements in evaluation of central hyperexcitability in CTTH.
Conclusions
We have provided evidence for generalized increased pain sensitivity in patients with CTTH and our data strongly suggest that this is caused by abnormal pain modulation in the CNS. Both female patients and healthy controls were more sensitive to suprathreshold stimulation than male patients and healthy controls. Furthermore, our data indicate that suprathreshold stimulation is more sensitive than threshold measurement for assessment of increased pain sensitivity in myofascial pain disorders.
Acknowledgements
We thank our laboratory technician Mrs Hanne Andresen for skilful technical assistance. The study was supported by the Danish Headache Society, the Foundation of Ludwig and Sara Elsa's and the Danish National Agency for Enterprise and Housing.
