Abstract
Recent data from the Women's Health Initiative have highlighted many fundamental issues about the utility and safety of long-term estrogen use in women. Current hormone replacement therapy for postmenopausal women incorporates progestin with estrogen, but it is uncertain if combined therapy provides major cerebrovascular risks or benefits to these women. No experimental animal stroke studies have examined combined hormone administration. The authors tested the hypothesis that combined hormone treatment reduces ischemic injury in middle-aged female rat brain. Reproductively senescent female rats underwent 2-hour middle cerebral artery occlusion (MCAO) followed by 22 hours reperfusion. Estrogen implants were placed subcutaneously at least 7 days before MCAO, and progesterone intraperitoneal injections were given 30 minutes before MCAO, at initiation, and at 6 hours of reperfusion. Rats received no hormone, a 25-μg estrogen implant, a 25-μg estrogen implant plus 5 mg/kg intraperitoneal progesterone, or 5 mg/kg intraperitoneal progesterone. Cortical, caudoputamen, and total infarct volumes were assessed by 2,3,5-triphenyltetrazolium chloride staining and digital image analysis at 22 hours reperfusion. Cortical and total infarct volumes, except in the acute progesterone-treated group, were significantly attenuated in all estrogen-alone and combined hormone-treated groups. There were no significant differences in caudoputamen infarct volumes in all hormone-treated groups as compared with untreated rats. These data have potential clinical implications relative to stroke for postmenopausal women taking combined hormone replacement therapy.
Estrogen has been extensively studied as a neuroprotective agent in women, animals, and a variety of in vitro models of neural injury and degeneration. Most data suggest that estrogen can benefit the brain confronted with an ischemic challenge and reduce cell death (for review, see Dhandapani and Brann, 2002; Green and Simpkins, 2000; Hurn et al., 1998; Hurn and Macrae, 2000; Murphy et al., 2003, 2004; Roof and Hall, 2000; Stein, 2001; Wise and Dubal, 2000). However, the total evidence for the clinical benefit of hormone replacement therapy (HRT) as an ischemic neuroprotectant is limited (for review, see Nelson et al., 2002). For example, recent data from the Women's Health Initiative (WHI) has brought many fundamental issues to light about the utility and safety of chronic estrogen use in women.
The goal of the WHI was to identify major causes of death and disability in postmenopausal women through prevention/intervention protocols and risk factor identification. HRT is one intervention under examination in WHI clinical trials (Rossouw et al., 2002). The combined estrogen plus progestin HRT trial, which was to have continued until 2005, was terminated in July 2002 based on recommendations by the WHI Data and Safety Monitoring Board. The board found that overall risks from use of combined HRT outweighed the benefits. In addition to an increased risk of breast cancer, other adverse effects included an increased stroke risk, with 8 more strokes (41%) per year for every 10,000 women in the combined HRT group (Rossouw et al., 2002).
These recent results emphasize that there are unanticipated and paradoxical effects of estrogen as it is currently administered in women. In light of the WHI, estrogen's neuroprotective properties and potential benefit in CNS ischemic injury must be reassessed. Although combined estrogen and progestin compounds, such as conjugated equine estrogen and medroxyprogesterone acetate, are the most commonly prescribed hormone regimen in the United States, it is not known whether progestins interact with estrogen and alter its neuroprotective effects. Progesterone has clearly been beneficial in experimental traumatic brain injury models (Bramlett and Dietrich, 2001; Galani et al., 2001; Roof and Hall, 2000; Stein et al., 1999; Stein, 2001; Vink et al., 2001; Wright et al., 2001). However, its effects on experimental ischemic brain damage have been more variable depending on gender, age, dose, or administration protocol (for review, see Roof and Hall, 2000; Stein et al., 1999; Stein, 2001; Stein and Hoffman, 2003). No experimental studies have examined combined hormone administration in animal stroke models. This emphasis on individual female sex steroids in experimental studies makes it difficult to study the interactive roles of estrogen and progestins.
The purpose of the present study was to define the effects of estrogen and progesterone treatments on transient brain ischemia in reproductively senescent female (RSF) rats. We tested the hypothesis that combined hormone treatment reduces ischemic injury in middle-aged female rat brain.
MATERIALS AND METHODS
This study was conducted in accordance with the National Institutes of Health guidelines for the care and use of animals in research. The Johns Hopkins University Animal Care and Use Committee approved all animal protocols. All methods are as previously described (Alkayed et al., 1998; Toung et al., 1998). Briefly, age-matched (14 to 18 months) RSF Wistar rats (Harlan, Indianapolis, IN, U.S.A.; Hsd:WI, 385 to 791 g) were used. All hormone implants (Innovative Research of America, Sarasota, FL, U.S.A.) were placed subcutaneously at least 7 days before middle cerebral artery occlusion (MCAO) and all progesterone intraperitoneal injections were given 30 minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion. Progesterone stock solution contained 50 mg/mL progesterone (USP; Sigma, St. Louis, MO, U.S.A.) with 10% benzyl alcohol (ACS reagent; Sigma) as preservative in sesame oil (Sigma) and was sterile-filtered before use. We chose doses of estrogen or progesterone alone that have previously been shown to be neuroprotective in both ischemic ovariectomized (Murphy et al., 2002; Rusa et al., 1999) and RSF (Alkayed et al., 2000) rat brain. Rats were randomly assigned to one of four treatment groups (n = 10 per group). RSF rats received either no hormone (Control), a 25-μg estrogen implant (Estrogen), a 25-μg estrogen implant + 5 mg/kg intraperitoneal progesterone (Estrogen + Progesterone), or 5 mg/kg intraperitoneal progesterone (Progesterone).
For MCAO, rats were anesthetized with halothane (4% to 5% induction 1.25% to 1.5% maintenance delivered via mask in O2-enriched air) and a femoral artery catheter was placed for continuous monitoring of MABP and measurement of arterial blood gases and blood glucose levels. Rectal and temporalis muscle temperature were continuously monitored with a Mon-a-therm system (Mon-a-therm model 6510; Mallinckrodt Medical, Inc., St. Louis, MO, U.S.A.) and maintained by a heating lamp. The effectiveness of vascular occlusion was determined by sustained reduction of laser-Doppler flow (LDF) signal (model MBF3D; Moor Instruments Ltd., Oxford, U.K.) obtained by probe placement 6 mm lateral and 2 mm posterior to bregma (Alkayed et al., 1998; Toung et al., 1998).
Focal cerebral ischemia was accomplished using a modified intraluminal filament technique as previously described (Longa et al., 1989). Briefly, the right common carotid and external carotid arteries were exposed and ligated. The right occipital artery was cauterized, then the right pterygopalatine artery was ligated. After the baseline LDF was determined, a 3–0 nylon monofilament surgical suture with a heat-blunted tip was introduced via the right common and internal carotid arteries until the LDF signal displayed an abrupt and significant reduction, confirming ongoing ischemia. Then the suture was secured in place. MCAO was maintained for 2 hours. LDF was measured during ischemia over 15 minute intervals and for the first 15 minutes of reperfusion to confirm cortical reperfusion when the occluding filament was removed. Five minute recording averages were analyzed for each time point. To ensure relative uniformity of the ischemic insult, animals were excluded from the cohort if mean ischemic LDF was more than 50% of baseline LDF. Intra-ischemic blood gases and blood glucose levels were measured 30 to 45 minutes after initiation of MCAO.
The animal was killed via decapitation under halothane anesthesia (4% to 5%) 22 hours after the 2-hour MCAO period, and the brain removed. The brain was then sliced into 2-mm-thick coronal sections (seven slices total) that were subsequently placed into a 1% solution of TTC (2,3,5-triphenyltetrazolium chloride; Sigma) and incubated at 37°C for 30 minutes. Stained sections were fixed in 10% neutral buffered formalin. Both sides of each stained coronal slice were photographed via a digital camera, then evaluated by digital image analysis (SigmaScan Pro; Jandel Corporation, San Rafael, CA, U.S.A.). The infarcted area is integrated across sections, and infarction volumes in cerebral cortex, caudoputamen, and hemisphere are expressed as percentage of the contralateral structure (cortex, caudate-putamen, hemisphere).
All values are reported as mean ± SD unless otherwise indicated. All repeated-measures data, such as MABP and LDF, during MCAO and early reperfusion were analyzed by two-way analysis of variance with post hoc Newman-Keuls for multiple comparisons (Jandel SigmaStat 2.0; Jandel). Differences in overall rectal and temporalis muscle temperatures, mean ischemic LDF, mean ischemic MABP, ischemic blood gases, ischemic blood glucose levels, and infarction volumes were determined by one-way analysis of variance with post hoc Newman-Keuls for multiple comparisons (Jandel SigmaStat 2.0; Jandel). The criterion for statistical significance was set at P < 0.05.
RESULTS
A total of 47 rats were used in this study. No animals died during surgery. However, seven animals (14%) died during the reperfusion period (four animals in the progesterone group and one animal in all of the other groups). Four of these animals had subarachnoid hemorrhages (two in the progesterone group and one in each of the other hormone treatment groups). Intraischemic mean MABP, blood gases, and blood glucose levels were equivalent among treatment groups (Table 1), except for PaO2, which was significantly higher in the estrogen plus progesterone group than in the control group. Rectal and temporalis muscle temperatures were monitored and controlled and were not significantly different between treatment groups across the study period (Table 1). MABP was also not different between groups at all time points evaluated.
Summary of ischemic physiologic parameters
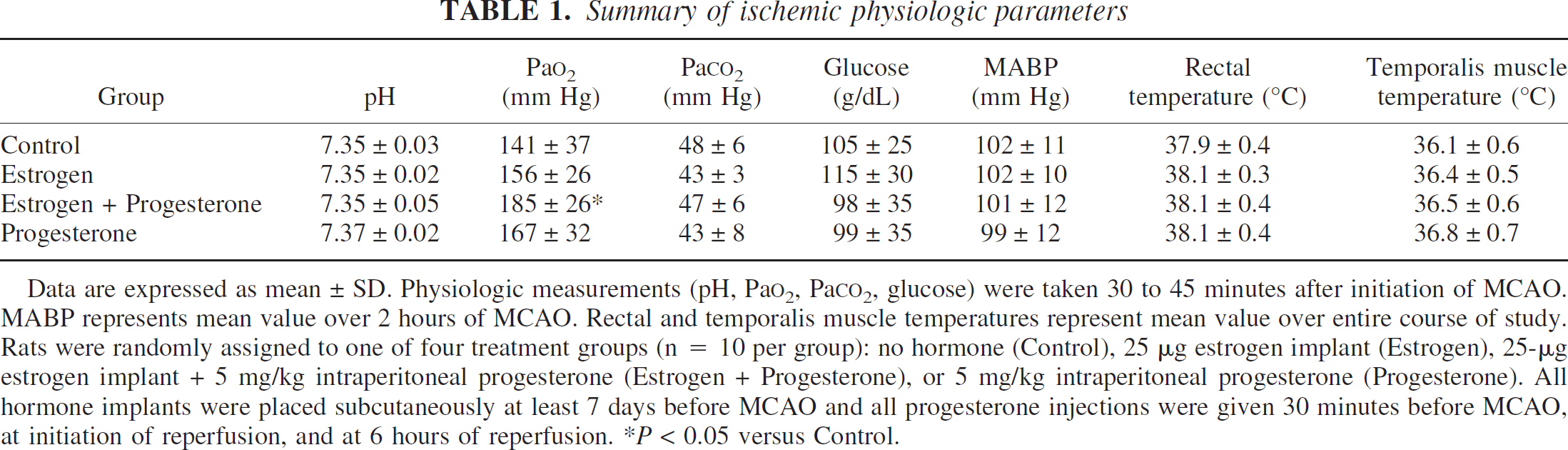
Data are expressed as mean ± SD. Physiologic measurements (pH, PaO2, PaCO2, glucose) were taken 30 to 45 minutes after initiation of MCAO. MABP represents mean value over 2 hours of MCAO. Rectal and temporalis muscle temperatures represent mean value over entire course of study. Rats were randomly assigned to one of four treatment groups (n = 10 per group): no hormone (Control), 25 μg estrogen implant (Estrogen), 25-μg estrogen implant + 5 mg/kg intraperitoneal progesterone (Estrogen + Progesterone), or 5 mg/kg intraperitoneal progesterone (Progesterone). All hormone implants were placed subcutaneously at least 7 days before MCAO and all progesterone injections were given 30 minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion. *P < 0.05 versus Control.
Figure 1 illustrates the effect of estrogen and progesterone, alone or combined, on infarction volume (percentage of contralateral structure). Cortical infarct volumes were significantly reduced in all estrogen alone (9% ± 16%) and combined hormone treatment groups (9% ± 10%) as compared with untreated animals (40% ± 28%). Total infarct volumes were also attenuated in all estrogen alone (9% ± 9%) and combined hormone treatment groups (8% + 6%) when compared to controls (24% + 13%). Acutely administered progesterone alone had no effect on either cortical (32% ± 25%) or total (19% ± 14%) infarct size as compared to untreated animals. Caudoputamen infarct volumes were not significantly different in all hormone treated groups (59% ± 40% in Estrogen; 46% ± 32% in Estrogen + Progesterone; 65% ± 40% in Progesterone) as compared with untreated RSF rats (87% ± 25%).
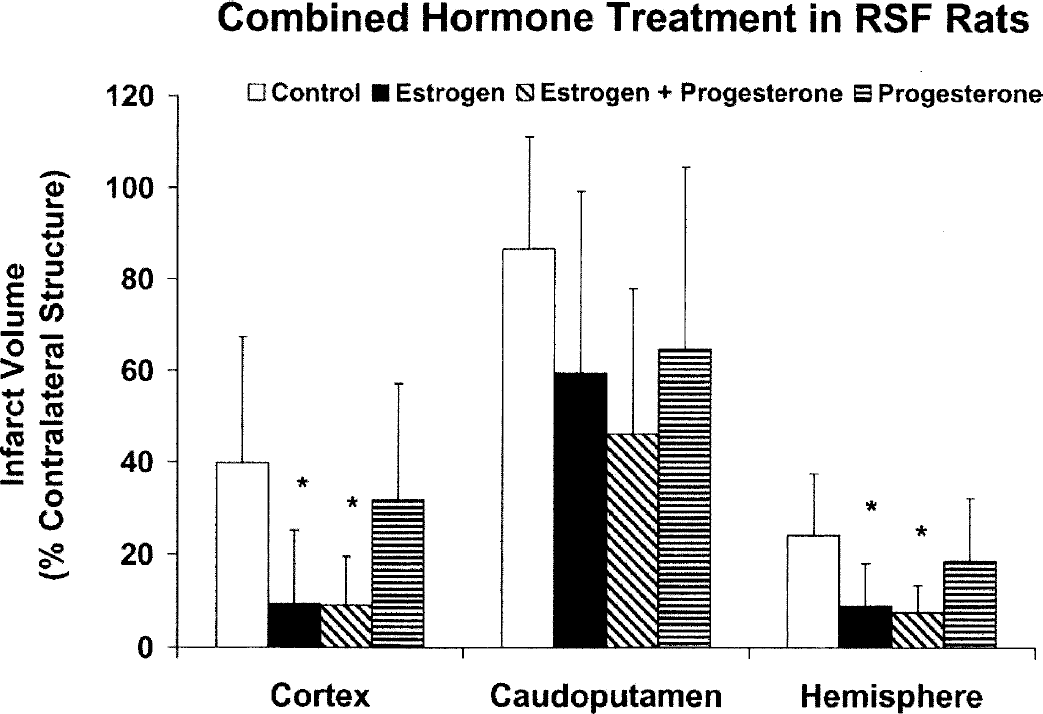
TTC-determined cortical, caudoputamen, and total hemispheric infarction volume (percentage of contralateral structure) at 22 hours of reperfusion after 2-hour MCAO in RSF rats (n = 10 per group) with no hormone (Control), 25-μg estrogen implant (Estrogen), 25-μg estrogen implant + 5 mg/kg intraperitoneal progesterone (Estrogen + Progesterone), or 5 mg/kg intraperitoneal progesterone (Progesterone). All hormone implants were placed subcutaneously at least 7 days before MCAO and all progesterone intraperitoneal injections were given 30 minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion. *P < 0.05 versus Control. All values are mean ± SD.
Figure 2 summarizes residual intraischemic LDF (expressed as a percent of baseline signal) and emphasizes the lack of difference in reduction of LDF signal among treatment groups. Reperfusion, as demonstrated by early recovery of LDF signal, occurred in all animals regardless of treatment group within minutes of occlusion release. Mean ischemic LDF (percentage of baseline signal) in RSF rats treated with either estrogen alone (29% ± 10%), progesterone alone (33% ± 10%), or combined hormone (33% ± 7%) was not different from that observed in untreated animals (28 ± 14%), further suggesting that the relative ischemic insult was equivalent among all groups.
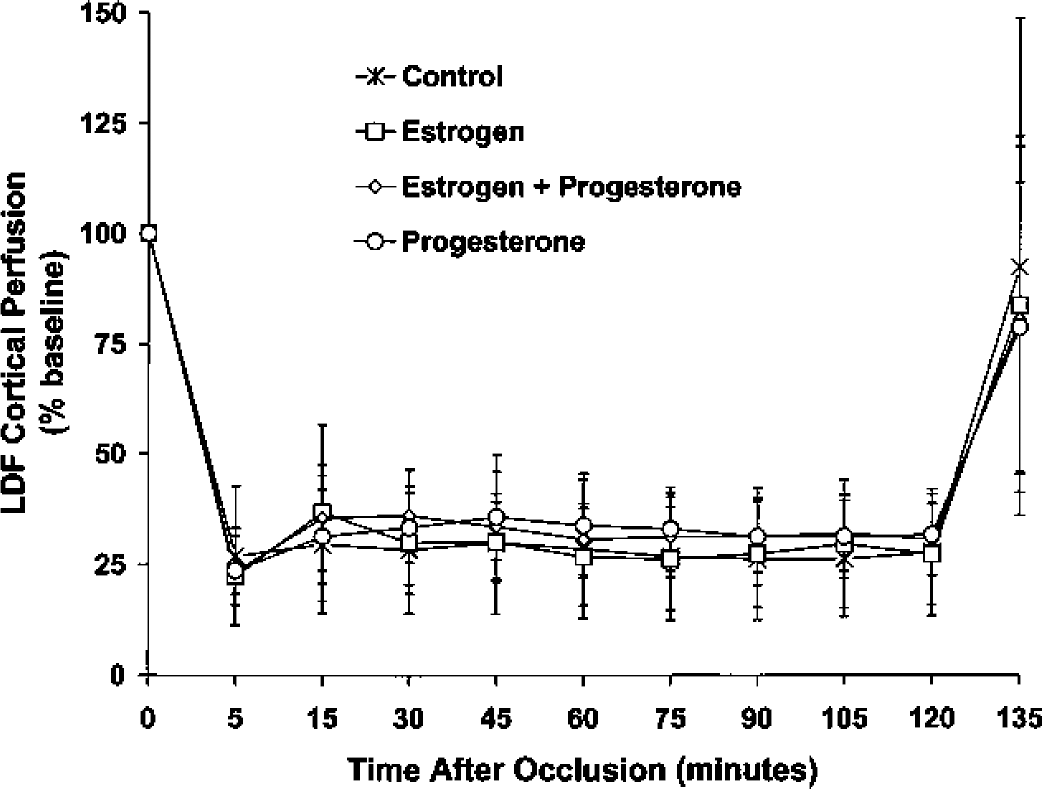
LDF cerebral cortical microvascular perfusion during 2 hours of MCAO and early reperfusion in RSF rats (N = 10 per group) with no hormone (Control), 25-μg estrogen implant (Estrogen), 25-μg estrogen implant + 5 mg/kg intraperitoneal progesterone (Estrogen + Progesterone), or 5 mg/kg intraperitoneal progesterone (Progesterone). All hormone implants were placed subcutaneously at least 7 days before MCAO and all progesterone intraperitoneal injections were given 30 minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion. LDF was measured over the ipsilateral parietal cortex and expressed as a percentage of baseline. There were no significant differences among treatment groups over time. All values are mean ± SD.
DISCUSSION
The present study has several findings. Combined hormone administration and chronically administered 17ß-estradiol alone reduced cortical and total brain ischemic injury. In contrast, acutely administered progesterone alone did not reduce cortical or striatal infarcts. These results suggest that in ischemic RSF rat brain, combined hormone administration reduces infarction volume and that progesterone does not attenuate estrogen's effect.
Reproductively senescent female rodents have been used to study neuroendocrine changes associated with menopause in women (Cooper et al., 1986; Meites et al., 1980; Wise, 1993, 1999; Wise et al., 1999). This is the first study to report the effects of combined hormone treatment on ischemic RSF rat brain. Our overall objective was to determine if combined hormone administration would reduce infarct injury in middle-aged female rats and if this effect was comparable with or greater than the effects observed with each hormone alone. Each treatment protocol used doses of estrogen or progesterone that, when given alone, are neuroprotective in ischemic ovariectomized (Murphy et al., 2002; Rusa et al., 1999) or RSF (Alkayed et al., 2000) ischemic rat brain. Administration of a 25-μg 17ß-estradiol pellet subcutaneously in ovariectomized females (Rusa et al., 1999) and RSF rats (Alkayed et al., 2000) resulted in increased estrogen plasma levels of 20 ± 25 pg/mL (n = 10) and 14 ± 9 pg/mL (n = 8), respectively versus untreated ovariectomized (3 ± 1 pg/mL, n = 10) and RSF animals (7 ± 6 pg/mL, n = 6). In ovariectomized female rats (n = 14 per group), the progesterone dose of 5 mg/kg intraperitoneal given at 30-minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion resulted in significantly (P < 0.05) higher progesterone plasma levels of 32 ± 13 ng/mL and 20 ± 18 ng/mL at 1 hour of ischemia and at 22 hours of reperfusion, respectively as compared with controls during ischemia (8 ± 4 mg/mL) and reperfusion (6 ± 5 ng/mL) (Murphy et al., 2002).
Although combined estrogen/progestin compounds are the most commonly prescribed hormone regimen in the United States, it is not known whether progestins interact with estrogen and diminish its neuroprotective effects. Experimental data suggest that progestins like medroxyprogesterone acetate can reverse the beneficial effect of estrogen seen on atherosclerotic plaque formation in nonhuman primates (Adams et al., 1997; Williams et al., 1994). However, recent clinical results argue against this hypothesis. The Estrogen Replacement and Atherosclerosis trial used estrogen with or without a progestin and found no benefit in coronary disease progression as measured angiographically in either treatment group (Herrington et al., 2000). Still, progestin type, formulation, and route and timing of administration are important factors to consider when determining whether progestins alone increase susceptibility or protection in cerebrovascular disease or ischemic brain injury. The role of medroxyprogesterone acetate in the suspended combined HRT Women's Health Initiative trial has yet to be elucidated (Rossouw et al., 2002). Progestins that might be detrimental, neutral, or beneficial when solely administered might prove to be antagonistic, neutral, or synergistic when combined with estrogen. Our findings suggest that progesterone did not antagonize estrogen's effects in ischemic brain.
In the present study, chronically administered estradiol reduced cortical and total hemispheric infarct but had no effects on striatal infarct in RSF rats. This is in contrast with our previous findings in RSF rats, in which the same dose (25-μg 17ß-estradiol) and method of delivery (subcutaneous implant) was used but both cortical and striatal infarct reduction was observed (Alkayed et al., 2000). Acutely administered progesterone alone (5 mg/kg intraperitoneal) administered before and after MACO had no effect on cortical or striatal injury in RSF rats. However, in young ovariectomized females, this same treatment protocol resulted in reduction in both cortical and total hemispheric infarct (Murphy et al., 2002). Previous work has shown that long-term progesterone treatment reduced cortical injury in RSF rats (Alkayed et al., 2000) but exacerbated striatal injury in young ovariectomized rats (Murphy et al., 2000). These observations suggest that outcome could be linked to dose and duration of hormone used, as well as differential effects linked to age.
The differences in our findings on individual female sex steroids from previous work may also be explained to some extent by temporalis muscle temperature. In our previous study (Alkayed et al., 2000), temporalis muscle temperature was reported to be between 36.5°C and 37.5°C in both control and only estrogen-treated RSF rats. However, in our current study, intraischemic temporalis muscle temperature was relatively lower in both control (36.1° ± 2.0°C) and estradiol-treated animals (36.3° ± 1.6°C). This could partly explain the lack of an effect of estradiol on striatal injury in the current study as compared with our previous work (Alkayed et al., 2000). Nonetheless, there were no differences in temporalis muscle temperature among the treatment groups in this study.
Differences in cortical injury in progesterone-treated animals between the current study and our previous study (Murphy et al., 2002) could be explained by differences in age (8 to 10 weeks versus 14 to 18 months). It is not known whether mechanisms of ischemic injury and neuroprotection are similar in young adult versus RSF rats. Outcome differences may also be a consequence of varying progesterone and estradiol levels in plasma in middle-aged animals versus ovariectomized rats. Most groups characterize RSF rodents in terms of vaginal cytology or gonadotrophin levels (Cooper et al., 1986; LeFevre and McClintock, 1988; Lu et al., 1994; Meites et al, 1980; Wise, 1993, 1999; Wise et al., 1999). Very few groups report hormone levels for RSF rodents. Our previous plasma estradiol measurements (Alkayed et al., 2000) as well as other laboratories characterizations based on vaginal cytology or gonadotrophin levels would suggest that RSF Wistar rats are a heterogeneous group in terms of endocrine status, as are perimenopausal women, thereby making the middle-aged or RSF rat a suitable model for perimenopausal women rather than aged rats. Progesterone metabolism in hypothalamus and pituitary can be altered with reproductive senescence (Hodges and Karavolas, 1992; Marrone and Karavolas, 1982). Therefore, differences in progesterone metabolism between RSF and younger animals may also be contributing to the observed differences in outcome.
Several mechanisms can be considered to explain the effect of combined hormone administration in ischemic RSF rat brain. Estrogen's proposed mechanisms of protection for brain and the cerebral vasculature are quite complex (for review, see McCullough and Hurn, 2003). There is evidence implicating, and refuting, the importance of nuclear hormone receptor signaling mechanisms to gender differences and to 17β-estradiol's antiischemic activity (Dubal et al., 2001; Sampei et al., 2000; Sawada et al., 2000; Simpkins et al., 1997). However, it is clear that rapid receptor-mediated and receptor-independent intracellular signaling is pertinent in neuroprotection and does not involve gene transcription (Falkenstein et al., 2000; Linford et al., 2000). These actions involve putative membrane estrogen receptors, kinase cascades, and intracellular signaling that activate ion channels, neurotransmitter receptors, and enzymes such as nitric oxide synthase. Such mechanisms may be critical to estrogen's protection in experimental stroke. Finally, many estrogens have potent, concentration-dependent lipid antioxidant activity (Kume-Kick and Rice, 1998; Mooradian, 1993). Estrogens likely act at multiple sites in injured brain and use signaling processes that are receptor-dependent, receptor-independent, and non-cell-type specific.
Current literature suggests several possible protective mechanisms for progesterone in brain. For example, progesterone reduced membrane lipid peroxidation in traumatic brain injury models (Roof et al., 1997; Roof and Hall, 2000; Stein et al., 1999). Progesterone also has direct effects on neurotransmission by altering GABA conduction (Majewska, 1992), upregulating GABAA receptor number (Weiland and Orchinik, 1995), increasing GABAA agonist binding affinity (Jussofie et al., 1995) and site number (Juptner et al., 1991), and by enhancing glutamic acid decarboxylase mRNA (Grattan et al., 1996; Weiland, 1992). Progesterone also moderates excitatory amino acid responsiveness (Smith et al., 1987; Smith, 1991). Thus, progesterone may ameliorate ischemia-induced excitotoxic injury through potentiation of inhibitory GABA receptor activation and/or inhibition of excitatory amino acid receptors. Progesterone receptors could also be involved in the response to vascular injury (Karas et al., 2001). The marginal effects on striatal injury by progesterone observed in this study and our previous studies (Alkayed et al., 2000; Murphy et al., 2002) may thus be related to differences in regional expression of GABA, glutamate, and progesterone receptor subunits/subtypes or to different ischemic mechanisms in the cortex versus the striatum. Lastly, progesterone's actions in injured brain may be mediated in part by locally produced metabolites such as 5α-dihydroprogesterone and 3α, 5α-tetrahydroprogesterone (Ciriza et al., 2004).
Potential cooperation in stroke mechanisms between progesterone and estrogen must also be considered. It is well known that the background steroid environment of the brain alters the ability of progesterone or estrogen to vary reproductive signaling and behavior (McCarthy, 1995; Smith, 1994). Our previous work and that of others indicate that estrogen does not require normal plasma progesterone levels to reduce stroke injury (Alkayed et al., 1998, 2000; Hurn et al., 1998; Rusa et al., 1999; Simpkins et al., 1997; Toung et al., 1998). However, estrogen priming is a requisite for many progesterone-mediated actions in normal brain (McCarthy, 1995; Smith, 1994; Tischkau and Ramirez, 1993). These two steroids also interact at the receptor level, because estrogen has been shown to upregulate progesterone receptor expression in brain (Moffatt et al., 1998). Therefore, progesterone may be most efficacious in an ischemic brain environment that has been estrogen primed. However, the results from this study do not support this supposition.
The widening gap between clinical trial results and experimental laboratory based data would suggest that our understanding of the cerebral ischemic pathophysiology and of estrogen's and/or progesterone's role as a cerebroprotectant is incomplete. The discrepancy between observational studies, preclinical data, and large randomized clinical trials (for review, see Murphy et al., 2003, 2004) emphasizes the need for further study of the mechanisms leading to the increased stroke incidence observed in postmenopausal women and the effects of HRT on stroke incidence and outcome. These data have potential clinical implications relative to stroke for postmenopausal women taking combined HRT.
