Abstract
Although progesterone is neuroprotective in traumatic brain injury, its efficacy in stroke is unclear. The authors determined whether there are infarction differences after middle cerebral artery occlusion (MCAO) in ovariectomized rats treated acutely with progesterone before MCAO or both pre- and postischemia. Rats received vehicle, 5 (P5), 10 (P10), or 20 (P20) mg/kg progesterone intraperitoneally 30 minutes before MCAO. In another cohort, animals received vehicle or 5 (P5R) mg/kg progesterone intraperitoneally 30 minutes before MCAO, at reperfusion initiation, and at 6-hour reperfusion. Animals underwent 2-hour MCAO by the intraluminal filament technique, followed by 22-hour reperfusion. Cortical (CTX) and caudate-putamen (CP) infarctions were determined by 2,3,5-triphenyltetrazolium chloride staining and digital image analysis. End-ischemic and early reperfusion regional cerebral blood flow (CBF) was measured by [14C]-iodoantipyrine quantitative autoradiography in vehicle- or progesterone (5 mg/kg)-treated rats. Cortical infarction (% contralateral CTX) was 31 ± 30% (vehicle), 39 ± 23% (P5), 41 ± 14% (P10), and 28 ± 20% (P20). Caudate-putamen infarction (% contralateral CP) was 45 ± 37% (vehicle), 62 ± 34% (P5), 75 ± 17% (P10), and 52 ± 30% (P20). In vehicle and P5R groups, CTX infarction was 37 ± 20% and *20 ± 17%, respectively (*P < 0.05 from vehicle). In vehicle and P5R groups, CP infarction was 63 ± 26% and 43 ± 29%, respectively. End-ischemic regional CBF and CBF recovery during initial reperfusion was unaffected by progesterone treatment. These data suggest that progesterone administration both before MCAO and during reperfusion decreases ischemic brain injury.
Premenopausal women have a lower risk of stroke compared with men of the same age, but vascular events increase in frequency after menopause (Wenger et al., 1993). Few studies have examined sex differences in cerebrovascular disease mechanisms or the importance of estrogen and progesterone in stroke morbidity. Although cardioprotection has been reported with postmenopausal estrogen or estrogen-progestin replacement (Grodstein et al., 1996, 2000; Hulley et al., 1998), a limited number of epidemiologic studies have evaluated progestins and stroke risk in humans. Recent studies have found that, in normotensive women, the use of oral or injectable progestogen-only contraceptives (World Health Organization, 1998) or combined oral (Gillum et al., 2000) or injectable contraceptives (World Health Organization, 1998) was not associated with significant changes in overall risk estimates for stroke.
Many experimental animal studies evaluating estrogen's effects in cerebral ischemia have suggested a neuroprotective role for this reproductive steroid (Hurn et al., 1998; Green and Simpkins, 2000; Roof and Hall, 2000; Stein, 2001). Progesterone's effects on experimental ischemic brain damage, however, have been more variable depending on sex, age, dose, or administration protocol. Previously, we have shown that chronic progesterone administration before ischemia exacerbates striatal injury in ovariectomized rats (Murphy et al., 2000). Others have reported neuroprotection due to progesterone in global ischemia (Gonzalez-Vidal et al., 1998; Cervantes et al., 2002) and in focal ischemic brain injury studies in male rats (Betz and Coester, 1990a, b ; Jiang et al., 1996; Chen et al., 1999; Kumon et al., 2000) and in older female rats (Alkayed et al., 2000). Beneficial effects of progesterone have also been described in traumatic brain injury (TBI) models in both male and female animals (Stein et al., 1999; Roof and Hall, 2000; Bramlett and Dietrich, 2001; Galani et al., 2001; Stein, 2001; Vink et al., 2001; Wright et al., 2001).
The purpose of the present study was to determine whether progesterone has neuroprotective qualities by evaluating the effect of exogenous progesterone replacement in young adult ovariectomized female rats. Progesterone was administered acutely either before experimental focal ischemia or during both pre- and post-ishemic periods. We tested the hypotheses that (1) progesterone improves stroke outcome in a dose- and/or time-dependent manner and (2) progesterone is neuroprotective through a blood flow-dependent mechanism.
MATERIALS AND METHODS
This study was conducted in accordance with the National Institutes of Health guidelines for the care and use of animals in research, and the protocols were approved by the Animal Care and Use Committee of the Johns Hopkins University. All methods are as previously described (Alkayed et al., 1998; Toung et al., 1998). Briefly, age-matched, sexually mature female Wistar rats (Hsd:WI, 200 to 225 g; Harlan, Indianapolis, IN, U.S.A.) were ovariectomized under halothane anesthesia (induction 4% to 5%, maintenance 1.25% to 1.5% delivered via mask in O2-enriched air) 1 to 3 weeks before vehicle/progesterone treatments and middle cerebral artery occlusion (MCAO). Progesterone stock solution contained 50 mg/mL progesterone (USP; Sigma, St. Louis, MO, U.S.A.) with 10% benzyl alcohol (ACS reagent; Sigma) as preservative in sesame oil (Sigma) and was sterile-filtered before use. Ovariectomized females received sesame oil (vehicle or P0), 5 (P5), 10 (P10), or 20 (P20) mg/kg progesterone intraperitoneally 30 minutes before MCAO (n = 8 per group). In an additional cohort, ovariectomized females were administered either sesame oil (P0R, n = 14) or 5 mg/kg progesterone (P5R, n = 14) intraperitoneally 30 minutes before MCAO, at initiation of reperfusion, and at 6 hours of reperfusion. Ovariectomized animals were randomly assigned to preischemic and pre-/postischemic treatments. Average number of days from ovariectomy until MCAO for each treatment group for both infarction outcome studies and regional CBF studies did not significantly differ.
For MCAO, the animal was anesthetized with halothane (induction 4% to 5%, maintenance 1.25% to 1.5% delivered via mask in O2-enriched air) and a femoral artery catheter was placed for continuous monitoring of MABP and measurement of arterial blood gases. Rectal and temporalis muscle temperature were monitored with a Mon-a-therm system (Mon-a-therm, model 6510; Mallinckrodt Medical, Inc., St. Louis, MO, U.S.A.) and maintained by a heating lamp. The effectiveness of vascular occlusion was determined by sustained reduction of laser-Doppler flow signal (LDF, model MBF3D; Moor Instruments Ltd., Oxford, U.K.) obtained by probe placement 6 mm lateral and 2 mm posterior to bregma (Alkayed et al., 1998; Toung et al., 1998).
Focal cerebral ischemia was accomplished using a modified intraluminal filament technique, as previously described (Longa et al., 1989). Briefly, the right common carotid and external carotid arteries were exposed and ligated. The right occipital artery was cauterized, and the right pterygopalatine artery was then ligated. After the baseline LDF was determined, a 4.0 nylon monofilament surgical suture with a heat-blunted tip was introduced via the right common and internal carotid arteries until the LDF signal displayed an abrupt and significant reduction, confirming ongoing ischemia. The suture was then secured in place. Laser Doppler flow was measured during ischemia over 15 to 30 minute intervals and for the first 15 minutes of reperfusion to confirm cortical reperfusion when the occluding filament was removed. Five-minute recording averages were analyzed for each time point. To ensure relative uniformity of the ischemic insult, animals were excluded from the cohort if mean ischemic LDF was greater than or equal to 40% of baseline LDF. The animal was euthanized via decapitation under halothane anesthesia (4% to 5%) 22 hours after the 2-hour MCAO period, and the brain removed. The brain was then sliced into 2-mm-thick coronal sections (7 slices total) that were subsequently placed into a 1% solution of 2,3,5-triphenyltetrazolium chloride (TTC; Sigma) and incubated at 37°C for 30 minutes. Stained sections were fixed in 10% neutral buffered formalin. Both sides of each stained coronal slice were photographed via a digital camera, and then evaluated by digital image analysis (SigmaScan Pro; SPSS Science, Chicago, IL, U.S.A.). The infarcted area is integrated across sections, and infarction volumes in cerebral cortex, caudate-putamen complex, and hemisphere expressed as percentage of the contralateral structure (cortex, caudate-putamen, hemisphere). Blood was obtained 1 hour into ischemia and at 22-hour reperfusion for steroid analysis. Plasma 17β-estradiol and progesterone levels were measured in duplicate by radioimmunoassay (Coat-a-count; Diagnostic Products Corp., Los Angeles, CA, U.S.A.), as previously described (Hurn et al., 1995). Regional CBF was measured at 2-hour MCAO (end-ischemia) and at 10 minutes of reperfusion in additional nonsurvival cohorts (n = 4 per group) of ovariectomized female Wistar rats with the use of quantitative [14C]-iodoantipyrine (IAP) autoradiography under halothane anesthesia, as described previously (Alkayed et al., 1998, 2000). Femoral vascular catheters and LDF monitoring were used in each animal, and MCAO was done as in previous cohorts. Animals received either progesterone (5 mg/kg) or vehicle (sesame oil) intraperitoneally 30 minutes before MCAO (end-ischemia group) and at the initiation of reperfusion (10-minute reperfusion group). Arterial blood gases were measured before MCAO, at 1-hour MCAO, and at 5 minutes before regional CBF measurements. At end-ischemia or at 10 minutes of reperfusion, 40 μCi of [11C]-IAP (New England Nuclear, Boston, MA, U.S.A.) in 0.8 mL of isotonic saline was infused intravenously over 45 seconds. During infusion, fifteen 20-μL samples of free-flowing arterial blood from the femoral artery catheter were collected at 3-second intervals in EDTA microtainer tubes (Becton Dickinson, Franklin Lakes, NJ, U.S.A.). The rat was decapitated 45 to 50 seconds after the start of infusion, and the brain quickly removed and frozen at −50°C in 2-methylbutane on dry ice. Each brain was sectioned by cryostat into 20-μm-thick coronal sections at −20°C and thaw-mounted onto cover glasses. Sections were apposed for 1 week to film (Bio-max MR; Eastman Kodak Company, Rochester, NY, U.S.A.) with [11C] standards. The concentration of [14C]-IAP in the blood samples was determined by liquid scintillation spectroscopy (LS 1800; Beck-man Coulter, Irvine, CA, U.S.A.) after decolorization with 0.2 mL of tissue solubilizer (Soluene-350; Packard Instrument Co., Meriden, CT, U.S.A.). Autoradiographic images representing different coronal levels were digitized, and regional CBF was determined by use of image analysis software (Inquiry, Loats Associates). Two methods of analysis were used to determine regional CBF, as previously described (Alkayed et al., 1998, 2000). First, CBF was measured by sampling 0.1-mm squares within core and penumbral regions affected by MCAO: frontal and parietal cerebral cortex and lateral and medial caudo-putamen. Flow rates were averaged within 6 to 9 consecutive brain slices at each of 3 coronal levels (+2.2, +0.2, −1.8 mm from the bregma). In the second method, areas perfused with predetermined CBF intervals were isolated by digital imaging scanning and summed to construct a histogram distribution of brain tissue over incremental ranges of blood flow rates. Areas were averaged among 3 sections from 5 coronal levels (+2.2, +0.2, −1.8, −3.8, −5.8 mm from the bregma) and then were numerically integrated to obtain an estimate of tissue volume for each CBF interval.
All values are reported as mean ± SD unless otherwise indicated. All repeated-measures data, including physiologic parameters, plasma hormone levels, and LDF, during MCAO and early reperfusion were analyzed by both repeated measures 2-way analysis of variance (ANOVA) and repeated measures 1-way ANOVA with post hoc Newman-Keuls test for multiple comparisons (SigmaStat Statistical Software, Version 2.0; SPSS Science). Differences in mean ischemic LDF, infarction volumes, and regional CBF measurements were determined by 1-way ANOVA with post hoc Newman-Keuls test for multiple comparisons (SigmaStat Statistical Software, Version 2.0; SPSS Science). The criterion for statistical significance is P < 0.05.
RESULTS
Baseline, intraischemic and reperfusion MABP, blood gases, and glucose levels were equivalent among preischemic treatment groups (Table 1). However, in the pre-/postischemic treatment groups, there was a significant but similar decrease in MABP during early reperfusion from baseline within both vehicle and progesterone treatment groups (Table 1). Mean intraischemic rectal and temporalis muscle temperatures were controlled in all groups. Mean intraischemic plasma estradiol values, as well as plasma estradiol levels at 22-hour reperfusion, were not different among groups (< 5 pg/mL). Progesterone levels were elevated in all progesterone-treated groups relative to vehicle-treated, ovariectomized animals within the ischemic period but fell to within ovariectomized baseline levels by 22 hours of reperfusion except in the P5R group (Table 1).
Summary of physiologic parameters
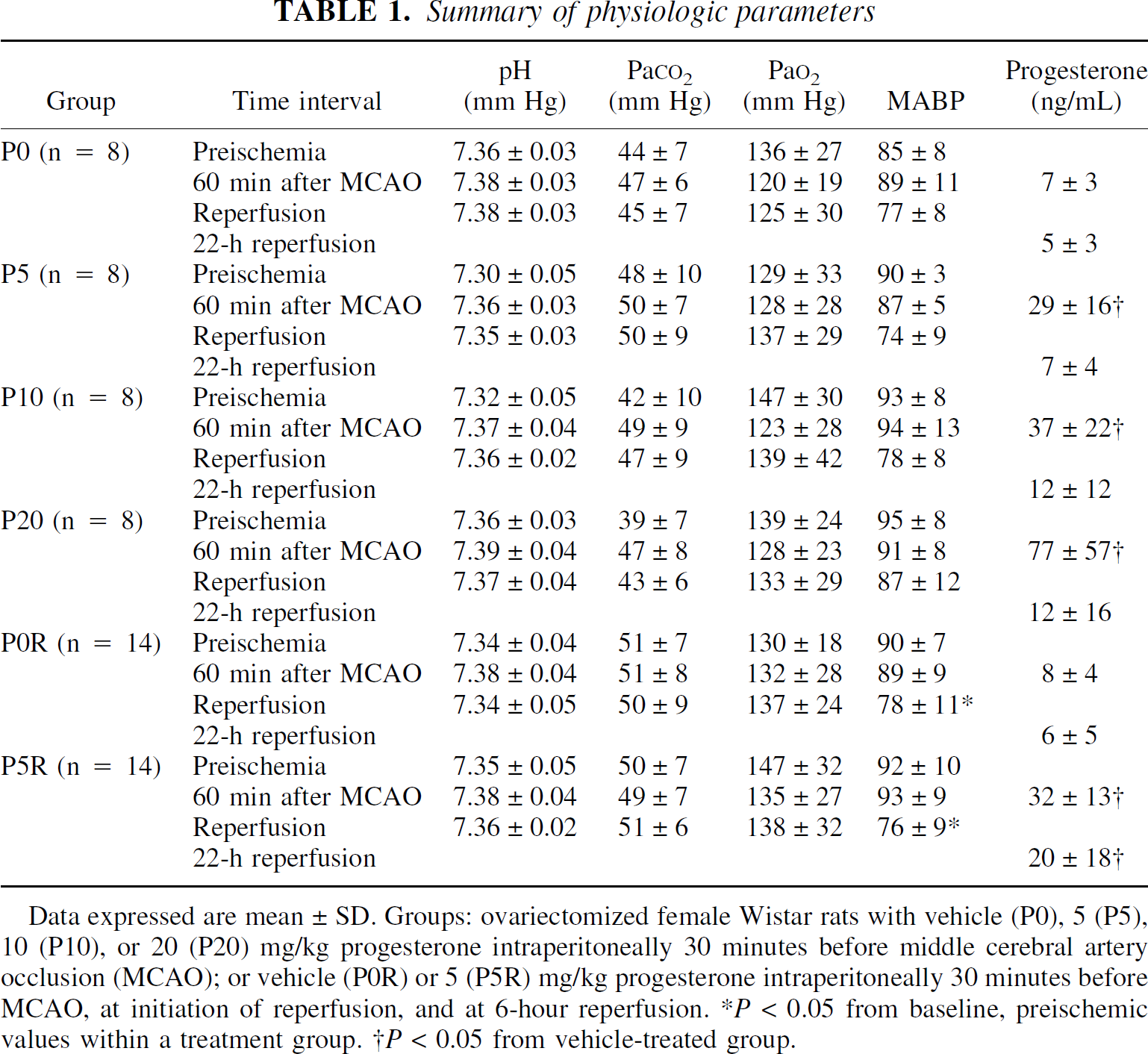
Data expressed are mean±SD. Groups: ovariectomized female Wistar rats with vehicle (P0), 5 (P5), 10 (P10), or 20 (P20) mg/kg progesterone intraperitoneally 30 minutes before middle cerebral artery occlusion (MCAO); or vehicle (P0R) or 5 (P5R) mg/kg progesterone intraperitoneally 30 minutes before MCAO, at initiation of reperfusion, and at 6-hour reperfusion. *P < 0.05 from baseline, preischemic values within a treatment group. †P < 0.05 from vehicle-treated group.
Figure 1 illustrates the lack of an effect of progesterone on cortical infarction volume (% contralateral cortex) at any of the acute preischemic doses tested, as compared with the vehicle-treated ovariectomized female rats. Similarly, acute preischemic treatment did not improve caudate-putamen (CP) injury. Cortical and total hemispheric damage, however, were decreased by pre-and postischemic progesterone administration (Fig. 2). Conversely, this treatment paradigm did not improve CP infarct size.
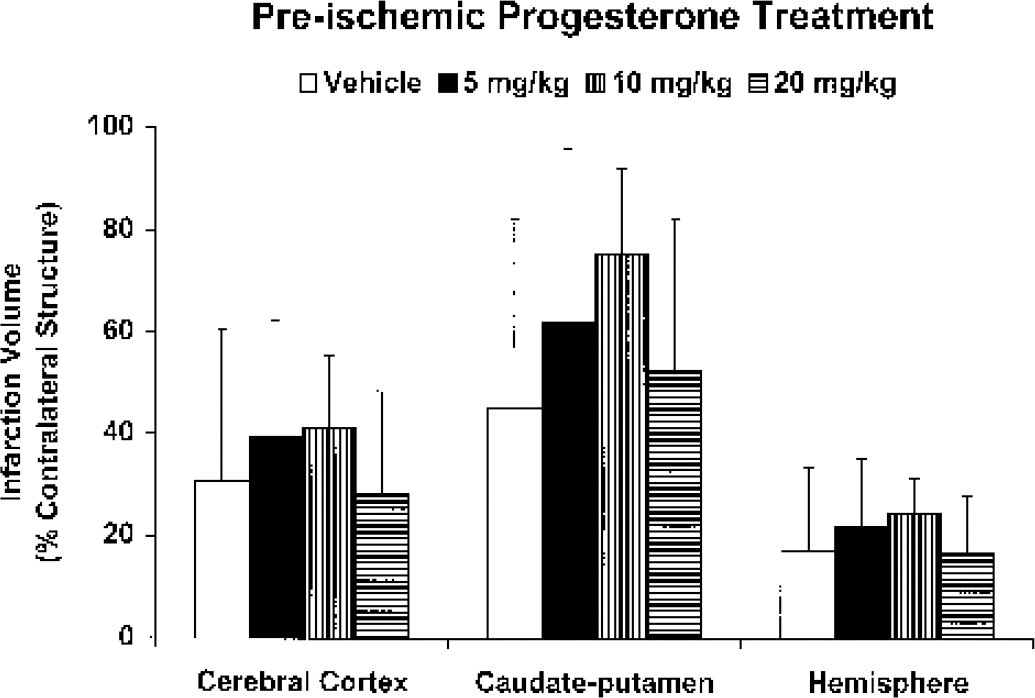
Cortical, caudate-putamen, and hemispheric infarction volumes (% contralateral structure) in ovariectomized female Wistar rats pretreated with either vehicle, 5 mg/kg, 10 mg/kg, or 20 mg/kg progesterone intraperitoneally 30 minutes before middle cerebral artery occlusion (n = 8 per group). All values are mean ± SD.
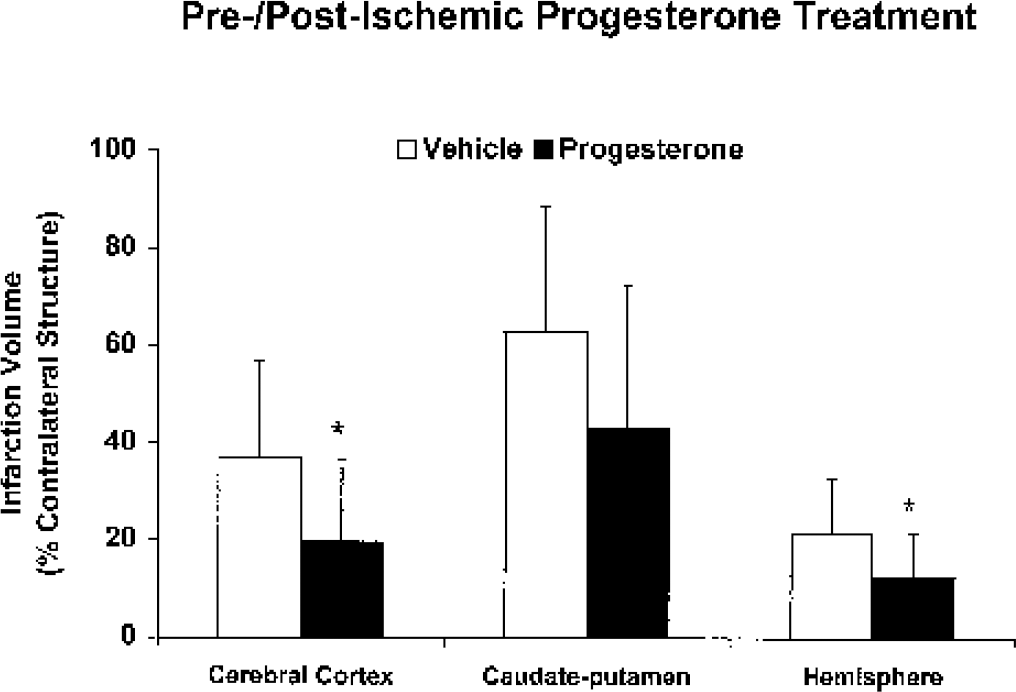
Cortical, caudate-putamen, and hemispheric infarction volumes (% contralateral structure) in ovariectomized female Wistar rats pretreated with either vehicle or 5 mg/kg progesterone intraperitoneally 30 minutes before middle cerebral artery occlusion, at initiation of reperfusion, and at 6 hours of reperfusion (n = 14 per group). All values are mean ± SD. *Significantly different from vehicle-treated animals (P < 0.05).
Mean ischemic LDF (% baseline signal) in ovariectomized rats with either acute progesterone replacement (25 ± 7% in P5; 25 ± 8% in P10; 26 ± 9% in P20) or repeated pre- and postischemic progesterone replacement (27 ± 7% in P5R) was not different from that observed in P0 (25 ± 8%) and P0R (26 ± 6%), suggesting that the relative ischemic insult was equivalent among all groups. Reperfusion, as demonstrated by early recovery of LDF signal, occurred in all animals regardless of treatment group within minutes of occlusion release.
CBF at end-ischemia and at 10 minutes of reperfusion was evaluated in separate cohorts of vehicle- or progesterone-treated animals. Figure 3 shows absolute regional CBF in affected brain regions of the middle cerebral artery territory (core and penumbral regions), as well as average CBF within the contralateral regions. No differences were seen between treatment groups at each time point evaluated except at end-ischemia in the contralateral, nonischemic lateral CP. As compared with end-ischemic CBF, regional CBF in ipsilateral cortical and striatal regions was increased at 10 minutes of reperfusion regardless of treatment group.
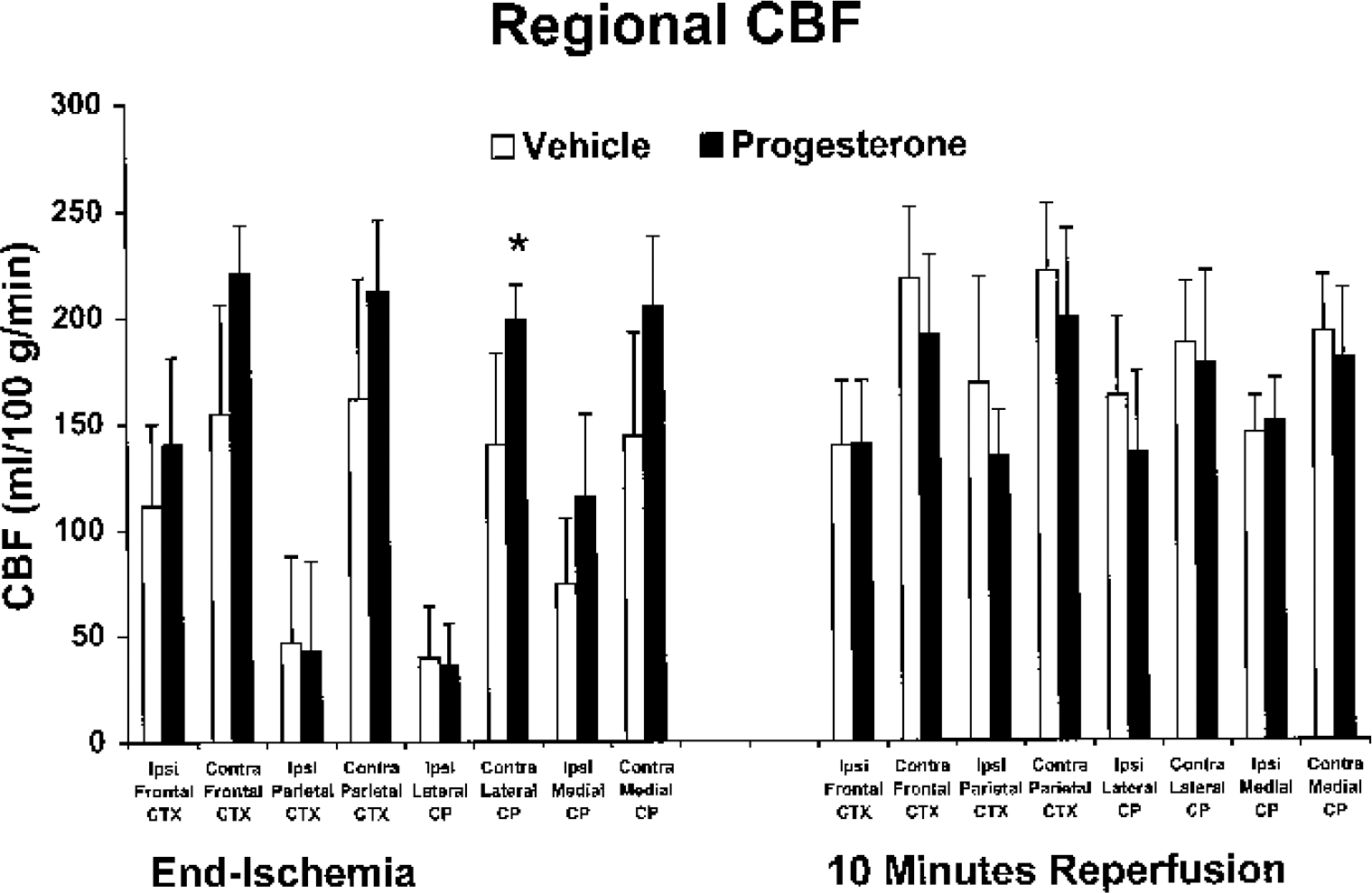
Effect of progesterone versus vehicle on cerebral blood flow (CBF) in 4 brain regions (frontal and parietal cortex [CTX]; lateral and medial caudate-putamen [CP]) within the ischemic, ipsilateral (ipsi), and nonischemic, contralateral (contra) middle cerebral artery territory at end-ischemia and at 10 minutes of reperfusion. Ovariectomized female Wistar rats were pretreated with vehicle or 5 mg/kg progesterone intraperitoneally 30 minutes before middle cerebral artery occlusion (MCAO; end-ischemia, n = 4 per group), or with vehicle or 5 mg/kg progesterone intraperitoneally 30 minutes before MCAO and at initiation of reperfusion (10 minutes reperfusion, n = 4 per group). All values are mean ± SD. *Significantly different from vehicle-treated animals (P < 0.05).
To further examine differences in end-ischemic and early reperfusion CBF distribution in these animals, we quantified total hemispheric brain tissue volume that experienced near-zero CBF (potentially ischemic core) as well as tissue volumes experiencing less severely reduced CBF (likely penumbra). We monitored LDF during occlusion and reperfusion up to within 5 minutes of IAP injection and decapitation to ensure uniformity of insult. There were no differences in either mean ischemic LDF or LDF assessed approximately 5 minutes before IAP injection/decapitation between treatment groups. Figure 4 shows the results of this partitioning of brain volumes into incremental levels of absolute CBF in progesterone-deficient versus progesterone-repleted animals at end-ischemia and at 10 minutes of reperfusion. There were no differences between treatment groups at any flow increment at either time point evaluated, suggesting that progesterone did not recruit tissue from a low “flow state” to a partially preserved flow state at end-ischemia or during early reperfusion.
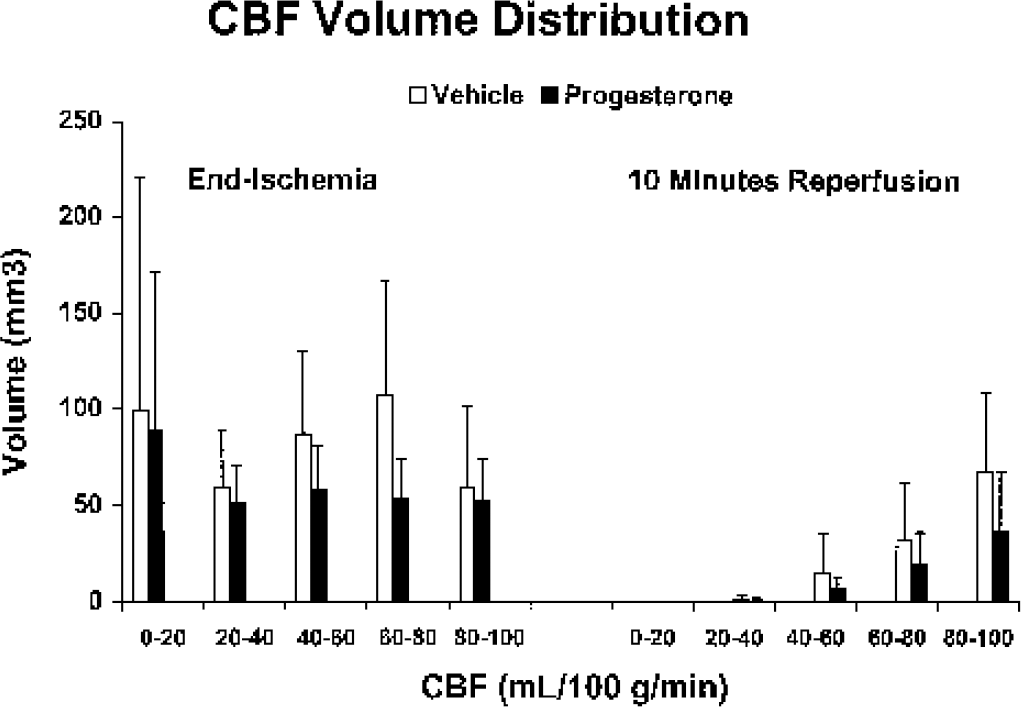
Effect of progesterone versus vehicle on tissue volume distribution when partitioned into cerebral blood flow (CBF) increments at end-ischemia and at 10-minute reperfusion. Ovariectomized female Wistar rats were pretreated with vehicle or 5 mg/kg progesterone intraperitoneally 30 minutes before middle cerebral artery occlusion (MCAO; end-ischemia, n = 4 per group), or with vehicle or 5 mg/kg progesterone intraperitoneally 30 minutes before MCAO and at initiation of reperfusion (10-minute reperfusion, n = 4 per group). All values are mean ± SD.
DISCUSSION
The present study demonstrates 3 important findings. First, acute exogenous preischemic progesterone treatment did not significantly alter brain infarction after vascular occlusion in young adult, ovariectomized female rats. Second, progesterone administration both before MCAO and during reperfusion decreased ischemic brain injury in ovariectomized rats. Third, exogenous progesterone does not protect by preserving intraocclusion CBF or by enhancing early reperfusion. These findings suggest that progesterone is neuroprotective in the ischemic female rat brain when physiologic plasma progesterone levels are sustained during the reperfusion period and that exogenous progesterone has a dose- and time-dependent neuroprotective action in experimental stroke.
Our overall research objective was to identify a neuroprotective dose for progesterone in ovariectomized rats in the MCAO model. We have previously observed either no neuroprotection or a deleterious effect on infarct volume in this same model (Murphy et al., 2000). We hypothesized that lower preischemic progesterone doses could be protective and accordingly evaluated several additional doses administered as a pretreatment. We then selected the lowest dose (one that did not exacerbate injury) and changed the treatment to exposure both during and after ischemia. This latter treatment paradigm was found to be efficacious. The dose selected for longer exposure studies was similar to those previously reported to be neuroprotective in ischemic male rats (Jiang et al., 1996; Chen et al., 1999; Kumon et al., 2000). Further, the time points for progesterone administration in the pre-/postischemic dosing regimen were adapted from previous studies in male rats subjected to 2-hour MCAO and rats exposed to TBI. All of these studies demonstrated either a reduction in infarct size or cerebral edema due to progesterone given before and after injury. In the first of these studies (Jiang et al., 1996), male rats were given 4 mg/kg progesterone 30 minutes before MCAO and/or at 2, 6, and 24 hours after MCAO. A reduction in infarct injury was observed 48 hours after MCAO. Plasma progesterone levels, however, were reported from only 2 animals (41.9 and 70.7 ng/mL 4 hours after initial injection). A separate study demonstrated that 8 mg/kg progesterone given at 2, 6, and 24 hours after MCAO was also neuroprotective in male rats (Chen et al., 1999). In another study, male spontaneously hypertensive rats given 8 mg/kg progesterone at 2, 6, and 24 hours after MCAO, then daily for 7 days, also showed reductions in lesion size (Kumon et al., 2000). In rat TBI, a 4 mg/kg progesterone dose given 1, 6, and 24 hours after injury, then daily, was shown to be effective in reducing edema (Roof et al., 1994, 1996; Asbury et al., 1998; Galani et al., 2001). In a more recent study in the rat TBI model (Wright et al., 2001), an inverse relation between serum progesterone levels and percent cerebral edema was found. Male rats were given 4 mg/kg progesterone 1, 6, and 24 hours after injury, with progesterone levels being measured 2, 6, 24, and 48 hours after the initial progesterone injection. Peak progesterone levels (∼ 25 to 30 ng/mL) occurred between 0 and 6 hours after the initial injection.
In the present study, combined pre- and postischemic progesterone treatment was associated with a reduction in both cortical and total hemispheric infarct but had no effects on striatal infarct in young ovariectomized female rats. This is in agreement with our previous findings in reproductively senescent female rats in which progesterone reduced cortical injury (Alkayed et al., 2000). Our present data are also in agreement with studies of brain injury in young animals (Stein et al., 1999; Roof and Hall, 2000; Bramlett and Dietrich, 2001; Galani et al., 2001; Stein, 2001; Vink et al., 2001; Wright et al., 2001). Exogenous progesterone given after TBI reduces secondary neuronal loss and attenuates brain edema, independent of estrogen, (Roof et al., 1994, 1996; Asbury et al., 1998; Galani et al., 2001; Wright et al., 2001), by an antioxidant mechanism (Roof et al., 1997; Stein et al., 1999). Further, our results with MCAO in ovariectomized females agree with observations in male animals. In male rats, pretreatment with progesterone (2 mg/kg 1 hour before MCAO) reduced brain edema during the early stages of ischemia (Betz and Coester, 1990a,b). Progesterone administered before and/or after the onset of transient focal cerebral ischemia decreases injury and neurologic deficits in males (Jiang et al., 1996; Chen et al., 1999; Kumon et al., 2000). In acute global cerebral ischemia in ovariectomized cats treated with 10 mg kg−1 d−1 for 7 days before and after ischemia, progesterone also greatly reduced ischemic cell damage (Gonzalez-Vidal et al., 1998; Cervantes et al., 2002).
Our present results, however, contrast with our previous findings in ovariectomized rats in which chronic preischemic hormone exposure exacerbated striatal infarction volume after reversible MCAO (Murphy et al., 2000). One explanation for the divergence of our present study from our previous outcomes in young, ovariectomized female rats could be linked to the dose and duration of progesterone used. In rat, plasma progesterone ranges from basal levels of 2 to 18 ng/mL to approximately 120 to 130 ng/mL in pregnancy, with intermediate values of 40 to 90 ng/mL during late proestrus (Wiest, 1970; Butcher et al., 1974; Sutter-Dub et al., 1974; Nequin et al., 1979). The progesterone doses used in this study were intended to achieve intraischemic plasma progesterone levels comparable to peak levels observed during proestrus. Although variations in plasma progesterone reflected individual differences in intraperitoneal absorption and metabolism of the injected drug, ovariectomized animals treated only in the preischemic period with progesterone demonstrated mean ischemic plasma progesterone levels ranging from 29 to 77 ng/mL. These same groups showed levels comparable to the vehicle-treated animals by 22 hours of reperfusion, ranging from 7 to 12 ng/mL on average. The P5R group, treated in both the pre- and postischemic periods, demonstrated average ischemic and 22-hour reperfusion progesterone levels of 32 and 20 ng/mL, respectively, in contrast to vehicle-treated animals which had average progesterone levels less than or equal to 8 ng/mL at those times.
In our prior study with ovariectomized rats treated chronically with progesterone before MCAO (30 mg kg−1 d−1 intraperitoneally for 7 to 10 days), we observed an abrupt and absolute average decrease of 125 ng/mL in plasma progesterone from ischemic values to those levels observed at 22 hours of reperfusion (Murphy et al., 2000). We speculated that the increased striatal injury we observed (Murphy et al., 2000) could be due to GABA current modulation and alterations in GABAA receptor a4 subunit levels as a result of sharply declining plasma progesterone levels (Moran and Smith, 1998; Smith et al., 1998) after ischemia. The present study showed relatively smaller drops, ranging from 22 to 65 ng/mL on average, in progesterone levels in pre-MCAO-treated animals at 22-hour reperfusion from ischemic levels but demonstrated sustained physiologic plasma progesterone levels at 22-hour reperfusion in animals dosed both during preischemia and early reperfusion. Our earlier study in reproductively senescent female rats, which also confirmed cortical protection with progesterone (10 mg 7 to 14 days before MCAO), used long-term steroid delivery via a subcutaneous hormone implant (Alkayed et al., 2000) rather than a repeated-injection protocol. This method of hormone administration ensured sustained plasma progesterone levels during the reperfusion period rather than the severely declining levels we observed in our prior study examining chronic daily preischemic progesterone administration (Murphy et al., 2000). When physiologic plasma progesterone levels are sustained during the reperfusion period, progesterone appears to be neuroprotective in the ischemic female rat brain.
Our findings are not explained by differences in physiologic parameters between treatment groups. Although there is little evidence that progesterone is vasoactive in the cerebral circulation (Lu et al., 1996), this steroid has been shown in vitro to induce coronary relaxation (Jiang et al., 1992) and to inhibit arterial smooth muscle cell proliferation (Lee et al., 1997). Our results, however, suggest that the beneficial effect of progesterone does not appear to be related to preservation of blood flow during MCAO or CBF recovery during early reperfusion. Laser Doppler flow signal was reduced by a similar percentage of baseline values in all groups, suggesting that the relative ischemic insult was equivalent among all groups. After the occluding filament was removed, reperfusion, as evidenced by early recovery of LDF signal, occurred in all animals regardless of treatment group.
Because LDF measures only relative changes in cortical perfusion, rather than absolute CBF in cortical and other brain regions, we quantified end-ischemic and early reperfusion CBF in separate cohorts of animals treated with the protective dose. Using [14C]-IAP autoradiography, we found no differences within the ipsilateral, ischemic brain regions within the middle cerebral artery territory at end-ischemia and early reperfusion CBF in progesterone- versus vehicle-treated animals. Similarly, end-ischemic and early reperfusion tissue volumes at near-zero, low, and relatively preserved flow were not altered by exogenous progesterone administration. We did observe a modest blood flow difference at end-ischemia in contralateral, nonischemic lateral CP between progesterone- versus vehicle-treated animals. Lack of blood flow differences in other nonischemic regions studied, however, is consistent with previous work (Alkayed et al., 1998) in which intact and ovariectomized, anesthetized female rats were found to have equivalent baseline CBF. Although the initial rate of CBF recovery in early reperfusion is not different in the progesterone group, our results cannot exclude that this female sex steroid may exert vasodilatory action later in reperfusion, should secondary hypoperfusion occur (Kagstrom et al., 1983). We should also point out that the third and final dose of progesterone, which was given at 6-hour reperfusion in our outcome studies measuring infarction volume, was not administered in the CBF studies owing to the selected time points examined.
The results of our study suggest that progesterone is neuroprotective in ischemic brain injury. Other mechanisms remain to be considered, however. Progesterone reduces membrane lipid peroxidation in TBI models, most likely through a membrane-stabilizing type of antioxidant action (Roof et al., 1997; Stein et al., 1999; Roof and Hall, 2000). Progesterone has also been shown to have direct effects on neurotransmission by altering GABA conduction (Majewska, 1992), upregulating GABAA receptor number (Weiland and Orchinik, 1995), increasing GABAA agonist-binding affinity (Jussofie et al., 1995) and site number (Juptner et al., 1991), and by enhancing glutamic acid decarboxylase (GAD) mRNA (Weiland, 1992; Grattan et al., 1996). Progesterone also moderates excitatory amino acid (i.e., glutamate) responsiveness (Smith et al., 1987; Smith, 1991). Progesterone may therefore ameliorate ischemia-induced excitotoxic injury through potentiation of inhibitory GABA receptor activation and/or inhibition of excitatory amino acid receptors. There may also be a direct role for progesterone receptors in the response to vascular injury. In a model of carotid artery injury, ovariectomized progesterone-receptor knockout mice had more pronounced vascular injury regardless of whether or not the animals received exogenous progesterone, whereas ovariectomized wild-type mice with intact progesterone receptors (PRs) showed increased vascular injury response when administered progesterone (Karas et al., 2001). This study would suggest that PRs regulate the vascular injury response in a complex manner, either augmenting or attenuating the degree of injury. Marginal effects on striatal injury by progesterone may therefore be related to differences in regional expression of GABA, glutamate, and progesterone receptor subunits/subtypes or to different ischemic mechanisms in the cortex versus the striatum.
Potential cooperation in stroke mechanisms between progesterone and estrogen must also be considered. From an endocrinological standpoint, it is well known that the background steroid environment of the brain alters the ability of progesterone or estrogen to alter reproductive signaling and behavior (Smith, 1994; McCarthy, 1995). Our previous work and that of others indicate that estrogen does not require normal plasma progesterone levels to reduce stroke injury (Simpkins et al., 1997; Alkayed et al., 1998, 2000; Hurn et al., 1998; Toung et al., 1998; Rusa et al., 1999). Estrogen priming, however, is a requisite for many progesterone-mediated actions in normal brain (Tischkau and Ramirez, 1993; Smith, 1994; McCarthy, 1995). We also know that these two steroids interact at the receptor level, because estrogen, alone or in combination with progesterone, has been shown to upregulate progesterone receptor expression in brain (Moffatt et al., 1998). Therefore, it may be that progesterone is most efficacious in an ischemic brain environment that has been estrogen primed.
In conclusion, we have demonstrated that progesterone therapy both before and after ischemia rather than preischemia alone reduces ischemic cortical injury in ovariectomized female rats. The neuroprotective mechanism may not be through preservation of ischemic or initial reperfusion regional CBF, but further studies are warranted to examine the effects of this hormone on CBF at later stages of reperfusion. These data have potential implications for clinical progestin-containing hormone replacement regimens and for an understanding of the relative importance of female sex steroids in stroke. Additional studies are required to further define dose- and time-response relationships in experimental stroke for progesterone alone or in combination with estrogen.
Footnotes
Acknowledgements
We would like to acknowledge the technical assistance of Jinyan Bo in the cryosectioning of brains for regional CBF studies and of Judith P. Klaus, R.N., during sample collection for regional CBF studies.
