Abstract
Molecular neuroimaging is a powerful tool for studying ovarian steroid effects on the brain. We systematically reviewed non-clinical studies of female reproductive transitions, ovarian suppression, or estradiol (E2) and/or progesterone (P4) administration. Most studies used ≤3T [1H]MRS to study neurometabolites or PET imaging of glucose metabolism and serotoninergic activity. Results suggest ovarian steroids dynamically influence neurometabolic activity and serotonin neurotransmission. Elevated E2, whether during the late follicular phase or with postmenopausal administration, enhanced glucose-related metabolic activity and excitatory serotonin signaling, while low postmenopausal E2 may shift metabolism away from glucose for energy production. Rising P4, whether during the luteal phase or with postmenopausal administration, attenuated regional energy storage potential and glucose metabolism, while amplifying excitatory serotonin signaling. The perinatal transition was less studied, mostly with [1H]MRS, and showed non-significant or transient effects. Studies examining outcomes related to neuroprotection, neuroinflammation, and hormone receptor density were limited. We highlight the need for further molecular neuroimaging, including multimodal approaches, to systematically characterize ovarian steroid targets and their molecular context. Advances in MRS and PET offer opportunities to study ovarian steroid effects on neuroplasticity, mitochondrial function, neuroprotection, and neuroinflammation, and there is a need for continued robust prospective longitudinal and experimental studies.
Introduction
A decade after the National Institutes of Health mandate to include sex as a biological variable in study designs, women remain underrepresented in neuroscience and biomedical research. 1 Studies focusing specifically on women’s brain health, such as those of the menstrual cycle, pregnancy, and menopause, constitute approximately 0.5% of human neuroimaging research. 2 To close this gap, new research initiatives are aimed at advancing scientific understanding related to women’s brain health.1,2 One important avenue for understanding conditions more common in women, including major depressive disorder and Alzheimer’s disease, 3 is to investigate the effects of ovarian steroid hormones on brain architecture and activity. 2 Human magnetic resonance imaging (MRI) studies have recently demonstrated dramatic effects of female reproductive transitions on structural volumes and functional connectivity.4–7 These findings, alongside advances in molecular neuroimaging techniques,8,9 have fueled interest in the use of human molecular neuroimaging to study sex hormone effects on neurochemical processes and neuroreceptor activity.8,10
Molecular neuroimaging provides powerful tools for examining biological processes in the brain, offering insights that are critical for understanding how reproductive hormones like estradiol (E2) and progesterone (P4) modulate brain function. Insights from molecular neuroimaging into specific neurobiological processes are largely determined by the imaging technology and radioligands selected.8,11,12 Historically, most magnetic resonance spectroscopy (MRS) research has used [1H]MRS at ≤3T to measure concentrations of metabolites involved in diverse cellular functions, like n-acetylasparatate (NAA), choline-containing compounds (choline), creatine/phosphocreatine (creatine), glutamate/glutamine (glutamate), and GABA. 12 However, MRS techniques like [31P]MRS, high resolution [1H]MRS, or [2H]MRS with [2H]substrate infusion—less commonly used than 3T [1H]MRS—can provide insights into specific processes such as oxidative stress, energy utilization, or glucose/TCA cycle metabolic rates 9 to elucidate the effects of ovarian steroids on energy metabolism and metabolic stress. Similarly, positron emission tomography (PET) neuroimaging has been most widely used for assessing neurometabolic activity, monoaminergic signaling, and neuropathology accumulation.8,11 However, newer radioligands enable the study of other processes that may be influenced by ovarian steroids, including synaptic density, 13 mitochondrial function, 14 and neuroinflammation. 15
We conducted a systematic review of molecular neuroimaging studies of female reproductive transitions as well as related experimental studies of ovarian steroid withdrawal or administration of estradiol (E2) with or without progesterone (P4), focusing our review on non-clinical cohorts. The purpose of this review is to 1) provide a summary and integrative review of the literature to date on normative neuromolecular effects of ovarian steroids and reproductive transitions on the female brain; 2) characterize, when possible, correspondence between different neuromolecular outcomes; and 3) highlight future research directions, capitalizing on advances in molecular and multimodal neuroimaging.
Methods
Systematic review methods followed PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). 16 The search, conducted in PubMed, Embase, Scopus, and APA PsychInfo, combined subject headings and keywords for three main concepts: ovarian hormones, molecular neuroimaging, and neurochemistry (Supplement S1). The search excluded animal studies, case reports, and non-English language publications. The initial search was on 9/27/22 and a final search was conducted on 6/3/24. Covidence software (Veritas Health Innovation, Melbourne, Australia; www.covidence.org) facilitated screening, full-text review, and data extraction, with decisions made by consensus amongst two independent reviewers, with third party arbitration as needed. A total of 49 articles met the inclusion criteria (Figure 1).
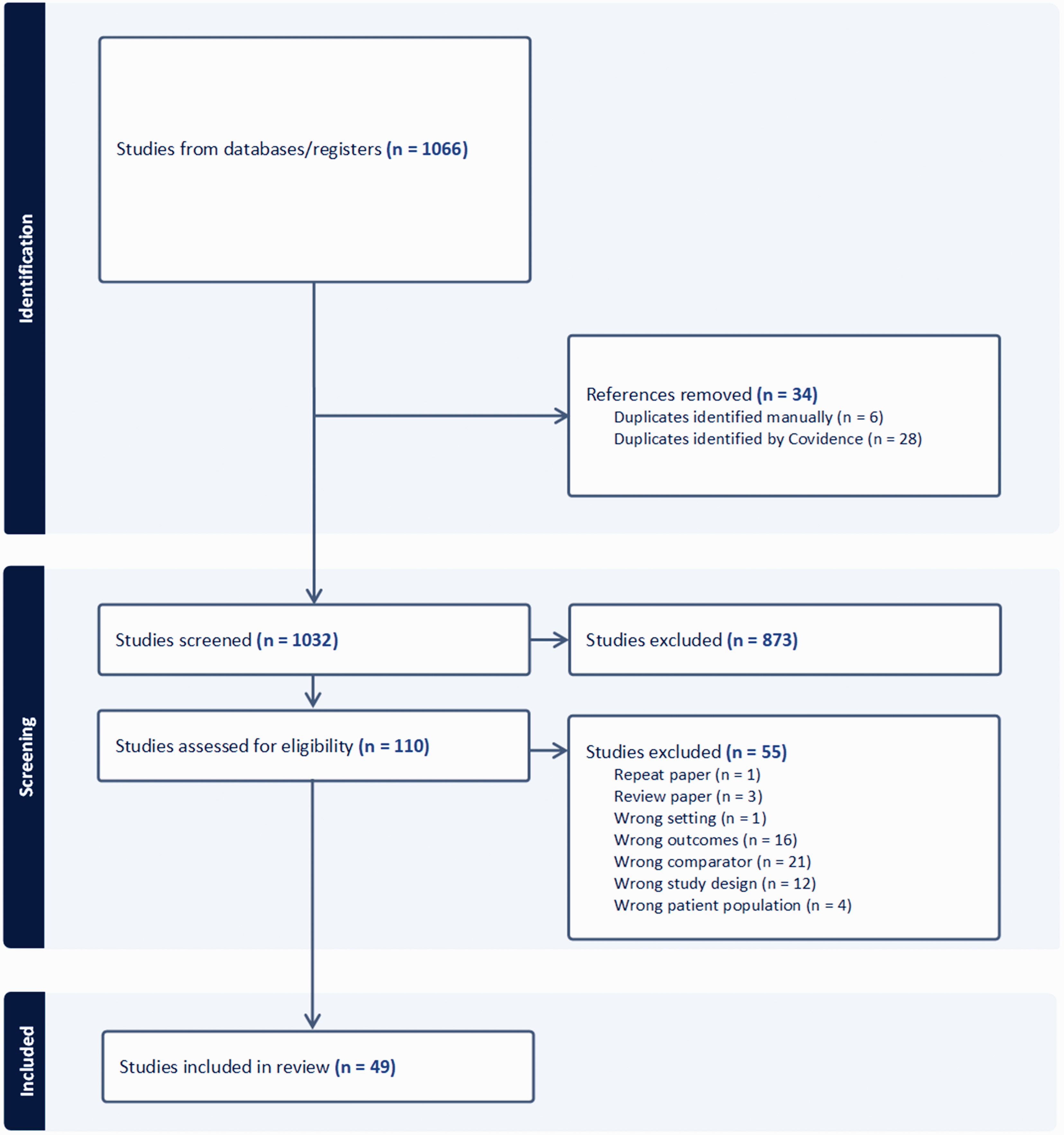
PRISMA flow chart.
Studies were included if they used 1) a placebo-controlled or open-label design to test the effects of ovarian steroid administration or withdrawal via gonadotropin-releasing hormone (GnRH) agonist or 2) a longitudinal or cross-sectional design to compare stages of reproductive transitions (puberty, menstrual cycle, perinatal transition, menopause transition). Accepted neuroimaging methodologies were MRS, PET, or single photon emission computed tomography (SPECT). Only non-clinical cohorts of biological females were included; studies focusing on clinical samples were excluded unless non-clinical comparison groups were separately analyzed. Non-clinical cohorts were defined as those without major psychiatric or neurological diagnoses or without major risk factors for such diagnoses (e.g., “at risk” for reproductive mood disorders or dementia).
Cross-sectional studies of menopausal hormone replacement or hormonal contraception were excluded due to their frequent prescription for clinical conditions, given the focus of this review on non-clinical cohorts. However, relevant experimental studies were included if conducted in non-clinical groups. Open-label or RCT studies of other ovarian steroids (non-E2 or P4) or related hormonal preparations (e.g., testosterone, 17 conjugated equine estrogens and medroxyprogestin acetate 18 ) were excluded, due to the focus on E2 and P4. Two [31P]MRS studies of cerebral magnesium are summarized in Supplement S2. Meta-analyses, systematic reviews, case studies, and studies with n < 5 per reproductive stage were excluded. Correlational studies were excluded, as they do not differentiate state and trait differences in hormone levels on the brain. We excluded studies of cerebral blood flow (i.e., 5O-H2O, SPECT – HMPAO, and ECD), as these primarily reflect perfusion rather than specific neurochemical processes. There were no restrictions on setting or publication date.
Relevant study design, sample characterization, reproductive staging, treatment details, neuroimaging methods, and results variables were extracted from each paper. The primary outcome was the difference or change in neuromolecular measures (metabolite concentrations, binding potential, volume of distribution, etc.). Quality metrics were extracted regarding study design, participant characterization, reproductive staging, neuroimaging methodology, statistical methods, bias assessment, and missing data. Key quality metrics incorporated into review tables were 1) study design (within-subjects [WS] vs. between-subjects [BS]) and 2) sample size. Three menstrual cycle studies were excluded for insufficient reporting for MRS 19 or reproductive staging methods to determine eligibility20,21; no other studies were excluded based on quality assessment, but relevant limitations are discussed in the Results section.
Results
Forty-nine molecular neuroimaging studies of reproductive transitions met inclusion criteria for review (Tables 1 to 3). These studies predominantly used either 1) PET imaging to estimate glucose metabolism and monoamine function or 2) ≤3T [1H]MRS to measure metabolite concentrations of creatine, glutamate, GABA, choline, and NAA. Results are organized based on reproductive transitions with the largest contrast in either E2, P4, or E2 + P4 levels to facilitate inferences about how changes in each ovarian steroid hormone (or their combination) modify molecular endpoints. Typical patterns of E2 and P4 changes during menstrual, perinatal, and perimenopausal transitions are shown in Figure 2.
Reproductive transitions characterized by marked changes in E2 levels.
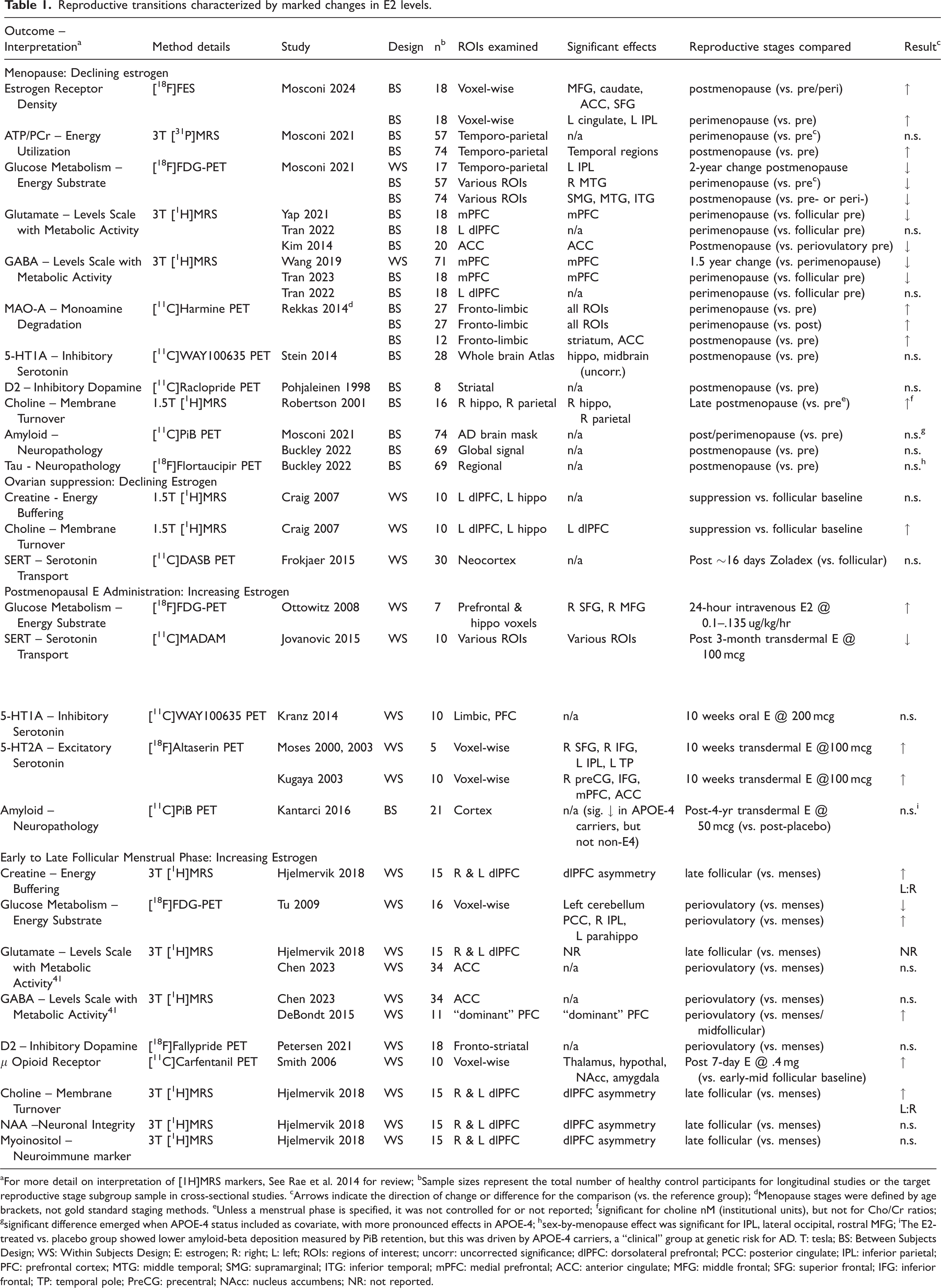
For more detail on interpretation of [1H]MRS markers, See Rae et al. 2014 for review; bSample sizes represent the total number of healthy control participants for longitudinal studies or the target reproductive stage subgroup sample in cross-sectional studies. cArrows indicate the direction of change or difference for the comparison (vs. the reference group); dMenopause stages were defined by age brackets, not gold standard staging methods. eUnless a menstrual phase is specified, it was not controlled for or not reported; fsignificant for choline nM (institutional units), but not for Cho/Cr ratios; gsignificant difference emerged when APOE-4 status included as covariate, with more pronounced effects in APOE-4; hsex-by-menopause effect was significant for IPL, lateral occipital, rostral MFG; iThe E2-treated vs. placebo group showed lower amyloid-beta deposition measured by PiB retention, but this was driven by APOE-4 carriers, a “clinical” group at genetic risk for AD. T: tesla; BS: Between Subjects Design; WS: Within Subjects Design; E: estrogen; R: right; L: left; ROIs: regions of interest; uncorr: uncorrected significance; dlPFC: dorsolateral prefrontal; PCC: posterior cingulate; IPL: inferior parietal; PFC: prefrontal cortex; MTG: middle temporal; SMG: supramarginal; ITG: inferior temporal; mPFC: medial prefrontal; ACC: anterior cingulate; MFG: middle frontal; SFG: superior frontal; IFG: inferior frontal; TP: temporal pole; PreCG: precentral; NAcc: nucleus accumbens; NR: not reported.
Reproductive transitions characterized by concurrent changes in Both E2 and P4 levels.
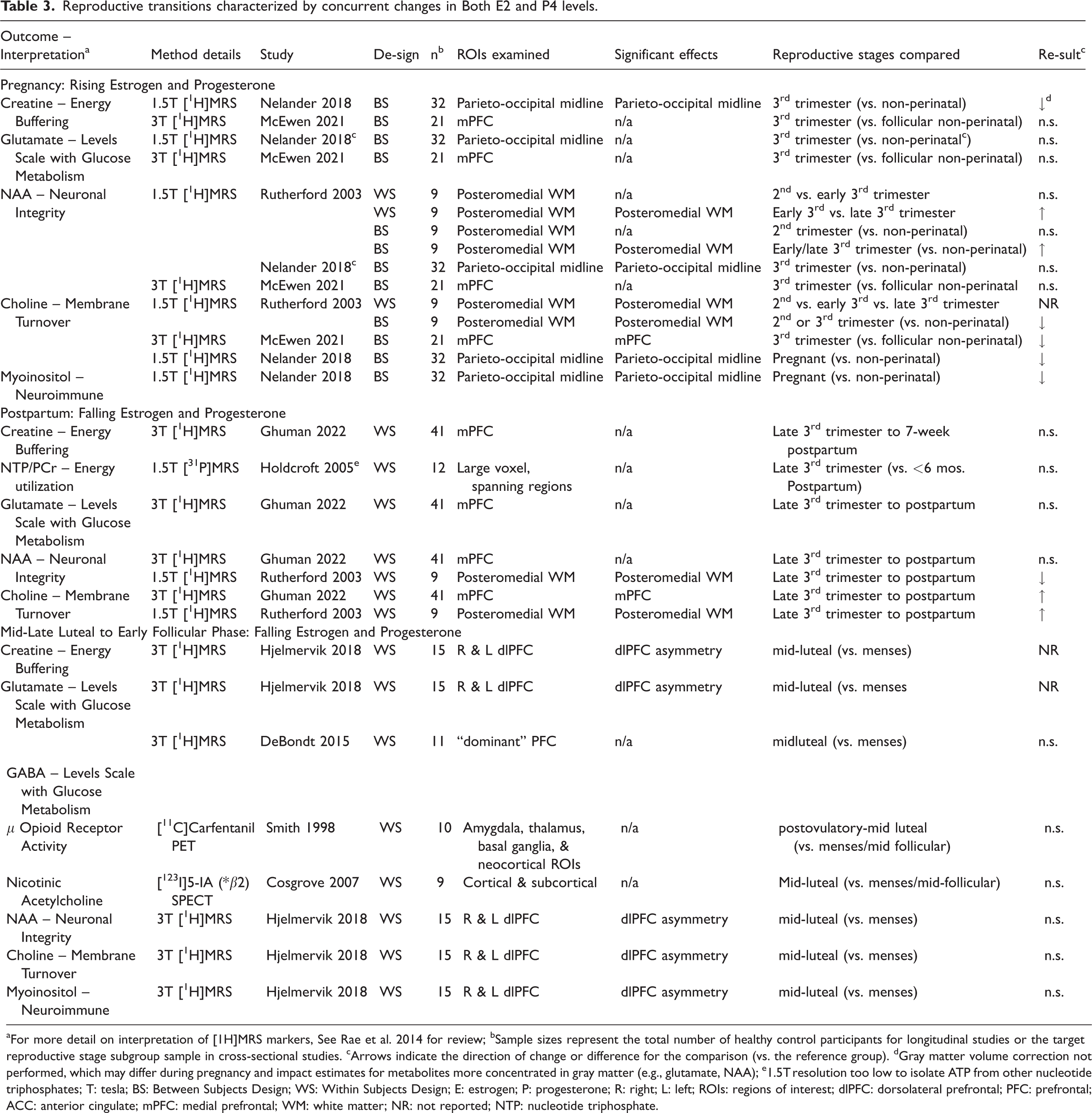
For more detail on interpretation of [1H]MRS markers, See Rae et al. 2014 for review; bSample sizes represent the total number of healthy control participants for longitudinal studies or the target reproductive stage subgroup sample in cross-sectional studies. cArrows indicate the direction of change or difference for the comparison (vs. the reference group). dGray matter volume correction not performed, which may differ during pregnancy and impact estimates for metabolites more concentrated in gray matter (e.g., glutamate, NAA); e1.5T resolution too low to isolate ATP from other nucleotide triphosphates; T: tesla; BS: Between Subjects Design; WS: Within Subjects Design; E: estrogen; P: progesterone; R: right; L: left; ROIs: regions of interest; dlPFC: dorsolateral prefrontal; PFC: prefrontal; ACC: anterior cingulate; mPFC: medial prefrontal; WM: white matter; NR: not reported; NTP: nucleotide triphosphate.
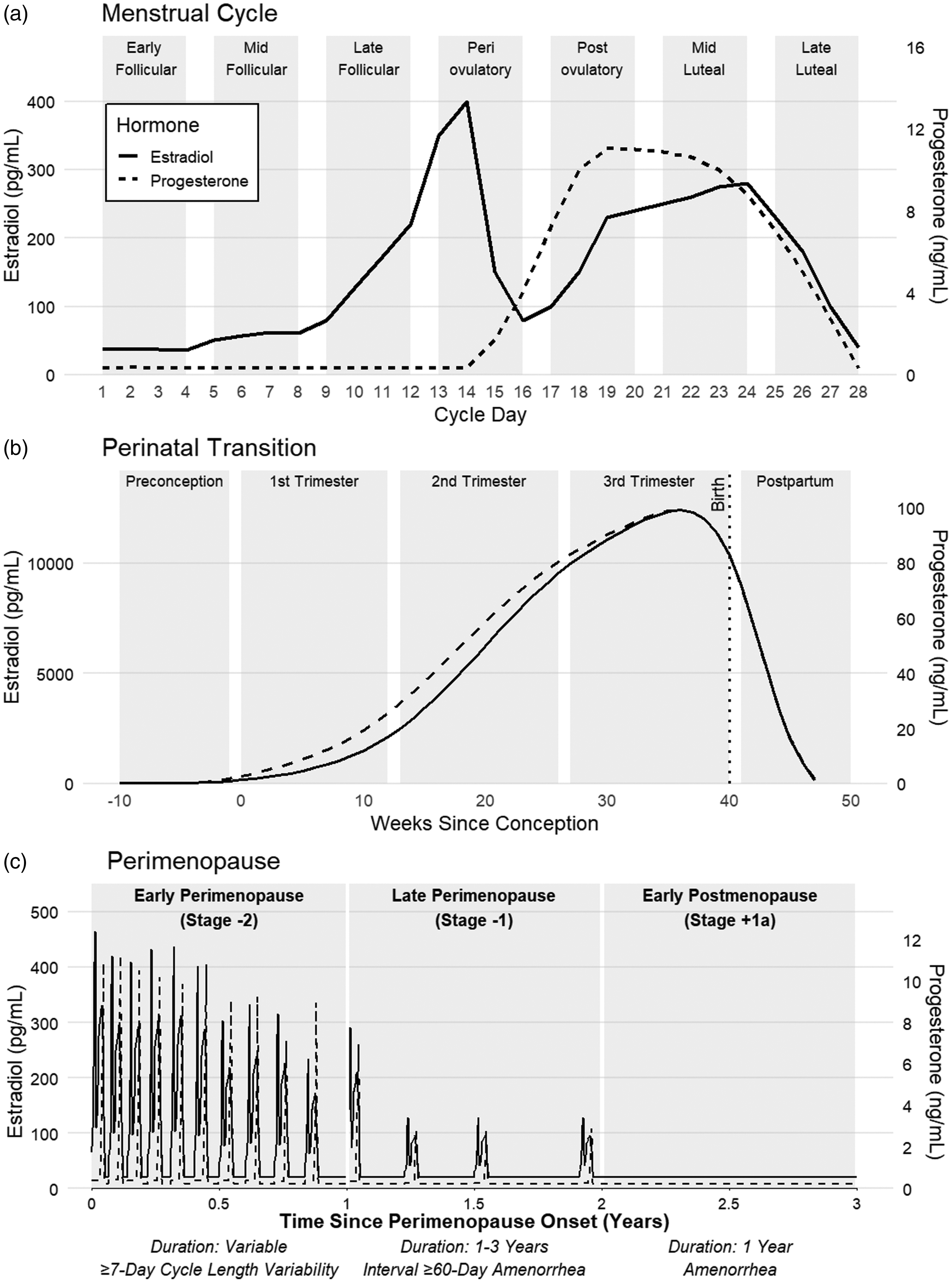
Illustration of estradiol (E2) and progesterone (P4) fluctuations across the menstrual cycle and the perinatal and menopause transition, utilizing simulated data. Panel a depicts the menstrual cycle, where E2 levels remain low in the early follicular phase and surge during the late follicular phase to induce ovulation. During the luteal phase, E2 and P4 rise and fall, precipitating menses and another menstrual cycle.5,23,56 Pregnancy (Panel b) is characterized by a gradual escalation in E2 and P4 levels, which decline precipitously at childbirth.23,121 For the menopause transition (Panel c), stages are defined by the Stages of Reproductive Aging Workshop – 10 (STRAW-10) criteria. 22 The early perimenopausal stage is marked by increased menstrual irregularity, specifically a ≥7-day difference in cycle length across consecutive cycles. The late perimenopausal stage begins after a ≥60-day amenorrhea interval and ends with the final menstrual period, lasting between 1–3 years. The early postmenopausal stage begins with 12 consecutive months of amenorrhea and represents the first several years of postmenopausal hormonal stabilization.22,23,122
Molecular neuroimaging of transitions with marked E2 change
Hormonal dynamics
The studies reviewed here examined various reproductive stages or hormone administration protocols relevant to the effects of E2 (Table 1). The following comparisons were considered:
Premenopause vs. Peri- or Post-Menopause (Figure 2, Panel c): During perimenopause, E2 levels can be erratic, but population mean E2 levels progressively decrease from pre- to peri- to post-menopause, eventually stabilizing at low levels during postmenopause,
22
typically below 20 pg/mL.
23
Comparisons of perimenopause to premenopause capture the effects of rapidly fluctuating or declining E2, while comparisons of postmenopause to premenopause reflect the impact of chronically low E2. Pre- to Post-Ovarian Suppression: Ovarian suppression, typically induced using gonadotropin-releasing hormone agonists (GnRHa), reduces E2 to postmenopausal levels after an initial stimulation phase.
24
The suppression phase is often compared with the early-to-mid follicular phase of the menstrual cycle, where E2 levels are relatively higher, around 40–100 pg/ml.
23
Depending on the duration of GnHRa administration, this contrast may reflect more acute (as in Frokjaer et al., 2015)
25
or chronic effects of E2 suppression (as in Craig et al., 2007).
26
Postmenopausal E2 Administration versus Placebo or Baseline: Administering E2 to postmenopausal women elevates serum E2 levels from low postmenopausal concentrations to levels comparable to the early follicular phase of the menstrual cycle.
23
Most studies assessed the chronic effects of ∼10–12 weeks of postmenopausal E2 treatment versus baseline or placebo.17,27–29 Late Follicular vs. Early Follicular Menstrual Phase (Figure 2, Panel b): During the early follicular phase (menses), E2 levels are at their lowest, usually around 40 pg/mL,
23
gradually increasing to a peak in the periovulatory phase, reaching concentrations around 200–400 pg/mL.
23
Thus, comparisons of the early follicular phase to the late follicular phase can be used to infer the acute effects of rising E2.
Notably, other hormones and biological processes apart from ovarian steroid hormones change during the menstrual cycle, the transition to menopause, and ovarian suppression. While their impact on neural endpoints is less well studied, factors like gonadotropins 23 may also play a role. Given that more is known about the effects of E2 and P4 on neurobiology than any other hormone or neurosteroid, this review focuses on ovarian steroid hormones.
Overview of studies
A total of 26 studies examined transitions marked by rising or falling E2 levels, with sample sizes ranging from 5 to 69 participants per reproductive stage comparison. Studies on the menopause transition typically used a cross-sectional design, while studies on the menstrual cycle, ovarian suppression, and postmenopausal E2 administration employed repeated assessments, usually without a control or placebo comparison group (with exceptions25,27,30–32) The most frequently used molecular neuroimaging outcomes included ≤3T [1H]MRS neurometabolite levels, [18F]FDG glucose metabolism, and PET studies of serotonin signaling.
Study findings
First, we summarize studies examining the effects of low or decreasing E2. Across two studies, postmenopause—a stage marked by chronically low E2 levels 33 —was associated with increased regional estrogen receptor density (n = 18 postmenopausal) 34 as well as decreased temporal glucose metabolism and increased temporal ATP utilization (n = 57 postmenopausal), 10 relative to premenopause. Compared with premenopause, perimenopause—a stage marked by highly variable but overall decreasing E2 levels22,23—was associated with lower medial prefrontal levels of glutamate (n = 18 perimenopausal) 35 and GABA (n = 18 36 ; n = 71 perimenopausal 37 ) metabolites that tend to scale with glucose metabolism. 38 The postmenopausal stage (n = 16) 39 and more chronic ovarian suppression (n = 10) 26 were also associated with regionally increased choline, a marker of membrane turnover. 40 Also elevated in peri- (n = 17) and postmenopausal-aged women (n = 12) was monoamine oxidase-A (MAO-A), 41 which degrades serotonin and other monoamines.
Other studies examined reproductive transitions marked by rising E2. In one study of early vs. late follicular menstrual phase (n = 16) 42 and one study of acute postmenopausal E2 infusion (n = 7), 43 increased regional glucose metabolism was found. The most prominent effects appeared in the default mode network during the menstrual follicular phase 42 and in the right prefrontal cortex following postmenopausal E2 infusion. 43 Certain neurometabolites were also regionally increased from the early to late menstrual follicular/periovulatory phase, including left prefrontal GABA (n = 11) 31 as well as creatine and choline (n = 15). 32 Studies of more chronic postmenopausal E2 administration (∼10–12 weeks) suggest a facilitative effect on synaptic serotonin, indexed by lower serotonin transporter (n = 10; SERT), 17 and increased excitatory 5-HT2A (n = 5 29 ; n = 10 28 ) particularly in frontal, limbic, and temporo-parietal regions. E2 administration at 400 mcg relative to early-mid follicular baseline in reproductive-aged women resulted in upregulation of limbic μ opioid receptor binding (n = 10). 44
In several studies, molecular endpoints did not significantly differ when comparing reproductive stages marked by differing E2 levels. For example, studies of menopause transition stages or ovarian suppression found non-significant differences in dorsolateral prefrontal (dlPFC) creatine (n = 10) 26 as well as glutamate and GABA levels (n = 18) 45 ; regional SERT (n = 30) 25 ; inhibitory serotonin (n = 28; 5-HT1A) 46 and dopamine (n = 8; D2) receptors 47 ; and markers of neuropathology (n = 69 for tau 48 ; n = 74 for amyloid 10 ) Furthermore, anterior cingulate GABA and glutamate (n = 34) 49 ; dlPFC immune markers (n = 15; myoinositol) 32 ; inhibitory serotonin (n = 10; 5-HT1A) 27 and dopamine (n = 16; D2) 50 receptor binding; and neuropathology markers (n = 21; amyloid) 30 were not significantly affected by either rising follicular E2 levels or postmenopausal E2 administration. Non-significant effects may result from several factors. For instance, in a study comparing pre- (n = 20) vs. perimenopausal (n = 18) dlPFC GABA and glutamate, there were not significant group differences in E2 levels, 45 potentially limiting power to detect E2-related effects. Similarly, while medial prefrontal GABA decreased across the menopause transition in one study (n = 71), 37 no changes were observed in a study comparing the early to late follicular menstrual phase (n = 34), 49 suggesting medial prefrontal effects may be more prominent once E2 declines beyond a certain threshold or chronically. Finally, while E2 administration reduced SERT in one study (n = 10), 17 acute ovarian suppression did not significantly impact SERT (n = 30). 25 However, this discrepancy may occur due to the relatively short suppression protocol, 25 while changes in SERT expression may require a longer timescale. 51 Finally, it’s important to note that E2 effects on neuropathology markers may be more pronounced in subgroups with genetic risk for Alzheimer’s disease, such as APOE-4 carriers. 30
Summary
Overall, findings suggest that very low E2 levels during postmenopause increase estrogen receptor density 34 —potentially to compensate for low circulating estrogen levels—and reduce regional glucose-related metabolic activity.10,35–37,52 There was also evidence suggesting increased ATP utilization ([31P]MRS ATP/phosphocreatine ratios), 10 phospholipid membrane turnover ([1H]MRS choline),26,39 and serotonin metabolism (MAO-A PET), 41 which may suggest an increase in metabolic stress 53 and lipid metabolism. 54 In contrast, reproductive transitions marked by rising E2 levels seem to have the opposite effect, with evidence of increasing regional glucose metabolism,42,43 regional neurometabolite levels,31,32 synaptic serotonin (SERT-PET) 17 and excitatory 5-HT2A signaling.28,29 Several molecular markers showed null findings of E2 transitions, potentially due to small sample sizes (n = 10–18) or low power in the context of multiple regional comparisons, 46 including myoinositol levels 32 and inhibitory monoamine receptor binding (5-HT1A,27,46 D2 50 ). Others showed variable results across studies, potentially reflecting distinct reproductive dynamics or methodological differences (e.g., SERT,17,25 creatine,26,32 medial prefrontal GABA37,49) Effects of menopause or E2 treatment on amyloid10,48 and tau 48 were non-significant, even in larger samples, though differences emerged in at-risk groups like APOE-E4 carriers (e.g., amyloid 10 ).
Molecular neuroimaging of transitions with marked P4 change
Hormonal dynamics
The studies reviewed in this section focused on the effects of rising P4 across the menstrual cycle and with postmenopausal hormonal administration (Table 2). It is important to note that P4 does not act in isolation in normal physiology—E2 facilitates P4 signaling, 55 so the impact of E2 + P4 is typically assessed relative to E2 alone, as P4 is rarely administered in isolation. The following comparisons were considered:
Reproductive Transitions with Marked Changes in P4 Levels.
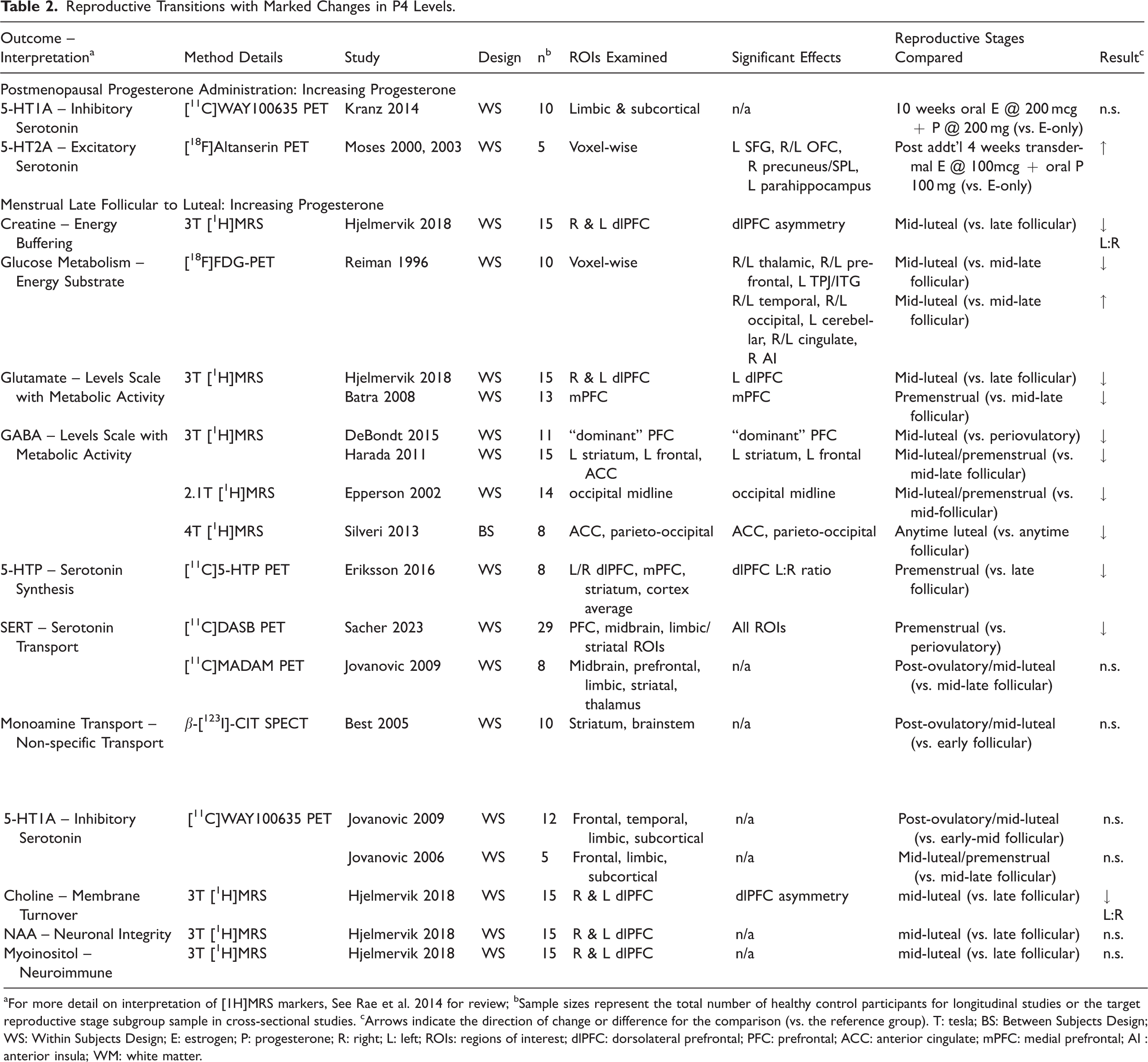
For more detail on interpretation of [1H]MRS markers, See Rae et al. 2014 for review; bSample sizes represent the total number of healthy control participants for longitudinal studies or the target reproductive stage subgroup sample in cross-sectional studies. cArrows indicate the direction of change or difference for the comparison (vs. the reference group). T: tesla; BS: Between Subjects Design; WS: Within Subjects Design; E: estrogen; P: progesterone; R: right; L: left; ROIs: regions of interest; dlPFC: dorsolateral prefrontal; PFC: prefrontal; ACC: anterior cingulate; mPFC: medial prefrontal; AI : anterior insula; WM: white matter.
Postmenopausal E2 + P4 Administration vs. E2 Alone: Administering P4 alongside E2 to postmenopausal women raises serum P4 levels from baseline low concentrations to those more typical of the luteal phase. 23 By comparing this to E2-only treatment, studies can isolate the additive effect of elevated P4 on molecular endpoints. In the two reviewed studies examining P4 administration, daily oral doses were administered for ∼2–10 weeks.27,29
Late Follicular/Periovulatory vs. Mid-Late Luteal Menstrual Phase (Figure 2, Panel a): In the menstrual cycle, the luteal phase is characterized by a substantial rise in P4 levels (alongside E2) following ovulation. In contrast, the late follicular phase features low P4 levels but high E2 levels. 56 This comparison allows for inference on how P4 more acutely modifies E2’s effects, either amplifying or attenuating them, relative to E2 alone.
While studies of postmenopausal E2 + P4 vs. E2 administration controlled for E2 levels through standardized dosing, E2 levels differ in magnitude between the late follicular and luteal phase, but this was not controlled in the reviewed studies. However, both types of studies yielded similar results for 5-HT1A,27,57,58 the only outcome that was commonly assessed. Patterns may therefore be similar across menstrual cycle and hormone administration studies, and we have included both study types in this synthesis.
Overview of studies
A total of 14 studies were included, two of P4 administration and 12 of the menstrual cycle, with sample sizes ranging from 5 to 29 participants. All but one study 59 used repeated measurement to evaluate reproductive stage or E2 and P4 administration effects, including one randomized, placebo-controlled trial (RCT). 27 The most frequently used molecular neuroimaging modality was ≤3T [1H]MRS, followed by PET studies of monoaminergic signaling, while only one study used [18F]FDG to assess glucose metabolism across the menstrual cycle. 60
Study findings
Herein, we review studies comparing reproductive transitions marked by rising P4. In a singular study (n = 10), 60 the mid-late follicular to mid-late luteal menstrual transition was associated with regional decreases in glucose metabolism, particularly affecting left frontoparietal regions, but also regional increases, especially in somatosensory and salience processing regions. Notably, this study also included women in the mid-follicular phase, where E2 levels are lower (Figure 2; Panel a), so it is uncertain if effects are due to relatively elevated luteal E2, P4, or their combination. In comparisons of the late follicular/periovulatory vs. mid-late luteal phase, glutamate and GABA showed regional reductions, including in the medial and left prefrontal glutamate (n = 13 61 ; n = 15 32 ) and left frontal and regional GABA (n = 8 59 ; n = 11 31 ; n = 14 62 ; n = 15 63 ) There was also a luteal phase attenuation in leftward dlPFC asymmetry for an energy buffering metabolite (creatine; n = 15), 32 with similar patterns found for glutamate and choline. 32 Parallel patterns of attenuated leftward dlPFC asymmetry were found for serotonin synthesis-related activity (n = 8), 64 accompanied by widespread reductions in luteal SERT binding (n = 29), 65 though significant effects were not observed in a smaller sample study (n = 8) 57 and a study using a non-selective monoamine transporter PET tracer (n = 10). 66 Furthermore, a study examining the additive effect of daily oral P4 to postmenopausal transdermal E2 found more widespread increases in 5-HT2A (n = 5). 29 Notably, transitions marked by rising P4 did not appear to impact inhibitory serotonin signaling (5-HT1A; n = 5 58 ; n = 10 27 ; n = 12 57 ) as well as a neuroimmune marker (myoinositol) and a marker of neuronal integrity (NAA; n = 15). 32
Summary
Overall, findings suggest the late follicular to mid-late luteal transition, marked by rising P4, attenuates regional glucose metabolism, particularly affecting left prefrontal cortex 60 as well as leftward prefrontal concentrations of energy buffering metabolites (creatine) 32 and left frontal neurometabolite levels for glutamate and GABA.31,32,63 Rising menstrual P4 may also attenuate leftward prefrontal serotonin synthesis. 64 Parallel patterns of leftward prefrontal attenuation of both creatine 32 and serotonin synthesis 64 may suggest that the latter plays a role in supporting mitochondrial function across reproductive transitions (e.g., via serotonin’s intracellular functions). 53 Studies also suggest that P4 enhances postsynaptic serotonin signaling, demonstrated by more widespread increases in luteal phase synaptic serotonin (evidenced by reduced SERT 65 ) and greater excitatory 5-HT2A signaling following the addition of P4 to postmenopausal E2 administration. 29 Several studies reported non-significant effects of P4 transitions on serotonergic activity and [1H]MRS metabolites—possibly due to small samples (n = 5–15)—including SERT57,66/5-HT1A binding27,57,58 and myoinositol/NAA levels. 32
Molecular neuroimaging of transitions marked by both E2 and P4 change
Hormonal dynamics
In some reproductive stage comparisons, changes in E2 and P4 could not be disentangled, necessitating a separate examination of transitions where both hormones rose or fell together. The following comparisons were considered:
Late-term Pregnancy vs. Early Pregnancy or Non-Perinatal Women (Figure 2, Panel b): Pregnancy is characterized by a sustained and dramatic increase in both E2 and P4, with levels far exceeding those observed in the menstrual cycle. This transition reflects the physiological effects of prolonged exposure to high E2 and P4 concentrations (and their neuroactive metabolites, like allopregnanolone
67
) Postpartum vs. Late-Term Pregnancy (Figure 2, Panel b): The postpartum period is marked by a rapid decline in both E2 and P4 following delivery.
23
When compared to late-term pregnancy, this transition captures either the acute or prolonged effects of a very large-magnitude decline in E2 and P4. Mid-Late Luteal Phase vs. Early Follicular Phase (Menses; Figure 2, Panel a): The luteal phase of the menstrual cycle is defined by relatively elevated E2 and P4 levels,
56
which decline sharply at the onset of menses, marking the transition to the early follicular phase (menses).
56
Like the postpartum transition, this comparison captures the acute effects of E2 and P4 withdrawal, but with a smaller magnitude of decline.
Notably, the perinatal transition is marked by concurrent hormonal changes and stressors that may impact neuromolecular endpoints (e.g., fetal development, fragmented sleep, stress, etc.), 23 which may be considered in the interpretation of results.
Overview of studies
A total of 14 studies were included, with sample sizes ranging from 9 to 41 participants per reproductive stage group. Most [1H]MRS studies used repeated measurement, with some exceptions,68,69 to characterize the perinatal transition and the transition from luteal phase to menses. A handful of PET studies also used cross-sectional designs to interrogate postpartum monoaminergic transmission relative to non-perinatal women.70–72
Study findings
Pregnancy, whether examined longitudinally (n = 9) 73 or in cross-sectional comparisons to non-perinatal women (n = 21 69 ; n = 32 68 ) consistently reduced choline, a marker of membrane turnover. 40 Furthermore, one longitudinal study of early vs. late pregnancy reported elevated white matter NAA, which may indicate re-myelination,74–76 specific to the late third trimester when E2 and P4 reach their peak (n = 9). 73 In converse, the postpartum stage, relative to late-term pregnancy, was associated with rising regional choline (n = 9 73 ; n = 41 77 ) and declining white matter NAA (n = 9). 73 Other regional metabolites were not affected in longitudinal studies across pregnancy or from late-term pregnancy to postpartum, including nucleotide trisphosphate ratios (n = 12) 78 and creatine, glutamate, or gray matter NAA (n = 41). 77 Similarly, a cross-sectional study of pregnant vs. non-perinatal women found no significant differences in glutamate, creatine, and NAA (n = 21) 69 ; however, another cross-sectional study found a significant pregnancy-associated reduction in creatine and myoinositol, though correction for pregnancy-related differences in gray matter volume was not applied (n = 32). 68 Finally, a [1H]MRS study (n = 15) 32 found no differences in dlPFC NAA, myoinositol, or choline when comparing the late luteal to menstrual phase; however, dlPFC hemispheric asymmetry was found in the luteal (rightward) but not menstrual phase for glutamate and in the menstrual (leftward) but not luteal phase for creatine, but phase comparisons were not directly assessed. Furthermore, two PET studies of neurotransmitter receptors (nicotinic acetylcholine, n = 9 79 ; μ opioid, n = 10 80 ) did not show significant differences when comparing the luteal to early follicular (menses) transition, wherein E2 and P4 levels decline, though to a lesser magnitude than the late-term pregnancy to postpartum transition.
Four studies compared postpartum to non-perinatal women (not included in Tables 1 to 3), either in the follicular phase or various menstrual stages. However, E2 and P4 levels were not consistently different between these stages, so these comparisons may reflect the effects of the postpartum decline in E2 and P4, or other postpartum hormonal and environmental variables. Among these studies, one [1H]MRS study found lower medial occipital GABA levels in <3-month postpartum women (n = 14), 62 who showed lower E2 levels, though P4 levels did not differ significantly. One PET study observed higher MAO-A density in very early postpartum women (<7 days; n = 15), 70 when E2 and P4 levels have fallen dramatically compared to pregnancy but are still relatively high compared to non-perinatal women. However, no differences in MAO-A were found in later postpartum (<18 months; n = 15) 71 compared to non-perinatal women, where E2 and P4 levels did not differ. Finally, another study found lower binding at the D2 dopamine receptor in postpartum women (<3 months; n = 13), 72 with no differences in E2/P4 levels between groups. These findings suggest that the early postpartum period may be characterized by a reduction in GABA levels and monoaminergic signaling, though these changes may stem from the sharp decline in E2 and P4 or other postpartum factors.
Summary
Overall, results from [1H]MRS studies suggest rising E2 and P4 during pregnancy may reduce membrane turnover (indexed by choline levels),68,73 perhaps due to elevated inhibitory tone. 81 Furthermore, high late-third trimester E2 and P4 levels may lead to elevated white matter NAA, 73 potentially reflecting its role in oligodendrocyte-driven myelin re-synthesis, a process observed in late-term pregnancy.74–76 However, rising E2 and P4 associated with pregnancy or falling E2 and P4 associated with postpartum or the premenstrual luteal to early follicular phase (menses) did not significantly impact levels of most other neurometabolites, including nucleotide triphosphates, 78 glutamate,68,69,77 GABA, 31 and gray matter NAA. 32 ,,68,69,77 and findings were inconsistent for creatine32,68,69,77 and myoinositol.32,68 Furthermore, GABA metabolite levels 82 and monoaminergic signaling70,72 may be lower during early postpartum when compared to non-perinatal women. Few studies used PET/SPECT to compare stages marked by elevated vs. low E2 and P4,79,80 except for two small-sample studies (n = 9–10) of nicotinic acetylcholine 79 or μ opioid receptors, 80 limiting conclusions about how these hormonal dynamics affect metabolic activity or neurotransmission.
Discussion
Molecular neuroimaging has begun to characterize the dynamic regional impact of ovarian steroids on neurometabolic activity and neurotransmission. In our review of non-clinical studies, transitions marked by elevated E2—whether rising during the follicular phase or with postmenopausal E2 administration—generally enhanced glucose metabolic activity (and related neurometabolite levels), particularly in default mode 42 and prefrontal regions. 43 In contrast, very low E2 (e.g., postmenopause) may lead to lower glucose metabolism 10 but higher levels of ATP utilization 10 and a marker of phospholipid membrane turnover (choline),26,39 potentially indicating a shift toward lipid metabolism for energy. 54 Higher E2 also enhanced serotoninergic signaling, as indicated by reduced SERT binding 17 and increased excitatory 5-HT2A receptor activity.28,29 Transitions marked by rising P4, particularly the luteal menstrual phase, were associated with reduced left fronto-parietal glucose metabolic activity 60 and attenuated leftward prefrontal asymmetry of an energy buffering metabolite (creatine), 32 suggesting a role in modulating regional energy storage and metabolism. P4 may also amplify the serotonin-enhancing effects of E2 on SERT 65 and 5-HT2A. 29 The perinatal transition showed more subtle or transient effects, assessed mostly by [1H]MRS—pregnancy did not significantly impact creatine or glutamate levels,69,77 but choline was reduced, and white matter NAA was increased late-term. 73 Findings on neuroprotection and neuroimmune function were sparse, with no clear impact of reproductive transitions on a marker of neuronal integrity (gray matter NAA)26,32,68,69,77 or a neuroimmune marker (myoinositol). 32 In the following sections, we synthesize the reviewed findings, highlighting converging themes related to ovarian steroid effects on metabolic activity and serotonin signaling, and suggest future directions in molecular and multimodal neuroimaging.
Characterizing reproductive transition effects on neurometabolism
Menopause, and likely other reproductive transitions, may alter the link between glucose metabolism and energy utilization in a regionally dependent manner. For instance, menopause was associated with lower temporal glucose metabolism but higher ATP utilization, 10 consistent with preclinical evidence showing E2-depletion-mediated decoupling of glucose metabolism from energy utilization.83,84 It remains uncertain if such metabolic decoupling occurs across other reproductive transitions, like the menstrual cycle or pregnancy. Methods used in reviewed studies, like [18F]FDG and [1H]MRS, offer valuable insights but lack the resolution to distinguish specific metabolic processes. For example, [1H]MRS creatine can only estimate potential for energy storage,12,40 and [18F]FDG measures cellular glucose metabolism, irrespective of its endpoint in energy production or other processes. 9 [31P]MRS at 3T, used in one menopause study, 10 can estimate ATP/phosphocreatine ratios, providing better insight into energy utilization, alongside multimodal investigation of energy substrate metabolism (e.g., [18F]FDG). Other methods, like [2H]MRS with substrate infusion 9 and the PET tracer [18F]BCPP-EF, 14 were not utilized in the reviewed studies, but could further enhance our understanding of changes in cerebral metabolic rates, mitochondrial efficiency, and alternate substrate utilization across reproductive transitions.
Reduced glucose metabolism and elevated ATP demand, akin to patterns observed in postmenopause, 10 may lower mitochondrial antioxidant capacity, 83 prompting alternative antioxidants, like serotonin, to be shunted intracellularly for antioxidation. 53 Supporting this hypothesis, MAO-A, which degrades serotonin that has been used for antioxidation, 53 was elevated in postmenopausal women. 41 Similarly, the reviewed studies showed parallel rightward dlPFC increases during the menstrual luteal phase in creatine levels, 32 a marker of energy buffering capacity, and intracellular serotonin synthesis, 64 potentially for antioxidant defense. One excluded study (no reproductive staging) also found that higher dlPFC glutathione levels were associated with lower E2 levels in a mid-to-older female sample, 85 suggesting reproductive transitions marked by E2 reduction, such as menopause, warrant more detailed investigation of oxidative stress. Future multimodal research will be useful for clarifying how reproductive hormones impact mitochondrial efficiency and antioxidation, such as studies using [18F]BCPP-EF PET 14 and glutathione [1H]MRS.
Impact of ovarian steroids on neurotransmission
Molecular imaging studies of neurotransmission have focused primarily on serotonin, suggesting that E2 reduces SERT binding 17 and upregulates excitatory 5-HT2A,28,29 with more widespread effects when E2 is combined with P4.28,29,65 In comparable studies of 5-HT1A, no effects survived statistical correction,27,57,58 suggesting receptor-specific effects on the serotonin system. Other neurotransmitter systems also showed minimal effects,50,79,80,86 except for a positive effect of relatively high-dose E2 administration on μ opioid receptor binding. 44 However, non-serotonergic systems were less robustly and systematically assessed, and thus, effects of ovarian steroids on these systems cannot be ruled out. Recent preclinical evidence suggests serotonin, acting through 5-HT2A receptors, regulates mitochondrial biogenesis and efficiency, thereby supporting neuronal energy regulation and neuroprotection against oxidative stress. 87 While selective modulation of SERT and 5-HT2A would be consistent with the role of E2 and P4 in modulating mitochondrial function55,88 (see Discussion 4.2), robust and systematic characterization of ovarian steroid effects on non-serotonergic receptors is warranted.
Neuromolecular changes during pregnancy and postpartum
Studying neuromolecular changes in pregnancy is challenging due to ethical considerations and PET imaging constraints. Most reviewed studies relied on MRS, though it can be limited in its neurophysiological specificity. 40 Findings suggest pregnancy is associated with regionally stable energy metabolites68,69,78; stable glutamate 89 ; decreasing choline69,73; and increasing NAA in white matter. 73 Though speculative, the latter findings may be consistent with evidence suggesting late-term pregnancy myelin remodeling. 90 Future research combining MRS and MRI could clarify the relationship between neurochemical changes and structural/functional brain alterations, including white matter microstructural integrity. Furthermore, the stability of neuroenergetic metabolites during pregnancy is surprising, given the magnitude of elevation in E2 and P4. Approaches with higher spectral resolution and sensitivity are warranted, such as ≥3T [ 31 P]MRS or MRS studies of metabolic rates. 9 Additionally, PET imaging may be used in experimental models of pregnancy 91 to study other changes related to ovarian steroid neuromodulation, such as neuroprotection, neurotransmission, and neuroimmune activity.
Methodological considerations
Study design
While our review grouped findings based on reproductive stage comparisons with the most significant changes in E2, P4, or E2 + P4, experimental studies are the gold standard for drawing conclusions about the specific effects of these hormones. RCTs are critical for isolating hormonal effects on neuromolecular endpoints by controlling for between-subject confounds and placebo effects. In this systematic review, parallel-group RCTs were used in only two reviewed studies, one of ovarian suppression 25 and one of postmenopausal E2 or E2 + P4 vs. placebo. 27 While no crossover RCTs were used, this design offers additional advantages by allowing each participant to serve as their own control and enhancing the ability to directly compare hormonal interventions—such as E2 alone vs. E2 + P4. An additional benefit of experimental studies is that they can model hormone transitions, such as pregnancy,91–93 which cannot be studied naturalistically with PET due to radiation exposure.
Prospective longitudinal studies across reproductive transitions, especially with robust staging methodology 56 and denser sampling protocols,56,94 can complement experimental studies, which may not fully recapitulate natural transitions. However, only a handful of longitudinal studies characterized trajectories at three or more timepoints32,62,77 and utilized a “control” group (e.g., biological males,32,63 hormonal contraceptive users 31 ) to account for measurement reliability and off-target changes (e.g., changes in age across the menopause transition). Controlled experimental and prospective longitudinal studies that include at least three timepoints and appropriate control groups are needed robustly characterize the effects of ovarian steroids on neuromolecular function.
Hormone measurement, metabolism, and signaling
Several studies measured changes in peripheral concentrations of E2 or P4 and relationships to neuromolecular change. However, few found significant associations, except in postmenopausal hormone administration studies finding correlations between changes in E2 and serotonin signaling/glucose metabolism.17,28,43 Notably, few studies measured peripheral concentrations of neurosteroid metabolites (e.g., allopregnanolone), with exceptions,62,82 despite evidence of their significant neuromodulatory effects.95,96 Ovarian steroid variability, beyond absolute levels, may also influence neurochemical processes. For instance, in one study, MAO-A levels were found to be highest during perimenopause, a time of erratic E2 fluctuations, 23 though they were also elevated during postmenopause, but to a lesser extent. 41 Daily urine sampling allows for capturing hormonal variability and cumulative exposure, providing additional insight into drivers of neurochemical change. 97 Future work examining trajectories of E2/P4, neurosteroid metabolites (e.g., via chromatography-mass spectrometry metabolic pathway profiling 98 ) and daily hormonal variability could help identify key hormonal drivers of neuromolecular change.
Parametric mapping & data integration
Few reviewed studies employed parametric mapping, but amongst those that did, several studies suggest ovarian steroid effects are region- and hemisphere-specific.28,29,34,43 For example, menopause may elevate estrogen receptor density most prominently in the bilateral middle frontal gyrus, left caudate, anterior cingulate, and right superior frontal gyrus. 34 These cortical ROIs were also targets of postmenopausal E2-mediated changes in glucose metabolism 43 and 5-HT2A activity.28,29 In future work, voxel-wise analyses could complement ROI-based approaches by offering detailed insights into regional hormone targets. Additionally, parametric maps can be correlated with molecular-genetic atlases, using tools like neuromaps and abagen, allowing for broader contextualization of ovarian steroid-related neuromolecular changes.99–101
Future opportunities in PET and multimodal molecular neuroimaging
GABA-A and NMDA receptors
Preclinical and clinical research is expanding in the area of reproductive transitions and their impact on GABA81,102 and glutamate receptors, 103 especially considering their role as treatment targets for postpartum and major depression.104,105 While [1H]MRS studies in this review explored GABA or glutamate concentrations, these neurometabolites are involved in multiple cellular processes, and their concentrations scale with metabolic activity. 40 PET studies are needed for direct insights into glutamatergic and GABAergic receptor activity. The lack of studies examining reproductive transition effects on glutamate NMDA or GABA-A receptors, despite available radioligands,106,107 represents a gap in the research.
Synaptic density
Although none of the reviewed studies employed methods to assess neuroplasticity, the positive impact of E2 on synaptic spine density is well-documented in preclinical research.108–110 In humans, reproductive transition effects on structural brain plasticity have been confirmed using MRI,5,111 but PET imaging of synaptic density 112 offers a way to directly study synaptic changes in future research.
Hormone receptors and neurosteroid synthesizing enzymes
Examining hormone receptor density and neurosteroid synthesis can clarify how the brain adapts to ovarian steroid flux. A single study used [18F]FES to image brain estrogen receptors and found higher distribution volume ratios relative to cerebellar gray matter after menopause. 34 However, future work may require novel tracers 113 due to low sensitivity of [18F]FES to detect estrogen receptor specific binding in the brain. 114 Aromatase binding, responsible for brain estrogen synthesis, 115 can also be imaged, 8 but no such studies met inclusion criteria for this review. However, a recent study showed that nicotine blocks limbic aromatase binding, 116 highlighting that common psychotropics can impact endogenous brain steroid signaling and potentially shape allostatic adaptations across reproductive transitions.
Neuroimmune activation
Preclinical evidence suggests that ovarian steroids influence the neuroimmune response. 117 However, only one [1H]MRS study reported myoinositol concentrations, a marker associated with glial cell activity, 40 and found no significant menstrual phase effects in the dlPFC. 32 It is important to note that myoinositol is involved in various cellular processes, 40 and its changes may not reflect glial activity related to the neuroimmune response. Novel radioligands allow more direct quantification of microglial and astrocyte activation tied to neuroimmune activity. 15 Although no studies using these methods met the inclusion criteria, one excluded study (due to lack of reproductive staging) observed female-specific, age-related increases in regional TSPO binding, 118 potentially linked to menopause. Understanding how ovarian steroids affect neuroimmune function could provide valuable insights into the increased vulnerability of women to neuropsychiatric and neurodegenerative diseases. 117
Multimodal PET-MRS opportunities
The exposure to ionizing radiation in PET studies imposes constraints on the number of scans that can be performed within a single subject and time period. MRS provides a complementary approach to PET, offering insights into metabolic function and the neurochemical environment without additional radiation exposure.9,12 For example, one reviewed study used [31P]MRS to estimate ATP synthesis alongside [18F]FDG-PET to measure glucose metabolism, 10 providing insight into the dissociation between energy production and glucose metabolism in the postmenopausal brain. However, multimodal molecular neuroimaging was utilized in only a handful of the reviewed studies.10,48,57 Combining these modalities can be especially beneficial in RCTs or longitudinal studies, where the challenge of repeated radiation exposure may limit the feasibility of multimodal PET assessments. Multimodal PET-MRS studies will be instrumental in elucidating the metabolic and neurochemical landscape underlying neuroreceptor changes during reproductive transitions and in response to hormonal treatments.
Conclusions
Molecular neuroimaging offers a promising yet underutilized method to examine neuromodulatory functions of ovarian steroid hormones. Studies to date of non-clinical groups have begun to explore how ovarian reproductive transitions affect neuromolecular function, including measurements of neurometabolite levels, glucose metabolism, and monoaminergic signaling. A small body of evidence suggests regional neuroenergetic shifts occur across the menstrual cycle 32 and with menopause, 10 but their impact on mitochondrial efficiency or oxidative stress is uncharacterized in humans. Furthermore, striking parallels were observed between 1) regional patterns of menstrual phase-related changes in energy buffering metabolites 32 and serotonin-synthesis activity, 64 and 2) peak regional effects of postmenopausal E2 on glucose metabolism 43 and 5-HT2A activity. 28 These similarities may indicate that the facilitatory effects of E2 and P4 on serotonin signaling contribute to sustaining mitochondrial efficiency and antioxidation—a hypothesis that can be explored in future multimodal studies. There were several [1H]MRS studies quantifying markers linked to metabolic activity (GABA/glutamate), 40 neuronal integrity (NAA), 40 or neuroimmune activity (myoinositol), 119 but none interrogated more specific markers of cellular function. Notably, two excluded studies (no reproductive staging) using more specific neuromolecular markers hint at higher oxidative stress (indexed by glutathione concentrations) 85 and microglial activity (index by 18 kDa translocator protein binding) 118 in association with lower E2 levels, highlighting ripe areas for ongoing inquiry. We recommend that future research 1) prospectively track across reproductive transitions longitudinally or conduct RCTs with hormone administration, 2) systematically assess neuromolecular targets in a priori ROIs and through voxel-wise approaches, and 3) integrate findings within a broader neuroscientific context using multimodal approaches or brain atlas integration tools (e.g., abagen, 120 neuromaps 99 ) Such work will enhance scientific understanding of ovarian steroid and reproductive transition effects on normative brain function as well as their role in vulnerability and resilience to female-biased neuropsychiatric and neurodegenerative diseases.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251348865 - Supplemental material for Molecular neuroimaging of ovarian steroid effects on the female brain: A systematic review of human non-clinical studies
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251348865 for Molecular neuroimaging of ovarian steroid effects on the female brain: A systematic review of human non-clinical studies by Melissa JM Walsh, Kathryn Gibson, Reese M Gray, Mila McNeal, Lucia S Lynch, Michelle Kang, James Brierley, Erin Bondy, Gabriel S Dichter and Crystal Edler Schiller in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Mental Health T32MH093315 and R01MH128238.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
