Abstract
Matrix metalloproteinase-9 (MMP-9) participates in the disregulation of blood–brain barrier during hemorrhagic transformation, and exacerbates brain injury after cerebral ischemia. However, the consequences of long-term inhibition or deficiency of MMP-9 activity (which might affect normal collagen or matrix homeostasis) remains to be determined. The authors investigated how MMP-9 gene deficiency enhances hemorrhage and increases mortality and neurologic deficits in a collagenase-induced intracerebral hemorrhage (ICH) model in MMP-9–knockout mice. MMP-9–knockout and corresponding wild-type mice at 20 to 35 weeks were used to model an aged population (because advanced age is a significant risk factor in human ICH). Collagenase VII-S (0.5 μL, 0.075 U) was injected into the right basal ganglia in mice and mortality, neurologic deficits, brain edema, and hemorrhage size measured. In addition, MMP-9 activity, brain collagen content, blood coagulation, cerebral arterial structure, and expressions of several MMPs were examined. Increased hemorrhage and brain edema that correlated with higher mortality and neurologic deficits were found in MMP-9–knockout mice. No apparent structural changes were observed in cerebral arteries, even though brain collagen content was reduced in MMP-9–knockout mice. MMP-9–knockout mice did exhibit an enhanced expression of MMP-2 and MMP-3 in response to ICH. The results indicate that a deficiency of MMP-9 gene in mutant mice increases collagenase-induced hemorrhage and the resulting brain injury. The intriguing relationship between MMP-9 deficiency and collagenase-induced ICH may reflect the reduction in collagen content and an enhanced expression of MMP-2 and MMP-3.
Spontaneous intracerebral hemorrhage (ICH) is one of the most serious stroke events, and results in high mortality and morbidity in patients (Bernardini and DeShaies, 2001; Panagos et al., 2002; Woo and Broderick, 2002). Mortality in ICH is often as high as 50% within the first month after ICH, with 50% of these deaths occurring within 48 hours (Broderick, 1993). The mechanisms of ICH-induced early brain injury (e.g. brain edema) have been investigated (Altumbabic et al., 1998; Del Bigio et al., 1999; Xi et al., 2002); however, the complete etiology of early brain injury in ICH is incompletely understood.
Matrix metalloproteinases (MMPs) are involved in the pathogenesis of cerebral vascular disorders, particularly the formation and rupture of aneurysms. For example, pro–MMP-2 has been implicated as a significant component within arterial walls of intracerebral aneurysms that leads to subarachnoid hemorrhage (Todor et al., 1998). Other studies also support elevated MMP (elastase and collagenase) activity in ruptured vessels in subarachnoid hemorrhage but not in unruptured aneurysms (Gaetani et al., 1999). MMPs are also involved in the hemorrhagic transformation after cerebral ischemia, especially after thrombolysis (Hosomi et al., 2001; Montaner et al., 2003; Sumii and Lo 2002). Hemorrhagic transformation is closely related to levels of plasma MMP-9 (Castellanos et al., 2003), and MMP inhibitors can reduce hemorrhage after thromboembolic stroke (Lapchak et al., 2000). MMPs are further associated with derangement of blood–brain barrier (Rosenberg et al., 1998), and MMP inhibition reduces brain edema after ICH (Rosenberg and Navratil, 1997). However, long-term deficiency of MMP-9 may carry unanticipated and undesirable effects that could predispose individuals to cerebral hemorrhage because MMP-9 deficiency could alter the collagen content in peripheral vascular tissues (Ducharme et al., 2000). In the present study, we measured mortality, neurologic deficits, hemorrhage size, brain edema, brain collagen content, cerebrovascular morphology, and MMP activity after collagenase-induced ICH in MMP-9–knockout and background-matched wild-type mice to consider what role MMP-9 might play in this model of ICH.
MATERIALS AND METHODS
MMP-9–knockout mice
This project followed an institutionally approved protocol (in compliance with the Guidelines established by the National Research Council Guide for the Care and Use of Laboratory Animals). MMP-9–knockout mice and their background-matched wild-type controls (Wang et al., 2000) were bred in the animal facility at Louisiana State University Health Sciences Center in Shreveport, Louisiana. The breeders were kindly provided by Dr. Eng H. Lo (Neuroprotection Research Laboratory, Harvard Medical School). All MMP-9–knockout and their sex/age-matched wild-type mice were studied at 20 to 35 weeks of age, which models the older human population with higher incidence of ICH.
Intracerebral hemorrhage induction
We adapted the collagenase-induced ICH model (Rosenberg et al., 1990) previously described in mice (Choudhri et al., 1997; Clark et al., 1998). Briefly, mice were anesthetized with an intraperitoneal injection of α-chloralose (40 mg/kg) and urethane (400 mg/kg) and positioned prone in a stereotaxic head frame (Stoelting, Kiel, WI, U.S.A.). An electronic thermostat-controlled warming blanket was used to maintain the core temperature at 37° ± 0.50°C. The calvarium was exposed by a midline scalp incision from the nasion to the superior nuchal line, and then the skin was retracted laterally. With a variable-speed drill (Dremel, Racine, WI, U.S.A.), a 1.0-mm burr hole was made 1.0 mm posterior to the bregma and 1.0 mm to the right of midline. A 24-G needle on a Hamilton syringe was inserted with stereotaxic guidance 4.0 mm into the right deep cortex/basal ganglia. The collagenase (VII-S, Sigma, St. Louis, MO, U.S.A.; 0.075 U in 0.5 μL of saline) in the syringe was infused into the brain at a rate of 0.25 μL/min over 2 minutes with an infusion pump (Bioanalytical Systems, West Lafayette, IN, U.S.A.). The needle was left in place for an additional 30 minutes after injection to prevent the possible leaking of collagenase solution. After removal of needle, the incision was closed and the mice were allowed to recover. Sham operation was performed with needle insertion only.
Animals were killed 24 hours after surgery for the recording of final mortality. Because most MMP-9–knockout mice died before 24 hours and hemorrhage has reached maximum in 5 hours (preliminary observation), animals were killed 5 hours after ICH to measure hemorrhage size, brain hemoglobin content, and brain water and sodium contents. For measuring the effect of ICH on MMP activity, brains were collected from mice at 0 (sham), 1, 5, and 24 hours after surgeries. Blood pressure was measured before and during surgery via a catheter in femoral artery in both MMP-9–knockout and wild-type mice.
In four mice (two each from MMP-9–knockout and wild-type groups), autologous blood was injected into basal ganglia to test whether blood injection activates MMP-9. We used an established blood-injection intracranial pressure (ICP) method as described by others (Xi et al., 2002). The brain samples were collected for zymography.
Neurologic deficits and mortality
Mice were assessed on a simple six-point scale based on level of consciousness, paresis, and circling behavior as previously described (Nehls et al., 1990). Neurologic deficit scores were assigned as follows: 0, no evidence of deficit; 1, mild deficit, such as the tendency to circle with stimulation; 2, definite circling with stimulation (gently pulling tail); 3, spontaneous circling; 4, spontaneous circling and brachial monoparesis; and 5, depressed level of consciousness. Neurologic evaluations were conducted at 5 hours and then at 24 hours after ICH. Because early mortality were high in MMP-9–knockout mice, only the neurologic scores obtained at 5 hours after ICH were shown (similar neurologic scores were obtained at 24 hours).
Hemorrhage area
Animals were decapitated, brains were removed, and six coronal brain sections were made at 5 hours after ICH. In all animals, the largest hematoma was near the needle insertion point/path. The areas of hematoma were scaled to five grades: 0 (no hemorrhage), 1 (<1 mm), 2 (1 to 2 mm), 3 (2 to 3 mm), 4 (3 to 4 mm), and 5 (>4 mm).
Hemoglobin assay
The ipsilateral cerebral hemisphere was collected at 5 hours after ICH from mice and distilled water (1 mL) was added to each hemisphere, followed by homogenization for 30 seconds, sonication on ice with an ultrasonicator for 1 minute, and centrifugation at 13,000g for 30 minutes. The hemoglobin-containing supernatant was collected and 80 μL of Darbkin's solution (Sigma) was added to a 20-μL aliquot. Fifteen minutes later, the optical density of solution at 540-nm wavelength was measured to assess hemoglobin content (Choudhri et al., 1997).
Anti-IgG staining: blood–brain barrier
Brain immunolocalization of IgG was studied as described previously (Richmon et al., 1998). Briefly, brain sections were collected at 5 hours after ICH and were incubated at room temperatures with 2.0% goat serum plus 0.2 Trioton-X-100 and 0.1% bovine serum albumin for 5 minutes, then with 10% goat serum plus 0.2% Triton-X-100 and 0.1% bovine serum albumin for 20 minutes. Brain slides were then incubated with biotinylated goat anti–rat IgG (1:200 or 1:1,000 Santa Cruz Biotechnology, Inc., Santa Cruz, CA, U.S.A.) for 2 hours, rinsed in phosphate-buffered saline (PBS), and incubated in ABC complex for 30 minutes. After the PBS rinse, the reaction product was visualized using 0.05% DAB in the presence of 0.02% H2O2. The sections were rinsed in PBS again, dehydrated, and coverslipped.
Brain water and sodium contents
The brain water and sodium content were measured as described previously (Yang et al., 1994; Xi et al., 2002). Briefly, mice were decapitated at 5 hours after ICH under deep anesthesia. The brains were immediately removed and divided into five parts: ipsilateral and contralateral basal ganglia, ipsilateral and contralateral cortex, and cerebellum. Cerebellum was used as internal control for brain water content. Tissue samples were weighed on an electronic analytical balance (model AE 100, Mettler Instrument Co., Columbus, OH, U.S.A.) to the nearest 0.1 mg to obtain the wet weight (WW). The tissue was then dried at 100°C for 24 hours to determine the dry weight (DW). Brain water contents (%) were calculated as [(WW—DW)/WW] x 100.
In another group of MMP-9–knockout and wild-type mice, the brains were separated into two hemispheres, the wet weights were measured, and the ratio of ipsilateral hemispheres that were divided by the contralateral hemispheres was used to represent brain swelling.
The dehydrated brain samples were digested in 1 mL of 1N nitric acid for 1 week. Sodium content was measured by flame photometry (Instrumentation Laboratory, Inc., Lexington, MA, U.S.A.) and expressed in microequivalents per gram of dehydrated brain tissue (μEq/g DW) as described elsewhere (Xi et al., 2001).
Matrix metalloproteinase zymography
To confirm the deficiency of MMP-9, blood samples were collected from tail veins, MMP-9 and MMP-2 gelatinase activities determined by zymography as described previously (Wang et al., 2000). Briefly, blood samples (60 μL) were lysed in lysis buffer including protease inhibitors on ice. Total protein concentrations were determined using the Bradford assay (Bio-Rad, Hercules, CA, U.S.A.). Equal amounts (50 μg) of total protein extracts were prepared and separated by 10% Tris-glycine gel with 0.1% gelatin as substrate (Bio-Rad). After separation, the gel was renatured and incubated with developing buffer (at 37°C for 24 hours). After developing, the gel was stained with 0.5% Coomassie Blue R-250 for 30 minutes and then destained. Gelatinolytic activity was determined as clear zones or bands at the appropriate molecular weights. Mouse MMP-9 and human MMP-2 (Chemicon, Temecula, CA, U.S.A.) were used as standards.
To determine the effect of collagenase-induced ICH on MMP-9 activity in the brain, mice were killed at 0 (sham), 1, 5, and 24 hours after surgery. The brains were removed quickly and ipsilateral hemispheres were homogenized in lysis buffer on ice. After centrifugation, the supernatant was collected and the total protein concentrations were determined. Zymography was again performed.
In addition, we performed zymography using protein extracts of ipsilateral hemispheres of mice at 5 hours after blood injection to test the effect of blood on MMP-9 activation in both MMP-9–knockout and wild-type mice. In additional experiments, we added collagenase directly to the gelatins and tested the effect of bacterial collagenase in zymography.
Western blots
Protein expression of MMP-2, MMP-3, and MMP-7 in brain tissues were determined with Western blot as previously described (Asahi et al., 2001; Ducharme et al., 2000). Similarly prepared protein samples (50 μg) (as in MMP zymography) were separated on 10% to 12% Tris-glycine gels and transferred to PVDF membranes. After blocking with 5% nonfat dry milk in PBS (pH 7.4) containing 0.1% Tween 20 (PBS-T), membranes were incubated overnight with antibody against MMP-2, MMP-3, and MMP-7 (Chemicon), followed by washing and incubation with a matching secondary antibody (Santa Cruz Biotechnology). Antigens were detected by enhanced chemiluminescence (Amersham Pharmacia Biotech, Amersham, U.K.).
Collagen assay
Total brain collagen content was determined by quantifying total soluble collagens using Sircol Collagen Assay kit (Biocolor, Newtownabbey, Northern Ireland) according to manufacturer's instruction. Brains were homogenized in 1 mL of 0.5-mol/L acetic acid containing protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, U.S.A.). Each sample was incubated for 24 hours at 4°C with stirring. After centrifugation, 100 μL of each supernatant was mixed with 1 mL of Sircol dye reagent for 30 minutes. After the reaction, the pellet was collected by centrifugation and then suspended in 1 mL of alkali reagent (0.5-mol/L NaOH). The optical density was evaluated at 540 nm with a spectrophotometer. The values from the test samples were compared to the values obtained with collagen standard solutions provided by the manufacturer that were used to construct a standard curve. The results are expressed as microgram per gram of brain weight (micrograms per gram).
Histological examination
Structural changes of basilar artery and brain were examined by hematoxylin and eosin (H&E) staining as described previously (Perkins et al., 2002). Animals were perfused with 10 mL 0.1 PBS and then with 20 mL 10% neutral buffered formalin, and the tissues post-fixed in the same fixative. Arteries were cut transversely (6 μm) and brain sections were cut coronally (10 μm) with a Cryostat (Leica CM 3050). Collagen accumulation in the basilar artery and aorta was assessed using Picrosirius Red Staining as described by Ducharme et al. (2000). Sections were rinsed with distilled water and incubated in Picrosirius red solution (1% Sirius red, Sigma) in saturated picric acid for 18 hours. Sections were rinsed twice with 0.01N HCL for 2 minutes, dehydrated, and mounted with Permount. Slides were observed by polarized light using an Olympus microscope and photographed with identical exposure for each section. Ponseau-S collagen staining was also used in brain slides to demonstrate collagen deposits in the basilar artery.
Coagulation and glucose
Coagulation was determined by blood clotting time that measured as described by Rubio-Poo et al. (1998). Samples of blood (25 μL) were collected from tails of unanesthetized mice into a microhematocrit glass capillary tube. Timing was started when blood first contacted the glass capillary tube. The blood was made to flow by gravity between the two marks of 45 mm apart until blood ceased to flow (reaction end-point).
Glucose concentrations were measured in blood samples from the tail of non-fasting animals using a Glucometer (Walgreen Co., Deerfield, IL, U.S.A.).
Statistical analysis
Quantitative data were expressed as mean ± SEM. Statistical comparisons were conducted using analyses of variance followed by Tukey-Kramer tests for intergroup comparisons. The Student's t-test was used in analysis of paired samples. Differences with P < 0.05 were considered statistically significant.
RESULTS
Neurologic function and mortality
Mortality (Fig. 1A) at 24 hours (including animals died within 5 hours after ICH) after collagenase-induced ICH was markedly increased in MMP-9–knockout mice (71%, 30 out of 42) when compared with that of wild-type mice (34.7%, 17 out of 49). For wild-type mice, 12 out of 17 mice survived to 5 hours (29.4% mortality) and 20 out of 32 survived to 24 hours (37.5% mortality). For MMP-9–knockout mice, 7 out of 20 survived to 5 hours (65% mortality) and 5 out of 22 survived to 24 hours (77% mortality).
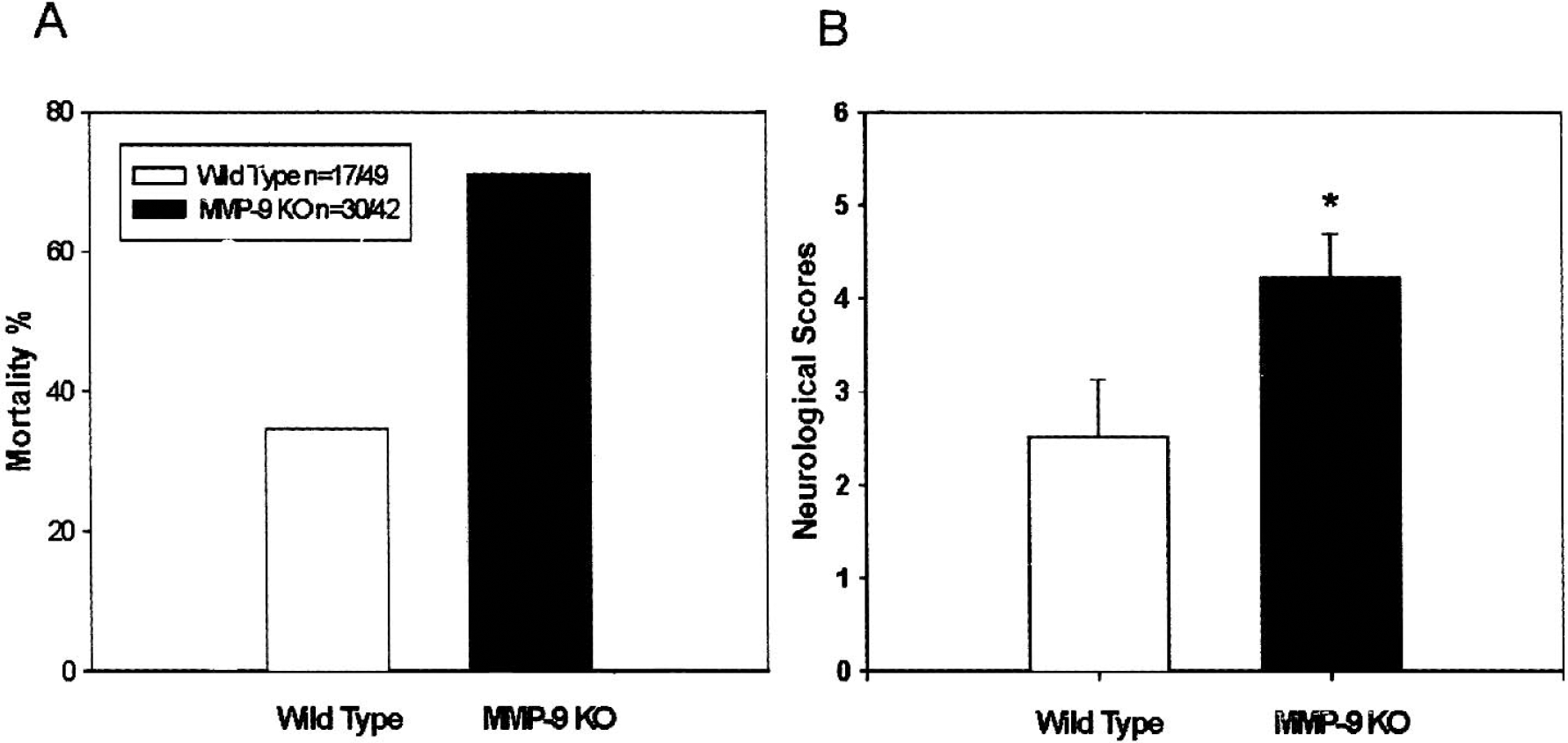
Mortality (
Neurologic deficits at 5 hours after collagenase-induced ICH were more severe in MMP-9–knockout mice (4.222 ± 0.465, n = 9) than in wild-type mice (2.375 ± 0.536, n = 8) (P < 0.05; Student's t-test, Fig. 1B). The timing of neurologic evaluation at 5 hours is adapted from Wang et al. (2002) because of the high mortality over 24 hours.
Hemorrhage size
Figure 2A shows representative samples of grade 0 (sham operated), grade 2 (wild type), and grade 5 (MMP-9 knockout). After collagenase infusion, severe ICH developed in the MMP-9–knockout mice. Enhanced hemorrhage was determined by an increased hematoma size: 3.778 ± 0.401 mm in MMP-9 knockouts (n = 9) and 2.380 ± 0.353 mm in wild types (n = 8) (P = 0.019; Student's t-test, Fig. 2B) and by brain hemoglobin content: OD540 = 0.311 ± 0.006 in MMP-9 knockouts (n = 9) and 0.231 ± 0.028 in wild types (n = 6) (P < 0.05; Fig. 2C). We used the OD540 value to represent hemoglobin content as reported previously (Choudhri et al., 1997; Kitaoka et al., 2002). Both hemorrhagic size and hemoglobin content were measured in brain samples collected at 5 hours after ICH because our pilot experiments determined that maximum bleeding occurred less than 5 hours after collagenase injection, and because of the high mortality among MMP-9–knockout mice within 24 hours.
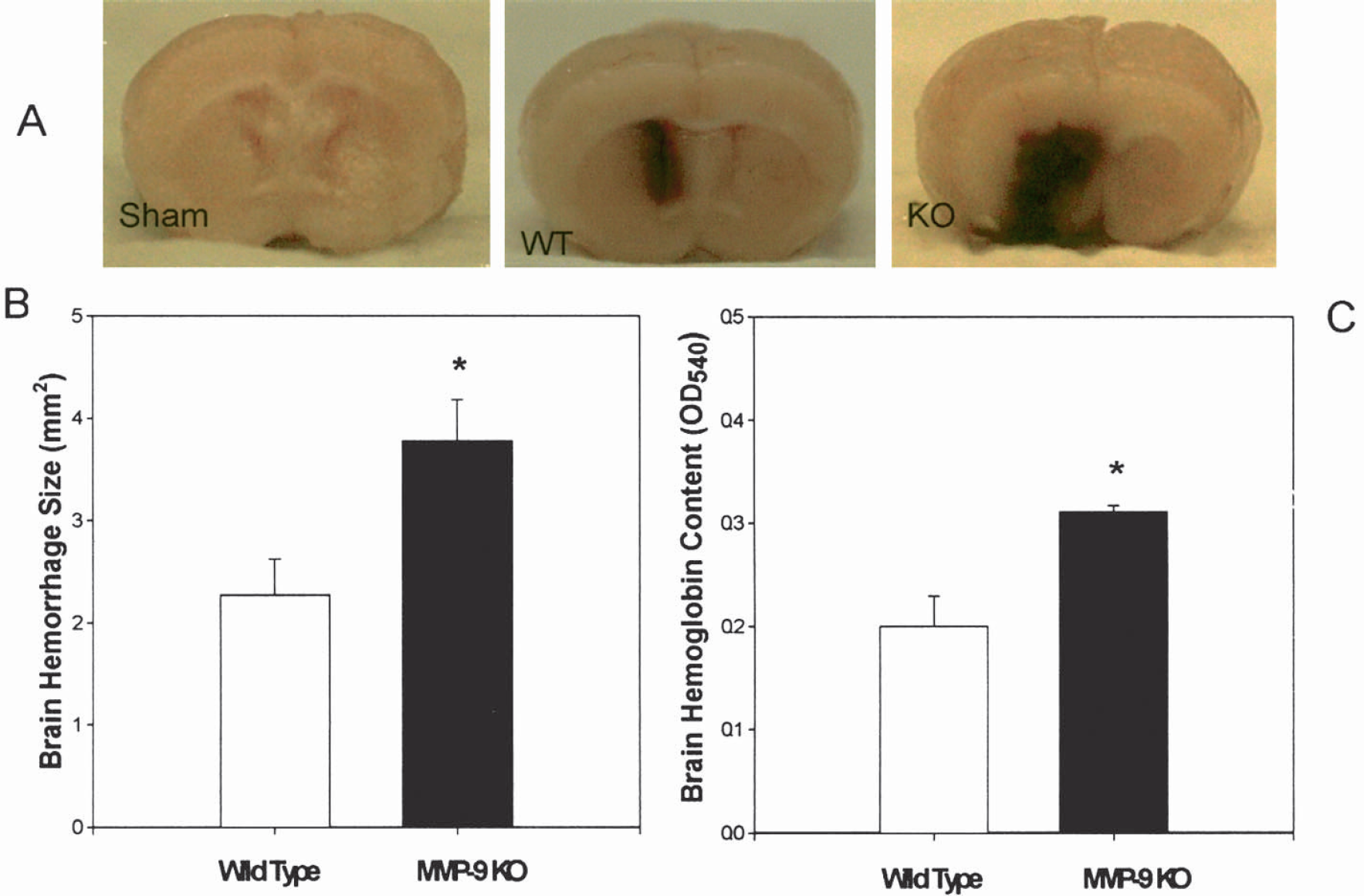
(
Hemorrhage under microscope at 5 hours after ICH was recorded by H&E staining in brain slides. Needle insertion did not produce hemorrhage either in wild-type (Fig. 3A1) or in MMP-9–knockout mice (Fig. 3C1). Collagenase injection produced hemorrhage in wild-type mice (Fig. 3B1) and massive hemorrhage in MMP-9–knockout mice (Fig. 3D1). Higher magnification showed bleeding from microvessels (Figs. 3B2 and 3D2).
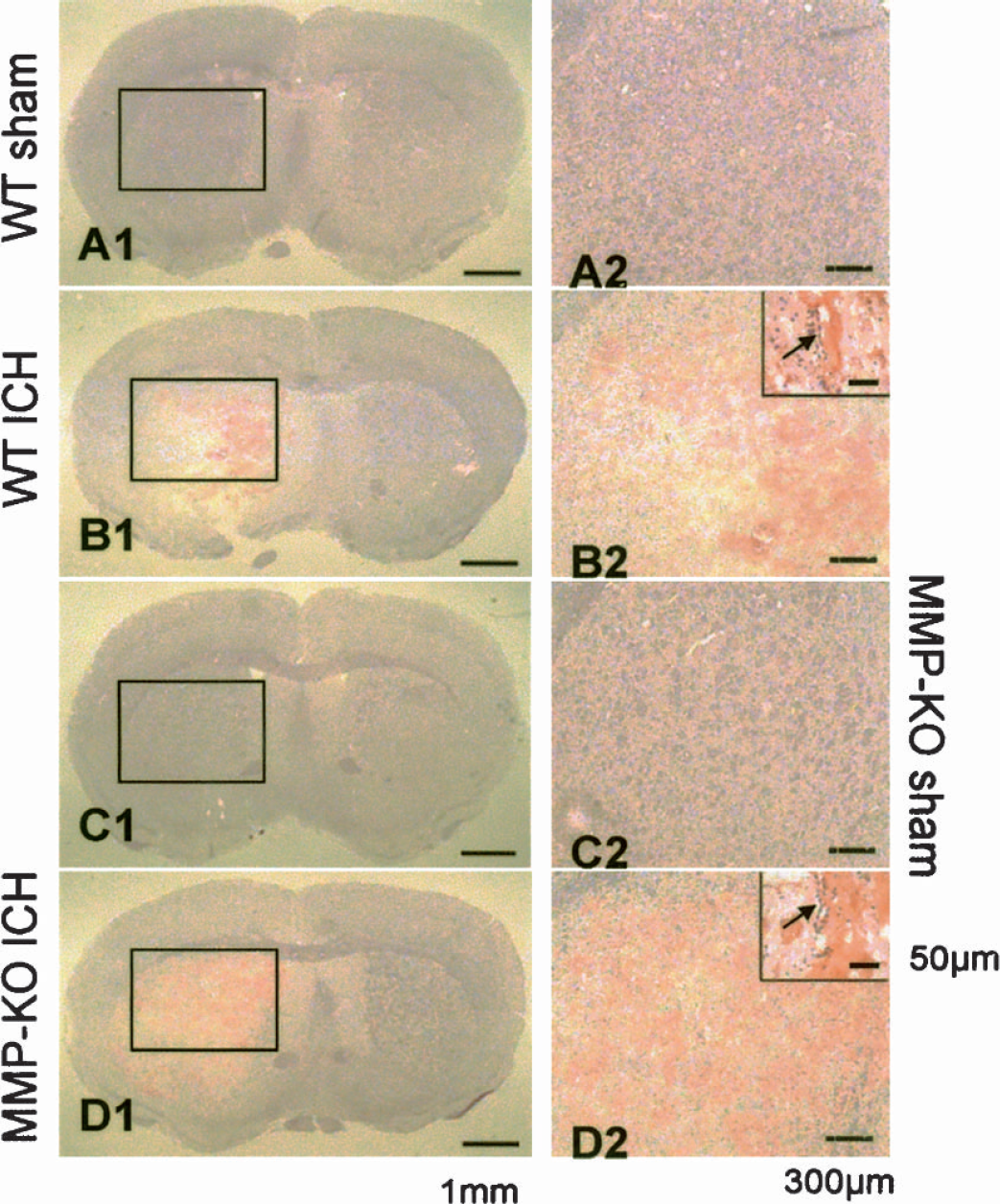
H&E staining illustrated hemorrhage from microvessels at 5 hours after collagenase-injection–induced ICH. Needle insertion failed to produce any hemorrhage in either wild type or MMP-9–knockout mice (
Brain IgG immunostaining
In normal mouse brain, only some tincture in periventricular structures were stained (Figs. 4A1, 4A2, 4C1, and 4C2). At 5 hours after collagenase injection, there was widespread IgG staining surrounding the entire hemorrhage area, indicating a breakdown of the blood–brain barrier allowing the IgG serum proteins to enter the brain parenchyma (Richmond et al., 1998). Even though bigger hemorrhage occurred in MMP-9–knockout mice (Fig. 4D1) compared with wild-type mice (Fig. 4B1), the total areas of IgG staining were comparable between wild-type and MMP-9–knockout mice. Higher magnification showed leaking of IgG around microvessels (Figs. 4B2 and 4D2).
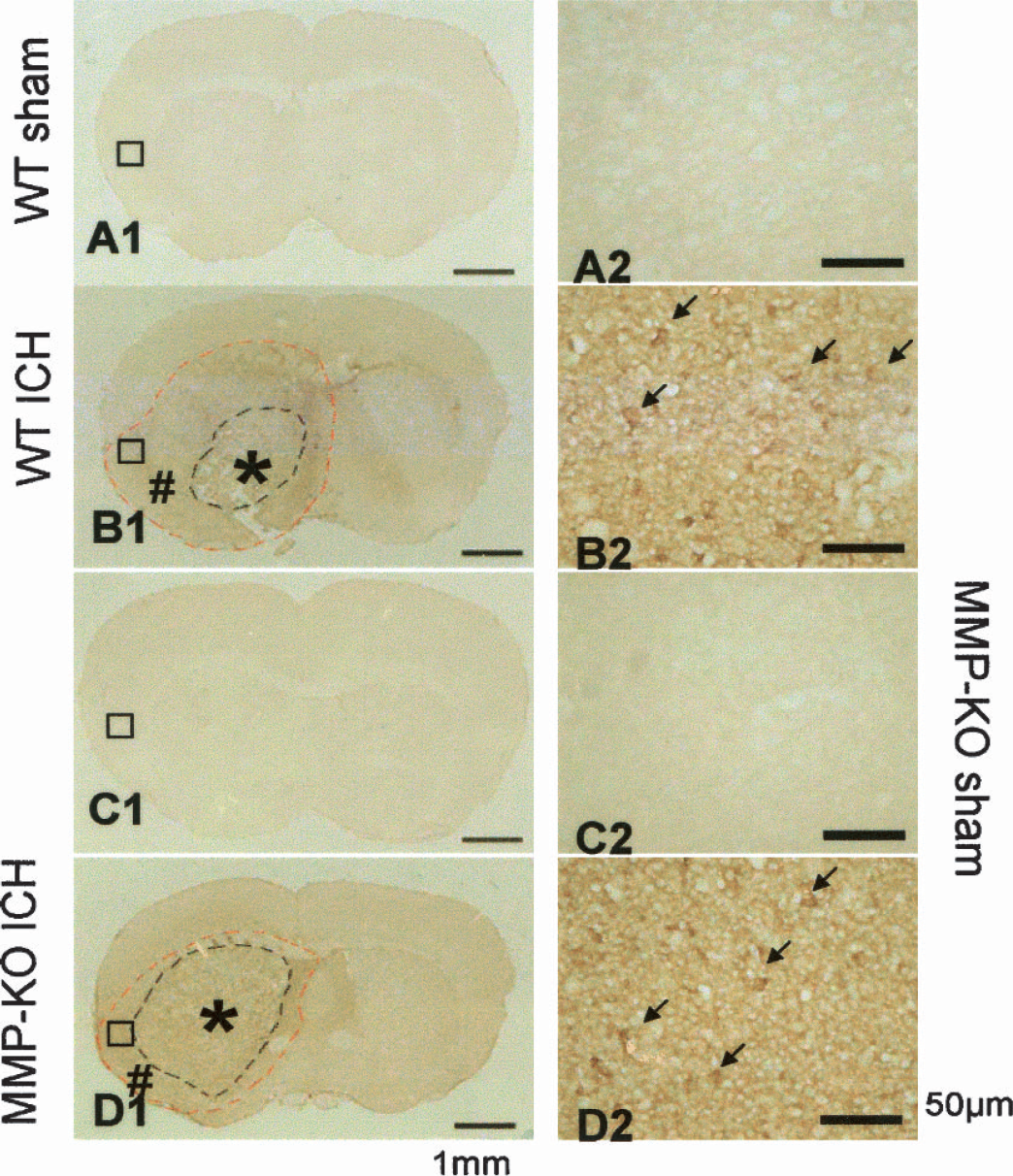
Immunostaining for anti-IgG as an indicator for blood–brain barrier rupture was performed at 5 hours after ICH. Negative staining was obtained in sham-operated mice (
Brain edema
Deletion of the MMP-9 gene enhanced ICH-induced brain edema (represented by increase of brain water content, Fig. 5A) in the ipsilateral cortex and basal ganglia regions (P < 0.05 vs. wild type, analysis of variance) at 5 hours after collagenase injection. The water content in the ipsilateral basal ganglia in wild-type mice was increased when compared with the contralateral basal ganglia (P < 0.05). No water content changes were observed in cerebellum, contralateral basal ganglia, or cortex between MMP-9–knockout and wild-type mice (P > 0.05, analysis of variance). Seven MMP-9–knockout animals and eight wild types were used for brain water content measurement.
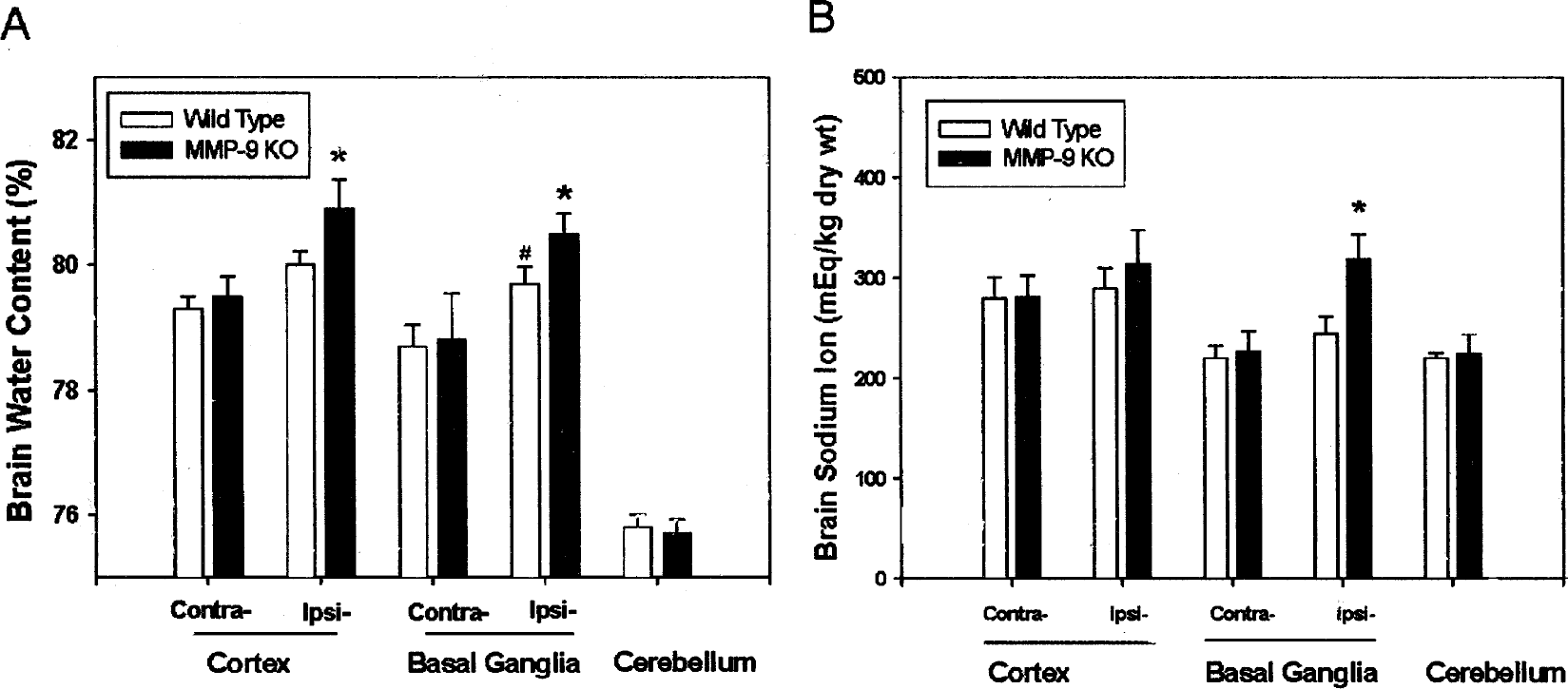
Brain edema (
The brain swelling at 5 hours after ICH was determined using the ratio of wet weight of the ipsilateral/contralateral hemispheres. The ratio in wild-type mice was 1.089 ± 0.0254 (n = 11); the ratio in MMP-9 knockouts was 1.187 ± 0.0378 (n = 8). The ratio of wet weight in MMP-9–knockout mice was significantly higher than that of wild-type mice (P = 0.039, Student's t-test).
In addition, tissue sodium content (Fig. 5B), another indicator of brain edema (Rosenberg and Navratil, 1997; Xi et al., 2002), increased significantly (P < 0.05) at 5 hours after ICH in the ipsilateral basal ganglia in MMP-9–knockout mice (n = 8) compared with wild-type mice (n = 11). No statistical significance was observed in the cerebellum, contralateral cortex, basal ganglia or ipsilateral cortices between MMP-9–knockout and wild-type mice (P > 0.05, analysis of variance).
MMP-9 enzymatic activity
MMP-9 gene deletion in MMP-9–knockout mice was confirmed by zymography using both blood (Fig. 6A) and spleen samples (Fig. 6B). Blood samples collected from mice were used to measure MMP-9 enzymatic activity by gelatin zymography. Both pro–MMP-9 (105 kd) and active MMP-9 (97 kd) were detected in the blood of wild-type mice, but such activity was not detected in the blood of MMP-9–knockout mice (n = 17, only 6 were shown) (Fig. 6A). This documents the deficiency of MMP-9 gene in MMP-9–knockout mice. No overcompensation of MMP-2 activity (72 kd) was noted in zymograms (Fig. 6A). Similar results were obtained from spleen samples: wild-type mice expressed both MMP-9 isoforms, but no bands were observed in MMP-9 knockouts (Fig. 6B). Overcompensation in MMP-2 activity may have been detected within spleens of MMP-9–knockout mice. This, however, was not confirmed by blood zymography (Figs. 6A and 6B).
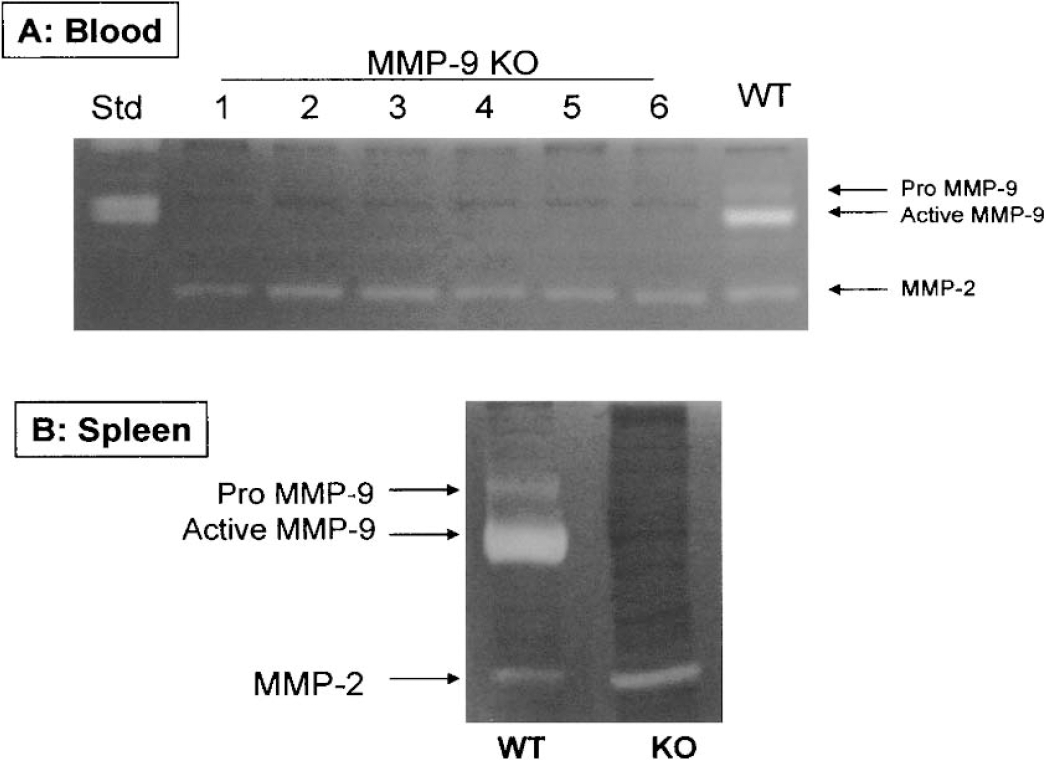
(
Brain MMP-9 activity after ICH
Ipsilateral hemispheres were collected from sham (killed at 5 hours) and ICH groups killed at 1, 5, and 24 hours after collagenase injection. The effect of collagenase-induced ICH on the enzymatic activity of MMP-9 was determined by zymography. Fig. 7A shows that the gelatinase activity of MMP-9 was increased at 1, 5, and 24 hours after collagenase-induced ICH in the brains of wild-type mice. Weak MMP-9–like gelatinase activity was observed in the brain of MMP-9–knockout mice at 1 and 24 hours after ICH. No MMP-9 activity was observed in the ipsilateral hemisphere of sham-operated MMP-9–knockout mice. No marked changes in MMP-2 activity were observed in wild-type and MMP-9–knockout mice. Figure 7A represents results from three separate experiments.
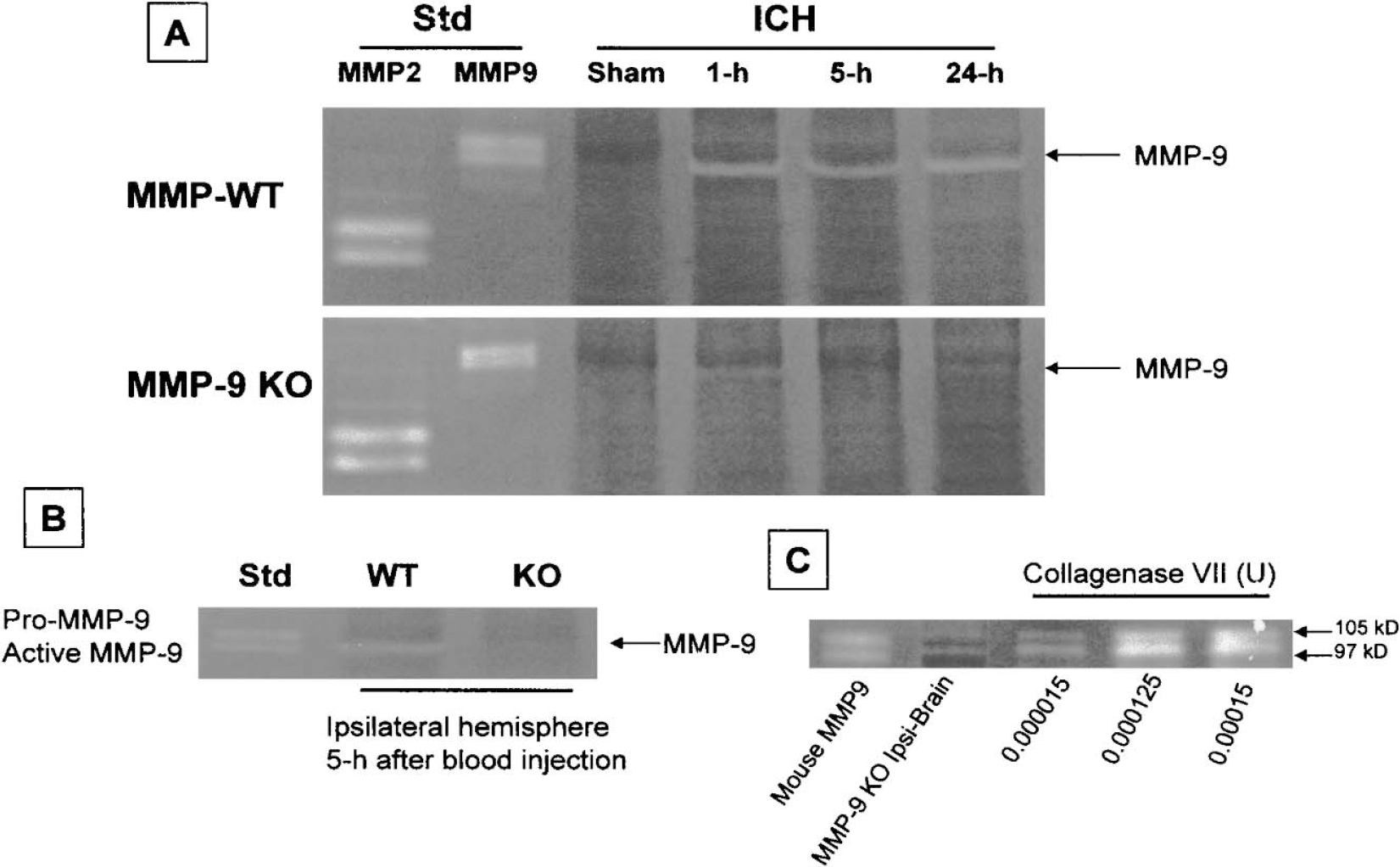
Zymography of brain samples after collagenase (
Furthermore, we measured MMP-9 activation in the ipsilateral hemispheres in MMP-9–knockout and wild-type mice after autologous blood injection (n = 3 from each group). Figure 7B shows that MMP-9 activity was increased at 5 hours after blood injection in the wild-type mice but not in the MMP-9–knockout mice.
In addition, we tested the direct effect of bacterial collagenase on the gelatins and found bacterial collagenase produced bands at the similar locations of MMP-9 (Fig. 7C).
Other MMP protein expression
The protein expression of several MMPs was measured to establish a possible overcompensation of other MMP isoforms after MMP-9 knockout. Western blotting was performed to measure expression of MMP-2, MMP-3, and MMP-7 in the ipsilateral hemispheres collected from sham-operated and ICH mice at 5 hours after collagenase injection (Fig. 8). No difference in the expression of MMP-2 and MMP-3 was observed in sham-operated brain. MMP-2 and MMP-3 were increased after ICH in both wild-type and MMP-9–knockout mice. The degree of expression of MMP-2 and MMP-3 in response to ICH was markedly enhanced in MMP-9–knockout mice compared with wild-type mice. MMP-7 was expressed and remained unchanged in sham-operated and ICH wild-type or MMP-9–knockout mice. These data are consistent with previous observations (Ducharme et al., 2000) where MMP-2 and MMP-3 increased in the myocardium after myocardial infarction, and suggest that MMP-9 may regulate expression of other MMPs. Figure 8 represents results from three separate experiments.
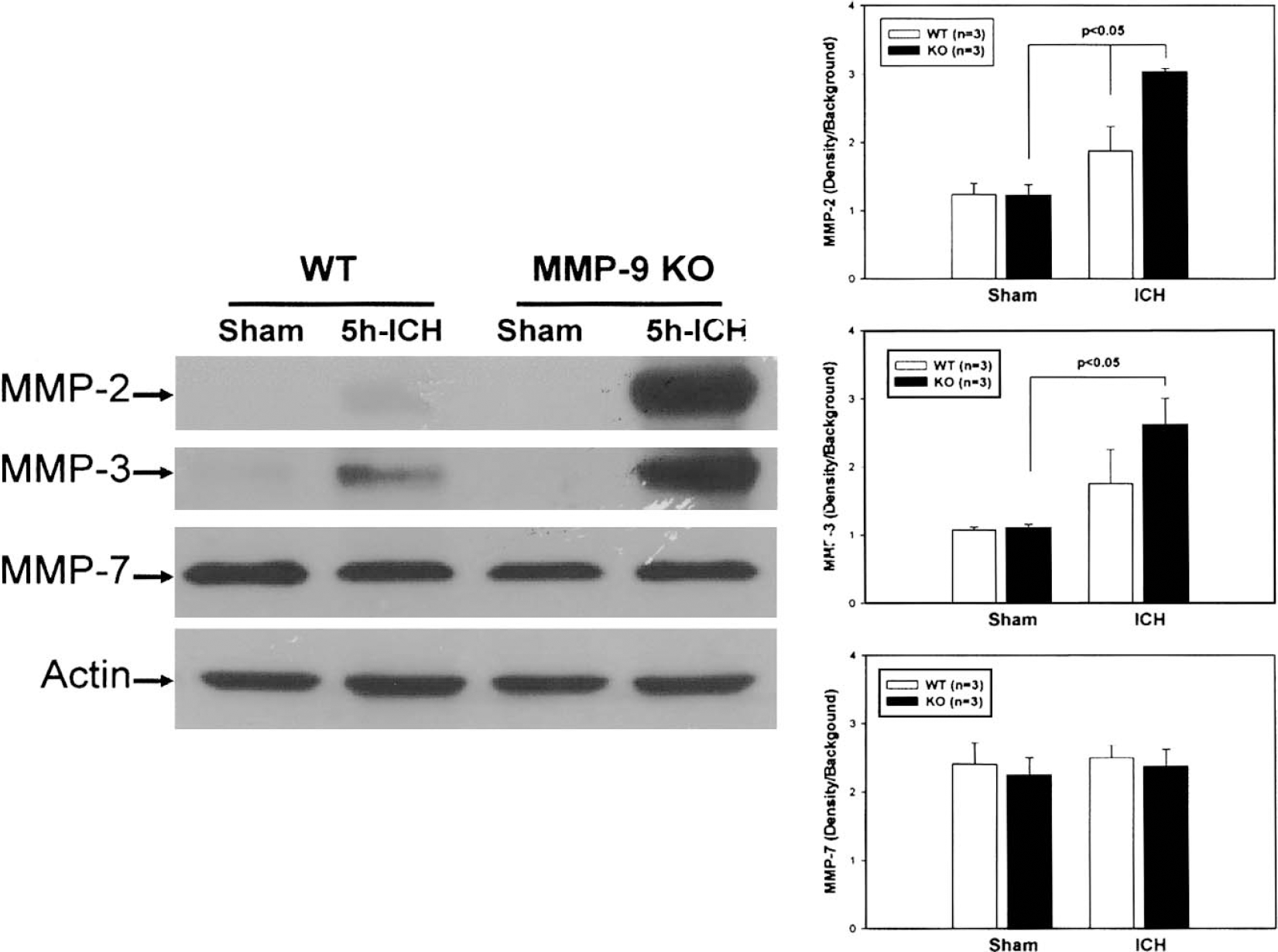
Western blot was performed to measure the expression of MMP-2, MMP-3, and MMP-7 in the ipsilateral hemispheres collected from sham-operated and ICH mice at 5 hours after collagenase injection. No difference in the expression of MMP-2 and MMP-3 was observed in sham brain. MMP-2 and MMP-3 were increased after ICH in both wild-type and MMP-9–knockout mice. The degree of expression of MMP-2 and MMP-3 in response to ICH was markedly enhanced in MMP-9–knockout mice compared with wild-type mice. MMP-7 was expressed and remained unchanged in sham and ICH wild-type or MMP-9–knockout mice.
Collagen staining and content
The Picrosirius red collagen staining (the light yellow in the vessel wall) showed a similar level of collagen accumulation in the basilar artery and aorta in both MMP-9–knockout and wild-type mice (Fig. 9). Figure 9 shows the collagen levels in the four segments of the aorta (Figs. 9A and 9B) and four segments of the basilar artery (Figs. 9C and 9D) from wild-type and MMP-9–knockout mice.
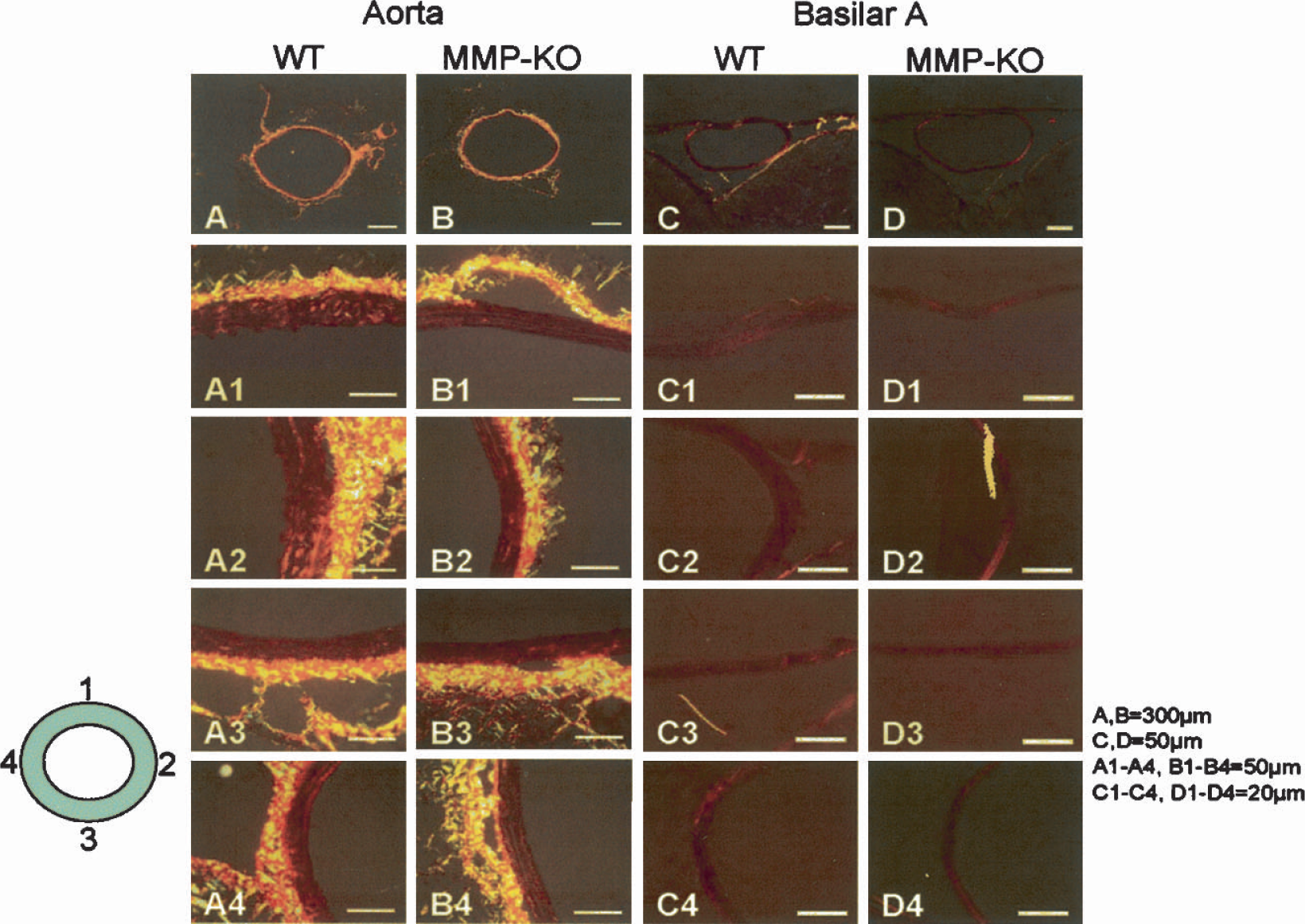
The Picrosirius red collagen staining (light yellow in the vessel wall) showed similar level of collagen accumulation in the basilar artery and aorta of either MMP-9–knockout or wild-type mice. Polarized light revealed collagen levels in the four segments of the aorta (
Ponseau-S collagen staining is shown in Fig. 10 (top). Similar levels of collagen (red in vessel wall) were observed in wild-types and MMP-9–knockouts. Pircosirius red collagen staining failed to shown collagen fibers in the parenchyma of the brains (Fig. 10, bottom left).
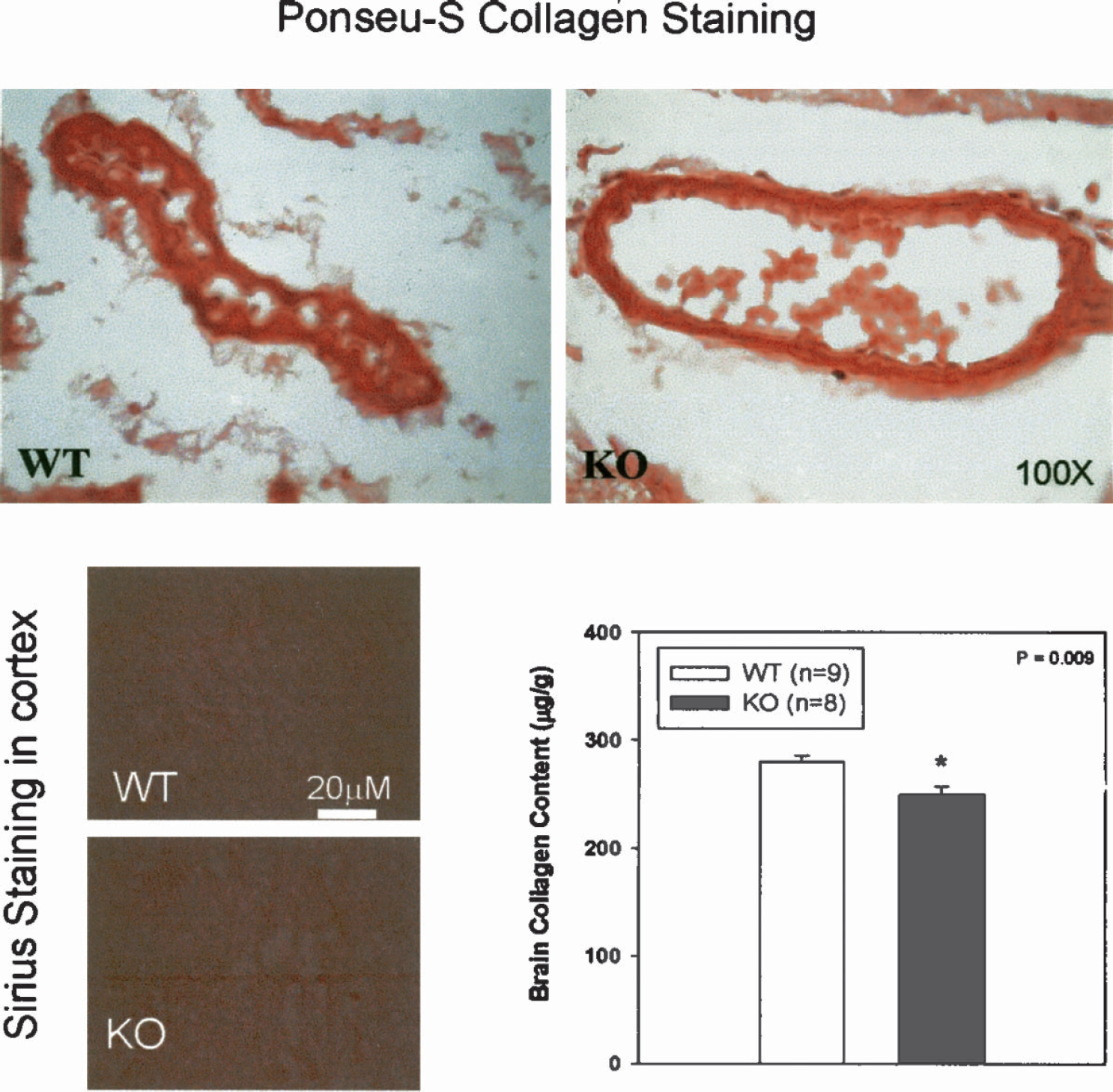
Ponseu-S collagen staining (top) showed that a similar level of collagen (red in the vessel wall) was observed in wild-type and MMP-9–knockout mice. Pircosirius red collagen staining failed to show collagen fibers in the parenchyma of the brains (bottom left). The Sircol Collagen Assay showed that the total soluble collagens in the brains of MMP-9–knockout mice were significantly decreased (P < 0.01; bottom right) compared with wild-type mice.
The Sircol Collagen Assay showed that the total soluble collagens in the brains of MMP-9–knockout mice (249.35 ± 8.28 μg/g, n = 8) were significantly decreased (P < 0.01; Fig. 8, bottom right) compared with those of wild-type mice (279.94 ± 6.11 μg/g, n = 9).
Cerebral arteries
The structures of the basilar artery and brain tissue were assessed by H&E staining. No structural difference was found in the brains of MMP-9–knockout mice when compared with that of wild-type mice (not shown, but see Figs. 7 and 8).
Blood pressure and coagulation and glucose
Blood pressures in MMP-9–knockout and wild-type mice were normal before the induction of ICH (82.5 ± 6.3 mm Hg for MMP-9–knockout mice (n = 5) and 81.4 ± 5.2 mm Hg for wild-type mice (n = 5); P > 0.05). Blood pressure varied during surgery between 70 and 90 mm Hg in both wild-type and knockout mice. Blood clotting time and blood glucose level were measured in wild-type (n = 6) and MMP-9–knockout mice (n = 11). Blood clotting time in MMP-9–knockout mice was significantly reduced (126.8 ± 5.01 seconds) compared with wild-type mice (149.0 ± 8.68 seconds, P < 0.05). The levels of glucose in wild-type (114.7 ± 7.27 mg/dL) and knockout mice (106.7 ± 2.33 mg/dL) were not different (P = 0.354).
DISCUSSION
Brain tissues express MMPs constitutively and alter MMP expression in response to cellular stress. Secreted as inactive enzymes, MMPs require activation by other proteases or free radicals (Lo et al., 2002; Rosenberg, 2002). There is a recent interest in the role of MMPs in intracerebral hemorrhage, especially in ischemic hemorrhagic transformation (Hosomi et al., 2001; Lapchak et al., 2000; Sumii and Lo, 2002). MMPs have been shown to degrade components of the basal lamina and to disrupt the blood–brain barrier, both of which contribute to the hemorrhagic transformation and associated brain injury (Lo et al., 2002; Rosenberg, 2002).
The use of transgenic mice is an important new development in ICH research, especially in terms of our ability to define molecular mechanisms in ICH. For example, spontaneous intracerebral bleeding occurs in mice lacking of αv integrins (McCarty et al., 2002) and in mice with cerebral amyloid angiopathy (Lee et al., 2003; Winkler et al., 2001). A double-blood injection model was attempted in mice and showed convincing results that closely parallel human ICH (Belayev et al., 2003). We selected the collagenase-induced ICH mouse model because collagenase-induced ICH in mice has a high mortality, which is age dependent, like human ICH, with marked brain edema. Second, the intriguing relationship between MMP-9 and collagenase-induced ICH led us to explore the relationships between MMP-9 deficiency and collagen content. Collagenase injection is a popular ICH model where collagenase injected into the basal ganglia produces hemorrhage (Rosenberg et al., 1990). Even though collagenase does not discriminate among different brain tissues, it produces massive bleeding and mortality, similar to that in human ICH. This model has been used extensively in investigations of ICH (Brown et al., 1995; DeBow et al., 2003; Del Bigio et al., 1996; Elger et al., 1994; Lyden et al., 1997; MacLellan et al., 2002; Matsushita et al., 2000; Rosenberg and Navratil, 1997; Rosenberg et al., 1992), including studies on the long-term deficits after collagenase-induced ICH (Chesney et al., 1995). Another animal model used to determine ICH pathophysiology and to evaluate therapeutic treatment (Andaluz et al., 2002) is the direct injection of autologous blood into the rat caudate nucleus (Nath et al., 1986), a model used in numerous studies (Bullock et al., 1984; Hua et al., 2002; Huang et al., 2002; Jiang et al., 2002; Mendelow, 1993; Nath et al., 1987; Thulborn et al., 1990; Xue and Del Bigio, 2000; Wu et al., 2002). This blood injection model was refined further into a double-hemorrhage model (Deinsberger et al., 1996), especially for the study of tissue injury after ICH (Felberg et al., 2002; Hickenbottom et al., 1999).
Our present study, using “aged” animals, contradicts some reports in the literature showing that MMP-9 deficiency or inhibition attenuates brain injury after ischemic/hemorrhagic stroke insults. We found that more bleeding occurred in MMP-9–knockout mice after collagenase injection, a more severe brain edema, greater deterioration of neurologic function, and dramatically increased mortality. We repeated experiments several times in the present study to (1) to reconfirm MMP-9 deficiency in the knockout mice by zymography, (2) evaluate the collagen content in the brain tissues, (3) compare the structure of cerebral arteries, (4) test the coagulation functions, (5) measure blood pressure, and (6) rule out “overcompensation” by other MMPs. All of these factors may be altered by MMP-9 gene deletion and could contribute to enhanced bleeding and mortality during collagenase-induced ICH (Belaaouaj et al., 2000; Ducharme et al., 2000; Salonurmi et al., 2004).
By examining blood MMP-9 enzymatic activity using zymography, we found absence of MMP-9 bands in all MMP-9–knockout mice tested. These observations were later confirmed by examining MMP-9 activity in the spleen, an organ with very high MMP-9 activity (Vu et al., 1998). In the brain tissues, strong bands appeared at MMP-9 locations in MMP wild-type mice at 1, 5 and 24 hours after collagenase-injection; however, only extremely weak bands were seen in MMP-9–knockout mice at 1 hour, and hardly any were observed at 5 or 24 hours. Even though these striking differences distinguish wild-type from MMP-9–knockout mice, the weak band seen at 1 hour (sometimes at 5 hours) after ICH in MMP-9–knockout mice is probably artifactual, based on zymographic and the genetic of these animals. One of the possibilities is that the weak “MMP-9” bands at 1 hour after ICH in MMP-9–knockout mice may not be the endogenous MMP-9 generated in brain tissues, because MMP-9 enzymatic activity was absent in both blood (Fig. 6A) and spleen samples (Fig. 6B). One of the likeliest sources for the weak MMP-9–like band in MMP-9–knockout mice could be derived from the bacterial collagenase, which might induce the signal at similar location, as indicated by Fig. 7C. Even though bacterial collagenases could modify our interpretations of the data, findings in wild-type and knockout mice, the striking difference as shown in Fig. 6A indicates that endogenous MMP-9 was formed extensively only in wild-type mice. Additional data confirming the deficiency of MMP-9 protein were obtained in blood-injection ICH mice (Fig. 7B). Injection of autologous blood activated MMP-9 in the brain of MMP wild-type mice; no equivalent activation was seen in MMP-9–knockout mice.
Another factor that might influence bleeding after collagenase injection is the possible changes in the collagen content. It has been reported in a model of myocardial ischemia that lower collagen accumulation (measured by Picrosirius red collagen staining) occurred in the infarcted myocardium area in MMP-9–deficient mice compared with wild-type mice (Ducharme et al., 2000). The lack of collagen may delay wound healing (Salonurmi et al., 2004). Enhanced MMP activity, especially MMP-2, was colocalized with macrophages and was associated with collagen degradation (Silence et al., 2002). In a model of carotid artery remodeling, MMP-9 deficiency increased interstitial collagen accumulation and impaired the capacity of smooth muscle cells to compact collagen (Galis et al., 2002). We speculate that low level of collagen in cerebral tissues, especially in vessels, may weaken these mice to collagenase exposure, and result in more severe bleeding. This presumption is supported by the present study. The brain collagen content (measured by Sircol Collagen Assay) showed a significant reduction in MMP-9–knockout mice (Fig. 10). It should be mentioned that the Sircol Collagen Assay kit measures all soluble collagens in the brain, including collagen type I though VI and type XIV, and we are not certain which collagen isoforms were reduced. Two other methods, Picrosirius red and Ponseau-S collagen staining, failed to distinguish the possible differences of collagen deposits in brain tissues, basilar arteries, and aorta (Figs. 9 and 10). We also could not determine whether the structure of cerebral arteries was affected by MMP-9 deficiency because major arteries (such as the basilar artery) retain similar structure under H&E staining. Some deformation of femoral arteries in MMP-9–knockout mice was seen during surgical procedures. It is also possible that changes in collagenase or elastase in the brain tissues including major arteries or microvessels contribute to this effect (Ducharme et al., 2000; Gaetani et al., 1999). Enhanced expression of collagenases (MMP-1, MMP-8, MMP-13, and MMP-18) and elastases (MMP-12) in the MMP-9–deficient mice might promote a more severe bleeding after external collagenase-injection, a possibility which requires further investigation.
MMP-9 deficiency might also influence blood coagulation and blood pressure to promote bleeding and brain injury. It has been reported that activation of multiple MMPs cleaves tissue factor pathway inhibitors, altering normal coagulation (Belaaouaj et al., 2000). We measured the clotting time in MMP-9–knockout and wild-type mice and obtained shorter clotting time in the knockout mice. Shortening of clotting time should promote faster clotting and reduce bleeding, contradictory to the observed enhanced bleeding in MMP-9 knockouts. Further, we are unaware of documented blood pressure changes related to MMP-9 expression (Wang et al., 2002). Blood pressures in both MMP-9–knockout and wild-type were similar before ICH (approximately 80 mm Hg) and remained similar (from 70 to 90 mm Hg) during collagenase injection. Hyperglycemia may enhance brain injury in this model (Sieber and Traystman, 1992), but similar glucose values were found in wild-type and knockout mice. It is thus unlikely that the enhanced bleeding in MMP-9–knockout mice was related to the hypertension (or hypertensive crisis during surgery), abnormal glucose, or coagulation abnormalities.
Finally, overcompensation of other MMPs could alter collagen or collagenase, induce microvessel wall malformations, and influence bleeding and the brain injury. Overexpression of MMP-2, MMP-3, and MMP-13 as well TIMP-1 in cardiomyocytes (Ducharme et al., 2000) but not in carotid artery or aorta (Galis et al., 2002) was observed in MMP-9–knockout mice. We examined the activity of MMP-2 (Gelatinase-A) and the protein expressions of MMP-2, MMP-3 (Stromelysin), and MMP-7 (Matrilysin). Even though MMP-7 was not changed before and after ICH (Fig. 8), the expressions of MMP-2 and MMP-3 were remarkably enhanced after collagenase injection, especially in MMP-9–knockout mice. Therefore, the activity of MMP-2 may not have overcompensated, but the expression of MMP-2 and MMP-3, but not of MMP-7, was enhanced by ICH in MMP-9–knockout mice. We acknowledge the low sensitivity of the method used in the present study for measuring MMPs because this method is better suited for tissues from rats than it is of tissues from mice. A possible overcompensation of MMP-2 activity may not be detected under this condition. Our data, however, could not support or exclude the possibility that overexpression (possible compensation) of MMP-2 and MMP-3 could affect collagen content to negatively influence cerebral vessel structural integrity and facilitate hemorrhage and brain injury after collagenase-induced ICH, a mechanism which deserves future study.
