Abstract
Both vascular endothelial growth factor (VEGF) and integrin αvβ3 play roles in angiogenesis. In noncerebral vascular systems, VEGF can induce endothelial integrin αvβ3 expression. However, it is unknown whether VEGF, like integrin αvβ3, appears in the initial response of microvessels to focal brain ischemia. Their coordinate expression in microvessels of the basal ganglia after middle cerebral artery occlusion (MCAO) in the nonhuman primate model was examined quantitatively. Cells incorporating deoxyuridine triphosphate (dUTP+) by the polymerase I reaction at 1 hour (n = 3), 2 hours (n = 3), and 7 days (n = 4) after MCAO defined the ischemic core (Ic) and peripheral regions. Both VEGF and integrin αvβ3 were expressed by activated noncapillary (7.5- to 30.0-μm diameter) microvessels in the Ic region at 1 and 2 hours after MCAO. At 7 days after MCAO, the number of VEGF+, integrin αvβ3+, or proliferating cell nuclear antigen-positive microvessels had decreased within the Ic region. The expressions of VEGF, integrin αvβ3, and proliferating cell nuclear antigen were highly correlated on the same microvessels using hierarchical log-linear statistical models. Also, VEGF and subunit αv messenger ribonucleic acids were coexpressed on selected microvessels. Here, noncapillary microvessels are activated specifically early during a focal cerebral ischemic insult and rapidly express VEGF and integrin αvβ3 together.
The appearance of new blood vessels (angiogenesis) is a relatively late response of the brain to focal ischemia (Krupinski et al., 1994; Chuaqui and Tapia, 1993). The signals, their timing, and sequence of presentation for angiogenesis after the initial ischemic events have not been established (Okada et al., 1996). The appearance of leukocyte adhesion receptors on microvascular endothelium (Okada et al., 1994; Haring et al., 1996) and changes in microvascular integrin expression (Wagner et al., 1997) within the first hours after middle cerebral artery occlusion (MCAO) suggest that signals for microvascular cell activation and angiogenesis may be generated early during ischemia.
Vascular endothelial growth factor (VEGF), also known as vascular permeability factor, has been implicated in endothelial cell proliferation, permeability, and angiogenesis (Dvorak et al., 1995; Senger, 1996). Expression of VEGF and its receptors, flt-1 and KDR, has been observed in developing organs during embryogenesis and in several pathologic conditions, including ischemic injury of the brain (Kovacs et al., 1996; Hayashi et al., 1997) and the myocardium (Li et al. 1996), wound healing (Brown et al., 1992), and in several inflammatory conditions (Detmar et al., 1994; Fava et al., 1994). In vitro studies show that VEGF stimulates the expression of integrin αvβ3 in microvascular endothelial cells in addition to tissue factor, osteopontin, and plasminogen activator expression (Clauss et al., 1996; Zucker et al., 1998; Pepper et al., 1991; Senger et al., 1996). In the rodent, VEGF antigen and messenger ribonucleic acid (mRNA) appear from 5 to 24 hours after MCAO, late after the onset of focal ischemia (Kovacs et al., 1996; Hayashi et al., 1997).
Integrin αvβ3, a cellular receptor for several Arg-Gly-Asp (RGD)–containing ligands, plays a significant role in the processes of angiogenesis (Brooks et al., 1994). Among the variety of ligands recognized by integrin αvβ3 are fibrin(ogen), von Willebrand factor, vitronectin, thrombospondin, osteopontin, and the extracellular matrix proteins laminin and collagen (Cheresh, 1993). In the vasculature, integrin αvβ3 is expressed by endothelial cells, smooth muscle cells (SMC), activated leukocytes, and macrophages and has been shown to mediate tumor angiogenesis. Vascular endothelial cell migration involves the ligation of integrin αvβ3 with vitronectin, whereas SMC migration involves the interactions of integrin αvβ3 with osteopontin and vitronectin (Leavesley et al., 1993). It has been demonstrated that integrin αvβ3 promotes adhesion-dependent survival in angiogenic blood vessels (Brooks et al., 1994). Okada et al. demonstrated that integrin αvβ3 (but not integrin αvβ5, the receptor for vitronectin) appears on noncapillary microvessels in the basal ganglia within 2 hours of MCAO (Okada et al., 1996). Its appearance was significantly associated with fibrin (one of its ligands) deposited within the microvessel lumina at all times after MCAO. Whether this significant association results from increased microvascular permeability, endothelial cell or SMC reactivity, or from separate unrelated events is important for understanding the fate of cerebral microvessels and neurons after MCAO (Okada et al., 1994; Okada et al., 1996; Tagaya et al., 1997).
The hypothesis tested by this study is that microvascular integrin αvβ3 expression is increased together with the appearance of VEGF and evidence of endothelial cell and SMC activation within the first moments after the onset of experimental focal brain ischemia. Furthermore, these changes are related to neuron injury. Here, to test the null hypothesis of no co-localization, the number of microvessels expressing proliferating cell nuclear antigen (PCNA), VEGF, and integrin αvβ3 within the ischemic and nonischemic regions were tabulated for each subject after MCAO (at 1 hour, 2 hours, and 7 days). Log-linear models then were fitted to pairwise frequencies in multi way tables to determine the significance of any relation among PCNA, VEGF, and αvβ3 expression within the subjects. This study is the first to demonstrate that microvascular expression of integrin αvβ3 is related to VEGF up-regulation in reactive cerebral microvasculature displaying PCNA in the early moments of focal cerebral ischemia. It also provides evidence of an ordered temporal and topographic connection between microvascular activation and neuron injury during early MCAO.
MATERIALS AND METHODS
Experimental focal cerebral ischemia
Brain tissues from 13 adolescent male baboons (Papio anubis/cynocephalus) were used for this study. All animals were neurologically normal before MCAO and apparently free of infections or inflammation during the experiments. All procedures in this study were approved by the Institutional Animal Research Committee and were performed in accordance with the standards published by the National Research Council (Guide for the Care and Use of Laboratory Animals) and the U.S. Department of Agriculture Animal Welfare Act. In compliance with these standards, efforts were made to ensure that the subjects were free of pain or discomfort. The principal investigator, veterinarians, and primate handling staff were present for all procedures.
In the following experiments, three cohorts of subjects underwent MCAO for 1 hour (n = 3), 2 hours (n = 3), or 7 days (n = 4), compared to a control group (n = 3), which underwent no procedure. The 7-day cohort comprised subjects that underwent MCAO during surgical implantation and displayed persistent unilateral neurologic defects thereafter. Preparation of the awake baboon MCAO stroke model has been described in detail elsewhere (del Zoppo et al., 1986; del Zoppo et al., 1991). In brief, under general anesthesia with isoflurane using a transorbital approach, an inflatable balloon catheter assembly was placed around the MCA proximal to the origin of the lenticulostriate arteries. The MCAO was achieved by inflating the balloon in the awake subject. All experiments were terminated under thiopental sodium anesthesia by transcardiac perfusion with an isosmotic perfusion fluid containing heparin (2000 IU/L), sodium nitroprusside (6.7 μmol/L), and bovine serum albumin (25 g/L). Tissue blocks (1.0 × 1.0 × 0.5 cm) from symmetrically located sites of basal ganglia were excised. Alternate blocks either were embedded in Tissue-Tek OCT compound (Miles, Inc., Elkart, IN, U.S.A.), frozen in isopentane/dry ice, and stored at -80°C until use or were fixed with 2% paraformaldehyde (PFA) for 24 hours and embedded in paraffin for the immunohistochemical and in situ hybridization studies.
Antibodies
Poly clonal antibodies against VEGF (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, U.S.A.) and integrins containing subunit αv (integrin αvβ3 and αvβ5) (Chemicon International, Inc., Temecula, CA, U.S.A.), and monoclonal antibodies against PCNA (PC-10) (Sigma BioSciences, St. Louis, MO, U.S.A.) and integrin αvβ3 (LM609) (Chemicon Interational, Inc.), were used in this study. The anti-VEGF polyclonal antibody was made against the NH3-terminal peptide (1 to 20 residues) of VEGF. The specificity of VEGF immunoreactivity was confirmed by disappearance of immunoperoxidase signals on blocking with the VEGF NH3-terminal peptide (Santa Cruz Biotechnology, Inc.) (data not shown). The antisubunit αv polyclonal antibody cross-reacted with integrin αvβ3 and αvβ3. Although the cross-reactivity of this antibody with αvβ3 might have led to overestimation of αvβ3 expression, a previous study with the same primate model demonstrated that αvβ5 was not expressed on micro vessels in the basal ganglia during ischemia (see Results) (Okada et al., 1996). This antibody also cross-reacts with platelet integrin αvβ3 in addition to microvascular αvβ3; however, early after MCAO, the platelet contribution was expected to be low based on previous data (Okada et al., 1994). Working dilutions of each antibody preparation were optimized in preliminary studies.
Immunohistochemistry
Immunoperoxidase studies were performed on either serial 10-μm frozen sections or serial 3-μm paraffin sections. Frozen sections were fixed with a 1:1 (volume/volume [v/v]) mixture of acetone and methanol for 5 minutes and treated with 10 μmol/L glycine in phosphate-buffered saline (100 mmol/L Na2HPO4/NaH2PO4 and 150 mmol/L NaCl adjusted to pH 7.4), then incubated with Blotto to block nonspecific antibody reactions (Johnson et al., 1984). The fixed sections then were incubated with the primary antibody at 37°C for 2 hours, followed by incubation with biotinylated horse (or goat) secondary antibodies to anti-mouse (or rabbit) IgG (Vector Laboratories, Burlingame, CA, U.S.A.) at 37°C for 30 minutes. Avidin–biotin complexes were generated with streptavidin–horseradish peroxidase (Vector Laboratories) and detected with 3-amino-9-ethyl carbazole (AEC kit, Biomeda Corp., Foster City, CA, U.S.A.). The sections were then counterstained with Mayer's hematoxylin (Biomeda Corp.) or were left unstained. Paraffin sections were subjected to the same procedures after deparaffinization. For immunoperoxidase studies with the antisubunit αv polyclonal antibody using paraffin sections, the sections were incubated initially with 1.2 mg/mL trypsin (Sigma BioSciences) at 37°C for 15 minutes to unmask the epitope before the blocking treatment. Routine controls for each experiment included deletion of the primary and secondary antibodies and the use of an irrelevant primary antibody.
DNA scission
Cells displaying DNA fragmentation were detected by incorporation of digoxigenin–deoxyuridine triphosphate (dUTP) with DNA polymerase I as previously described (Tagaya et al., 1997). After fixation with acetone and methanol, frozen sections were incubated with 0.1 U/μL DNA polymerase I (Promega, Madison, WI, U.S.A.) and digoxigenin DNA labeling mixture (Boehringer Mannheim Corp., Indianapolis, IN, U.S.A.) in translation buffer (50 mmol/L Tris-HCl [pH 7.5], 10 mmol/L MgSO4, and 50 μg/mL bovine serum albumin) at 37°C for 2 hours. The sections were subsequently incubated with horseradish peroxidase–conjugated anti-digoxigenin antibody (Boehringer Mannheim Corp.) for 1 hour. The peroxidase signal was developed with 3-amino-9-ethyl carbazole (AEC kit, Biomeda Corp.). Paraffin sections were subjected to the same procedures after deparaffinization and pretreatment with 2 μg/mL proteinase K for 5 minutes. In each section, regions defined by cells incorporating dUTP (dUTP+) and regions peripheral thereto (dUTP−) were defined and designated ischemic core (Ic) and peripheral (Ip), respectively. Nonischemic regions were taken from stereotaxically identical sites in the contralateral basal ganglia.
In situ hybridization
In situ hybridization was performed as described previously using 35S-UTP cyclic RNA probes on paraffin sections only (Seiffert et al., 1991). Both sense and antisense probes labeled with 35S-UTP (Amersham Life Sciences, Arlington Heights, IL, U.S.A.) were synthesized from linearized subcloned plasmids using specific RNA polymerases. A 568-base pair (bp) fragment from residue 89 to 657 of mouse VEGF cDNA (GenBank M95200, kindly provided by Dr. Jeffrey Isner, St. Elizabeth's Medical Center, Boston, MA, U.S.A.) was subcloned into pCR II (Invitrogen Corp., San Diego, CA, U.S.A.), linearized with Not I, and transcribed with SP6 RNA polymerase for the antisense probe, or linearized with BamH I and transcribed with T7 RNA polymerase for the sense probe. A 401-bp fragment from residue 3075 to 3476 of human αv integrin cDNA (GenBank J02826, kindly provided by Dr. Ingrid Stuiver, The Scripps Research Institute, La Jolla, CA, U.S.A.) was subcloned into pGem72f+, linearized with EcoR I, and transcribed with SP6 RNA polymerase for the antisense probe, or linearized with Hind III and transcribed with T7 RNA polymerase for the sense probe.
Deparaffinized PFA-fixed sections were incubated in 0.2 mol/L HCl for 10 minutes and digested with 5 μg/mL proteinase K for 10 minutes. After incubation in prehybridization buffer (50% formamide [v/v], 0.3 mol/L NaCl, 20 mmol/L Tris-HCl [pH 8.0], 5 mmol/L ethylenediamine tetraacetic acid [EOTA], 1% Oenhardt's solution, 10% dextran sulfate (weight/volume), and 10 mmol/L dithiothreitol] at 42°C for 3 hours, the sections were hybridized with 1 × 106 cpm of 35S-labeled riboprobe at 55°C for 18 hours. After hybridization, the sections were washed twice in 2 × sodium chloride/sodium citrate (SSC)/2 mmol/L EOTA, treated with RNase A (5 to 20μg/mL) for 30 minutes, and washed twice again in 2 × SSC/2 mmol/L EDT A. A high-stringency wash of 0.1 × SCC/%bT–mercaptoethanol/EDTA was performed at 55°C for 2 hours followed by washing four times in 0.5 × SSC. The sections were coated with NTB2 emulsion (Kodak, New Haven, CT, U.S.A.) and exposed in a sealed box at 4°C for 4 to 6 weeks. After development, the sections were counterstained with Richardson solution.
Quantitative analysis
The absolute number and size (the minimum transverse diameter) of microvessels that demonstrated signals by immunohistochemical study or by in situ hybridization were quantified with the aid of computerized video imaging microscopy (del Zoppo et al., 1991). A matrix of 100 nonoverlapping microscopic fields at 400× magnification comprised each 6.0-mm2 region of interest. Two identical regions of interest were chosen within the Ic and Ip region, and one region of interest was chosen within the nonischemic region and in the left basal ganglia of control subjects. These regions of interest were superimposed in consecutive sections (immunostaining or in situ hybridization) and used to assess coexpression of the epitopes of interest on single microvessels or in groups of microvessels.
Statistical analysis
All values are expressed as the mean ± SO. Within each animal, microvessels were cross-classified according to the presence or absence of PCNA, VEGF, and integrin αvβ3 antigen or transcript using appropriate probes at 1 hour, 2 hours, or 7 days after MCAO. The corresponding counts were tabulated in multi way frequency tables. Hierarchical log-linear models, a standard statistical technique for categorical data analysis (Bishop et al., 1975), were used to analyze relations among these factors, and, in particular, to assess the null hypothesis that PCNA, VEGF, and αvβ3 expression were independent of one another. The quality of the fit of the log-linear models was assessed with deviance and chi-square statistics, with larger (nonsignificant) P values connoting adequate fits. Calculations were performed in Systat 6.0 for Windows (Wilkinson, 1996). Otherwise, significance was set at 2P < 0.05.
RESULTS
Patterns of nuclear DNA scission after middle cerebral artery occlusion
Cellular dUTP incorporation defined the regions of ischemic injury in the ipsilateral basal ganglia of all MCAO animals. Neurons, astrocytes, and microvascular cells labeled by digoxigenin-dUTP appeared in topographic patterns similar to those previously reported (Tagaya et al., 1997). At 1 and 2 hours after MCAO, dUTP was incorporated into nonvascular cells in clusters but into few microvascular cells within the Ic region. From a total of 66.1 ± 25.8 dUTP+ cells/mm2 in the Ic region at 2 hours after MCAO, only 0.8 ± 0.2 dUTP+ cells/mm2 were associated with microvascular structures. By 7 days after MCAO, the number of dUTP+ microvascular cells had increased to 5.2 ± 2.4 dUTP+ cells/mm2. At any time point, dUTP incorporation was associated predominantly with capillaries and small noncapillary microvessels with a mean diameter of 8.4 ± 3.9 μm (Fig. 1).

Microvascular distribution of deoxyuridine triphosphate (dUTP), proliferating cell nuclear antigen (PCNA), vascular endothelial growth factor (VEGF) and integrin αvβ3 by transverse diameter (μm) in the ischemic core (Ic) and peripheral (Ip) regions of ischemic subjects at various periods after middle cerebral artery occlusion (MCAO) (1 hour [n = 3], 2 hours [n = 3], and 7 days [n = 4]). Deoxyuridine triphosphate and PCNA were expressed by endothelial cells, smooth muscle cells, or both. VEGF and αvβ3 were seen in the myointima. □, 4.0 to 7.5 μm; ■, 7.5 to 30.0 μm;  30.0 to 50.0 μm;
30.0 to 50.0 μm;  more than 50 μm; C, control.
more than 50 μm; C, control.
PCNA expression in microvessels after middle cerebral artery occlnsion
Among all experiments, no detectable evidence of PCNA, integrin αvβ3, or VEGF was seen in any non ischemic basal ganglia. However, PCNA was found within the ischemic basal ganglia at both 1 or 2 hours after MCAO, predominantly in the Ic region. The PCNA+ nonvascular cells were clustered predominantly within the border zone of the Ic region, whereas in noncapillary microvessels PCNA was observed in the endothelium and the medial SMC, where it co-localized equally with VEGF or integrin αvβ3 in the Ic region (Fig. 1, Table 1). The number of microvessels displaying PCNA increased from 1 to 2 hours after MCAO (2.1 ± 1.9 to 6.0 ± 2.4 vessels/mm2, 2P = 0.144), but this difference did not reach significance (Table 2). In the Ip region, only 0.3 ± 0.3 microvessels/mm2 displayed PCNA immunoreactivity at 2 hours after MCAO. By 7 days after MCAO, the number of PCNA immunoreactive microvessels in both the Ic and Ip regions was low (0.4 ± 0.4 and 0.7 ± 0.4 vessels/mm2, respectively).
Colocalization of PCNA with VEGF or integrin α v >β 3 >
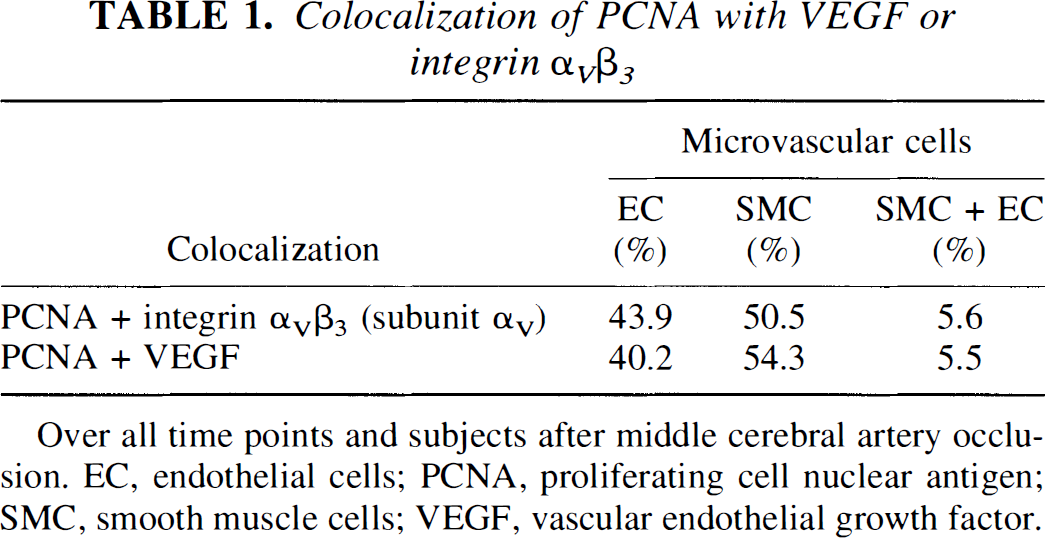
Over all time points and subjects after middle cerebral artery occlusion. EC, endothelial cells; PCNA, proliferating cell nuclear antigen; SMC, smooth muscle cells; VEGF, vascular endothelial growth factor.
Density of microvessels expressing dUTP, PCNA, VEGF, and integrin α v >β 3 >
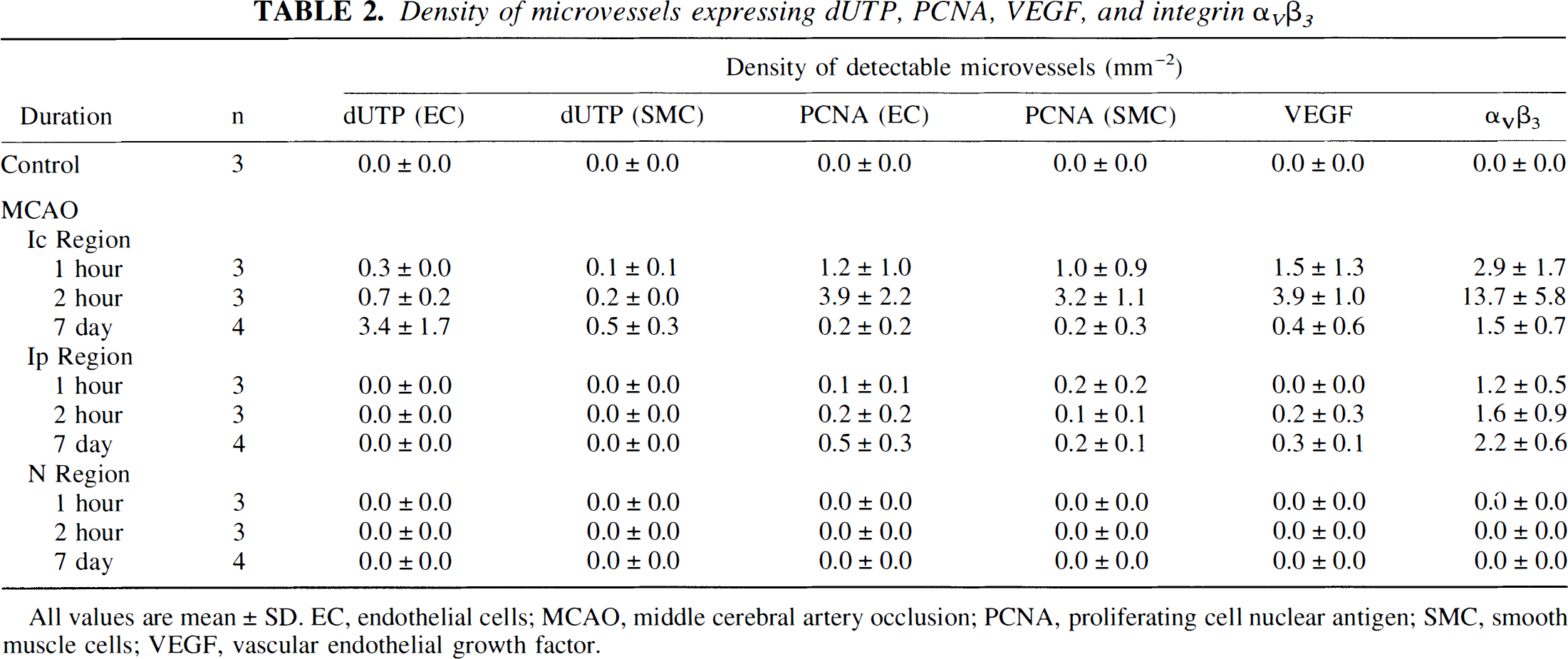
All values are mean ± SD. EC, endothelial cells; MCAO, middle cerebral artery occlusion; PCNA, proliferating cell nuclear antigen; SMC, smooth muscle cells; VEGF, vascular endothelial growth factor.
Integrin αvβ3 expression in microvessels after middle cerebral artery occlusion
Because LM609 consistently cross-reacts with integrin αvβ3 only in frozen tissue, a polyclonal antibody against αv, which cross-reacts with αvβ3 (and αvβ5), was used against the PFA-fixed tissue. A significant cross-correlation between the number of microvessels identified by both antibodies in frozen tissues (n = 10, r = 0.9421, 2P < 0.0001) and between LM609 (frozen) and the polyclonal antibody (PFA-fixed) (n = 10, r = 0.8940, 2P < 0.0001) was shown in preliminary experiments. It was concluded that the polyclonal antibody could identify microvascular αvβ3 expression in PFA-fixed tissue in a nearly equivalent manner to LM609 in the frozen preparations (see Materials and Methods).
No microvessel-associated αv antigen was detected in nonischemic tissues. In the ischemic basal ganglia at 1 and 2 hours after MCAO, subunit αv antigen appeared in the media of 7.5- to 30.0-μm diameter microvessels as previously shown (Okada et al., 1996) (Fig. 1). In the Ic region, the number of αv-immunoreactive microvessels increased from 1 to 2 hours after MCAO (2.9 ± 1.7 vessels/mm2 to 13.7 ± 5.8 vessels/mm2, 2P = 0.065) (Table 2). By comparison, a few microvessels expressing αv antigen also were found in the Ip region at 2 hours after MCAO. By 7 days after MCAO, the number of subunit αv-immunoreactive microvessels in the Ic region also had decreased but was not different from those in the surrounding Ip region (Table 2).
VEGF expression in ischemic microvessels after middle cerebral artery occlusion
At 1 and 2 hours after MCAO, VEGF also appeared in the media of 7.5- to 30.0-μm diameter microvessels in both the Ic and Ip regions (Fig. 1). Within the Ic region, the area of VEGF+ microvessels was completely encompassed by the area of microvessels expressing integrin αvβ3 at all time points (Fig. 2). Here, microvascular VEGF expression increased from 1 to 2 hours MCAO (1.5 ± 1.3 to 3.9 ± 1.0 microvessels/mm2, 2P = 0.105) (Table 2), but there were far fewer VEGF+ microvessels in the Ip region at 2 hours after MCAO (0.2 ± 0.3 microvessels/mm2). By 7 days after MCAO, the number of VEGF-expressing microvessels in the Ic region had decreased to 0.4 ± 0.6 vessels/mm2.

Distribution of microvessel-associated immunoreactive VEGF (black) and integrin αv (gray) in the ischemic basal ganglia of each subject after MCAO. Notice the larger fields of distribution of subunit αv (integrin αvβ3) than VEGF. The numbers below each basal ganglia designate the individual subjects. ( ) αvβ3; (■) VEGF; (
) αvβ3; (■) VEGF; ( ) Overlap.
) Overlap.
The VEGF antigen also appeared in nonvascular cells at 1 and 2 hours after MCAO, including small clusters of astrocytes in the Ip region and in the Ic/Ip border zone. Only within the Ic region, polymorphonuclear (PMN) leukocytes within or adjacent to microvessels or associated with hemorrhage were another nonvascular source of VEGF antigen (data not shown).
Co-localization of PCNA, VEGF, and integrin αvβ3
Proliferating cell nuclear antigen, VEGF, and integrin αvβ3 were detected together on selected microvessels followed through multiple serial sections only within the ischemic basal ganglia (Fig. 3). To determine the strength of the interaction of the three antigens, log-linear models of the three pairs of antigens appearing on the same microvessels in the fixed samples were prepared. With data from the Ic region, it was found that the log-linear models with subjects, and PCNA, VEGF, or αvβ3 expression constituting first-order effects, together with second-order interactions (pairwise interactions between the first-order factors) fit the cell frequencies remarkably well (Table 3). In this context, the significance of pairwise interactions connotes the lack of independence of PCNA, VEGF, and integrin αvβ3 expression. Furthermore, each of the three models (Table 3) was parsimonious in that deletion of any of the second-order interactions (or first-order effects) resulted in a significantly poorer fit.
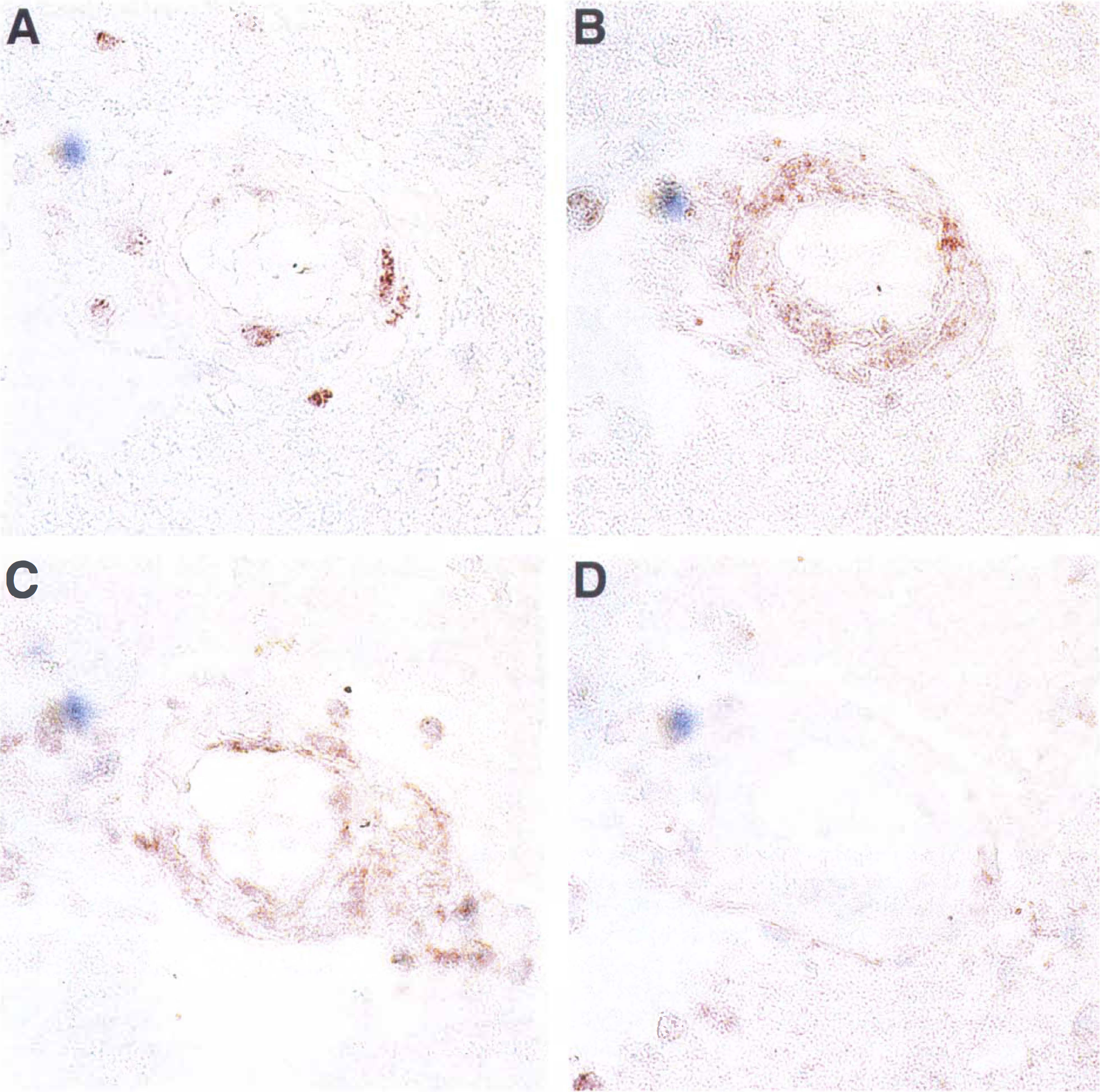
Co-localization of PCNA, VEGF, and αv antigens in a representative 25-μm diameter microvessel at 2 hours after MCAD in serial 3-μm sections (transverse view). The PCNA immunoreactivity
Pairwise interactions

The χ2 statistics are deviances, with nonsignificant values indicative of better fits. PCNA, proliferating cell nuclear antigen; VEGF, vascular endothelial growth factor.
Despite substantial animal-to-animal heterogeneity of expression, subjects constituted a highly significant main effect in this log-linear modeling. The subjects could not be placed into relatively homogeneous subgroups by time (1 hour, 2 hours, or 7 days), as might have been anticipated at the beginning of the study, however. On the other hand, the subjects were found to cluster into three relatively homogeneous subgroups independent of time based on joint PCNA, VEGF, and integrin αvβ3 expression in the Ic region (Fig. 4). Notably, in the Ip region, there was no significant relation among the microvascular expressions of PCNA, VEGF, and integrin αvβ3.
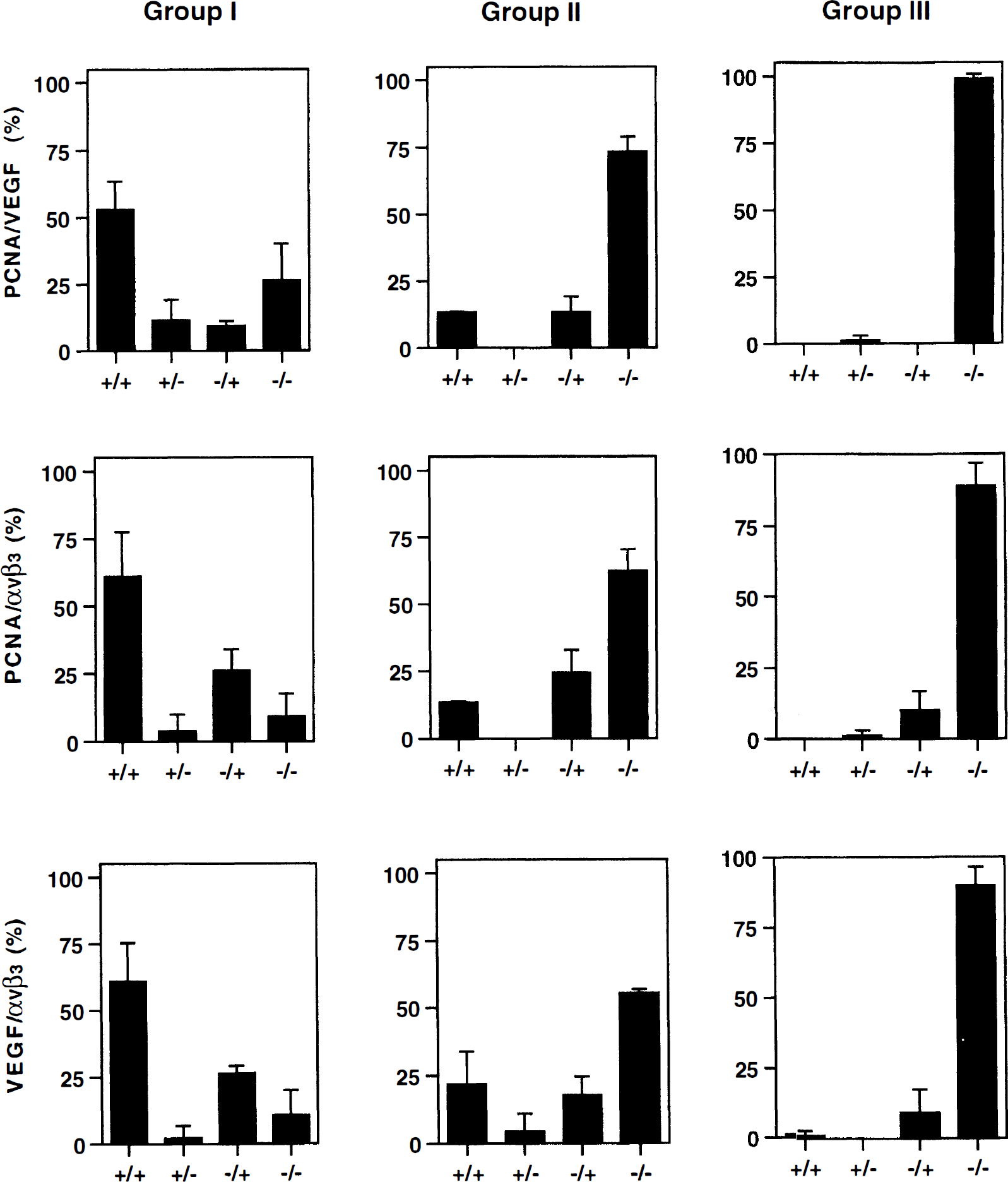
Co-localization of PCNA, VEGF, and integrin αv in microvessels of the Ic region. Microvessels were cross-classified according to the presence or absence of the three antigens. The 10 subjects were distributed into three subgroups. Group I (relatively high expression of PCNA, VEGF, and integrin αvβ3) consisted of five subjects that underwent 1 hour (n = 2) or 2 hours (n = 3) of MCAO. Group II (intermediate group between groups I and III) consisted of two subjects at 7 days of MCAO. Group III (almost no expression of any of PCNA, VEGF, and integrin αvβ3) consisted of three subjects at 1 hour (n = 1) and 7 days (n = 2) of MCAO.
Expression of subunit αv and VEGF mRNA
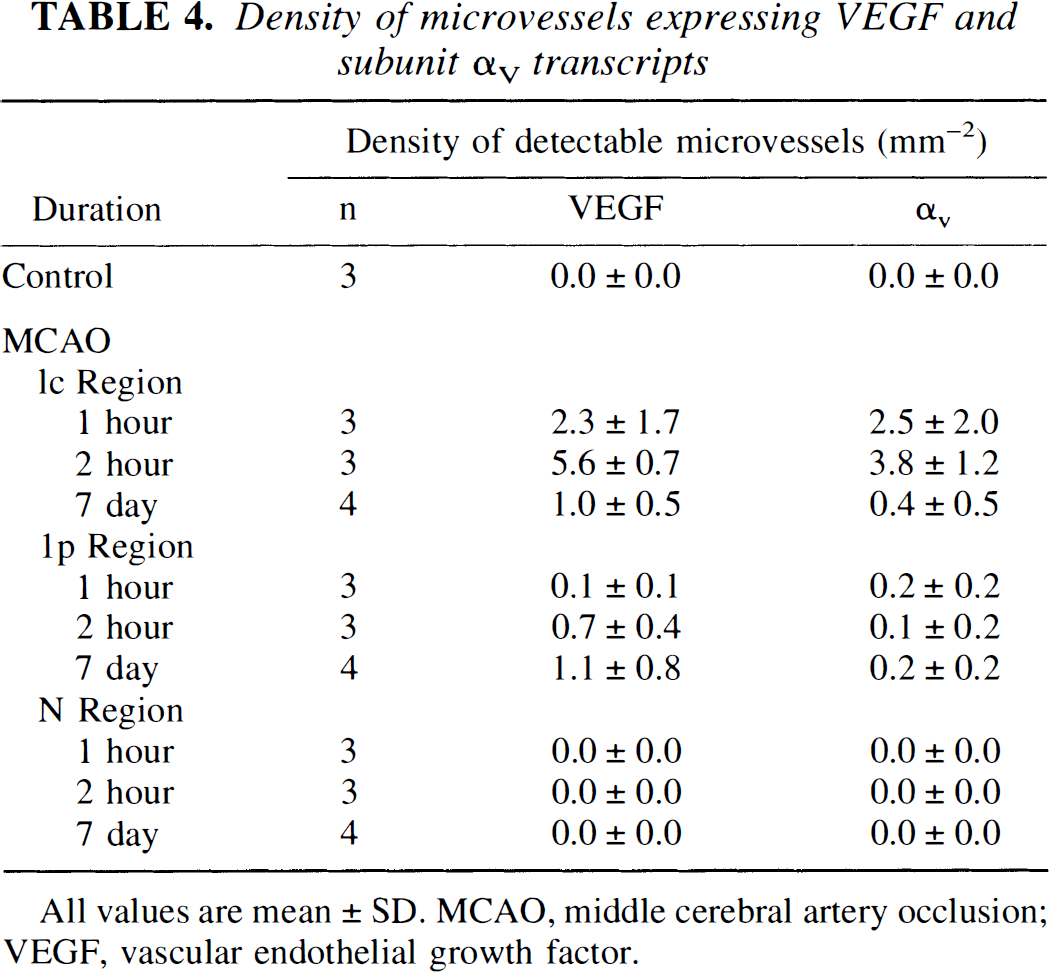
No VEGF or subunit αv mRNA transcripts were detected in the nonischemic regions of any subject. In the ischemic basal ganglia, VEGF and subunit αv mRNA were found on the same microvessels followed through multiple serial sections (Fig. 5). Primarily in the Ic region, subunit αv and VEGF transcripts were detected together in the media of noncapillary microvessels of 7.5- to 30.0-μm diameter, like αvβ3 antigen (Okada et al., 1996) (Fig. 6). From 1 to 2 hours after MCAO, a modest increase in the number of microvessels displaying subunit αv and VEGF mRNA was seen only in the Ic region (Table 4), which paralleled antigen expression. As with group I, which displayed significant co-localization of PCNA, VEGF, and integrin αvβ3 antigens (Fig. 4), the relation of αv subunit to VEGF transcripts in the vascular media of each subject also was significant (2P < 0.05).
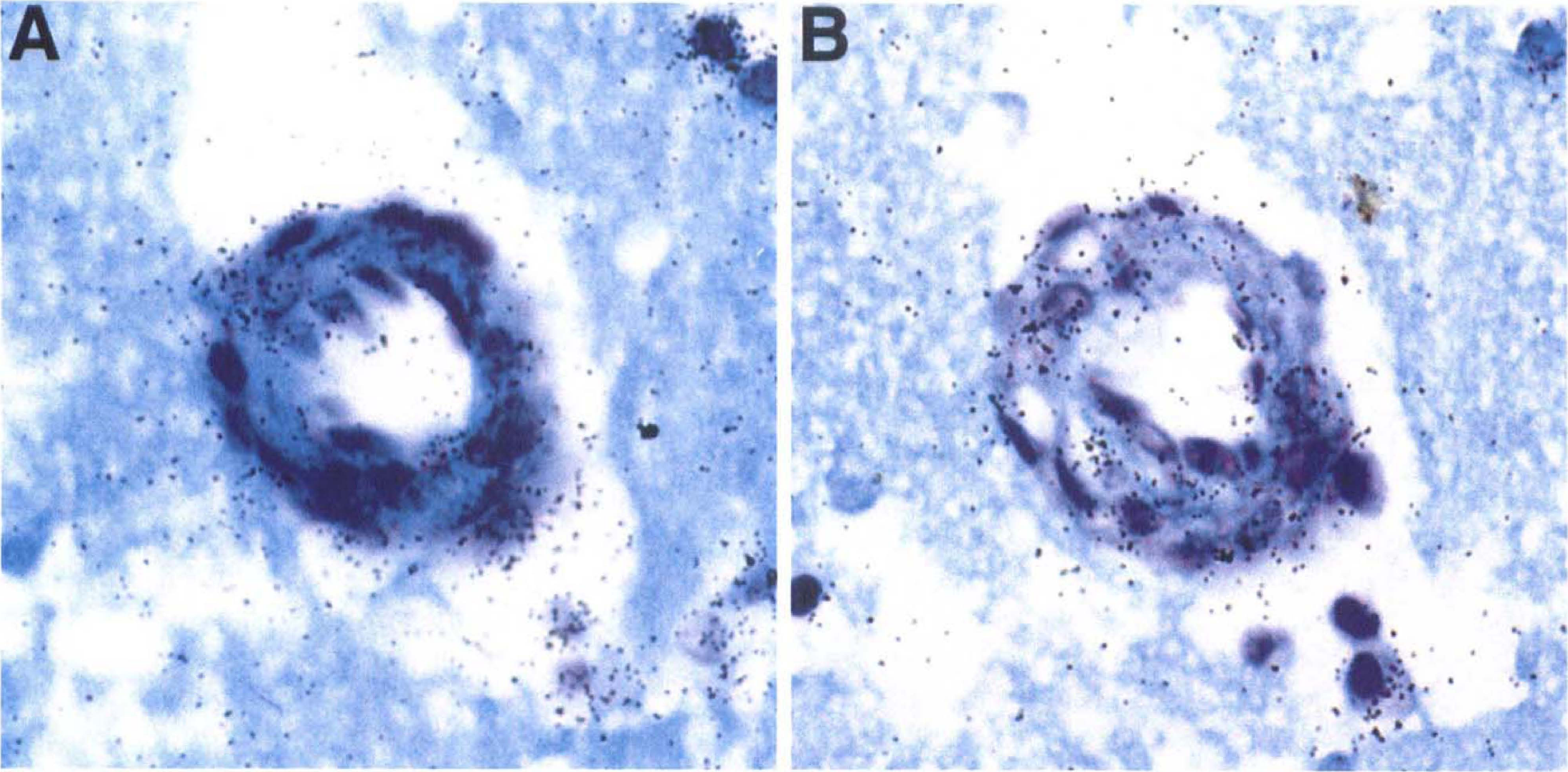
Co-localization of mRNA transcripts of the integrin αv subunit and VEGF in a 22-μm diameter microvessel at 2 hours' MCAO in serial transverse sections. VEGF transcripts

Distribution of microvessel-associated integrin subunit αv and VEGF mRNA transcripts by transverse diameter (μm) in the Ic and Ip regions of ischemic subjects after MCAD (1 hour [n = 3], 2 hours [n = 3], and 7 days [n = 4]). □, 4.0 to 7.5 μm; ■, 7.5 to 30.0 μm;  , 30.0 to 50.0 μm;
, 30.0 to 50.0 μm;  , more than 50 μm; C, control.
, more than 50 μm; C, control.
Density of microvessels expressing VEGF and subunit αvtranscripts
All values are mean ± SD. MCAO, middle cerebral artery occlusion; VEGF, vascular endothelial growth factor.
The VEGF mRNA transcripts also were expressed by astrocytes and PMN leukocytes in the ischemic basal ganglia in a pattern similar to VEGF antigen. At 1 and 2 hours after MCAO only, astrocytes in the Ip region and the Ic/Ip border zone showed VEGF mRNA transcripts.
Interestingly, a relatively strong VEGF mRNA signal also was associated with cells next to some microvessels in the Ic region displaying VEGF antigen in the media. These VEGF-expressing cells had the morphologic features of PMN leukocytes that had penetrated the vascular wall (Fig. 7) or histiocyte-appearing cells localized within the vascular wall (Kato et al., 1996).
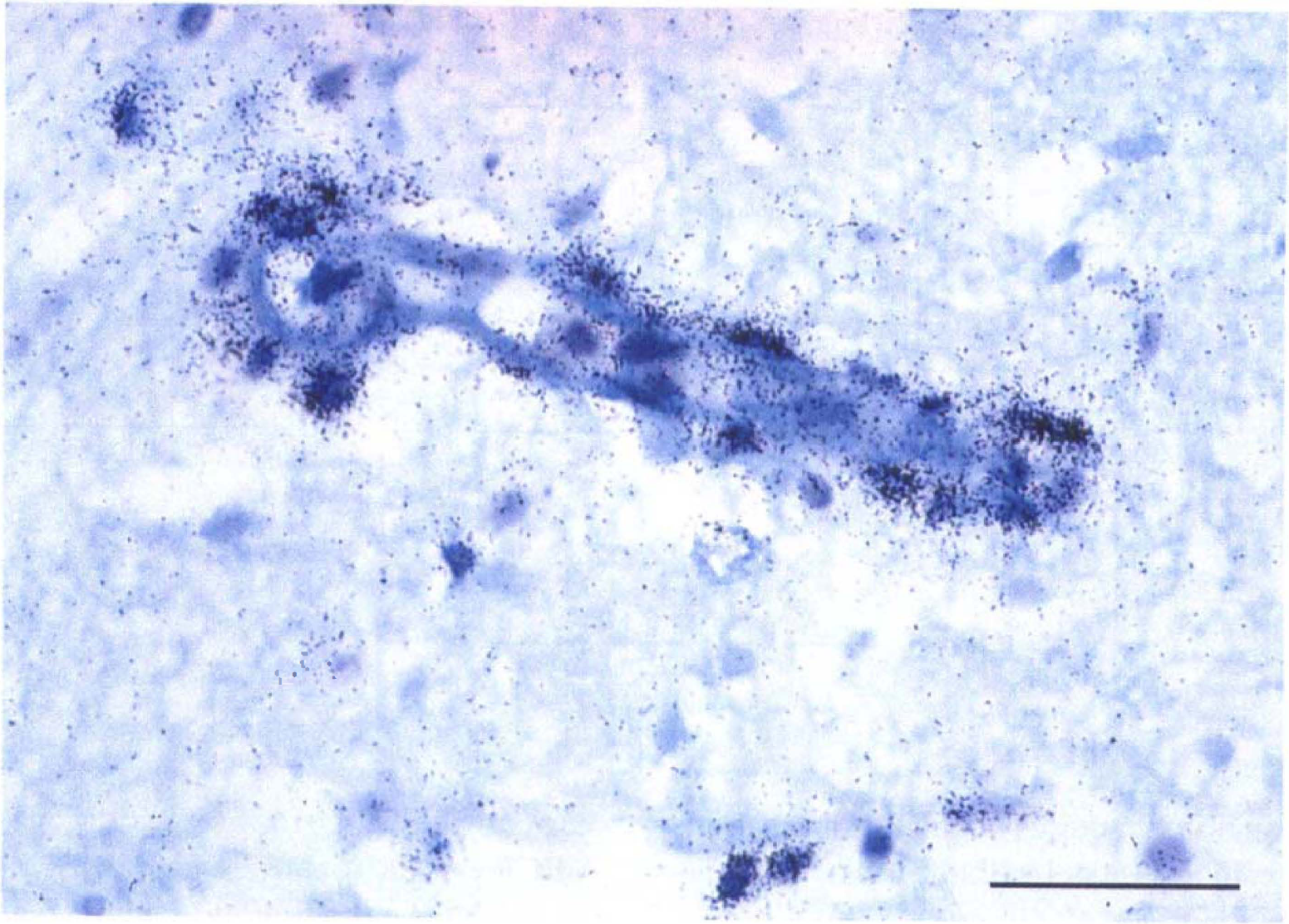
Cells with the morphologic makeup of polymorphonuclear (PMN) leukocytes displaying VEGF transcripts adjacent to a 14.6-μm diameter microvessel in the Ic region at 2 hours' MCAO.
DISCUSSION
The association of integrin αvβ3 with the microvascular deposition of fibrin after MCAD demonstrated by Okada et al. (1996) highlights one rapid response of the cerebral microvasculature to ischemia. Whether and how the presentation of the ligand, fibrin(ogen) in this instance, stimulates microvascular integrin αvβ3 expression is not apparent. One explanation is that integrin αvβ3 up-regulation is one event associated with microvascular activation stimulated by ischemia, and that ligand presentation occurs by independent but parallel processes. Alternatively, integrin αvβ3 presentation may be stimulated by an activator that also increases endothelial cell permeability, exposing the plasma to the tissue factor surrounding the blood vessel (del Zoppo et al., 1992). Senger et al. (Senger, 1996) have shown that exogenous VEGF can induce integrin αvβ3 expression in microvascular endothelial cells in vitro. Also, VEGF is known to increase endothelial cell permeability. Furthermore, both integrin αvβ3 and VEGF up-regUlation have been shown to prevent endothelial cell apoptosis in vitro (Spyridopoulos et al., 1997; Stromblad et al., 1996). Those observations suggest that a specific relation between VEGF and integrin αvβ3 could exist in the responses of microvessels to ischemia in the brain. If true, VEGF and integrin αvβ3 should coincide with evidence of microvascular activation in the initial response of the microvasculature to focal ischemia, and these responses could be time dependent. In this study, the corresponding null hypothesis of no co-localization of PCNA, VEGF, and integrin αvβ3 expression was effectively refuted by the log-linear modeling. Analysis of pairwise associations in individual microvessels demonstrated that a highly significant and coordinated expression of VEGF, integrin αvβ3, and PCNA was provoked in the microvessels of the Ic region beginning within 1 hour after MCAO.
The appearance of PCNA, a 29-kd accessory protein for DNA polymerases δ and ε (Shivji et al., 1995), in microvascular endothelium and SMC suggests a significant immediate microvascular cell response to focal ischemia. Expression of PCNA in the nucleoplasm clearly reflects proliferative responses of cells to growth factors, DNA injury or repair, and other initiators of cell activation during G1 and S phase (Kelman, 1997; Kurki et al., 1988). Expression of PCNA, then, is closely related to cell cycle initiation and could reflect an activated cell proliferative response after MCAO. There has been no report of microvascular or cellular presentation of this antigen in acute cerebral ischemia. Integral to chromosomal DNA replication (polymerase δ) and repair (polymerase ε) (Shivji et al., 1995), PCNA expression may be involved in the processes of DNA repair (Kelman, 1997). As VEGF and integrin αvβ3 expression may be involved in suppression of the cell death pathway (Spyridopoulos et al., 1997; Strombladra et al, 1996), their expression with PCNA may contribute to the preservation of cerebral vessels under ischemic conditions. In contrast to the absence of PCNA in microvascular endothelial cells and pericytes before 2 days in a rodent middle cerebral artery ligation model (Liu, 1994 and Chen, 1994), the current experiments confirm that PCNA expression is one of the earliest cerebral microvascular endothelial cell and SMC responses to ischemia.
Vascular endothelial growth factor acts directly on endothelial cells to stimulate their replication (Connolly et al., 1989). Whereas VEGF did not increase proliferation of aortic SMC in culture (Weatherford et al., 1996) or in large-artery injury models (Asahara et al., 1996), exogenous VEGF has been shown to increase endothelial cell and SMC proliferation in 20 to 100 μm collateral arterioles of the ischemic lower limb in the rabbit (Takeshita et al., 1995; Tsurumi et al., 1996). The cellular responses to VEGF in the skeletal muscle collaterals are consistent with the finding of VEGF in endothelial cells and SMC in PCNA+ cerebral microvessels after MCAO.
Whereas the expressions of VEGF and integrin αvβ3 antigens on microvessels in the Ic region were significantly related, the number and area of distribution of microvessels expressing integrin αvβ3 consistently exceeded those expressing VEGF antigen (Fig. 2, Table 2). This pattern may result from differences in antibody sensitivity, contributions of VEGF from extravascular cellular sources, or the effects of stimulation of integrin αvβ3 by non-VEGF agonists such as basic fibroblast growth factor. Fibroblast growth factor is reported to increase cell surface expression of the integrin αv and β3 subunits in cultured endothelial cells (Sepp et al., 1994).
The consistently larger area of expression of integrin αvβ3 than VEGF also suggests that the two vascular elements may reflect differential responses to one or more products of ischemia. Induction of VEGF, and, perhaps, integrin αvβ3 on microvessels probably results from local hypoxia. Under hypoxic conditions, increased VEGF expression has been detected in cultured astrocytes (Ijichi et al., 1995), tumor cell lines (Minchenko et al., 1994; Berse et al., 1992), and arterial SMC (Brogi et al., 1994). The VEGF and subunit (v genes both contain a hypoxia-inducible factor-1 (HIF-1) response element in their promoter regions, which is activated by the binding of HIF-1 under hypoxic conditions in vivo (Forsythe et al., 1996; Gleadle and Ratcliffe, 1997). The presence of the HIF-1 target sequence 5′-GACGTGGA-3′ (bp 791 to 798 of αv integrin promoter cDNA, GenBank U07375) in both genes implies that induction of VEGF and perhaps subunit αv may be independently regulated by an HIF-1 response. Further proof of the dependence of integrin αvβ3 on VEGF expression requires detailed examination of HIF-1 expression in this model with the use of specific inhibitors.
We failed to find any time relation of joint microvascular PCNA, VEGF, and integrin αvβ3 expression in the Ic region. The known relation of VEGF expression to HIF-1 induction hints at a basis for the lack of temporal relation in these studies. It is likely that in the injured territory of each animal subject, subregions within Ic experience ischemic injury at various times based on the changes or shunting of blood flow during MCAO. The resulting topographic heterogeneity of expression means that the appearance of all three elements by 1 to 2 hours MCAO in a given subject would be “averaged” so that any time dependence of expression, if present, would be obscured.
Two previous studies of VEGF expression in experimental focal cerebral ischemia in rodents differ from the current results. Kovacs et al. describe the appearance of VEGF on macrophages, neurons, glial cells, and endothelial cells in the central ischemic region (from 18 hours to 5 days) and in the peripheral area (from 18 hours to 14 days) after irreversible MCAO in the rat (Kovacs et al., 1996). Separately, Hayashi et al. describe up-regulation of VEGF antigen and mRNA by neurons and glial cells at 1 to 3 hours reperfusion after 1-hour MCAO (Hayashi et al., 1997). The more rapid appearance of microvascular VEGF in the primate basal ganglia is consistent with the increased susceptibilities of neurons to ischemic injury (Tagaya et al., 1997). Other forms of injury to the CNS may trigger VEGF. After cold injury and stab injury in rodent models, VEGF expression in astrocytes was maximal at 3 to 4 days (Papavassiliout et al., 1997), whereas VEGF appeared at various times in PMN leukocytes (1 hour to 1 day), astrocytes (3 to 8 days), macrophages (3 to 8 days), and vascular smooth muscle cells (6 hours to 10 days) (Nag et al., 1997). The reasons for these temporal differences in VEGF appearance in individual cell groups of nonprimate species are not known but may represent differences in species, model preparation, and injury type. In any event, the early coexpression of VEGF and integrin αvβ3 in the primate represents a prompt response of noncapillary cerebral microvessels to focal ischemia that may have a broad temporal presentation with persistent cerebral injury.
One possible effect of VEGF and integrin αvβ3 expression is the stimulation of angiogenesis downstream. However, there is limited information about the late vascular responses to focal cerebral ischemia. Tsutsumi et al. demonstrated capillary bud formation by 7 days after MCAO in the infarct zone in a canine stroke model (Tsutsumi, 1986; Tsutsumi et al., 1986). Chuaqui and Tapia subsequently demonstrated that in the infarcted zone, whereas most brain microvessels disappeared by 7 days after stroke onset, a subgroup of small normal-appearing muscular arterioles was frequently preserved within the first 4 weeks, followed by an increase in their number (Chuaqui and Tapia, 1993). In stroke patients who survived from 5 to 92 days, the number of microvessels within the infarcted hemisphere was seen to increase (Krupinski et al., 1994). In the current study, there was no clear evidence that microvessel density increased in either the Ic or the Ip region of the ischemic basal ganglia by 7 days MCAO (data not shown). However, the presence of VEGF and integrin αvβ3 within Ic suggests that signals associated with vascular proliferation are generated early. The precise mechanism of VEGF and integrin αvβ3 up-regulation and of their relation to neovascularization after focal cerebral ischemia are not known.
This report indicates that the early appearance of integrin αvβ3 and VEGF correlate with microvascular activation and the activation of intravascular coagulation, leading to fibrin formation (Okada et al., 1996). Thrombin generation may provide a link between microvascular reactivity and permeability, and the local cleavage of fibrinogen (Aschner et al., 1997; Garcia et al., 1996). It has been reported that osteopontin, a ligand for integrin αvβ3, is up-regulated late (more than 5 days) after MCAO in the cerebral parenchyma of the rat (Wang et al., 1998). Ligation of osteopontin to integrin αvβ3 is enhanced when osteopontin is cleaved by thrombin (Senger et al., 1996). Although osteopontin may induce migration of both endothelial cells and SMC, no relation to the early microvascular responses to ischemia has been reported.
The rapid appearance of PCNA, VEGF, and integrin αvβ3 on microvessels in the ischemic core also coincides with evidence of neuron injury in the nonhuman primate (Tagaya et al., 1997). Possible relations that may determine whether the activation of the cerebral microvessels are linked to neuron injury have yet to be explored. However, a highly significant correlation between neuron injury (or size of ischemic injury) and the appearance of pro-MMP-2 beginning within 1 hour after MCAO in this model has been shown (Heo et al., 1999). Zucker et al. have shown that exogenous VEGF stimulates endothelial cells to generate MMP-2 (Zucker et al., 1998). In the presence of coagulation factors, VEGF also may generate thrombin, known to activate pro-MMP-2 (Zucker et al., 1998). It has been proposed that the effects of MMP-2 on the microvascular basal lamina may be related to neuron injury (Heo et al., 1999). With the association of TIMP-1 and MMP-1 expression to VEGF stimulation in both tumor and endothelial cell culture systems (Unemori et al., 1992; Yoshiji et al., 1998; Zucker et al., 1998), additional pathways to microvascular remodeling and thrombin generation may contribute to neuron injury.
To explore the downstream events from integrin αvβ3 expression, interventional approaches using peptides containing the RGD sequence, which effectively block integrin αvβ3 function in tumor angiogenesis, may be tried (Brooks et al., 1994). Similarly, functional inhibitors of VEGF may prove useful to specifically define its relation to integrin αvβ3 expression and microvascular responses to ischemia. Ultimately, it is of interest whether the coexpression of VEGF and integrin αvβ3 on activated cerebral microvessels leads to changes in microvascular integrity during ischemia, to microvascular remodeling and the generation of new vessels, and to alterations in neuron integrity.
