Abstract
Perinatal hypoxic-ischemic (H/I) brain injury remains a major cause of neurologic disability. Because we have previously demonstrated that this insult depletes cells from the subventricular zone (SVZ), the goal of the present investigation was to compare the relative vulnerability to H/I of neural stem cells versus progenitors. The dorsolateral SVZs of P6 rats were examined at 2 to 48 hours of recovery from H/I using hematoxylin and eosin, in situ end labeling (ISEL), terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate-biotin nick end labeling (TUNEL), electron microscopy, and immunofluorescence. Pyknotic nuclei and ISEL+ cells were observed by 4 hours of recovery, peaked at 12 hours, and persisted for at least 48 hours. Many active-caspase3+ cells were observed at 12 hours and they comprised one third of the total TUNEL+ population. Electron microscopy revealed that hybrid cell deaths predominated at 12 hours of recovery. Importantly, few dying cells were observed in the medial SVZ, where putative stem cells reside, and no nestin+ medial SVZ cells showed caspase-3 activation. By contrast, active-caspase-3+/PSA-NCAM+ progenitors were prominent in the lateral SVZ. These data demonstrate that early progenitors are vulnerable to H/I, whereas neural stem cells are resilient. The demise of these early progenitors may lead to the depletion of neuronal and late oligodendrocyte progenitors, contributing to cerebral dysgenesis after perinatal insults.
Hypoxic-ischemic (H/I) brain injury as a result of asphyxia at term remains a major cause of neurologic disability. The prominent histopathological features of perinatal H/I are germinal matrix infarction, periventricular hemorrhage, intraventricular hemorrhage, as well as diffuse and focal gray and white matter damage (Towbin, 1998; Volpe, 2001). Despite major advances in our understanding of CNS development and perinatal diseases, the data are incomplete regarding the mechanisms leading to brain injury and failure of recovery.
Perinatal insults occur during an epoch in CNS development when there is extensive proliferation, migration, and cellular differentiation. A prominent structure in the immature brain is the germinal matrix, which is a general term that encompasses the precursors in both the ventricular and subventricular zones. In the 29th to 40th week of gestation of the human brain, the germinal matrix is largely comprised of cells of the subventricular zone (SVZ). The precursors that reside in the SVZ generate interneurons, astrocytes, and oligodendrocytes during the late fetal and newborn periods (Brazel et al., 2003). In light of our recent data demonstrating that SVZ cells are affected by H/I (Levison et al., 2001; Rothstein and Levison, 2002), we investigated which cells in the SVZ are vulnerable to H/I, whether the cells die classic apoptotic or necrotic deaths, when they die, and finally whether caspase-3 participates in their demise.
To study the effects of perinatal H/I on the cells of the SVZ, we used a modification of the widely used Rice/Vannucci postnatal rat model to produce unilateral brain damage (Rice et al., 1981; Vannucci et al., 1996). Here we show that there is a variety of cell deaths related to the interval of recovery from the insult. Interestingly, at the peak of cell death, the majority of the dying cells in the SVZ exhibit features of hybrid forms of cell death likely resulting from excitotoxicity brought about from this injury (Portera-Cailliau et al., 1997a, 1997b). Importantly, we show that few cells die in the most medial aspect of the SVZ, which is where the neural stem cells are located. By contrast, cell death is most prominent in the lateral SVZ, and many of these cells stain for active caspase-3 and the early progenitor cell enriched protein, polysialic acid neural cell adhesion molecule (PSA-NCAM). Taken together, these data demonstrate that neural stem cells are resistant to injury after perinatal H/I, whereas PSA-NCAM+ early neural progenitors are vulnerable. The demise of these early progenitors may lead to the depletion of neuronal and late oligodendrocyte progenitors, contributing to cerebral dysgenesis after H/I.
MATERIALS AND METHODS
Reagents
Unless specified, standard laboratory reagents were obtained from VWR (Bridgeport, NJ, U.S.A.) and Fisher Scientific (Pittsburgh, PA, U.S.A.). Paraformaldehyde and glutaraldehyde were purchased from Electron Microscopy Sciences (Fort Washington, PA, U.S.A.). Normal goat serum was purchased from Gibco (Grand Island, NY, U.S.A.). Rabbit primary antibodies raised against active caspase-3 were purchased from R&D Systems (Minneapolis, MN, U.S.A.) and Cell Signaling Technology (of New England BioLabs, Beverly, MA, U.S.A.). Mouse IgM monoclonal antibodies against PSA-NCAM were purchased from Chemicon (Temecula, CA, U.S.A.). Mouse IgG1 monoclonal antibodies raised against nestin (Rat 401) were purchased from the Developmental Studies Hybridoma Bank. Appropriate biotin-labeled secondary antibodies or fluorochrome conjugated secondary antibodies were purchased from Southern Biotechnology (Birmingham, AL, U.S.A.), Jackson Immunoresearch (West Grove, PA, U.S.A.) or Molecular Probes (Eugene, OR, U.S.A.). The Klenow fragment of DNA polymerase I was purchased from Worthington Enzymes (Freehold, NJ). Terminal transferase and digoxigenin reagents were purchased from Roche (Indianapolis, IN, U.S.A.). Horseradish peroxidase substrate Nova RED was purchased from Vector (Burlingame, CA, U.S.A.).
Perinatal hypoxia ischemia model
Timed Pregnant Wistar rats were purchased from Charles River Laboratories (Charles River, Wilmington, DE, U.S.A.), housed in individual cages and fed high-fat lab chow. After normal delivery, the litter size was adjusted to 10 pups per litter. Three different groups of animals were used: Controls, hypoxia controls, and experimental hypoxic/ischemic animals. Cerebral H/I was produced in male and female 6-day-old rats (day of birth being P0) by a permanent unilateral common carotid ligation followed by hypoxia (Rice et al., 1981; Vannucci et al., 1996). Briefly, pups were lightly anesthetized with halothane (4% induction, 1.5% maintenance). Once fully anesthetized, a midline neck incision was made and the right common carotid artery was identified. The common carotid artery was separated from the vagus nerve and then ligated using 3–0 silk. Animals were returned to the dam for 2 hours. Before exposure to hypoxia (8% O2/92% N2), the pups were prewarmed in jars for 20 minutes in a 37°C water bath. The pups were then exposed to 1.5 hours of hypoxia. After hypoxia, the pups were returned to their dam for recovery periods of 4, 12, 24, and 48 hours, at which time they were anesthetized and killed by intracardiac perfusion. Hypoxia controls received 1.5 hours of 8% oxygen balance nitrogen without common carotid artery ligation. Other controls were untouched.
Intracardiac perfusions, tissue fixation, and sectioning
Animals were perfusion fixed with 3% or 4% paraformaldehyde in 0.1 mol/L phosphate buffer. Brains were removed and immersion fixed in the same fixative overnight at 4°C. The brains were then coronally blocked and prepared for paraffin embedding using standard methods or frozen for cryostat sectioning. For frozen sections, the brains were coronally blocked and placed in 30% sucrose (with 0.05% sodium azide) overnight (with 2 changes). The following day, the tissue was mounted in cassettes with O.C.T. and frozen over a bath of ethanol and dry ice. Coronal sections were cut 15-μm thick at the level of the anterior commissure, -0.3 to -0.6 bregma and held at -80°C until used. Six-micron-thick paraffin sections were cut at the level of the anterior commissure, -0.3 to -0.6 bregma. All sections were mounted onto superfrost plus slides.
Terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate-biotin nick end labeling and in situ end labeling
Paraffin-embedded sections were deparaffinized and rehydrated through an alcohol series (50%, 70%, 95%, and 100%; 2 minutes each) and rinsed twice in Tris-buffered saline. Next, sections were incubated in 5 μg/mL proteinase K (Roche), followed by a 10-minute incubation of 1% peroxides in MeOH, and incubated for 20 minutes in terminal transferase buffer. Sections were incubated for 1 hour at room temperature in 300 U/mL terminal transferase enzyme and 10 mmol/L DIG DNA labeling mixture in terminal transferase buffer and then incubated in terminal transferase stop solution for 10 minutes (Gavrieli et al., 1992). Sections were rinsed, incubated for 10 minutes at room temperature in TGB superblock (Tris buffered, 10% Goat serum, 10% Bovine serum albumin), and then incubated overnight at 4°C in horseradish peroxidase conjugated sheep anti-digoxigenin in TGB diluent. Sections were rinsed extensively and the staining was visualized with VECTOR Nova Red horseradish peroxidase substrate kit and briefly counterstained in Mayer's hematoxylin (20 seconds), then dehydrated through an ascending alcohol series ending with xylene and coverslipped with cytoseal (Stephens Scientific, Kalamazoo, MI, U.S.A.). In situ end labeling (ISEL) was performed as described previously (Wijsman et al., 1993; Fehsel et al., 1994; Santarosa et al., 1994; Levison et al., 2001).
Immunofluorescence
Immunofluorescence was performed using standard methods. After dewaxing, slides were incubated in 10 mmol/L citrate buffer (pH 6.0) at 80°C for 1 hour and then cooled to room temperature for 2 hours. After blocking, the sections were incubated in antibodies against active caspase-3 (1:250 [R&D]; 1:1,000 [Cell Signaling], nestin [Rat 401] [1:2], or PSA-NCAM [1:2,000]). After extensive washes, the sections were incubated in appropriate secondary antibodies with or without biotynlated-tyramide signal amplification (Perkin Elmer Life Sciences, Boston, MA, U.S.A.). All slides were counterstained with 4'6'–Diaminido-2 phenylindole (DAPI) signal amplification. All slides were counterstained with DAPI and coverslipped with GEL/MOUNT (Biomeda, Foster City, CA, U.S.A.). Images were captured using a Photometrics cooled charged coupled device camera (Tucson, AZ, U.S.A.) interfaced with IP Lab scientific imaging software (Scanalytics Fairfax, VA, U.S.A.). In addition, z-series images of caspase-3+/PSA-NCAM+ cells were captured using a Leica SP2 confocal microscope Leica Microsystems, Exton, PA, U.S.A.).
Cell counts
Two methods were used to quantify cell death within the dorsolateral SVZ (SVZDL). One method quantified the incidence of cell death within a cross section of the SVZDL, whereas a second method quantified cell death within specific regional domains. Because the entire SVZDL cannot be easily demarcated, and because our goal was to compare the vulnerability of cells within subregions of the SVZDL, profile counts were performed, adapting the methods of stereology (Guillery and Herrup, 1997). Animals with a damage score of 10 to 15 were used for profile counts in the SVZDL (Towfighi and Mauger, 1998). To analyze the incidence of cell death across the dorsolateral SVZ, the SVZDL was separated into three distinct regions: medial, mediolateral, and lateral. We defined the medial region as the highly dense region of SVZDL cells that extends approximately 80 μm from the lateral ventricle. The mediolateral region extends from approximately 80 μm to 200 μm from the lateral ventricle, and the lateral tail extends from 200 μm to the most lateral tip of the SVZDL. First the total number of pyknotic or ISEL+ cells were counted in the SVZDL at 60X under oil immersion. Next cell profiles were counted at 60X using a Photometrics CCD camera interfaced with IP Lab scientific imaging software. A grid was placed over the monitor and all cell profiles within the grid border were counted. Cell profiles intersecting left-side and top grid borders were counted, while cell profiles that intersected either of the other two grid borders were not counted (Kroustrup et al., 1988). The average cell density value for the SVZDL was multiplied by the total area of the SVZDL to determine the cellularity of the SVZDL. The total number of pyknotic nuclei and ISEL+ cells was then calculated as a percentage of the average cellularity of the SVZDL. All values represent the average counts taken from two sections per brain with three to eight animals assessed at each time point. A second method was used to assess cell death within four regionally distinct fields within the SVZDL. Four fields within the SVZDL were capture at 60X (Fig. 2). Each field represented an area of 80 μm × 100 μm. The total number of dying cells was counted for each field. Next, the total number of cells per field was quantified.
Electron microscopy
Specimens were prepared for electron microscopy as described previously (Levison et al., 2001; Rothstein and Levison, 2002). An analysis similar to that described earlier was performed to assess regional differences in the vulnerability to H/I and to characterize the types of cell deaths. Images from the medial, mediolateral, and lateral SVZDL were assessed for occurrence of apoptotic, necrotic, and hybrid cell types. The data are presented as (1) the percentage of each type of cell death with respect to the total number of dying cells observed, and (2) the average number of that specific cell type reported regionally in the SVZDL.
Statistical analyses
Data were evaluated for statistical relevance by analysis of variance (ANOVA) using Stat View software (SAS Institute Inc, Carey, NC, U.S.A.) with Bonferroni/Dunn post-hoc analysis. Values provided are means ± SD. Results are considered significant when P < 0.05.
RESULTS
The perinatal SVZDL is vulnerable to hypoxic—ischemic insults
The goals of this study were to determine the relative vulnerability of SVZDL cells to perinatal hypoxia/ischemia, the time course and types of cell death caused by this injury, and the mechanism(s) for these deaths. To assess the time course of cell death after perinatal H/I, the fraction of pyknotic nuclei at 4, 8, 12, and 24 hours of recovery within the SVZDL was assessed (Fig. 1). Pyknotic cells were defined as cells with small, round, darkly stained nuclei with eosinophilic cytoplasm (Fig. 1B). A significant increase in the number of pyknotic cells in the ipsilateral SVZDL was apparent as early as 4 hours of recovery when compared to the contralateral SVZDL. The occurrence of pyknotic nuclei in the ipsilateral hemisphere tripled by 8 hours of recovery, and peaked at 12 hours of recovery (achieving approximately a fivefold increase in pyknotic cells in the ipsilateral SVZDL versus the contralateral SVZDL). The incidence of pyknotic cells began to decrease at 24 hours of recovery. At all time points, pyknotic cells were less abundant in the contralateral SVZDL than in the ipsilateral SVZDL (Fig 1 and Fig. 2). To determine whether the slight increase in pyknotic cells observed in the contralateral hemisphere could be attributed to a bystander effect, we compared the incidence of pyknotic cells in the damaged hemisphere versus naïve and sham-operated control hemispheres at 8 and 12 hours of recovery (Fig. 2). At both time points, there was an increase in the number of pyknotic nuclei observed in the contralateral SVZDL. A statistical analysis, however, revealed that the slight increase in pyknotic cells in the contralateral hemispheres was insignificant. By contrast, there were approximately 6 times more pyknotic cells in the ipsilateral SVZDL at 12 hours of recovery than in the contralateral SVZs of sham-operated and naïve animals, which is an increase that is comparable to that determined by assessing the ipsilateral versus contralateral SVZs of an H/I animal (Fig. 2).
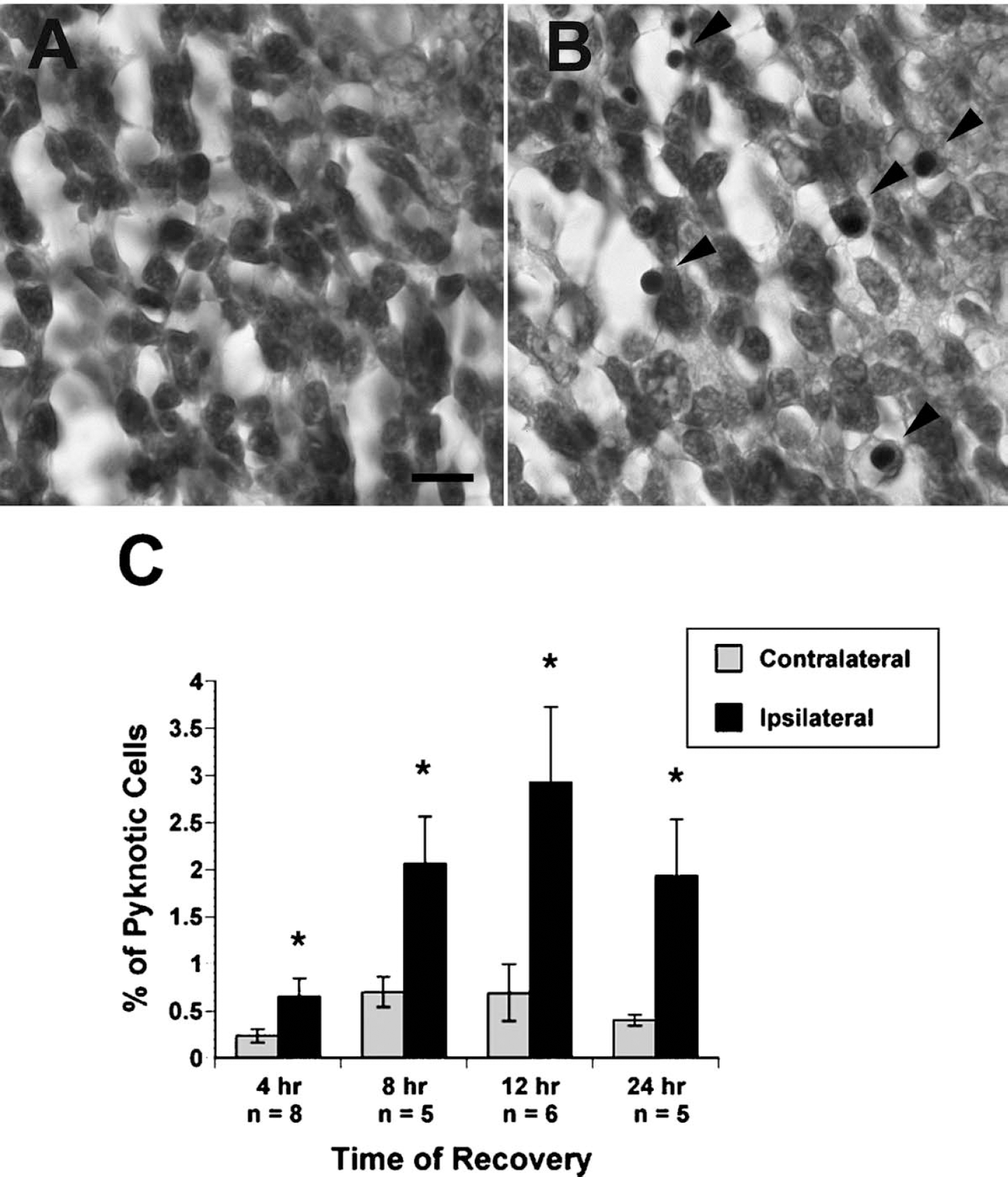
Pyknotic nuclei increase in the ipsilateral dorsolateral subventricular zone (SVZDL) after 90 minutes of hypoxic-ischemic (H/I) and peak at 12 hours of recovery. Six-micron paraffin-embedded tissue sections taken at the level of the anterior commissure (−0.6 bregma) were stained with Mayer's hematoxylin and eosin. (
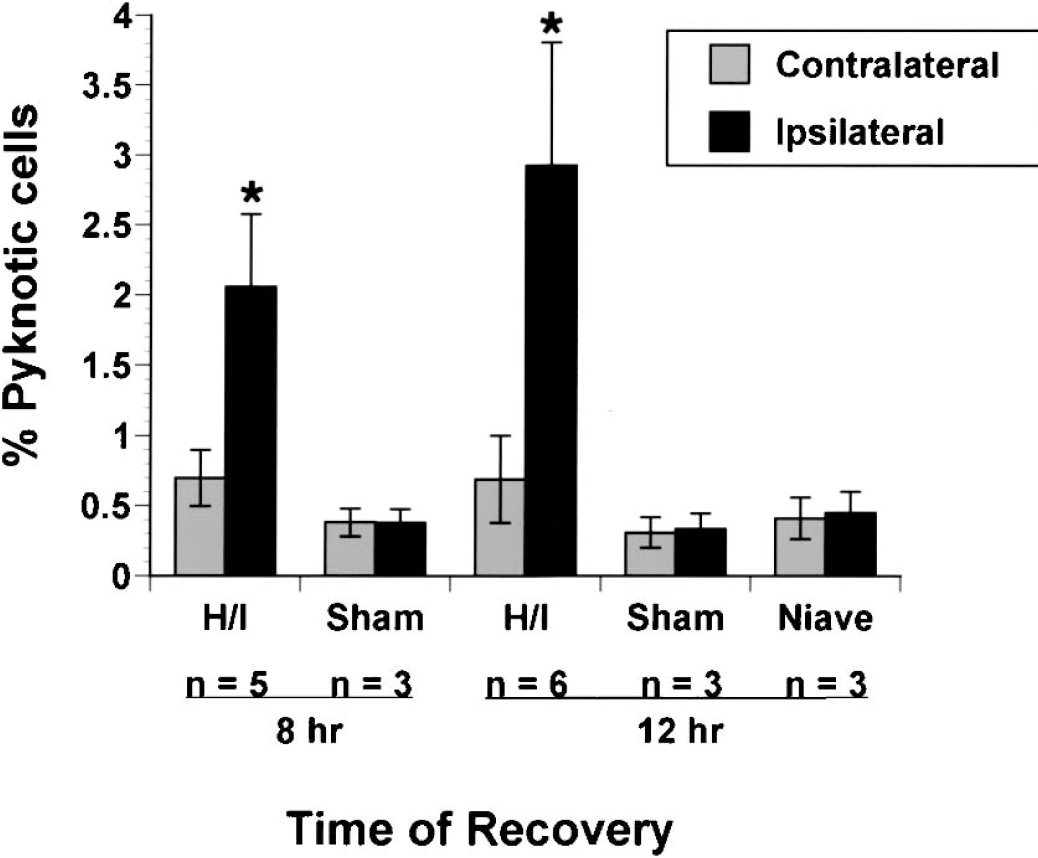
The percentage of pyknotic nuclei increases in the ipsilateral dorsolateral subventricular zone (SVZDL) when compared to contralateral, sham, and naive control SVZs. The number of pyknotic nuclei was counted from 6-μm paraffin-embedded sections taken at the level of the anterior commissure (−0.6 bregma) and stained with Mayer's hematoxylin and eosin. Pyknotic cells were counted at 60x under oil immersion from 2 non-adjacent sections and presented as the percent pyknotic cells ± SD with respect to the total number of cells in the SVZDL. Number of animals (n) analyzed at each time point is indicted. *Significantly different from the contralateral SVZDL by analysis of variance followed by Bonferroni/Dunn post hoc t-test P < 0.0008.
ISEL+ cells comprise a subset of cells in the early stages of cell death after the injury
We next used ISEL to more specifically assess the occurrence of early apoptotic deaths at 4, 12, 24, and 48 hours of recovery. As shown in Fig. 3, there was a significant increase in the incidence of ISEL+ cells in the ipsilateral SVZDL at all time points when compared to the contralateral SVZDL. In addition, the majority of cells stained by ISEL were pyknotic, but not all pyknotic cells were labeled by this method (Fig. 3B). A significant elevation in early apoptotic events was detected by 4 hours of recovery in the ipsilateral SVZDL, and the incidence of ISEL labeled cells in the ipsilateral SVZDL peaked at 12 hours of recovery when there was an approximately fivefold increase in ISEL+ cells (Fig. 3C). ISEL+ cells persisted to 24 hours in the ipsilateral SVZDL and then began to decrease by 48 hours, but ISEL+ cells remained elevated in the ipsilateral versus contralateral SVZDL (Fig. 3C).
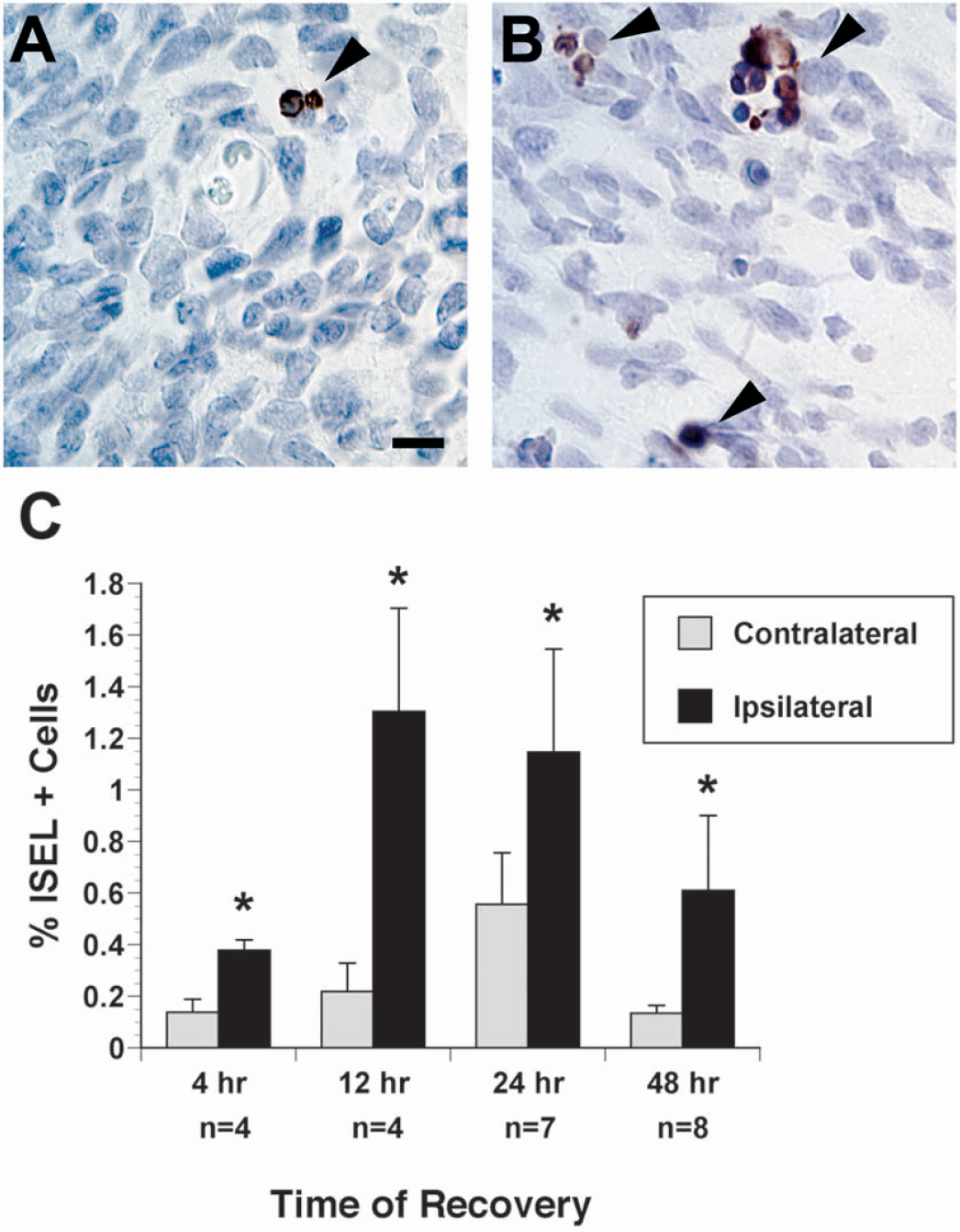
In situ end labeling+ cells become prevalent in the ipsilateral dorsolateral subventricular zone (SVZDL) after 90 minutes of hypoxic-ischemic (H/I) and peak at 12 hours after the insult. Six-micron paraffin-embedded tissue sections taken at the level of the anterior commissure were stained using ISEL, visualized with Nova Red horseradish peroxidase substrate and counterstained briefly in Mayer′s hematoxylin. Depicted are representative examples from the contralateral hemisphere
We should emphasize that these counts only indicate the level of instantaneous cell death that is occurring at any given time point. Because dying cells within the SVZ have a clearance rate of 2.4 hours, our estimates of cell death are clearly underestimates, which explains why there is a net reduction in the cellularity of the SVZ over time (Thomaidou et al., 1997; Levison et al., 2000, 2001).
Caspase-3 is regionally activated in the SVZDL at 12 hours of recovery from perinatal H/I
Caspase-3 activation is implicated as a common effector of cell death after brain injury; therefore, we used immunofluorescence for active-caspase-3 at 12 hours of recovery, when pyknotic nuclei and ISEL staining were maximal. Using two different anti-active caspase-3 antibodies, cells with strong cytoplasmic labeling could be identified within the ipsilateral SVZDL (Fig. 4A). Because it is becoming well established that there are subdomains with the SVZDL, cell counts were taken from four defined fields with the SVZDL: dorsal and ventral regions of the medial SVZDL, the mediolateral, and the most lateral aspect of the SVZDL. The percentage of active caspase-3+ cells in each given field was assessed at 12 hours of recovery (Fig. 4D). Active caspase-3 activation was detected in both the contralateral and ipsilateral SVZs, achieving a greater than twofold increase in the entire ipsilateral SVZDL versus the entire contralateral SVZDL after 12 hours of recovery (Fig. 4D). Importantly, more cells in the lateral aspects of the ipsilateral SVZDL stained for active-caspase 3 than those in the more medial fields. In the most medial fields (#1 and #2) there was no significant difference in the percentage of active-caspase-3-positive cells in either the ipsilateral or contralateral SVZs (Fig. 4D). In the more lateral regions (fields #3 and #4), a threefold increase in active-caspase-3-positive cells was observed (Fig. 4D). In summary, these data show that caspase-3 activation is one mechanism that mediates the cell death in the SVZDL after H/I. Moreover, cells residing in the most lateral aspect of SVZDL are most vulnerable to H/I.
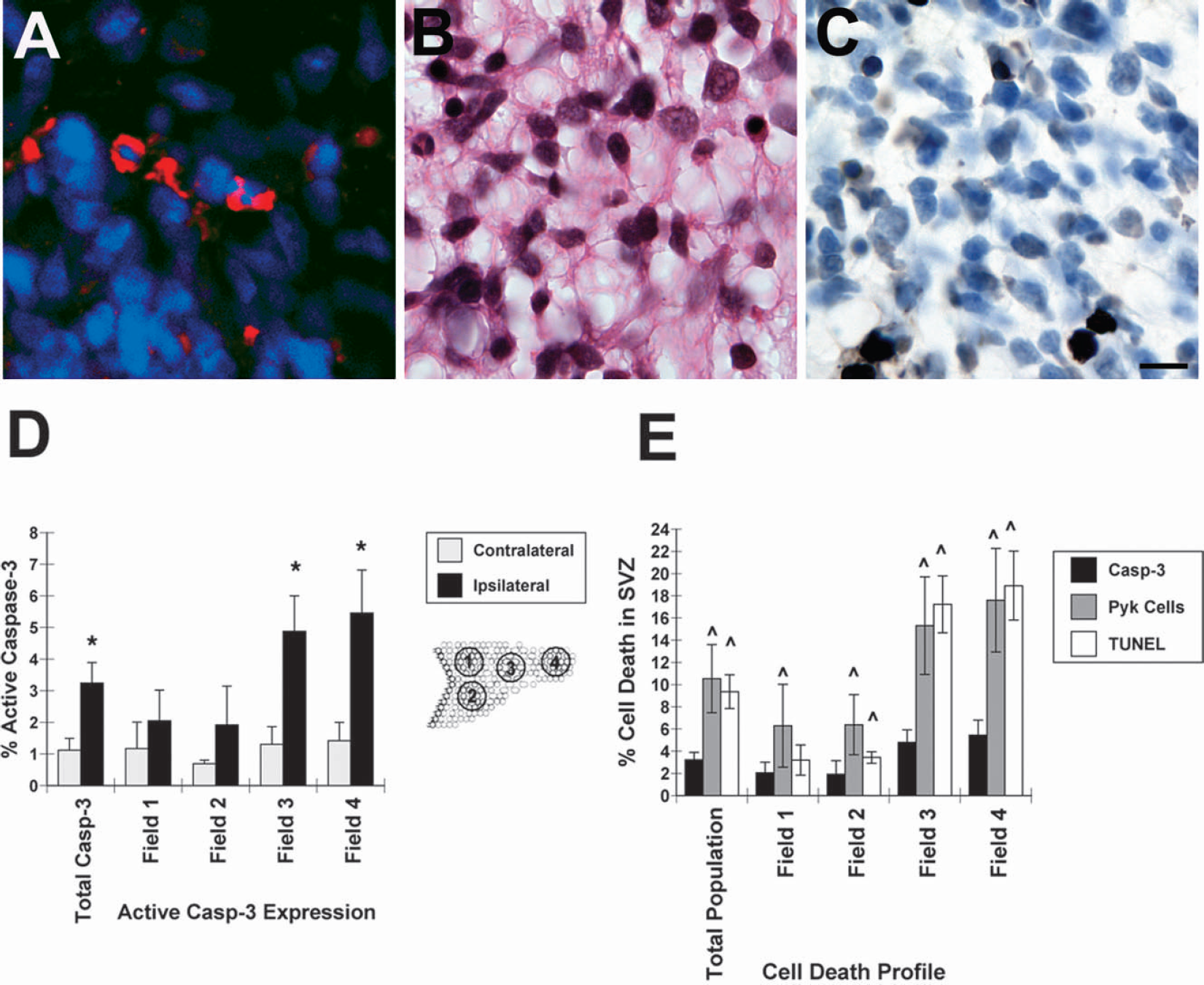
Activated caspase-3, terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate-biotin nick end labeling (TUNEL) staining, and pyknotic nuclei are elevated in the ipsilateral dorsolateral subventricular zone (SVZDL) at 12 hours after 90 minutes hypoxic-ischemic (H/I). Six-micron paraffin-embedded tissue sections taken at the level of the anterior commissure from 5 H/I brains at 12 hours of recovery were stained for active caspase-3
Caspase-3 is a death effector implicated in approximately one-third of the cell death observed at 12 hours of recovery
We sought to determine what percentage of cell death is mediated by caspase-3 activation. To accomplish this, tissue from the brains of 5 animals harvested at 12 hours of recovery were assessed for the occurrence of pyknotic, TUNEL+, and active caspase-3+ cells (Fig. 4B,Fig. 4C). Unlike the ISEL method, which labels nicks in double-stranded DNA, the TUNEL method labels free 3′ ends and, therefore, labels a much broader population of dying cells. Interestingly, regardless of the subdomain within the SVZDL, the number of TUNEL+ and pyknotic cells was always higher than the incidence of active caspase-3+ cells (Fig. 4E). For instance, in the mediolateral region (field #3) the number of caspase-3+ cells represented approximately 4.8% of the total cells in this region; however, the percentage of TUNEL-labeled and pyknotic nuclei is approximately 4 times greater (17.2% and 15.3%, respectively) (Fig. 4E). This discrepancy was also observed in the lateral tail region of the SVZDL (field #4), where active-caspase-3+ cells represented 5.5% of the total cell population, whereas 18.92% and 17.61% of the total cells were TUNEL labeled or pyknotic, respectively (Fig. 4E). On average, active caspase-3+ cells comprise one third of the reported cell deaths observed in the SVZDL versus TUNEL labeling and pyknotic nuclei counts. In agreement with the regional differences in caspase-3 activation noted earlier, there was an increased incidence in both pyknotic cells and TUNEL+ cells in the more lateral regions of the SVZDL.
PSA-NCAM+ progenitors but not neural stem cells are vulnerable to perinatal H/I
Because the SVZDL is known to harbor both neural stem cells and progenitors, we next performed analyses to determine whether any of the caspase-3+ cells might be stem cells or progenitors. First we compared the regional distributions of the stem cell—enriched marker, nestin, and the progenitor cell—enriched marker, PSA-NCAM in the SVZDL. Filamentous nestin labeling was observed throughout the SVZDL, but the strongest nestin labeling was seen in those cells closest to the lateral ventricles (Fig. 5A). By contrast, PSA-NCAM was not present in the most medial regions of the SVZDL, whereas it was strongly expressed in more lateral regions (Fig. 5B). Double immunofluorescence for active-caspase-3 and either PSA-NCAM or nestin was performed on tissue sections at 12 hours of recovery to determine whether those cells with active-caspase-3 might preferentially label for either nestin or PSA-NCAM. Cells double-positive for PSA-NCAM were seen especially in the mediolateral and lateral regions of the SVZDL, and these cells represented a subset of all of the active-caspase-3-positive cells (Fig. 5C–Fig. 5E). Nestin immunofluorescence increased within the ipsilateral SVZDL, and nestin+ cells remained more prevalent in the most medial aspects of the SVZDL; however, nestin/active caspase-3 double-positive cells were never observed (Fig. 5G). In fact, although some active-caspase-3+ cells could be observed in the medial region, those cells that showed caspase-3 activation were not nestin+ (Fig. 5G inset).
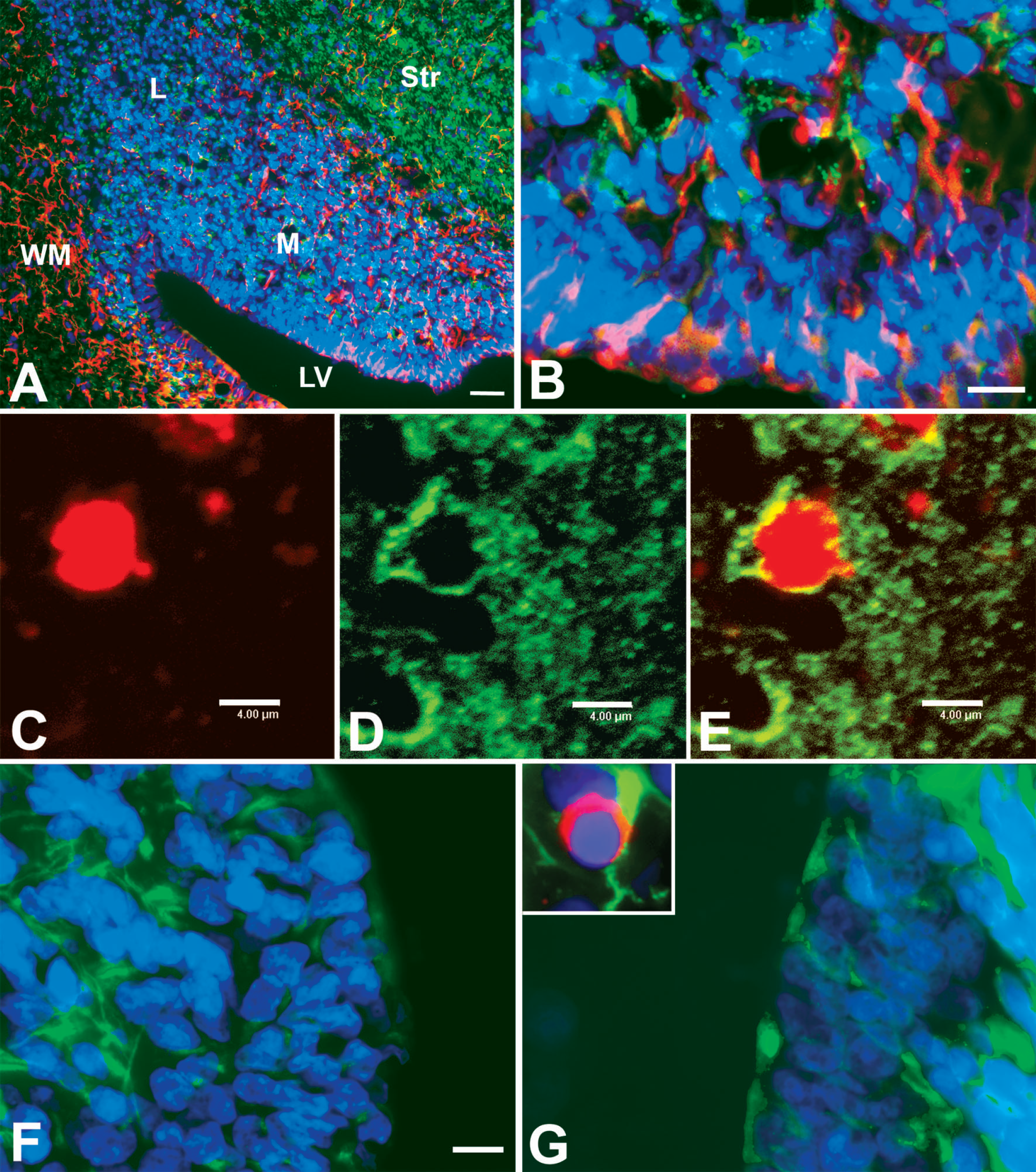
Nestin+ cells in the medial SVZDL are not active-caspase-3+. Cryostat sections taken at the level of the anterior commissure from hypoxic-ischemic animals at 12 hours of recovery were stained for nestin (Rhod) and polysialic acid neural cell adhesion molecule (PSA-NCAM) (Alexa 488) and counterstained with DAPI
Hybrid cell deaths predominate at 12 hours after perinatal H/I
To more clearly characterize the nature of cell death in the SVZDL, electron microscopy was used at 12 hours of recovery. By electron microscopy, apoptotic and necrotic figures as well as cells possessing features of both types of cell death (hybrids) could be observed (Fig. 6A). Interestingly, the majority of cells in the SVZDL at 12 hours of recovery could be classified as hybrid cell deaths because these cells exhibited evenly condensed chromatin (typical for apoptotic cells) with electrolucent cytoplasm, swollen organelles, and loss of membrane integrity (typical for necrotic cells) (Fig. 6D). Cells characterized as hybrids comprised 61% of the total dying cells observed (Fig. 6E and Table 1). A subset of dying cells exhibited classic apoptotic features (Fig. 6C), and a minority showed clear necrotic characteristics (Fig. 6B) (27.8% and 10.8%, respectively). Importantly, there were a number of healthy cells present in the injured SVZDL, some of which were actively dividing or phagocytosing dead or damaged cells. Additionally, there was evidence of macrophage and microglial infiltration into the SVZDL to aid in the phagocytosis of dead cells. Confirming our light microscopic observations, very little cell death was observed in the most medial region of the SVZDL.
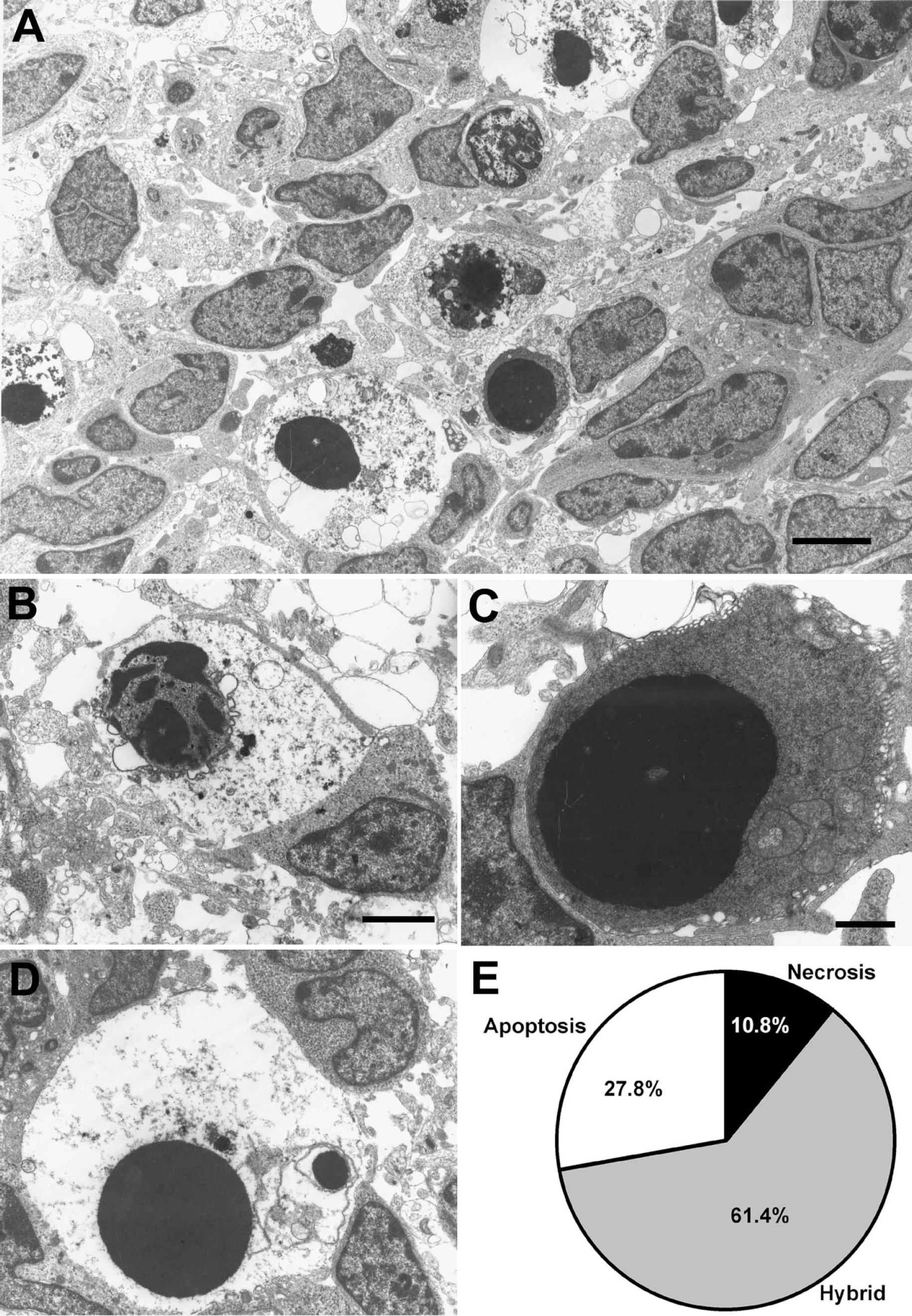
Electron microscopy shows prevalence of hybrid cells in the ipsilateral dorsolateral subventricular zone (SVZDL) after 90 minutes of hypoxic-ischemic. Ipsilateral SVZs at 12 hours after the insult were prepared for electron microscopy. (A) Low-power picture revealing many dying cells with assorted features.
Regional distribution of cell death types in the SVZDL at 12 h of recovery as assessed by electron microscopy after 90 min perinatal hypoxia/ischemia
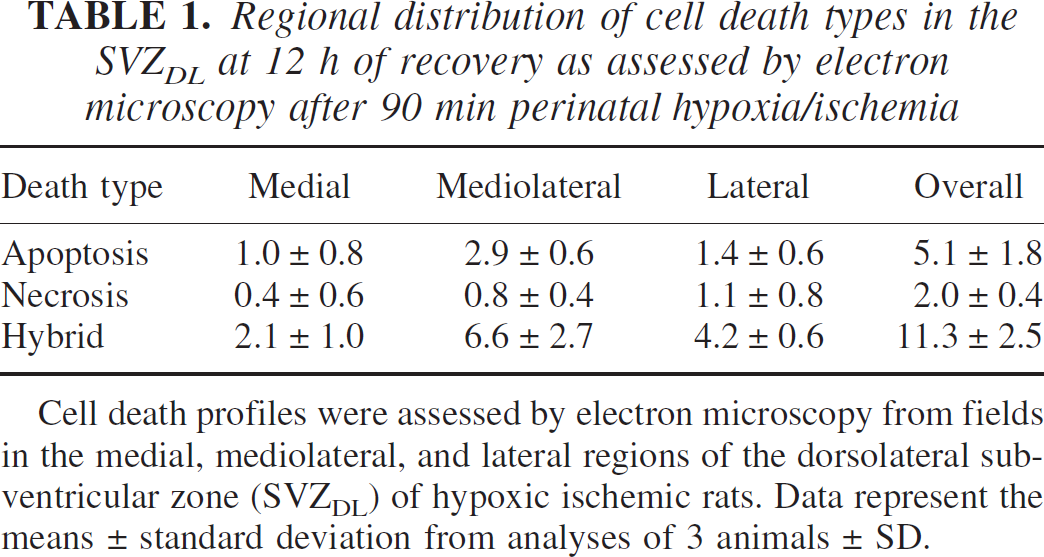
Cell death profiles were assessed by electron microscopy from fields in the medial, mediolateral, and lateral regions of the dorsolateral subventricular zone (SVZDL) of hypoxic ischemic rats. Data represent the means ± standard deviation from analyses of 3 animals ± SD.
DISCUSSION
The goal of the present investigation was to determine the relative vulnerability of the cells within the SVZDL to a perinatal hypoxic-ischemic insult. Here we report that (1) there is selective cell death within the SVZDL; (2) PSA-NCAM+ progenitors in the lateral SVZDL are vulnerable, whereas nestin+ cells in the medial SVZDL are resilient; (3) the majority of cell deaths occur at 12 hours of recovery, and these cells have a hybrid morphology; and (4) approximately one third of all cell deaths are mediated by caspase-3 at 12 hours of recovery. Although we had previously reported a depletion of neural stem cells as a consequence of H/I by 3 weeks of recovery, data provided here suggest that these cells are not eliminated during the early recovery period.
Neural stem cells in the medial SVZDL resist injury during early recovery from perinatal H/I
Neural stem cells are undifferentiated, nestin+, slowly dividing cells that give rise to PSA-NCAM enriched progenitors that mature into the neurons and glia of the CNS. Evidence that neural stem cells are located within the medial SVZDL comes from progenitor depletion and repletion experiments, microdissection, immunopanning, and promotor-based selection experiments (Chiasson et al., 1999; Johansson et al., 1999; Kawaguchi et al., 2001; Kirschenbaum et al., 1999). Previously we have shown that ependymal cells swell by 4 hours of recovery after an H/I insult (Rothstein and Levison, 2002). Other studies using this model have shown that cells of the ependymal and subependymal layers are in a state of high oxidative/hypoxic stress as visualized by EF5 adduct immunostaining (Bergeron et al., 1999). However, despite these indications that the cells in the medial SVZDL are under considerable stress as a consequence of H/I, the ependymal cells and those cells immediately subjacent to them within the SVZDL resist damage (Rothstein and Levison, 2002). At the light and electron microscopy levels, ependymal cells and those SVZDL cells immediately subjacent were rarely apoptotic or necrotic. Furthermore, active caspase-3+ cells were extremely rare among these cells, and no nestin+ cells in the most medial region of the SVZ showed pyknotic morphology. Indeed, the number of active caspase-3+ cells in the medial SVZDL of the ipsilateral hemisphere was equivalent to the number of active-caspase-3+ cells in the contralateral medial SVZDL. Importantly, double immunofluorescence showed no caspase-3 activation in nestin+ cells in the medial SVZDL. Furthermore, using a neurosphere assay to quantify the number of sphere-forming cells in the ipsilateral and contralateral hemispheres, we find no decrease in the number of sphere-forming cells in the ipsilateral hemisphere between 24 and 72 hours of recovery (Snyder, 2001). Taken together, these data strongly suggest that neural stem cells are resistant to injury during the early stages of recovery from perinatal H/I.
Neural stem cells may be intrinsically resilient to death effectors during the early stages of recovery
The low number of dying cells within the medial SVZDL suggests that these cells may be intrinsically resilient to death effectors. Observations that support this hypothesis are as follows: (1) Glycogen granules are found within ependymal and subependymal cells, which would serve as an energy substrate during H/I, thus enabling these cells to survive this insult (Blakemore, 1969); (2) Ependymal and subependymal cells greatly rely upon anaerobic respiration, which also renders them resistant to H/I (Cammermeyer, 1965; Shimizu et al., 1957); (3) Neural stem cells have higher levels of anti-apoptotic Bcl-2 and Bcl-XL, which would endow them with resistance to cell death stimuli (Brazel, 2003). Additionally, large blood vessels are prominent in the medial regions of the germinal matrix of the human as well as in the rodent SVZDL, affording rapid reperfusion during recovery (Towbin, 1998). The hypothesis that these cells are not as severely affected by the insult is less tenable because those cells situated in the most medial region of the SVZDL become significantly swollen early after perinatal H/I, indicating that they are indeed affected by this insult (Rothstein and Levison, 2002). Previously, at much later time points of recovery (3 weeks), we reported a depletion and reduction of the SVZ. We hypothesize that astrocyte production and gliosis associated with recovery at this time may lead to a terminal recruitment of stem cells to an astrocytic fate. Our best hypothesis is that as a consequence of increased production of cytokines, changes in cell-to-cell contacts, and altered growth and trophic factor production, the SVZ stem cells terminally differentiate into astrocytes, resulting in their depletion.
PSA-NCAM+ progenitors in the lateral SVZDL are vulnerable to perinatal H/I
PSA-NCAM plays a critical role in morphogenesis of the nervous system and is expressed on a subset of migrating progenitors that give rise to both neurons and glia (Blass-Kampmann et al., 1994; Doetsch et al., 1997; Doetsch et al., 1999; Grinspan and Franceschini, 1995; Lois and Alvarez-Buylla, 1993). Consistent with previous studies, we found that PSA-NCAM is present throughout the developing cortex, white matter, and striatum of the postnatal day 6 to 8 rat brain with no differences on either ipsilateral and contralateral hemispheres at 12 hours of recovery (data not shown). SVZDL cells also express PSA-NCAM, but not uniformly. Importantly, ependymal cells and medial SVZDL cells do not label for PSA-NCAM, whereas PSA-NCAM is expressed in the lateral SVZDL (Szele et al., 1994). Previous studies also reported that putative neural stem cells do not express PSA-NCAM (Doetsch et al., 1999). Because the majority of damaged cells are located in the lateral aspect of the SVZDL, and because we found active caspase-3+/PSA-NCAM+ cells in the mediolateral and lateral SVZDL, these observations suggest that progenitors in the more lateral aspects of the SVZDL are vulnerable to perinatal H/I injury.
Late oligodendrocyte progenitors are highly vulnerability to perinatal H/I brain injury. In addition, here we show that cells at an earlier stage of restriction than previously characterized also are vulnerable. We reported that immature oligodendrocytes and their progenitors, expressing the markers Yp and NG2, died in the periventricular white matter at 12 hours after H/I (Levison et al., 2001; Ness et al., 2001). We also showed progressive dysmyelination and subsequent gliosis in the periventricular white matter along with the permanent depletion of the SVZDL after long-term recovery (Levison et al., 2001). Our results are in general agreement with those of Back and colleagues, that H/I affects latestage oligodendrocyte progenitors in both rodents and humans in this disease model (Back et al., 1998, 2001, 2002). Consistent with these observations, the expression of both myelin basic protein and proteolipid protein are reduced in a mouse model of perinatal H/I (Skoff et al., 2001). The PSA-NCAM+ progenitor cells in the lateral SVZDL that are vulnerable to perinatal H/I are likely neuronal progenitors and oligodendrocyte preprogenitors, based on their abundance and location (Doetsch and Alvarez-Buylla, 1996; Grinspan and Franceschini, 1995). Their vulnerability has not been previously suspected. Although the P7 rat model produces more extensive forebrain damage than is seen in infants with periventricular leukomalacia, the demonstration that these preprogenitors are depleted provides a new avenue for exploring the mechanisms of cerebral dysgenesis and the progressive dysmyelination associated with periventricular leukomalacia (Back et al., 2001; Huppi et al., 2001; McQuillen et al., 2003; Sizonenko et al., 2003; Volpe, 2001). It will be most interesting to establish whether these preprogenitors also are depleted in the P2 rat model, where the damage is more restricted to white matter (Sheldon et al., 1996).
Caspase-3 is a likely mediator of progenitor cell death after perinatal H/I
Caspase-3 is a major death effector in perinatal brain damage (Gill et al., 2002; Hu et al., 2000; Northington et al., 2001; Wang et al., 2001; Zhu et al., 2000). We report a large increase in the numbers of SVZDL cells expressing active-caspase-3 at 12 hours of recovery after the insult. We also have observed strong caspase-3 activation in the neocortex and striatum by 12 hours of recovery, consistent with previous reports (Gill et al., 2002; Hu et al., 2000; Northington et al., 2001; Wang et al., 2001; Zhu et al., 2000). A critical role for caspases in perinatal brain damage is supported by studies reporting retention of neural tissue by intraventricular infusion of the pan-caspase inhibitor BAF after a perinatal H/I insult (Cheng et al., 1998). Our investigations more clearly define the therapeutic window whereby the administration of anti-apoptotic agents would be most potent in preventing cell death and subsequent tissue loss.
Hybrid cell deaths predominate during the peak of cell death, which suggests excitotoxic injury
Electron microscopy revealed a range of cell death morphologies after perinatal H/I. In our previous analysis, necrosis predominated during early recovery, but with reperfusion, cells having characteristics of both necrotic and apoptotic cells appeared and at later intervals pure apoptotic figures were observed (Levison et al., 2001). Here we show that at the time of maximal cell death within the SVZDL, the majority of the dying cells within the SVZDL have the features of hybrids. The nature of hybrid cell deaths previously described were attributed to excitotoxicity brought about by the increase in extracellular excitatory amino acids as a result of the injury (Portera-Cailliau et al., 1997a, 1997b). H/I causes increased release of excitatory amino acids neurotransmitters that open ionotropic channels allowing an influx of calcium, other cations, and water into receptive cells. Increased intracellular calcium is observed after perinatal H/I in the organelles and cytoplasm of affected neurons and oligodendrocytes, with many calcium-laden mitochondria rupturing by 3 hours of recovery (Puka-Sundvall et al., 2000). Elevated cytosolic calcium will activate m-calpain, which can then activate caspase-3. Calpain activity, as visualized by fodrin cleavage, precedes caspase activation, and evidence of calpain activation is seen in cells with active-caspase-3. Consistent with this scenario, attenuating calpains with CX295, 2-leuaminobutyricacid-CONH(CH2)3-morpholine decreases caspase-3 activation in a model of perinatal H/I (Blomgren et al., 2001). Given this result, and our data showing a predominance of hybrid cells at 12 hours of recovery in the SVZDL, we also hypothesize that, as in the cortex, calcium dysregulation and calpain activation leads to caspase-3 activation and cell death in the PSA-NCAM+ progenitors. However, because caspase-3 activation only accounted for one third of the total TUNEL+ cell deaths, other cell death effectors may be involved that may act independent of caspase-3 (Han et al., 2001; Harrison et al., 2001).
CONCLUSIONS
These data show that perinatal H/I results in death of progenitors within the lateral SVZDL, which peaks at 12 hours and persists to 48 hours of recovery. By contrast, nestin+ cells in the more medial SVZDL show no evidence of caspase-3 activation or pyknosis, which strongly suggests that neural stem cells are more resilient. Many of the dying cells found more laterally in the SVZDL were PSA-NCAM+ and are likely early neural progenitors. The demise of these early progenitors may reduce the numbers of later progenitors. The demise of these neuronal and glial progenitors may contribute to the diseases of periventricular leukomalacia and cerebral dysgenesis associated with injury to the term infant.
Footnotes
Acknowledgments
The Rat-401 hybridoma developed by Susan Hockfield was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, IA 52242.
