Abstract
Many studies showed beneficial effects of either erythropoietin (EPO) or mesenchymal stem cells (MSCs) treatment in cerebral ischemia. In addition to a neuroprotective role, not only EPO but also MSC favors neurogenesis and functional recovery. In an attempt to further improve postischemic tissue repair, we investigated the effect of a systemic administration of MSC, in the presence or not of EPO, on neurogenesis and functional recovery in a transient focal cerebral ischemia model in the adult rat. Twenty-four hours after ischemia, the rats were divided into four groups, namely vehicle, MSC, EPO, and MSC + EPO, and received a single intravenous injection of MSC (2times106 cells) and/or a repeated intraperitoneal administration of EPO (1,000 UI/kg) for 3 days. The lesion volume, the MSC outcome, neurogenesis, and functional recovery were assessed 51 days after ischemia. The results showed that cellular proliferation and neurogenesis were increased along the lateral ventricle wall in the MSC + EPO group, whereas no significant effect was observed in groups receiving MSC or EPO alone. This effect was accompanied by an improvement of mnesic performances. Mesenchymal stem cells expressing neuronal or glial markers were detected in the ischemic hemisphere. These results suggest that EPO could act in a synergistic way with MSC to potentiate the postischemic neurogenesis.
Introduction
Despite intense research efforts, stroke remains a major cause of morbidity and mortality. Mesenchymal stem cells (MSC) are known to have therapeutic benefits after cerebral ischemia (see for review Phinney and Isakova, 2005). In addition to their differentiation into osteoblasts, chondroblasts, adipocytes, and hematopoiesis-supporting stromal cells, MSC seem to have the potential to differentiate into neurons and glia
In addition to its neuroprotective effect in the treatment of stroke (see for review Grasso et al, 2007), erythropoietin (EPO) also plays a role in the proliferation and differentiation of neuronal progenitors and stem cells. Indeed,
Preclinical data showed that some combined treatments after cerebral ischemia could be an interesting approach and could be more efficient than a cellular or a molecular treatment alone (Chu et al, 2005; Hanabusa et al, 2005; Zhang et al, 2007). We thus hypothesized that EPO administration in combination with MSC may have synergistic effects on neurogenesis and functional recovery after ischemia. In this context, we investigated the effects of a cotreatment with MSC and EPO, administered 24 h after cerebral ischemia in rats. A low dose of EPO was administered to limit its effect on hematopoiesis. Furthermore, EPO was injected late after ischemia, that is beyond its neuroprotective therapeutic window (Brines et al, 2000), to focus only on its effects on brain regeneration. This experimental design of administration may be of importance for the clinical needs. Indeed, most patients cannot be treated with thrombolysis due to a late diagnosis. In general, 24 h after occlusion, the ischemic lesion is maximal and a regenerative therapy would be of interest.
We evaluated the lesion volumes at different times using magnetic resonance imaging after cerebral ischemia in rats and the functional recovery over a period of 51 days. Cellular proliferation and neurogenesis were also assessed at 51 days after occlusion. In addition, we evaluated the outcome of MSC administered 24 h after ischemia in the brain.
Materials and methods
Animals
Experiments were carried out on male Sprague—Dawley rats (CERJ, France), weighing 250 to 350 g at the time of ischemia. All procedures were approved by the regional ethics committee (protocol agreement number N/03-04-07/03/06-10) and were in accordance with the French legislation. The experimental design is presented in Figure 1.
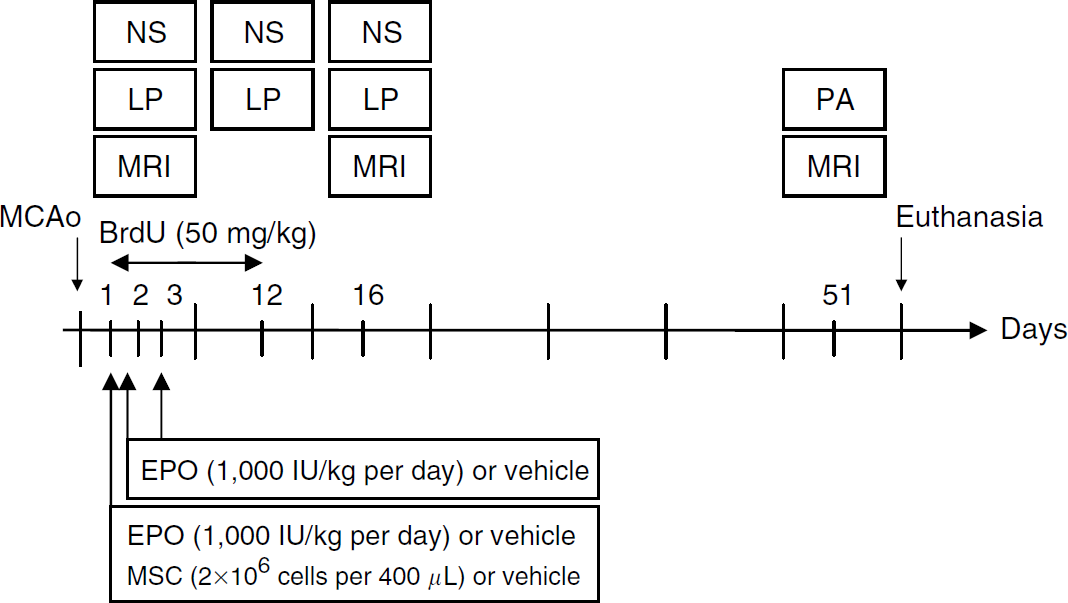
Experimental study design. MCAo, middle cerebral artery occlusion; NS, neurological score; LP, limb placing test; PA, passive avoidance test; MRI, magnetic resonance imaging. The day 0 refers to the day of MCAo.
Transient Focal Cerebral Ischemia
Rats were anesthetized with isoflurane (induction: 5% and maintenance: 2%) in a mixture of 70% N2O and 30% O2. Body temperature was monitored with a rectal probe and maintained with a heating pad at approximately 37°C throughout the experiment. Cerebral blood flow (CBF) was continuously recorded by laser Doppler flowmetry (Moor Instruments MoorLAB, Paris, France) from 10 to 15 mins before and 5 mins after ischemia (Figure 2A). Transient focal cerebral ischemia (90 mins) was induced by intraluminal occlusion of the right middle cerebral artery (Freret et al, 2006a). After a midline incision in the neck, the right common carotid artery, the external carotid artery, and the internal carotid artery were isolated. The common carotid artery was then ligated and the external carotid artery was electrocoagulated 4 mm distally from its bifurcation from the common carotid artery. The occluding device was made up of a nylon thread (0.18 mm diameter) with the extremity coated with translucent hot melt adhesive to constitute an embolus (4 mm length, 0.36 to 0.38 mm diameter). The embolus was inserted through a small incision into the external carotid artery and was gently advanced into the internal carotid artery until the CBF decreased (between 25% and 75%) or a slight resistance was observed. During surgery, the decrease of CBF at the time of occlusion was not always observed or followed by a spontaneous reperfusion in 15% of rats. These animals were excluded from the experiment. After the intraluminal thread was secured, the neck incision was sutured. The laser Doppler probe and the rectal temperature probe were removed. The rats recovered from anesthesia and were returned to their home cages. Ninety minutes later, rats were reanesthetized. The embolus and the ligation of the common carotid artery were removed to allow reperfusion. The rectal temperature was measured. The wounds were sutured and rats were placed back in their home cages. All animals received an intraperitoneal administration of physiologic saline (1 mL) shortly after surgery as well as daily for 5 days to prevent postsurgical dehydration.
(
Mesenchymal Stem Cell Preparation
Mesenchymal stem cell preparation was performed according to a modified version described by Hanabusa et al (2005). Bone marrow cells were extracted from femurs and tibias of adult rats by flushing bones with αMEM (Eagle's minimal essential medium; Eurobio, Courtaboeuf, France) and cultured in this medium supplemented with 20% fetal calf serum and 2 mmol/L glutamine (Sigma, I'Isle d'Abeau Chesnes, France). Nonadherent hematopoietic cells were removed by replacing the medium. After 3 to 4 passages, adherent cells were collected using 0.05% trypsin and 2% EDTA (Gibco, Cergy Pontoise, France) for 3 mins at 37°C. We verified that cells were multipotent and thus able to differentiate into osteocytes and adipocytes (data not shown).
Mesenchymal Stem Cells and Erythropoietin Administration
The selection of the dose of EPO (1,000 IU/kg per day, H Lundbeck A/S, Copenhagen, Denmark) and of the concentration of MSC (2 times 106 cells) was based on the literature data (Chen et al, 2001; Sola et al, 2005). Twenty-four hours after cerebral ischemia, animals received a single intravenous injection of MSC (2 times 106 cells suspended in 400 μL of phosphate-buffered saline (PBS) or 400 μL of PBS alone) into the tail vein and an intraperitoneal injection of EPO (1,000 IU/kg per day) or vehicle (PBS), repeated daily for 2 days.
Experimental Design
In the first experiment, rats were ranked according to their neurological score, measured 24 h after ischemia, just before the administration of MSC and/or EPO (Figure 1). This procedure, already used in a recent published study (Freret et al, 2006b), ensures that groups are similar in terms of initial impairment. Animals were assigned to one of the following four experimental groups, namely, vehicle (
In the second experiment, animals (
Hematocrit
Blood samples were taken via a tail vein at 1, 3, 8, 15, 28, and 51 days after ischemia for hematocrit determination.
Measurement of Infarct Size
The volume of the lesion was evaluated by magnetic resonance imaging (7T, PharmaScan, Bruker BioSpin, Ettlingen, Germany). T2-weighted imaging was performed for each animal at different times after occlusion (3, 16, and 51 days). T2-weighted imaging was obtained with a rapid acquisition with relaxation enhancement (RARE) sequence (RARE factor of 8; repetition time (TR) = 5,000 ms; echo time (TE) = 56; 4 averages; field of view (FOV) = 3.5 times 3.5 cm; matrix = 256 times 192 zero-filled to 256 times 256; 15 slices of 1.5 mm thickness). The lesion volumes were analyzed with ImageJ software (Rasband, 1997–2006). Areas were delineated on the sections and volumes were calculated by multiplying the total area by the distance between sections. Because of the disappearance of the necrotic area and the appearance of a cystic area, the lesion volume was measured according to an indirect method. Lesion volumes were calculated as follows:
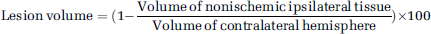
Immunohistochemistry
Anesthetized rats were perfused transcardially with saline followed by 4% paraformaldehyde (Sigma) 51 days after middle cerebral artery occlusion. The brains were removed and coronal sections (50 μm) were cut on a vibratome. The slices were kept at −20°C in a cryoprotective mixture (30% glycerol, 20% ethylene glycol, and 50% PBS).
Single immunohistochemical staining for BrdU was used to quantify cellular proliferation and identify MSC. The slices were heated for 2 h at 65°C in 50% formamide in 2X SSC, treated for 30 mins in 2N HCl at 37°C, and the reaction was neutralized in 0.1 mol/L boric acid (pH 8.5) for 10 mins (Sigma). Thereafter, the coronal sections were washed in PBS, incubated for 20 mins in 3% H2O2 in PBS, and washed again in PBS. After blocking the nonspecific binding for 2 h with PBS/3% BSA/0.3% Triton X-100, the slices were incubated overnight with monoclonal mouse anti-BrdU antibody (Chemicon, Paris, France) diluted in PBS/3% BSA/0.3% Triton X-100. Primary antibody was detected by incubation with anti-mouse biotinylated antibody (Sigma) diluted in PBS/3% BSA/0.3% Triton X-100 for 2 h. Staining was visualized after 2 h of incubation in an avidin—biotin complex (Vector, Paris, France) prepared in PBS/0.3% Triton X-100 with diaminobenzidine nickel (Vector).
Double immunohistochemical staining was used to quantify neurogenesis and to identify the potential of the differentiation of MSC into neurons or astrocytes. Slices were incubated overnight with primary antibodies (monoclonal rat anti-BrdU antibody, dilution 1:100, Abcys, Paris, France; monoclonal mouse anti-NeuN, 1:1,000, Chemicon; and polyclonal rabbit anti-glial fibrillary acidic protein (GFAP), 1:5,000, Sigma). Then, the sections were incubated for 2 h with FITC-conjugated donkey anti-rat (Jackson ImmunoResearch, Suffolk, UK), Alexa 555 anti-mouse (Molecular Probes, Cergy Pontoise, France), or Alexa 555 anti-rabbit (Molecular Probes).
Cell counting (cellular proliferation and neurogenesis) was performed from photographs taken on a Leica DM6000B. Optronic camera and the Metaview (version 6.3r7) software were used. For cellular proliferation, cells that were detected on single immunohistochemical staining were counted in three areas for each ipsilateral and contralateral region (along the lateral ventricle wall and in the dentate gyrus (DG)) on three different slices (
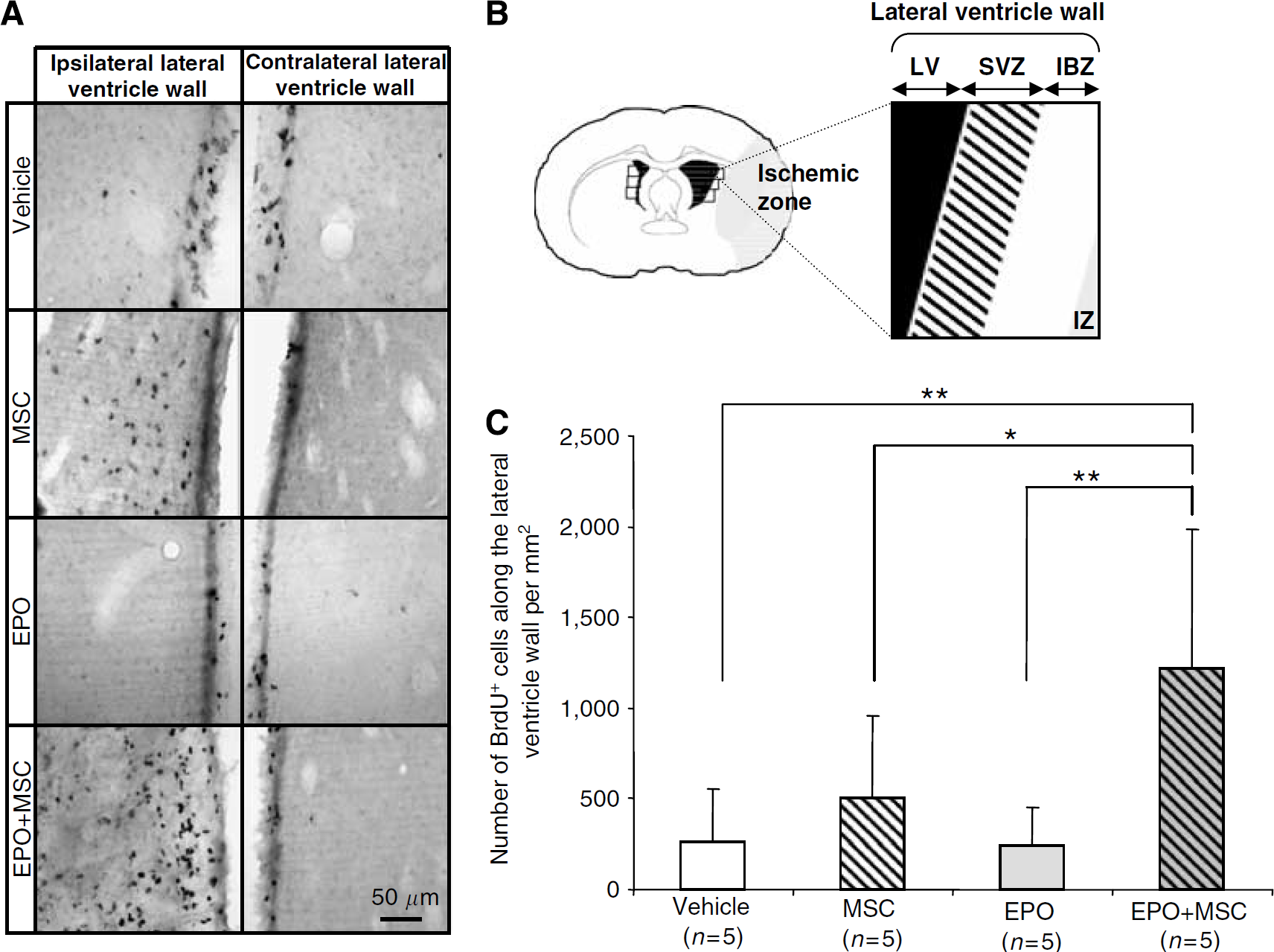
The cotreatment MSC + EPO increased cellular proliferation along the lateral ventricle wall. (
Behavioral Tests
The neurological score and the limb placing test evaluate neurologic and sensorimotor deficits, which appear early after cerebral ischemia and disappear 1 month after cerebral ischemia (Freret et al, 2006b). Thus, these two tests, previously validated in this model of ischemia by Freret et al (2006a), were performed for 3 weeks after cerebral ischemia.
The neurological score was performed according to a modified version of the method of Bederson et al (1986). The test consists of 14 subsets. Spontaneous walking and circling toward the paretic side were first observed. Then, the rat, held by the tail, was placed on a rough surface and pushed gently consecutively toward the ipsilateral and contralateral sides to assess resistance to push. Finally, the rat was suspended by the tail, sequentially by the right and the left hand of the experimenter, and lifted above the bench to assess the body rotation and flexions of the forelimbs and hindlimbs. Each subtest was graded on a scale from 0 to 2 (0 = no response or totally abnormal response; l=weak or abnormal response; and 2= normal response). Absence of deficit was assigned a maximal neurological score of 28.
The limb placing test was performed according to De Ryck et al (1989). This test consisted of four tasks assessing the sensorimotor integration of limb responses to visual, tactile, proprioceptive, and vibrissae stimulation. Both sides were studied separately.
For visual stimulation, the rat was maintained a few centimeters above a table and gently approached the surface. Normal rats extended and placed both forepaws on the table. For tactile stimulation, the rat was held by the investigator so that the paws were free. Contact was made between the dorsal surface of the forepaw and the edge of the table. Normal rats spontaneously placed both forelimbs on the surface of the table. The same stimulation was performed for the hindlimbs. For proprioceptive stimulation, the rat was placed on the top of a table. Each of its forelimbs and hindlimbs were approached toward the edge of the table and pulled down. Normal rats drew back the limbs and placed them on the table. For vibrissae stimulation, the investigator brought the rat toward the edge of a table top so that its limbs were free and several vertical movements were applied so that only the vibrissae touched the table. Normal rats placed the forelimbs on the side ipsilateral to vibrissae stimulation. The maximal score was 22, attesting the absence of deficit. To measure the effect of the different treatments on long-term deficits induced by cerebral ischemia, mnesic performances were evaluated late, 48 and 50 days after cerebral ischemia, using a modified version of the passive avoidance test (Yonemori et al, 1999). Thus, this design protocol not only allows the measurement of mnesic deficits at a time when animals have recovered their initial sensorimotor and motor performances but also the evaluation of potential regenerative effect of MSC and/or EPO, which is a long-term process. The apparatus (Bioseb, Chaville, France) consisted of two compartments, namely an illuminated chamber (21 times 21 times 16 cm) and a dark one (31 times 31 times 32 cm), separated by a guillotine door. During the acquisition trial, rats were placed in the light compartment. After 60 secs of habituation, the guillotine door was opened and the door was shut as soon as all four legs of the rat had entered the dark compartment. The step-through latency, that is the latency to enter the dark compartment, was recorded at this acquisition session. A 0.8 mA foot shock was subsequently delivered through the grid floor for 2 secs. After 48 h, the step-through (retention) latency was measured in the same way as in the acquisition trial, but foot shock was not delivered and the latency time was recorded to a maximum of 180 secs.
Statistical Analysis
Data obtained from the experiments were expressed as mean ± s.d. Physiologic parameters (temperature, hematocrit, and body weight), volume of lesion, and behavioral results (neurological score and limb placing test) were analyzed by a two-way analysis of variance (ANOVA) with repeated measurements followed by one-way analyses and
Results
Physiologic Parameters
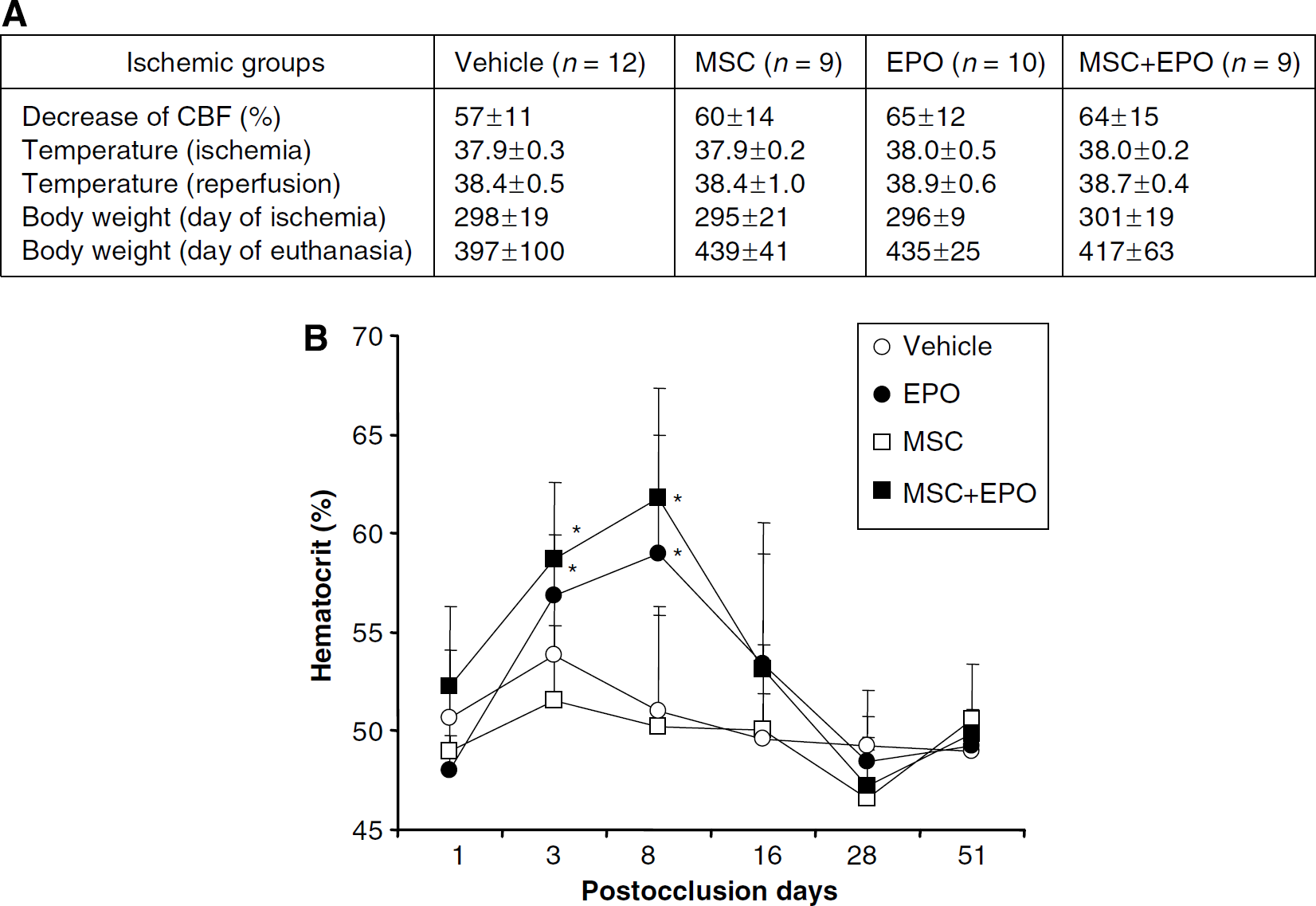
During ischemia, the decrease of CBF at the moment of middle cerebral artery occlusion did not differ between groups (one-way ANOVA,
Measurement of Hematocrit
The hematocrit was measured at different times after occlusion to verify the efficiency of EPO on hematopoiesis (Figure 2B). The hematocrit was significantly increased after cerebral ischemia in the groups receiving EPO during 3 days as compared with the vehicle group on day 3 (EPO: 59% ± 4% and MSC + EPO: 57% ± 3% versus vehicle: 52% ± 4%,
Lesion Volume
The lesion volumes were measured by magnetic resonance imaging on different times after occlusion (Figure 3A). On day 3, the percentage of lesioned tissue in the four groups was not significantly different (MSC: 20% ± 10%; EPO: 26% ± 10%; and MSC + EPO: 21% ± 11% versus vehicle: 27% ± 11%,
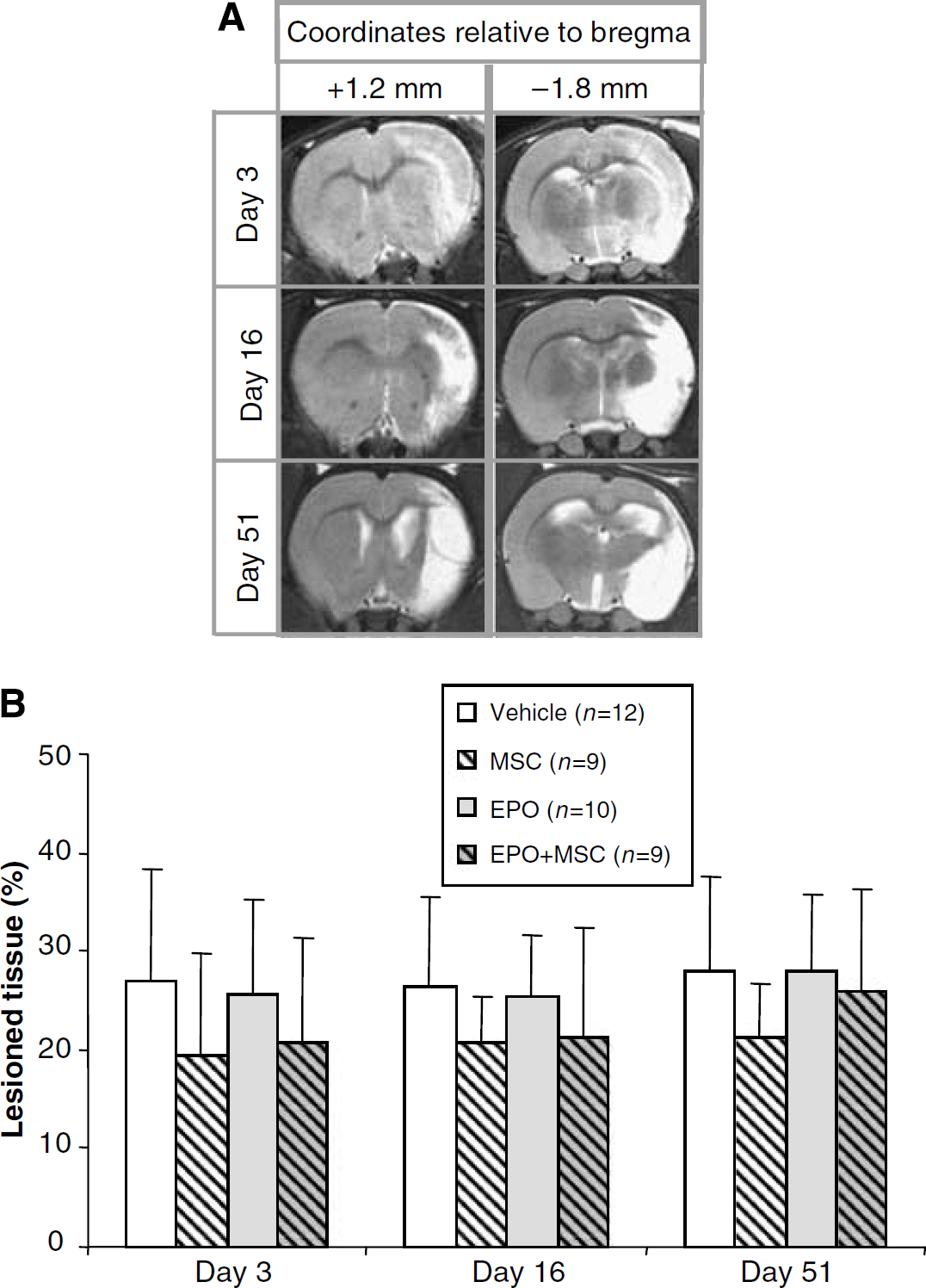
Administration of the different treatments had no effect on the lesioned tissue. (
Neurogenesis
Countings of BrdU-positive cells were performed in the neurogenic zones, that is the DC and along the lateral ventricle wall (Figures 4A and 4B). The number of BrdU-positive cells was expressed as the difference between that number in the ipsilateral and contralateral regions. Cellular proliferation was first assessed in control rats (nonischemic rats) (
In the striatal ischemic boundary zone (IBZ), the number of BrdU/NeuN-positive cells was expressed as the difference between the numbers in the ipsilateral and the contralateral regions (Figure 5A). The number of BrdU/NeuN-positive cells was not different in the MSC and in the EPO groups as compared with the vehicle group (MSC: 3 ± 9.9; EPO: −0.6 ± 2.0 versus vehicle: 1.9 ± 4.3) but was significantly increased in the MSC + EPO group as compared with the vehicle group (MSC + EPO: 28.3 ± 34.0 versus vehicle: 1.9 ± 4.3;
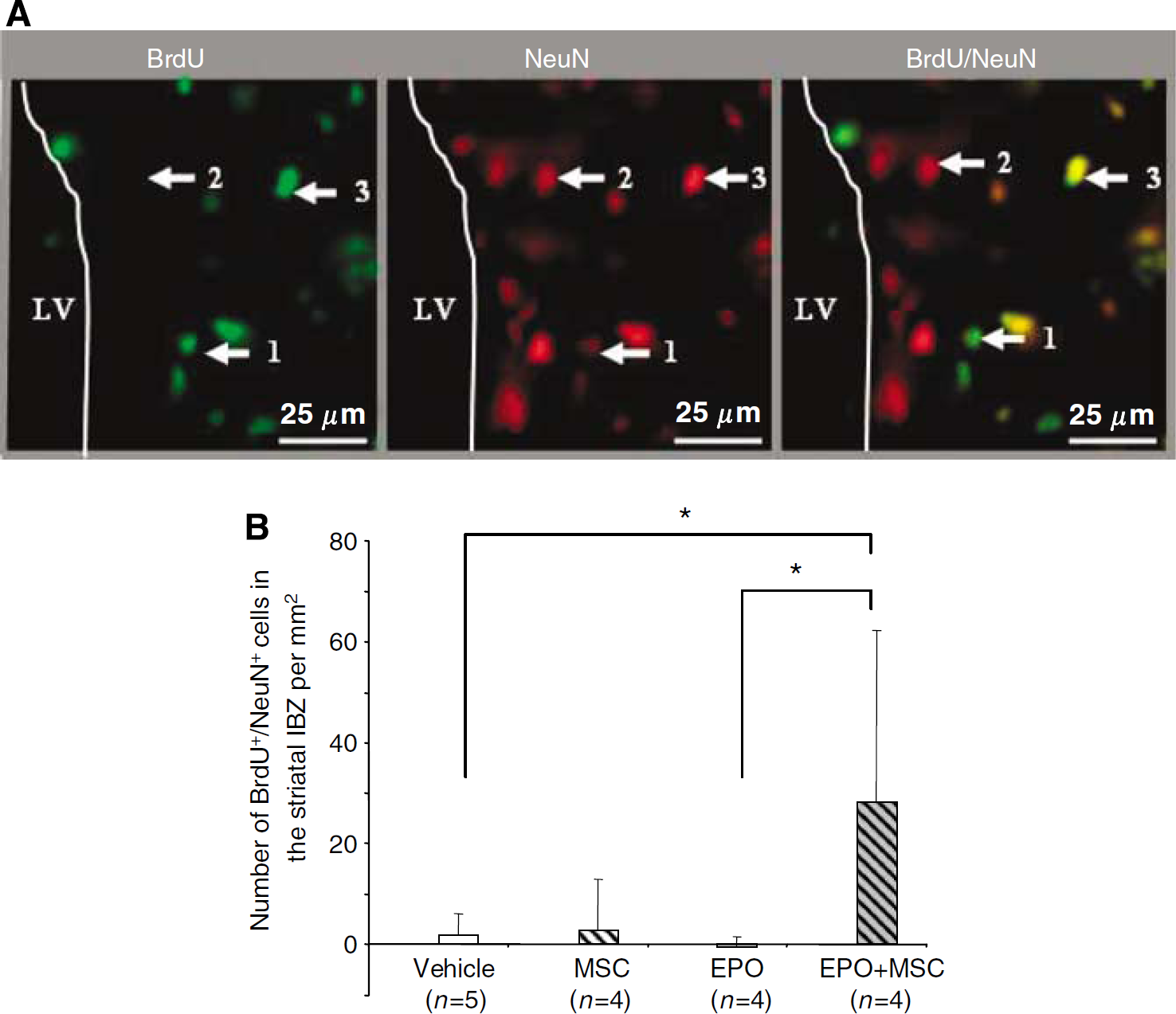
The cotreatment MSC + EPO increased neurogenesis in the striatal IBZ. (
Mesenchymal Stem Cell Outcome
Cells were prelabeled
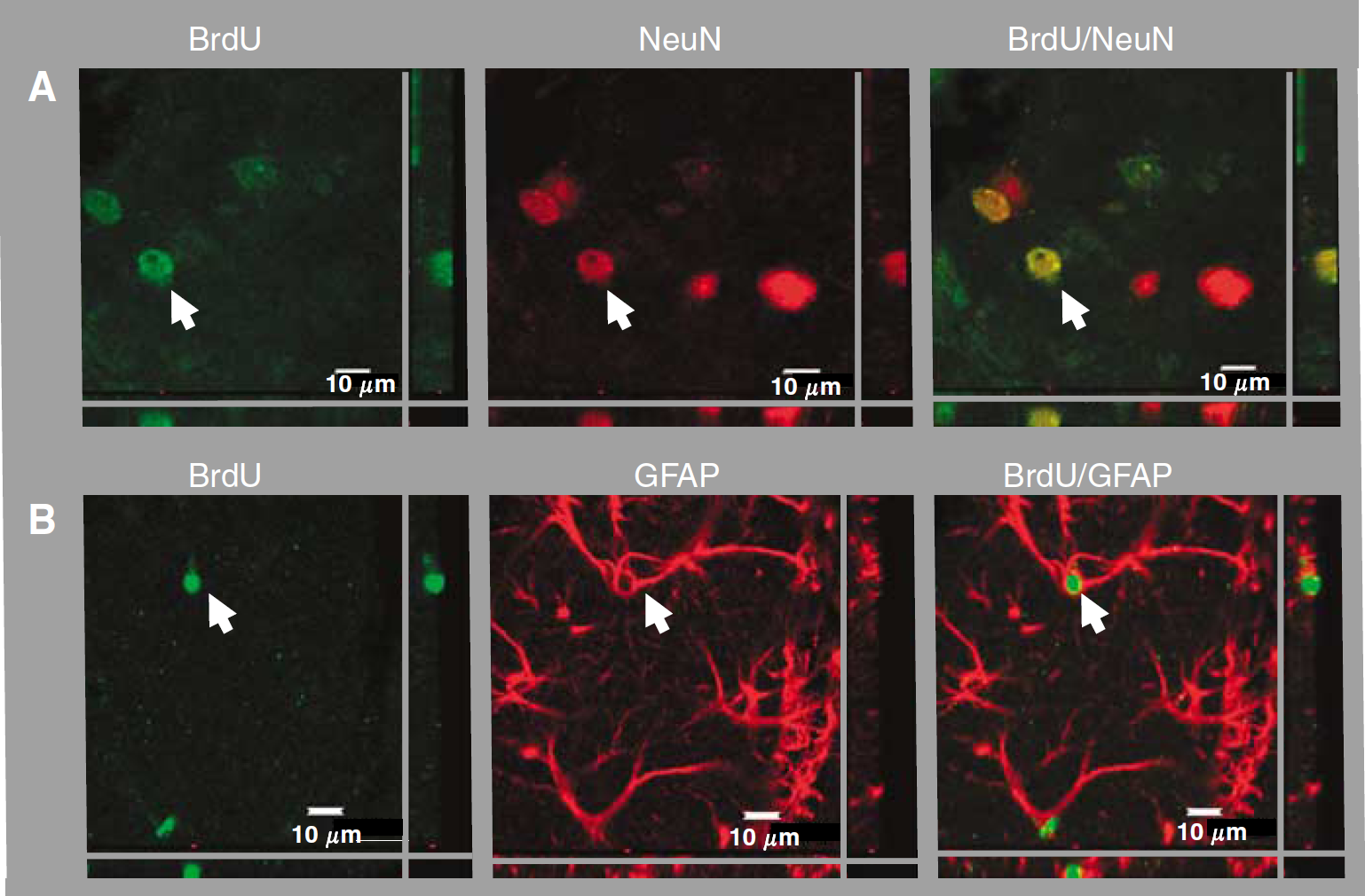
For both MSC and MSC+ EPO groups, BrdU-labeled MSCs were observed in the IBZ, that is in the striatum and in the cortex. Photographs illustrate MSC expressing the neuronal marker NeuN (
To evaluate the potential beneficial effect induced by MSC and/or EPO, functional recovery was assessed using sensorimotor and cognitive tests.
Behavior
The neurological score (Figure 7A) and the limb placing (Figure 7B) tests were performed during the 3 weeks after cerebral ischemia. These two tests, previously described by Freret et al (2006a) in this model, allow functional deficits induced by middle cerebral artery occlusion to be assessed. In the first week following ischemia, the neurological score was low in the four tested groups (score = 18 to 19) and it slowly increased during the second (score = 22 to 23) and the third (score = 24 to 25) weeks. However, the animals had not completely recovered 3 weeks after cerebral ischemia (Univariate
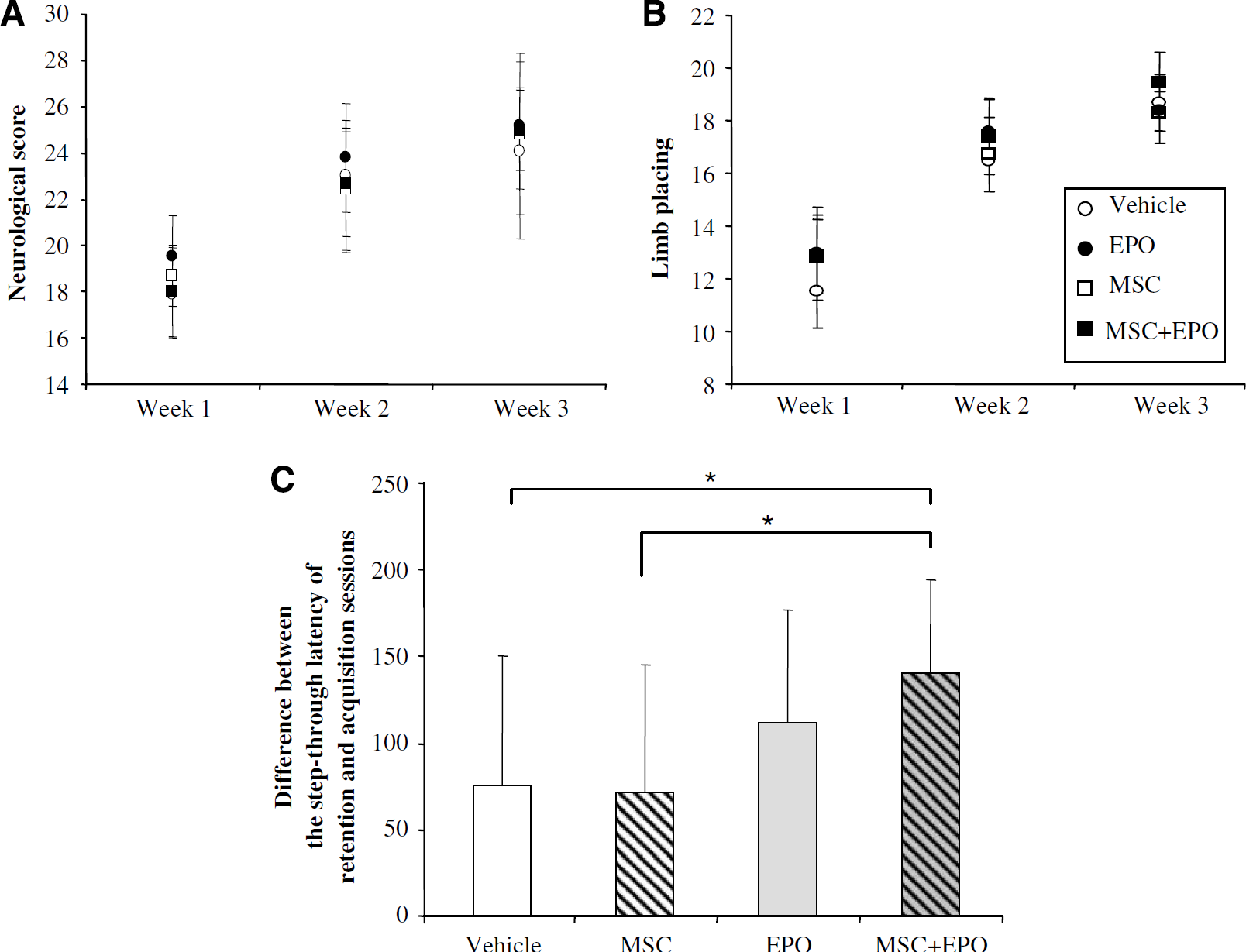
(
For the passive avoidance test, animals were examined between 48 and 50 days after cerebral ischemia (Figure 7C). The test was previously validated in our laboratory (data not shown). During the acquisition session, the step-through latency was not different in the four ischemic groups. The difference between the step-through latency at the acquisition and retention sessions was significantly increased in the group treated with MSC + EPO as compared with the vehicle group (MSC + EPO: 140 ± 54 versus vehicle: 75 ± 75,
Discussion
In this study, a late administration of MSC and/or EPO (i.e., 24 h after brain ischemia) had no significant effects on the volume of lesion, as expected. However, we showed that cotreatment with MSC and EPO increased cellular proliferation along the lateral ventricle wall and neurogenesis in the striatal IBZ and that this effect was accompanied by an improvement of cognitive function, whereas administration of MSC or EPO alone had no significant effects.
The absence of a neuroprotective effect of these different treatments is in accordance with previous studies showing that late administration of MSC or EPO alone did not reduce the volume of infarct (Wang et al, 2004; Chopp and Li, 2002). Thus, in the absence of neuroprotective effects of MSC and/or EPO, we focused on their effect on brain regeneration after cerebral ischemia and in particular on neurogenesis.
Cellular proliferation and neurogenesis were quantified in the neurogenic zones 51 days after cerebral ischemia. In the DC, cellular proliferation was unchanged after cerebral ischemia or administration of the different treatments. It seems that cellular proliferation in the DC is not dependent on ischemic hippocampal damage (Liu et al, 1998); nevertheless, published studies after focal cerebral ischemia described divergent results (Zhang et al, 2001; Arvidsson et al, 2001). Indeed, after focal cerebral ischemia in which hippocampus was not lesioned, Zhang et al (2001) showed that the number of BrdU-immunoreactive cells in the DC was unchanged, whereas Arvidsson et al (2001) observed an increase of cellular proliferation. Erythropoietin alone had no effect on cellular proliferation along the lateral ventricle wall and on neurogenesis in the striatal and cortical IBZ in contrast with a previous study showing that a long-term administration of EPO with a higher dose (5,000 IU/kg per day) and a longer time (7 days) increased the number of BrdU-, nestin- (marker of stem cells), and doublecortin-(Dcx) immunoreactive cells in the SVZ 28 days after cerebral ischemia (Wang et al, 2004). In our study, administration of MSC alone had no effect on cellular proliferation along the lateral ventricle wall and on neurogenesis in the striatal and cortical boundary zones of the ischemic hemisphere. In accordance with our data, a previous study reported that MSC administered 24 h after cerebral ischemia had no effect on neurogenesis in the SVZ 28 days after cerebral ischemia (Zhang et al, 2006). Mesenchymal stem cells seem to induce cellular proliferation and neurogenesis when these cells are administrated at a higher dose after cerebral ischemia (Chen et al, 2003; Baker et al, 2007). Interestingly, combined treatment with MSC and EPO increased the number of BrdU-positive cells along the lateral ventricle wall and the number of BrdU/NeuN-positive cells in the striatal IBZ but not in the cortical IBZ. These results suggest that this combined treatment may accelerate proliferation and/or differentiation of endogenous neural stem cells.
To discriminate the potential effect of MSC on endogenous neurogenesis from their own differentiation into neurons, MSC outcome was analyzed 51 days after cerebral ischemia. Mesenchymal stem cells were retrieved in the ischemic hemisphere, particularly around the SVZ and the ischemic zone. These results are in accordance with published data and confirm that MSC can migrate through the ischemic region when they are injected intravenously 24 h after cerebral ischemia (Chopp and Li, 2002). Furthermore, some MSCs expressed the mature neuronal marker NeuN and the astroglial marker GFAP 51 days after cerebral ischemia, as described in previous studies (Hanabusa et al, 2005; Chen et al, 2001; Li et al, 2001). However, administration of EPO in combination with MSC did not modify the survival of MSC and their outcome. These results indicated that beneficial effects observed after administration of MSC could be due to the supply of different cytokines and trophic factors by MSC in the ischemic area, as suggested in the literature (Chen et al, 2002). For example, MSC cultured in supernatant derived from ischemic brain extracts produced more neurotrophins and angiogenic growth factors (Chen et al, 2002).
To evaluate whether the beneficial effects induced by the cotreatment on neurogenesis may improve neurobehavioral outcome after stroke, functional recovery was assessed by the neurological score and the limb placing test. The neurological score is classically used to evaluate the general state of the animal after ischemia (Bederson et al, 1986) and the limb placing test allows an assessment of sensorimotor functions (De Ryck et al, 1989). The different treatments did not have any effect in these two tests. Our results are in contradiction with previous studies showing an increase of the neurological score 2 weeks after cerebral ischemia after intravenous administration of MSC (Chen et al, 2001). In our experiment, the treatments, however, seem to act on long-lasting memory. Long-term memory was assessed 50 days after cerebral ischemia with the passive avoidance test. Transient focal cerebral ischemia is known to induce long-term memory dysfunction in the rodent (Yamamoto et al, 1988), which seems to be associated with damages in the parietal cortex, the cortico-hippocampal connections (entorhinal and perirhinal cortex), and the striatum (Yonemori et al, 1999; Bouët et al, 2007). In our study, the passive avoidance test revealed that cotreatment MSC + EPO decreased the mnesic deficits induced by cerebral ischemia. Previous data showed a beneficial effect of EPO or MSC on long-term memory. Indeed, the long-term administration of EPO, starting 24 h after cerebral ischemia, improved spatial memory in the Morris water maze in a model of traumatic brain injury (Lu et al, 2005). Another study reported an improvement of cognitive function in the Morris water maze after the administration of MSC 3 days after cerebral ischemia (Sokolova et al, 2006). In this study, the cotreatment ameliorates long-term memory in ischemic rats, whereas the administration of MSC or EPO alone had not significant effect.
Although MSCs seem to be an interesting approach in the treatment of brain injuries and numerous other disorders (see for review Picinich et al, 2007), their therapeutic potency may be increased by combination with a molecular treatment. Indeed, the beneficial effect of MSC on cerebral ischemia was enhanced by administration of adrenomedullin (Hanabusa et al, 2005). Moreover, it has been reported that VEGF (vascular endothelial growth factor) could act synergistically on neural stem cells (Chu et al, 2005). Furthermore, MSCs have shown efficiency as therapeutic vectors. Indeed, these cells, genetically modified to overexpress, for example, brain-derived neurotrophic factor (Kurozumi et al, 2004; Nomura et al, 2005), glial cell line-derived neurotrophic factor (Kurozumi et al, 2005), and fibroblast growth factor (Ikeda et al, 2005), were therapeutically more efficient than nonmodified MSC. Recently, a study reported beneficial effects of combined treatments, that is bone marrow cells and adenovirus overexpressing metalloproteinase inhibitors (Baker et al, 2007).
Our data suggest that combined treatment with MSC and EPO improved endogenous cell proliferation and neurogenesis, which may account for the increase in long-term memory observed after cerebral ischemia. Many studies have reported a correlation between neurogenesis and cognitive function, indicating a link between these two processes (see for review lledo et al, 2006). Indeed, the acquisition on spatial memory in the Morris water maze correlated with the proliferation and survival of newborn neurons in the DC according to mouse strains (Kempermann and Gage, 2002). In the same manner, an odor-enriched environment was correlated to an increase of newborn cells survival in the olfactory bulb with an improvement of the olfactory memory (Rochefort et al, 2002). After focal cerebral ischemia, Liu et al (2007) described an improvement of sensorimotor and mnesic deficits associated with an increase of neurogenesis in the DC after a long-term treatment. These results suggest that EPO in combination with MSC could ameliorate mnesic performances in increasing neurogenesis in the striatal IBZ after transient focal cerebral ischemia. However, we cannot exclude a potential effect of the co-treatment MSC + EPO on the neurogenesis in the DC, although cellular proliferation was not improved in this region. Further studies on neurogenesis in the DC should be realized to clearly identify by which mechanisms long-lasting memory is improved.
In conclusion, combined treatment with MSC and EPO seems to be an interesting approach not only for the treatment of cerebral ischemia but also in other central nervous system disorders such as cognitive dysfunction.
Footnotes
Acknowledgements
We thank H Lundbeck A/S (Copenhagen, Denmark) for the gift of rhEPO.
