Abstract
The identification of genes differentially regulated by ischemia will lead to an improved understanding of cell death pathways such as those involved in the neuronal loss observed following a stroke. Furthermore, the characterization of such pathways could facilitate the identification of novel targets for stroke therapy. We have used a novel approach to amplify differential gene expression patterns in a primary neuronal model of stroke by employing a lentiviral vector system to specifically bias the transcriptional activation of hypoxically regulated genes. Overexpression of the hypoxia-induced transcription factor subunits HIF-1α and HIF-2α elevated hypoxia-mediated transcription of many known HIF-regulated genes well above control levels. Furthermore, many potentially novel HIF-regulated genes were discovered that were not previously identified as hypoxically regulated. Most of the novel genes identified were activated by a combination of HIF-2α overexpression and hypoxic insult. These included several genes with particular importance in cell survival pathways and of potential therapeutic value. Hypoxic induction of HIF-2α may therefore be a critical factor in mediating protective responses against ischemic injury. Further investigation of the genes identified in this study may provide increased understanding of the neuronal response to hypoxia and may uncover novel therapeutic targets for the treatment of cerebral ischemia.
The interruption of blood flow to the brain, such as occurs in stroke, can result in extensive cell loss in affected brain regions with often devastating consequences. The extent of damage and mechanisms of cell death within the ischemic region depend largely upon the length and severity of the insult, however it is now well documented that a region of necrotic cell death occurs in the core region of the infarct and this is surrounded by an apoptotic penumbra (Heiss and Graf, 1994). Elucidation of pharmacological agents that mediate protection from such ischemic cell loss has so far proven unsuccessful (Clark et al., 2000).
Previous attempts to elucidate patterns of differential gene expression in stroke models have mainly focused on animal models of cerebral ischemia, however, these have proven relatively unsuccessful in identifying potential therapeutic targets (Bates et al., 2001; Read et al., 2001; Sharp et al., 2000). Interpretation of differential gene expression data from such models has proven difficult, due to the genetic heterogeneity and diversity of the infarct pathophysiology between models together with spatial and temporal variations (Read et al., 2001; Sharp et al., 2000; Tang et al., 2002). The majority of animal studies, due to their nature, have investigated expression changes at relatively late timepoints following the onset of ischemia and this may have prevented the identification of early and potentially critical postischemic changes in gene expression (Kobayashi et al., 2000). Furthermore, expression changes in neuronal cell types are subtle and therefore it is critical that highly sensitive methods of detection are employed.
Hypoxia is a cardinal feature of ischemic stroke. Changes in gene expression during hypoxia are mediated by a family of transcription factors termed hypoxia inducible factors (HIF) (Semenza, 2001a). These are heterodimers consisting of an α and a β subunit, both belonging to the basic-helix-loop-helix (bHLH)-PAS protein superfamily (Crews and Fan, 1999). Although the β subunit (HIF-1β) is constitutively and stably expressed, stable expression of the α subunits is generally reliant on hypoxic conditions within the cell (Huang et al., 1996; Jiang et al., 1997). Under normoxia the α-subunits are rapidly degraded via a ubiquitination mechanism which is mediated by the von Hippel-Lindau tumor suppressor protein and HIF-mediated transcription is prevented (Cockman et al., 2000; Maxwell et al., 1999; Tanimoto et al., 2000). Although HIF expression is tightly regulated by the oxygen tension within the cell, other conditions have also been implicated in modulating this response in certain systems. Signals such as insulinlike growth factor-1 (Feldser et al., 1999), insulin (Zelzer et al., 1998), interleukin-1 (Thornton et al., 2000), tumor necrosis factor (Sandau et al., 2001), angiotensin (Richard et al., 2000) and epidermal growth factor (Zhong et al., 2000) have been demonstrated to lead to the stabilisation of HIF protein predominantly in tumor cells. However, the mechanisms by which this occurs remain controversial (Laughner et al., 2001; Hudson et al., 2002).
The most well characterized of the HIF family is HIF-1 and over forty HIF-1 regulated genes have been identified to date (Semenza, 2001b). HIF-1 has previously been implicated in mediating neuroprotective gene expression since hypoxic preconditioning has been observed to induce tolerance against focal transient and permanent cerebral ischemia in mice (Bernaudin et al., 2002b; Miller et al., 2001). Furthermore, HIF-1 has previously been well documented to mediate transcriptional activation of genes such as vascular endothelial growth factor (VEGF) and erythropoietin (EPO) which mediate neuroprotection in ischemic models (Bernaudin et al., 2002a). Conversely reports have also documented a requirement of HIF-1α in p53 mediated cell death, and expression of a dominant negative form of HIF-1α was observed to reduce hypoxia mediated neuronal death (Halterman and Federoff, 1999). In addition, embryonic stem cells with targeted disruption of the HIF-1α gene were observed to be resistant to hypoxia and hypoglycemia induced apoptosis (Carmeliet et al., 1998).
HIF-2α shares 48% sequence identity with HIF-1α but is much less well characterized. Like HIF-1α, protein expression is stabilized under hypoxic conditions and has been demonstrated to mediate induction of several HIF-1α target genes (Kappel et al., 1999; Wiesener et al., 1998). The identification of HIF-2α-specific target genes and their downstream functions is poorly defined, however gene inactivation studies have demonstrated that HIF-2α expression is an important requirement for cardiovascular development and angiogenesis (Favier et al., 2001; Peng et al., 2000; Tian et al., 1998). A role for HIF-2α in mediating apoptotic processes has also been hypothesized and some evidence suggests that HIF-2α may be involved in the cellular response to hypoglycemia rather than hypoxia (Brusselmans et al., 2001). More recent studies have demonstrated a predominant role of HIF-2α for activating hypoxic gene expression in the macrophage (White et al., 2003).
Minimal lentiviral vectors based on the equine infectious anemia virus (EIAV) have previously been described and these have proven extremely efficient at transducing primary neurons both in vitro and in vivo with minimal impact on target cell phenotype (Azzouz et al., 2002; Mazarakis et al., 2001; Mitrophanous et al., 1999). We have developed a novel approach to amplify gene expression patterns in a physiological response pathway by lentiviral-mediated overexpression of key factors regulating that pathway. In this study we utilized this approach in a primary neuronal model of hypoxia by overexpression of transcription factors involved in mediating the hypoxic response. Overexpression of HIF-1α and HIF-2α resulted in an increased activation of gene expression of known hypoxically regulated genes under oxygen deprivation compared with controls. More importantly we identified nearly 50 potentially novel HIF-1α and HIF-2α responsive genes that have not been previously identified in differential screens of hypoxia or ischemia. Further characterization of these and other genes may enable a better understanding of the mechanisms involved in the cellular adaptation to hypoxia and may uncover potential therapeutic targets for the treatment of stroke and other hypoxia-related disease.
METHODS
Cloning of viral constructs
The genes encoding the human versions of the hypoxically activated transcription factors HIF-1α and HIF-2α were amplified using standard techniques and generated to express a C-terminal Flag epitope tag. The primers used for amplification were as follows: HIF-1α (Forward) GGATCCGCTCACTTGTCATCGTCGTCCTTGTAGTCGTTAACTTGAT and (reverse) ATCAAGTTAACGACTACAAGGACGACGATGACAAGTGAGCGGATCC; HIF-2α (forward) GCGGCCGCTCACTTGTCATCGTCGTCCTTGTAGTCGGTGGCCTGGT and (reverse) ACCAGGCCACCGACTACAAGGACGACGATGACAAGTGAGCGGCCGC. These were subcloned into the previously described minimal EIAV vector genome pONY8.0GFP (Mazarakis et al., 2001) under the control of the cytomegalovirus promoter (pONY8.0HIF1α and pONY8.0HIF-2α). A control vector was also generated consisting of an identical EIAV vector genome but with no transgene of interest cloned downstream from the CMV (pONY8.0MCS). The sequence of HIF-1α and HIF-2α was verified by comparison to the published cDNA nucleotide sequences (Genbank accession U22431 and U81984).
In vitro transductions
Cultured primary neurones were maintained in vitro 7 days before transduction with viral vectors. Culture medium was reduced to approximately half the starting volume and the conditioned medium was retained. Transduction was performed by diluting the appropriate amount of concentrated virus (multiplicity of infection (MOI) of 10 transducing units/cell) in supplemented Neurobasal medium and adding directly to the culture medium of the cells. At 6 hours post transduction half of the culture medium containing virus was removed and replaced with the conditioned medium removed previously. Cells were maintained as described above and experiments were performed at least 72 hours post transduction.
MTT assay
3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) was added to the culture media of primary neuronal cultures to a final concentration of 0.5mg/ml. Following incubation for 2 hours under standard conditions culture media was removed from the cells and the purple formazan product solubilized using isopropanol. Absorbance at 570nm was then measured using a microplate reader.
Hypoxic challenge
Hypoxic gene expression was activated by incubating cells in a hypoxic incubator (0.1% O2, 5% CO2, 37°C). The total volume of culture media was reduced by half prior to hypoxic incubation to decrease the deoxygenation time. Following hypoxic incubation total RNA was isolated immediately to avoid reperfusion effects using RNeasy kits (QIagen, Crawley, West Sussex, UK).
Quantitative RT-PCR
RNA as prepared above was used as a template for synthesizing cDNAs using oligo (dT) primers with the Superscript II cDNA synthesis kit (Roche Diagnostics, Basel, Switzerland). The reaction mixture was incubated at 42°C for 50 minutes and terminated by heating to 70°C for 15 minutes. Real time quantitative PCR was performed using the ABI Prism 7700 detection system with a SYBR green DNA detection kit (Applera, Norwalk, CT, USA). Expression of the housekeeping gene β-actin was also quantified and used for normalization. All primers were designed using Primer Express 2.0 (Applera) and used at a concentration of 300nM. Standard cycling conditions were used.
Microarray analysis
For microarray analysis each experiment consisted of a sample set of 8 independent conditions. Untransduced and pONY8.0MCS, pONY8.0HIF-1α or pONY8.0HIF-2α transduced neurons (MOI of 10 transducing units/cell) were subjected to either normoxia or 8 hours of hypoxia. The experiment was performed in quadruplicate. Microarray expression analysis was performed according to the Affymetrix expression analysis technical manual (Affymetrix, UK Ltd.supporteurope@affymetrix.com). Briefly, double stranded cDNA was reverse transcribed from total RNA (10μg) using a cDNA synthesis kit (Invitrogen) in the presence of an oligo dT-T7 primer. Following phenol/chloroform extraction and ethanol precipitation the cDNA pellet was resuspended in 11μl of RNAse-free H2O. This was then used for the in vitro transcription amplification reaction, in the presence of biotynlated nucleotides (Enzo BioArray High yield RNA Transcript labeling kit). Labeled cRNA (15μg) was fragmented by incubation at 95°C for 35 minutes in fragmentation buffer (40 mM Trisacetate, pH 8.1, 100 mM KOAc, and 30 mM MgOAc) and the fragmented cRNA was then hybridized against the Affymetrix RG-U34 A-C oligonucleotide arrays. The arrays were scanned using the Agilent Gene Array laser scanner at 570nm. Quality of the cRNA was verified using TEST3 chips ensuring that the 3'–5’ signal ratios of GAPDH and β-actin were <2 and that the scaling factors were similar.
Analysis of differential gene expression
Microarray data were scaled in MicroArray Suite 5.0 (Affymetrix) and then exported into GeneSpring software (Silicon Genetics). Two methods were used for analysis of differential expression data and identification of upregulated genes. Firstly, the expression data for each gene were averaged across the 4 experiments to produce a merged data set. Genes were selected that were at least 2-fold upregulated by any condition and that were designated an Affymetrix confidence call of ‘present’ in at least 2 out of 4 experiments. One problem with a merged data set is that the mean expression profile of a gene could be skewed by one extreme result. Therefore, a second level of analysis was incorporated whereby the profiles for each gene from the 4 independent experiments were investigated and genes selected that were at least 2-fold upregulated in any condition and called ‘present’ in at least 2 of the 4 experiments. The results of the differential data analyses were merged. Expression data were normalized to the expression levels from the normoxic pONY8.0MCS transduced controls.
Annotation of ESTs:
The sequence similarity of the untranslated regions (UTRs) between rat and human genes is poor making identification of human orthologues from rat expressed sequence tag (EST) sequences difficult. However, there is significant similarity between the UTRs of mouse and rat, enabling reliable comparisons between these organisms to be made. The rat ESTs identified from the above analysis were compared with the mouse genomic sequence via the online BLAST-like alignment tool (BLAT) in order to identify full length genes. BLAT enables the visualization of the alignment of multiple mRNAs from multiple organisms against the mouse genomic sequence. Using this alignment as a guide, where possible, each of the rat ESTs was annotated with an accession number and gene name for the best-annotated reliable orthologue of the EST query. These orthologues were generally mouse sequences but there were several occasions where the mouse sequence was truncated in comparison to the human or other species and in these cases an alternative accession number from a different organism was been identified. Of the ESTs identified in this study, the “gene names” of approximately 65% of these were determined.
RESULTS
Model of hypoxia in primary cortical neurons
Optimisation of the most suitable timepoint of hypoxic exposure for these experiments was investigated by analyzing neuronal viability and expression patterns of known hypoxically regulated genes. Primary neurons remained viable following 8 hours of hypoxic insult but showed significant reduction in viability following 12 hours hypoxia, as assessed by MTT assay (Fig. 1A). It is interesting that following 48 hours of reperfusion in standard conditions the MTT value returned to normal levels suggesting that the cells had not irreversibly committed to death at this timepoint. However, after 16 hours of hypoxia the cells showed no recovery of viability following reperfusion. To determine whether cell viability under hypoxia was altered by overexpression of HIF-1α or HIF-2α, transduced cultures were also subjected to 8 hours of hypoxic insult and MTT assays performed. No significant differences in viability were observed between control transduced cultures and those overexpressing HIF-1α or HIF-2α indicating that under these conditions transgene expression was did not alter the survival pattern of these cells. Expression of the known hypoxically regulated genes VEGF, c-Fos and Glut3 was assessed under conditions of normoxia, 8 and 12 hours of hypoxia (Fig. 1B). Expression of each of these genes was induced following 8 hours of hypoxia, however, no significant increase in expression levels were observed after 12 hours. Since cell viability was potentially compromised immediately following 12 hours hypoxia we chose 8 hours as the most appropriate timepoint for maximizing hypoxic gene expression but minimizing cell death.
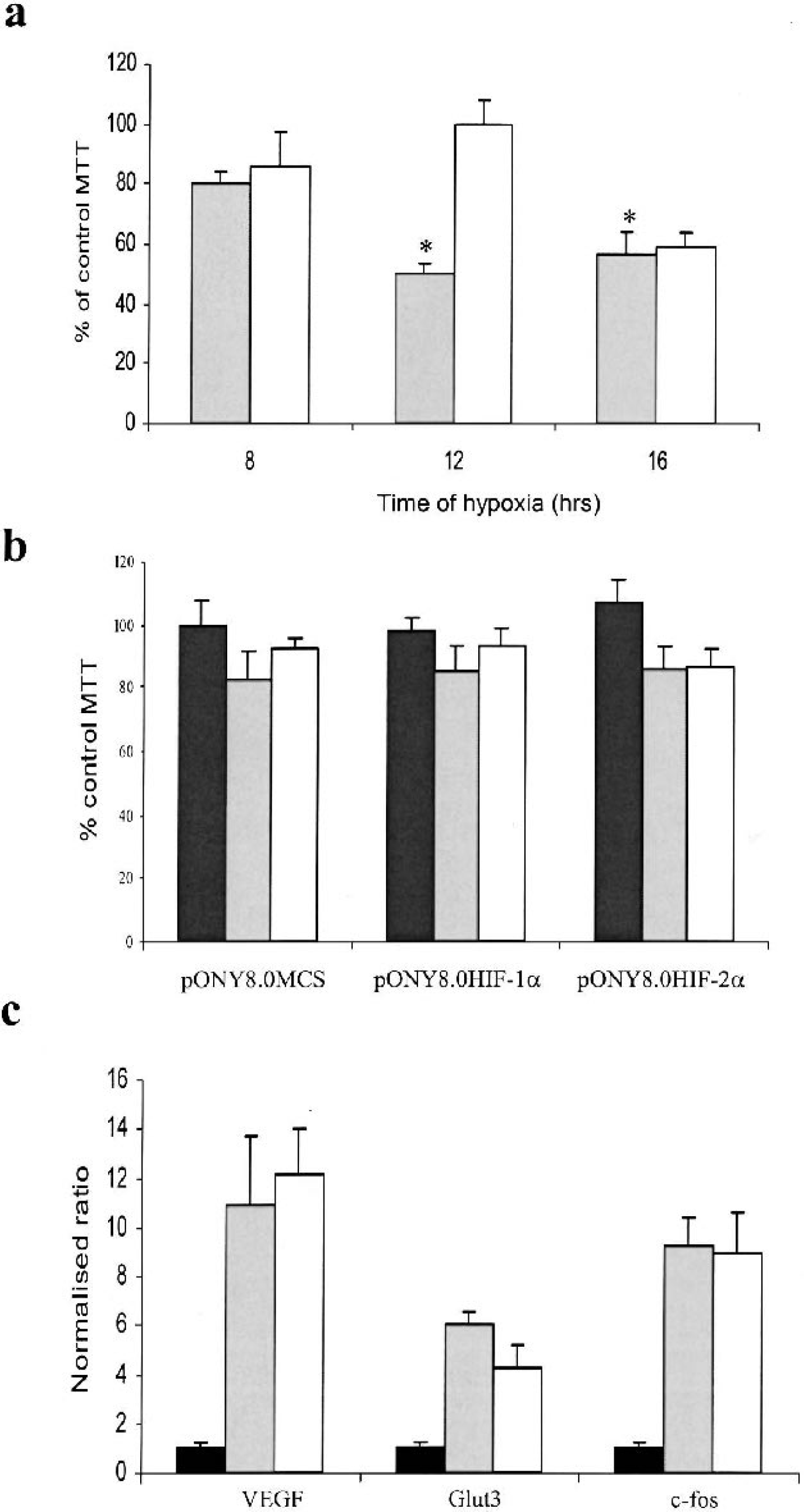
Optimization of a hypoxia model in primary cortical neurons. a) Viability of cortical cultures was assessed immediately following incubation in a hypoxic chamber (0.1% O2) using MTT assay. Data was expressed as a percentage of the normoxic control MTT value for all timepoints; b) Viability of transduced cortical cultures following 8 hours hypoxia. Cells were transduced 7 days previously with EIAV vectors as shown; c) Expression of known hypoxically regulated genes was investigated following hypoxia using Taqman Q-PCR analysis. White bars indicate data for normoxic samples, grey bars indicate data for hypoxic samples and black bars indicate data for hypoxic samples with 48 hours of reperfusion. * represents a significant difference in MTT compared to control levels, (P < 0.05) as assessed by two-tailed Students t-test. n = 4–6 for all experiments.
Proof of principle
In order to assess whether lentiviral mediated overexpression of HIF-1α and HIF-2α would amplify the hypoxic gene response we investigated the expression of known hypoxically regulated genes in transduced primary neuronal cultures following hypoxic challenge. Viral transduction using an MOI of 10 was routinely demonstrated to give a transduction efficiency of approximately 50% in these cultures (data not shown). Furthermore, transduction of pONY8.0HIF-1α and pONY8.0HIF-2α mediated overexpression of HIF-1α and HIF-2α to levels greater than 40-fold over endogenous expression as assessed using Taqman Q-PCR analysis of mRNA levels (data not shown).
Expression of Glut3 and VEGF was induced by 1.3 and 3.9 fold respectively following hypoxic challenge in cultures transduced with the control vector pONY8.0MCS using Taqman Q-PCR analysis. Overexpression of both HIF-1α and more significantly HIF-2α amplified the hypoxic induction of both genes to 3.8 and 14.2 fold respectively (Fig. 2). It is interesting to observe that the expression of a known hypoxically regulated gene such as Glut3 can fall below the 2-fold threshold of induction that is routinely used as a ‘cut off’ for studies of differential gene expression using microarray technology. Therefore, Glut3 provides a good example of a hypoxically regulated gene that would not have been identified as such in a differential screen of primary neurons using Affymetrix technology without the overexpression of HIF-1α or HIF-2α.
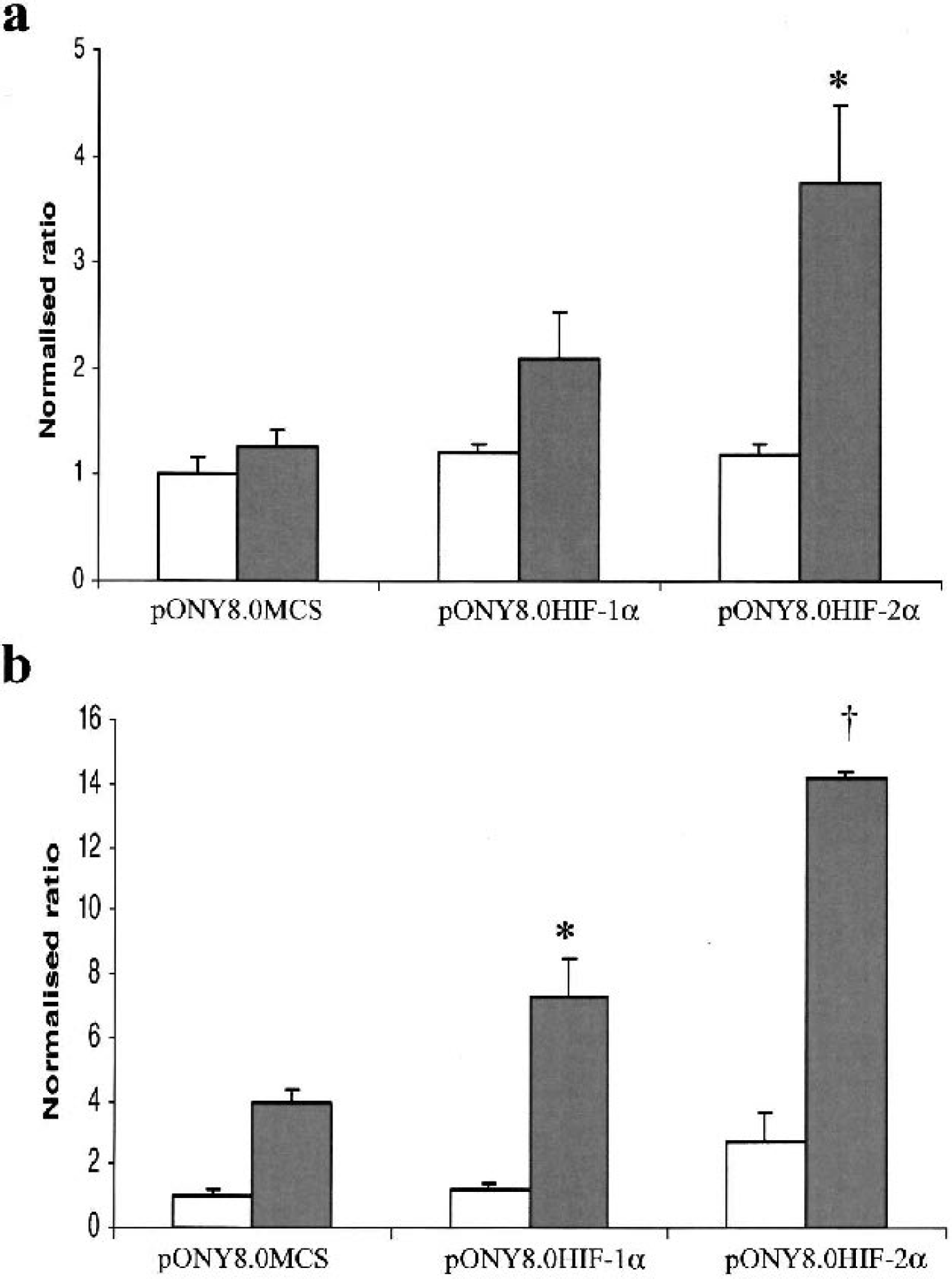
HIF-mediated amplification of hypoxic gene expression in transduced cortical neurons. Expression of known hypoxically regulated genes a) Glut3 and b) VEGF was assessed using Taqman Q-PCR. Cells were previously transduced with Lentivectors pONY8MCS (control), pONY8HIF-1α or pONY8HIF2-α as indicated and incubated in normoxic (white bars) or hypoxic (shaded bars) conditions for 8 hours. * and ‡ indicate a significant difference in gene expression compared to control levels under hypoxia (* P < 0.05; † P < 0.001 as assessed by two-tailed students t-test,. n = 4).
Analysis of differential expression
Following the microarray experiments genes were classified as hypoxic if they were upregulated greater than 2 fold in control transduced cultures following hypoxia. Genes were termed HIF-1α or HIF-2α requiring if expression was less than 2-fold upregulated in control transduced samples following hypoxia but greater than 2-fold upregulated in pONY8.0HIF-1α or pONY8.0HIF-2α transduced cultures. Genes were eliminated from the list of HIF-1/2α requiring genes if their expression was upregulated greater than 2-fold in untransduced cultures following hypoxia. In total, 226 individual genes were identified to be upregulated greater than 2-fold following 8 hours hypoxia with an approximately equal split between known named genes and ESTs (Table 1). A further 69 genes were upregulated above the 2-fold threshold following hypoxia combined with the overexpression of either HIF-1α or HIF-2α. This represents an additional 31% increase in total genes identified under hypoxia. Most of the genes identified in this manner were HIF-2α regulated (~90%) although some of these genes (~20%) were also upregulated by HIF-1α. Of the named genes identified as HIF-1α or HIF-2α requiring more than half have not been previously cited as hypoxically or ischemically regulated in the current literature (Table 2). Of the ESTs identified by these parameters over 80% have not previously been documented as being hypoxically regulated (Table 3). In total 46 potentially novel hypoxically regulated genes were identified by these studies.
Taqman validation of Affymetrix data
To validate the Affymetrix data using a non-hybridisation based methods and verify that the genes we identified were indeed HIF-1/2α requiring, Taqman Q-PCR analysis of mRNA levels was performed using the same RNA samples that were used for the microarray experiments (Fig. 3). Six genes with potential roles in cell death mechanisms that were hypoxically regulated in the presence of HIF-2α overexpression were selected for this analysis. The expression levels for these genes encompassed a broad range of values such that we could quantify differences between Affymetrix and Taqman data over a range of expression levels. In general the Q-PCR Taqman data were highly similar to the results from the Affymetrix analysis when relative ratios were compared following normalisation of expression levels to those of control transduced normoxic samples. The Taqman results for Pentraxin (Fig. 3B), a gene with very low expression levels (Affymetrix raw signal of 10.6 in normoxia, data not shown) were remarkably similar to the microarray data suggesting that even when assessing genes with very low raw signals the sensitivity of the Affymetrix analysis was very high. Genes with higher expression levels such as Lipocalin 2, (Affymetrix raw signal of 31.7 in normoxia, data not shown) were also demonstrated to have minimal differences between microarray and Taqman results (Fig. 3D). Two of the genes analyzed showed significantly different (Sphingosine Kinase, P < 0.05 and UNC5H2 receptor, P < 0.005) normalised ratios between the microarray and Taqman data in the hypoxia HIF-2α samples. However, a similar trend in the expression profile of these genes was observed, with some hypoxic regulation of expression evident in the control samples and elevation of this hypoxic induction following overexpression of HIF-2α (Fig. 3B, Fig. 3). Overall these results indicate that high the Affymetrix microarray system provides highly sensitive detection of differential expression of genes with a broad range of expression levels.
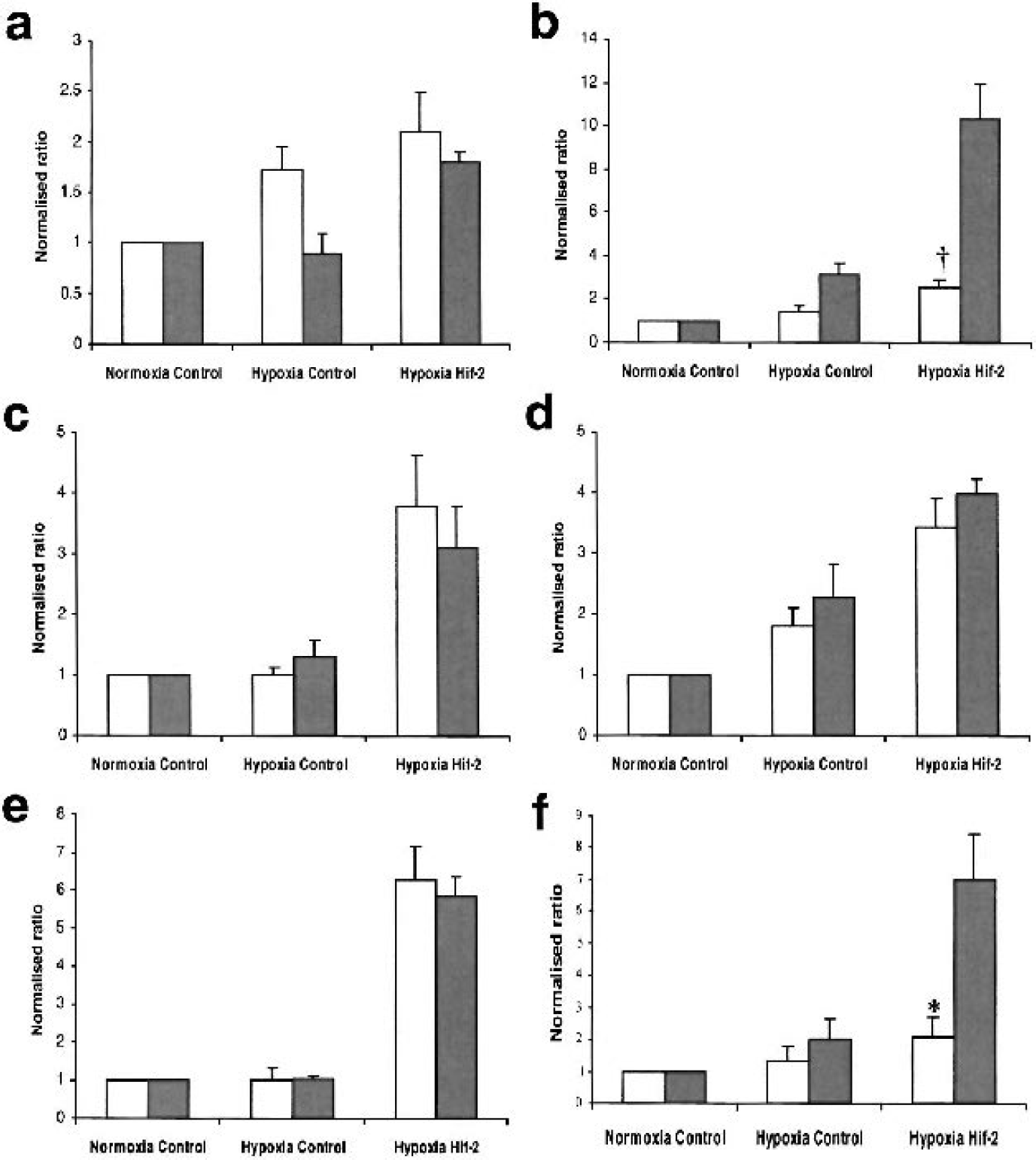
Taqman Q-PCR validation of HIF-requiring genes identified in this study. Relative expression levels of a) Pentraxin; b) UNC5H2 Receptor; c) Chemokine Orphan Receptor 1; d) Lipocalin; e) Gamma glutamyl cysteine synthetase; f) Sphingosine kinase as assessed by microarray analysis (light bars) and Taqman Q-PCR (shaded bars). * and ‡ represent a significant difference between Q-PCR and microarray data (* P < 0.05; † P <: 0.005 using two-tailed students t-test, n = 4 for all data).
DISCUSSION
Following cerebral ischemia induction of the hypoxically regulated transcription factor subunits HIF-1α and HIF-2α occurs in the apoptotic penumbra region bordering the ischemic infarct (Marti et al., 2000). The downstream transcriptional response of these factors is considerable and a number of HIF-1 regulated genes have previously been identified (Semenza, 2001b). These include genes involved in glucose metabolism, angiogenesis, erythropoiesis and cell survival (Semenza, 2001b). Although the importance of HIF-2α in vascular development has been implicated (Peng et al., 2000), the target genes of HIF-2 induced transcription are relatively unknown. Furthermore, the downstream mechanisms of HIF-1α and HIF-2α induction in response to cerebral ischemia have yet to be fully elucidated.
Number of hypoxia-regulated genes identified by microarray analysis of differential expression in primary cortical neurons
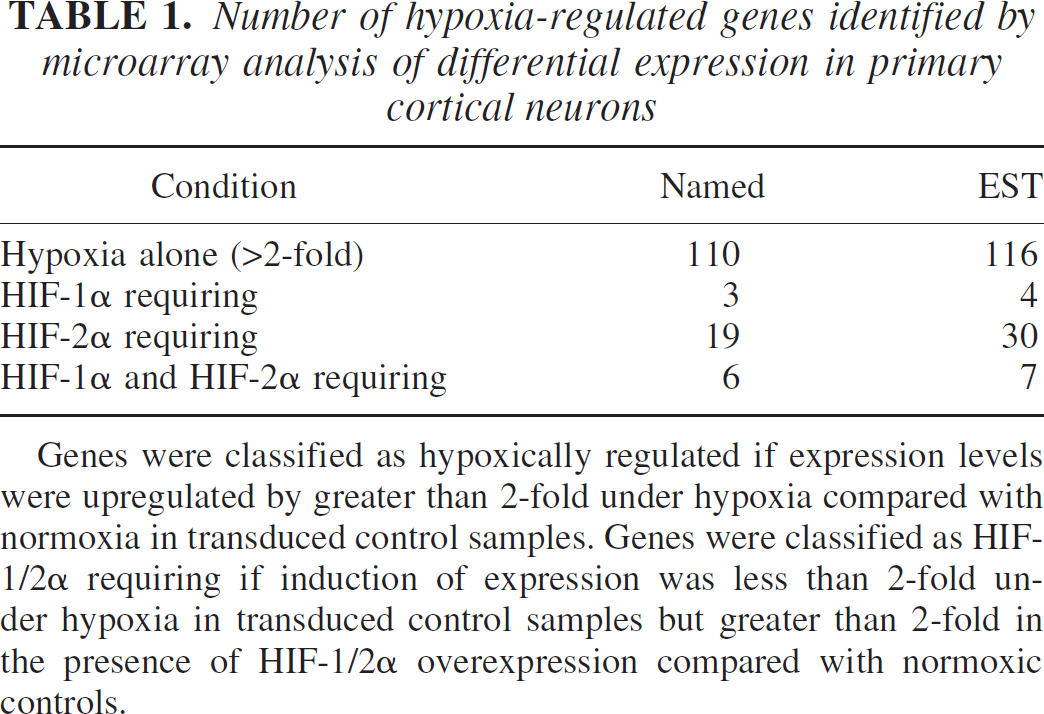
Genes were classified as hypoxically regulated if expression levels were upregulated by greater than 2-fold under hypoxia compared with normoxia in transduced control samples. Genes were classified as HIF-1/2α requiring if induction of expression was less than 2-fold under hypoxia in transduced control samples but greater than 2-fold in the presence of HIF-1/2α overexpression compared with normoxic controls.
Summary of named genes identified as HIF-1α or HIF-2α requiring
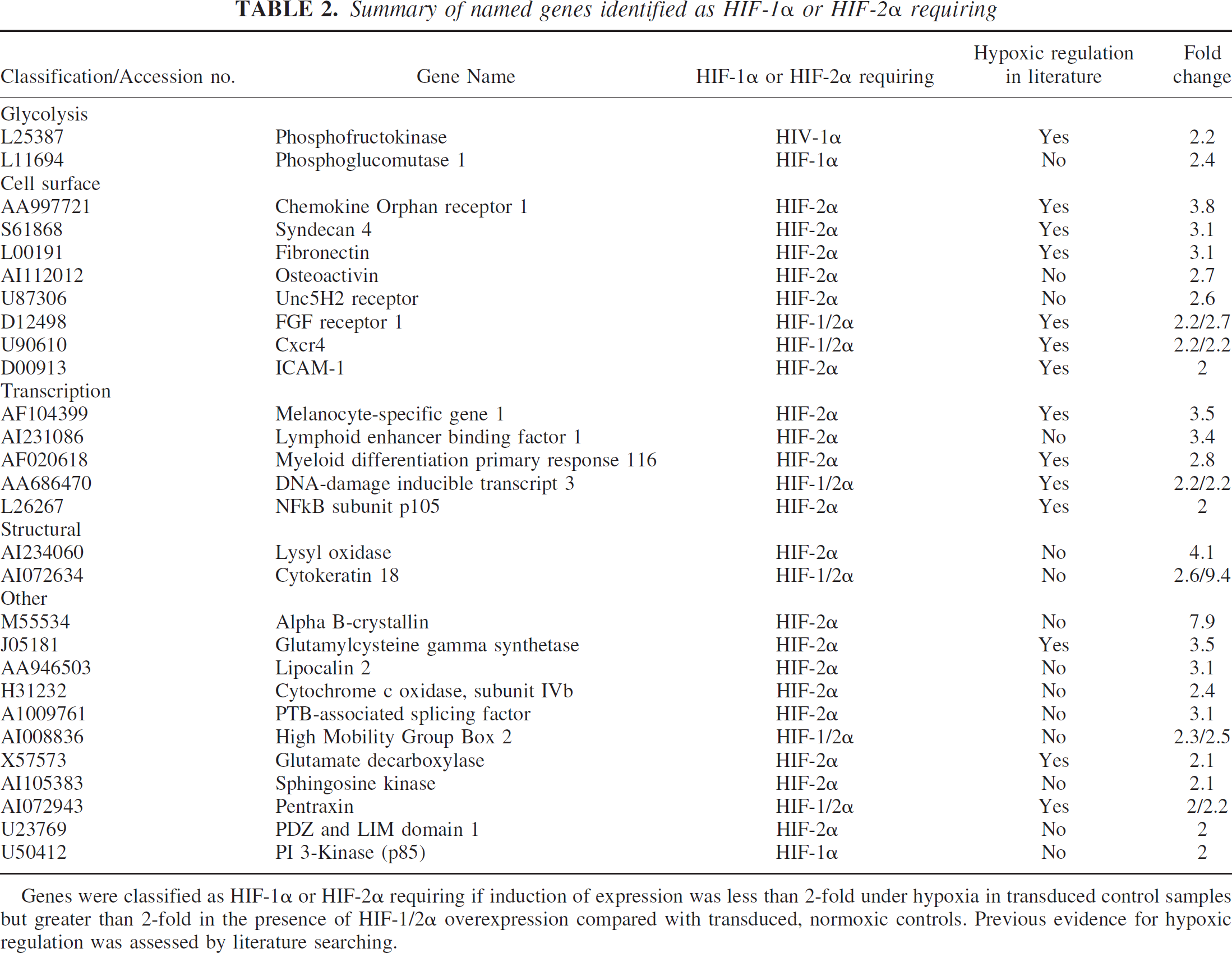
Genes were classified as HIF-1α or HIF-2α requiring if induction of expression was less than 2-fold under hypoxia in transduced control samples but greater than 2-fold in the presence of HIF-1/2α overexpression compared with transduced, normoxic controls. Previous evidence for hypoxic regulation was assessed by literature searching.
Following exposure to 8 hours hypoxia, induction of many known hypoxically regulated genes such as VEGF, glucose transporter 1 (Glut1) and hexokinase was observed (data not shown). The level of hypoxic induction of many of these genes was only just above the 2-fold threshold reiterating previous studies investigating differential expression patterns in neurons where subtle changes in gene expression were observed (Read et al., 2001). Indeed, induction of some well-characterized hypoxia-induced genes such as phosphofructokinase and Glut3 was not sufficient to classify these genes as hypoxically regulated when using a 2-fold cut off threshold. In order to maximize identification of HIF regulated genes we attempted to bias HIF-mediated transcription by overexpression of HIF-1α and HIF-2α in primary neurons under hypoxia. Taqman Q-PCR analysis of several known HIF-regulated genes including Glut3 and VEGF demonstrated a proof of principle and HIF-2α overexpression significantly increased the response to hypoxia of both these genes.
Named genes relevant to apoptosis
Summary of HIF-1α or HIF-2α requiring EST orthologues
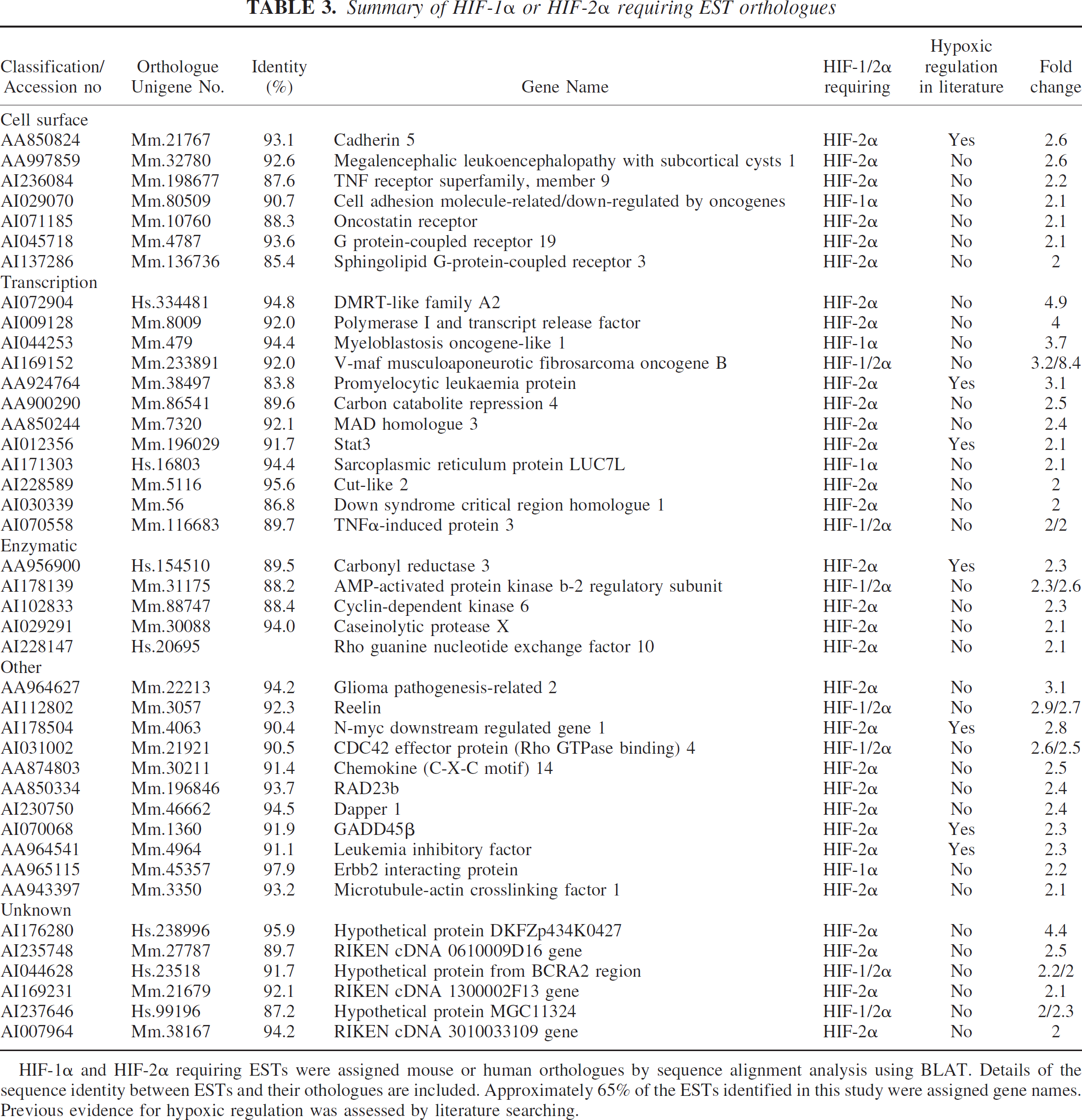
HIF-1α and HIF-2α requiring ESTs were assigned mouse or human orthologues by sequence alignment analysis using BLAT. Details of the sequence identity between ESTs and their othologues are included. Approximately 65% of the ESTs identified in this study were assigned gene names. Previous evidence for hypoxic regulation was assessed by literature searching.
Of the named genes identified as requiring HIF-1α or HIF-2α overexpression for detection under hypoxia, approximately half have previously been cited as hypoxically or ischemically regulated. This strongly suggests that true hypoxically regulated genes were identified from this study. These include a few genes that were previously demonstrated to be HIF regulated such as phosphofructokinase, however most have not previously been directly linked to HIF-1 or HIF-2 regulation. A number of interesting cell surface proteins were identified from the named gene list with possible implications in cell death pathways and neuroprotection. These included the Chemokine Orphan Receptor 1 that was slightly hypoxically regulated (<2-fold) in controls but showed greater hypoxic induction (3.8-fold) in the presence of HIF-2α overexpression. This gene encodes a putative 7 transmembrane receptor protein and the human orthologue was previously identified as the G-protein coupled receptor RDC1 (Libert et al., 1990). This protein was originally described as a receptor for a neuropeptide with regenerative and neuroprotective properties, vasoactive intestinal peptide (Sreedharan et al., 1991). More recent work however, has implicated RDC1 as a receptor for the hypotensive peptide adrenomedullin (ADM, Kapas and Clark, 1995; Ladoux and Frelin, 2000). Expression of ADM was previously demonstrated to be hypoxically regulated via a HIF-1 dependent mechanism (Ladoux and Frelin, 2000) and this effect was confirmed in our study. Upregulation of adrenomedullin expression was also observed in the ischemic cortex of rats following MCAO and administration of ADM before and after MCAO increased the degree of ischemic injury (Wang et al., 1995). A critical role of ADM in focal brain ischemia and stroke has therefore been suggested. Although RDC-1 has also previously been demonstrated to be upregulated by hypoxia in astrocytes, HIF-2 regulation has not been reported (Ladoux and Frelin, 2000). The role of RDC1 as a receptor for ADM remains controversial (Chakravarty et al., 2000; Kennedy et al., 1998) however, the importance of this protein in understanding ischemic cell death and as a potential therapeutic target is evident.
Expression of the receptor UNC5H2 was also identified as HIF-2α requiring in this study. This receptor plays an important role in axonal guidance and neuronal migration during development by acting as a receptor for the chemo-attractant/ chemo-repellent, netrin-1 (Tessier-Lavigne and Goodman, 1996). Interestingly, in terms of apoptotic function, UNC5H2 contains an intracellular death domain, a domain usually found within members of the death receptor family such as Fas and TNF receptor (Hofmann and Tschopp, 1995; Leonardo et al., 1997). UNC5H2 has also previously been demonstrated to be a dependence receptor. Such receptors create cellular states of dependence on their ligands by inducing apoptosis when unoccupied by ligand but inhibiting apoptosis in the presence of ligand (Llambi et al., 2001). Netrin-1 itself was not upregulated under hypoxic conditions in these studies. Induction of UNC5H2 could, therefore, play an important role in induction of apoptosis following a hypoxic or ischemic insult and as such provides a novel potential target for therapeutic design.
Although there has been some evidence for upregulation of the Fibroblast Growth Factor Receptor 1 (FGFR1) following ischemia in the brain (Liu and Zhu, 1999; Masumura et al., 1996) this receptor has not previously been identified as being HIF-regulated. In this study FGFR-1 was moderately upregulated by hypoxia alone (<2-fold), however in the presence of HIF-1α or HIF-2α overexpression induction was increased above the 2-fold threshold. Since FGF is effective at reducing post-ischemic neuronal damage in animal models of middle cerebral artery occlusion this result suggests that FGFR-1 upregulation may be an important factor in mediating such neuroprotection (Li and Stephenson, 2002; Song et al., 2002). Interestingly the expression profile of syndecan 4 is highly similar to FGFR1. Syndecan-4 is a transmembrane heparan sulphate proteoglycan (HSPG), a family of proteins that have been shown to act as acceptors of ligands such as ECM proteins and soluble growth factors such as FGF and VEGF (Hartmann and Maurer, 2001; Rapraeger, 2000). Binding of FGF to heparan sulphate proteoglycans such as syndecan-4 promotes ligand-receptor binding and enhances downstream signaling by promoting receptor multimerization (Rapraeger, 1995). Upregulation of syndecan 4 could potentially be a critical factor involved in FGF-mediated neuroprotection against ischemia.
Other named genes identified as HIF-2α-requiring and with potential relevance to ischemic cell death include lipocalin 2 and sphingosine kinase. Neither of these genes has previously been reported to be hypoxically regulated. The lipocalins are a family of small-secreted polypeptides with diverse biological functions (Flower, 1996). Lipocalin 2 was previously demonstrated to be upregulated and to mediate a strong pro-apoptotic effect in hematopoietic cells following cytokine withdrawal (Devireddy et al., 2001). The pro-apoptotic effect of lipocalin 2 was observed to be cell-type specific and to be dependent on binding to a cell surface receptor that has yet to be elucidated (Devireddy et al., 2001). The expression of lipocalin 2 in the brain and potential role in mediating apoptosis in neuronal cells has not previously been reported. These studies suggest that lipocalin 2 is expressed in cortical neurons and could potentially be involved in apoptotic pathways following hypoxia.
Sphingosine kinase catalyzes the phosphorylation of sphingosine-1-phosphate (SPP) a member of an important class of lipid mediators that influence signaling pathways involved in cell survival and proliferation (Maceyka et al., 2002). Activation of sphingosine kinase and subsequent phosphorylation of sphingosine-1-phosphate was previously demonstrated to prevent TNF induced apoptosis (Xia et al., 2002) and overexpression of sphingosine kinase mediated protection against trophic factor withdrawal in neuronal cell types (Edsall et al., 2001). Interestingly, activated sphingosine kinase has also been demonstrated to cause downregulation of ceramide, another member of the sphingolipid metabolite family that promotes apoptosis in neurons (Brugg et al., 1996; Hartfield et al., 1997; Mitoma et al., 1998). Sphingosine kinase has not previously been observed to be hypoxically regulated and may prove to be a key regulator of hypoxically mediated cell survival. The identification of enzymes involved in neuroprotection is of particular importance because of the established rationale for developing enzyme inhibitors and substrates as therapeutic agents.
EST-derived genes relevant to apoptosis
Similarly to the named genes classified as HIF-requiring, the ESTs identified correspond to a diverse set of genes with a wide range of biological functions including several with potential importance in ischemic disease. Furthermore, the majority of these ESTs have not previously been cited as hypoxically regulated and hence represent potentially novel hypoxia induced factors.
Several cell surface receptors were identified with particular relevance to cell death mechanisms. These included Sphingolipid G-protein-coupled receptor 3 (Edg-3), a receptor for SPP that was demonstrated to be HIF-2α requiring in this study and has been previously implicated in cell proliferation and survival signaling pathways (An et al., 2000). A second G-protein couple receptor, GPCR19 was also identified as HIF-2α-requiring. This has been demonstrated to be abundantly expressed in the brain (O'Dowd et al., 1996), however the function of this receptor has yet to be elucidated.
The largest group of gene orthologues identified from the HIF-requiring ESTs represented transcription factors. Several of these have particular relevance to cell survival mechanisms and provide clues for potential therapeutic targeting strategies. Promyelocytic leukaemia protein (PML) is a tumor/growth suppressor that has previously been demonstrated to be a requirement for induction of apoptosis by a wide range of apoptotic stimuli (Salomoni and Pandolfi, 2002; Wang et al., 1998). The pro-apoptotic mechanisms that are exerted by PML have yet to be fully elucidated, however, recent work has demonstrated that PML contributes to TNF-α induced apoptosis by inhibiting the NF-kB survival pathway (Wu et al., 2003). Although PML has previously been demonstrated to be upregulated in animal models of MCAO, HIF-mediated regulation of expression has not been reported (Hayashi et al., 2001). In our model PML was upregulated greater than 3-fold under hypoxia in the presence of HIF-2α overexpression. Hypoxic regulation of PML may play an important role in mediating post hypoxic/ischemic cell death and provides a potential protein for therapeutic targeting.
The gene encoding the AMP-activated protein kinase (AMPK) β-2 regulatory subunit was responsive to both HIF-1α and HIF-2α under hypoxia. AMPK is a member of a metabolite-sensing kinase family that plays important roles in response to metabolic stress and ATP depletion (Kemp et al., 1999). Although AMPK activation has been observed in response to ischemia and nutrient starvation, regulation by HIF has not previously been demonstrated. High expression levels of AMPK subunits have been reported in the brain and activation of AMPK was previously demonstrated to protect neuronal cells against death induced by glucose deprivation, chemical hypoxia and glutamate exposure (Culmsee et al., 2001). HIF-1/2α activation of AMPK-β2 may, therefore, initiate a neuroprotective response to hypoxic injury in the brain and may provide a novel target for therapeutic intervention possibly using a gene therapy based approach.
Tumor necrosis factorα-induced protein 3 (or A20) is a zinc finger protein that is rapidly upregulated in response to a variety of stimuli including cytokines such as tumor necrosis factor (Song et al., 1996). Induction of A20 has been demonstrated to inhibit nuclear NF-kappa B activity and TNF-mediated apoptosis (He and Ting, 2002; Lee et al., 2000; Song et al., 1996). In this study A20 expression was slightly upregulated (<2-fold) by hypoxia alone but was enhanced above the 2-fold threshold by the overexpression of HIF-1α or HIF-2α. Interestingly, growth arrest and DNA damage inducible gene 45β (GADD45β) was also identified as a potential HIF-regulated gene in this study. GADD45β expression is upregulated following stress in a NF-kB dependent mechanism and has been demonstrated to downregulate the pro-apoptotic JNK-signaling pathway induced by TNFα (De Smaele et al., 2001; Jin et al., 2002). Although expression levels of both A20 and GADD45β appear low in neuronal cell types (Takekawa and Saito, 1998) HIF-mediated gene expression may play an important factor in regulating this cell death pathway and may provide clues for generation of potential therapeutic strategies for abrogating hypoxic injury.
Cell death pathways
The genes identified in this study are diverse in their actions with respect to mechanisms of cell death, highlighting the complexity of the pathways involved in mediating the responses to hypoxia in neuronal cells. At the center of many of these response pathways is the transcription factor NF-κB that has been demonstrated to be activated in the brain following ischemic insult and may play a critical role in determining cell fate (Mattson et al., 2000). Recent studies have identified HIF-1α as a unique component of the NF-κB-mediated inflammatory/survival response pathway and TNFα induced accumulation of HIF-1α protein was demonstrated to be dependent upon NF-κB-driven transcription (Jung et al., 2003). In vitro protection studies have shown that activation of NF-κB rescues neurons against excitotoxic and metabolic insults relevant to the pathogenesis of stroke, (Cheng et al., 1994; Yu et al., 1999). Further evidence that NF-κB plays a protective role in neurons is provided by in vivo studies that have demonstrated increased expression levels of a κB-responsive protein, neuronal inhibitory protein-1 (NAIP) in neurons resistant to ischemic brain injury, and that overexpression of NAIP increases resistance of neurons to ischemia-mediated cell death (Xu et al., 1997). Proteins involved in NF-κB signalling are therefore potentially important targets for therapeutic intervention in neurological disorders such as stroke. Several of the genes identified above have been implicated in NF-κB signalling pathways and as such provide candidate targets for therapeutic design. These include pro-apoptotic genes such as PML and TNF-α induced-3 that have been shown to increase TNF induced apoptosis through inhibition of NF-κB pathways (Lee et al., 2000; Song et al., 1996; Wu et al., 2003) and genes such as GADD 45β and sphingosine kinase that mediate anti-apoptotic effects through NF-κB activation (De Smaele et al., 2001; Jin et al., 2002; Xia et al., 2002). Additionally, the netrin-1 receptor UNC5H2 contains a death domain that is related to that of TNF receptor and Fas and as such could potentially mediate its effects through NF-κB pathways (Tessier-Lavigne and Goodman, 1996).
Concluding remarks
Overexpression of HIF-1α and HIF-2α enabled the identification of far more differentially expressed genes under hypoxia than was achieved in untranduced or control transduced neuronal cultures. Robust validation of the expression data obtained was achieved by performing Taqman Q-RT PCR analysis of some of the more relevant genes identified.
Interestingly, the data strongly implies that dimerization of ARNT with HIF-1α or HIF-2α results in the activation of different sets of target genes. Indeed very few genes were identified that were activated by both factors under hypoxia and furthermore no genes were identified to be HIF-2α requiring that were previously cited in the literature to be HIF-1α regulated. This implies different roles for HIF-1α and HIF-2α in mediating the cellular response to hypoxia.
Many more HIF-2α requiring genes were identified than HIF-1α requiring genes, and of the latter the majority of genes were previously implicated as being regulated by hypoxia or ischemia. This is understandable since HIF-1 regulated gene expression has been far more comprehensively studied than HIF-2. Many of the genes identified as HIF-2α requiring displayed particular relevance to cell survival pathways implicating a role for HIF-2α in regulating neuroprotective and/or neurotoxic responses to hypoxia. Previous reports have observed that the HIF-2α protein is more stable than HIF-1α under normoxia and implied that HIF-1α is more important in mediating the transcriptional response to hypoxia (Bernaudin et al., 2002b). Our study has demonstrated that overexpression of HIF-2α under normoxia was insufficient to drive expression of HIF-2α target genes (data not shown). However, a combination of HIF-2α expression and hypoxic challenge resulted in significant activation of many target genes. This suggests that even if HIF-2α were more stable than HIF-1α under normoxia, transcriptional activation of target genes is still dependent on a hypoxic environment.
There is strong evidence that the genes identified in this study are true HIF regulated genes and not the result of a stress response to viral transduction. The expression profiles for each gene were normalized to control levels from cells that were transduced with control lentiviral vectors. Furthermore, as mentioned above, the activation of potential HIF target genes was only observed in cells that were subjected to a combination of HIF overexpression and hypoxia and not in cells overexpressing HIF in a normoxic environment. The majority of the genes identified as HIF-requiring demonstrated some upregulation in untransduced and control transduced samples following hypoxia, however, the induction of gene expression was not sufficient to classify these genes as differentially regulated using the standard cut off threshold of 2-fold. Many of these genes were probably not previously identified as hypoxically regulated due to this moderate hypoxic response. Nonetheless such genes could have major importance in understanding hypoxic cell death mechanisms and identification of novel therapeutic targets for treating ischemic disease. A very small number of genes showed no hypoxic regulation whatsoever in control samples, however, were upregulated by a combination of HIF overexpression and hypoxia. An example of such a gene is gamma-glutaylcysteine synthetase (GGCS) that was highly upregulated above threshold levels (>6-fold) by HIF-2α overexpression under hypoxia but showed no upregulation in hypoxic controls. These genes could represent those that respond to hypoxia in non-neuronal cell types and require levels of HIF expression greater than those observed in neuronal cells. Indeed, GGCS for example has previously been demonstrated to be upregulated in hypoxic tumor cells (O'Dwyer et al., 1994).
In summary, lentiviral vectors were used to bias gene expression in a physiological pathway relevant to stroke. We have identified many novel HIF regulated genes with diverse functions in hypoxia mediated signaling and survival pathways. Further investigation will reveal the importance of these genes in mediating cellular responses to hypoxic stress and as potential targets for the development of therapeutic strategies for the treatment of ischemic disease such as stroke (Karakesisoglou et al., 2000).
