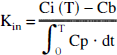Abstract
Recent results have demonstrated that the spin trapping agent α-phenyl-N-tert-butyl nitrone (PBN) reduces infarct volume in rats subjected to 2 hours of middle cerebral artery occlusion, even when given 1 to 3 hours after the start of recirculation. In the current study, the authors assessed the effect of NXY-059, a novel nitrone that is more soluble than PBN. Loading doses were given of 0.30, 3.0, or 30 mg · kg−1 followed by 0.30, 3.0, or 30 mg · kg−1 · h−1 for 24 or 48 hours. Dose–response studies showed that when treatment was begun 1 hour after recirculation, 0.30 mg · kg−1 had a small and 30 mg · kg-i a marked effect on infarct volume. At equimolar doses (3.0 mg · kg−1 for NXY-059 and 1.4 mg · kg−1 for PBN), NXY-059 was more efficacious than PBN. Similar results were obtained when a recovery period of 7 days was allowed. The window of therapeutic opportunity for NXY-059 was 3 to 6 hours after the start of recirculation. Studies of the transfer constant of [14C]NXY-059 showed that, in contrast to PBN, this more soluble nitrone penetrates the blood-brain barrier less extensively. This fact, and the pronounced antiischemic effect of NXY-059, suggest that the delayed events leading to infarction may be influenced by reactions occurring at the blood–endothelial interface.
A spin trap nitrone, a-phenyl-N-tert-butyl nitrone (PBN), has previously been shown to ameliorate ischemic brain damage. The original observations were made in gerbils subjected to 5 to 10 minutes of forebrain ischemia (Oliver et al., 1990; Phillis and Clough-Helfman, 1990). In this model, damage primarily affects hippocampal CA1 neurons, which become necrotic after a delay of 1 to 3 days. Although a similarly delayed CA1 cell death is observed after forebrain ischemia in the rat, PBN failed to ameliorate the damage in that species, suggesting species differences in pathophysiologic mechanisms (Pahlmark and Siesjö, 1996).
Recent results demonstrate, though, that PBN markedly reduces infarct volumes in rats subjected to long periods of focal ischemia induced by middle cerebral artery (MCA) occlusion. The results emanate from experiments on permanent distal occlusion of the MCA (Cao and Phillis, 1994), or from transient (proximal) MCA occlusion of 2-hour duration (Zhao et al., 1994). In the first of these studies, infarct volume, as assessed by 2,3,5-triphenyltetrazolium chloride (TTC) staining after 2 days of occlusion, was reduced even if administration of PBN (100 mg · kg−1, 4 intraperitoneal doses, 12 hours apart) was delayed for 5 hours, with some remaining effect after 12 hours (Cao and Phillis, 1994). In the second, the window of therapeutic opportunity was 3 hours after the initiation of reperfusion (Zhao et al., 1994).
These results put PBN in the focus of interest, since they suggest that the nitrone acts within a therapeutic window of clinical relevance. Reperfusion after transient MCA occlusion was shown to be accompanied by an initial recovery of the bioenergetic state of focal and perifocal (“penumbral”) areas, followed by secondary deterioration after 4 to 6 hours, the latter being ameliorated by PBN (Folbergrová et al., 1995). Studies of mitochondrial function in vitro gave similar results, suggesting that the secondary deterioration of the bioenergetic state was caused by mitochondrial dysfunction, which was mitigated by PBN (Kuroda et al., 1996). As is discussed later, it is less likely that PBN improves microcirculation.
We have explored the effect of a newly synthesized nitrone, NXY-059, which is more soluble than PBN, studying dose-response effects, windows of therapeutic opportunity, and recovery for 7 days with a histopathologic endpoint. In all experiments, MCA occlusion of 2-hour duration was used. The results demonstrate that NXY-059 works within the same window of therapeutic opportunity as PBN, but is more efficacious than PBN, and can be infused to give a lasting effect at 7 days of recovery. A novel finding is that NXY-059 has this marked effect, although it turned out to be sparingly permeable through the blood–brain barrier (BBB). The implication of this finding for the pathophysiologic mechanism of focal ischemic damage is discussed here.
MATERIALS AND METHODS
Chemicals
NXY-059 (disodium 4-[(tert-butylimino)methyl]benzene-1,3-disulfonate N-oxide), a nitrone, was developed and kindly given to us by Centaur Pharmaceuticals, Inc. (Sunnyvale, CA, U.S.A.). Its chemical structure is shown in Fig. 1. NRB-022 ([14C]NXY-059) (molar activity 2.2 mCi · mmol−1) was a kind gift of Astra Arcus AB (Södertälje, Sweden). The PBN was purchased from Sigma Chemical Co. (St. Louis, MO, U.S.A.).
Chemical structures of two spin trap nitrones, α-phenyl-N-tert-butyl nitrone (PBN) and disodium 4-[(tert-butylimmo)-methyl]benzene-1,3-disulfonate N-oxide (NXY-059).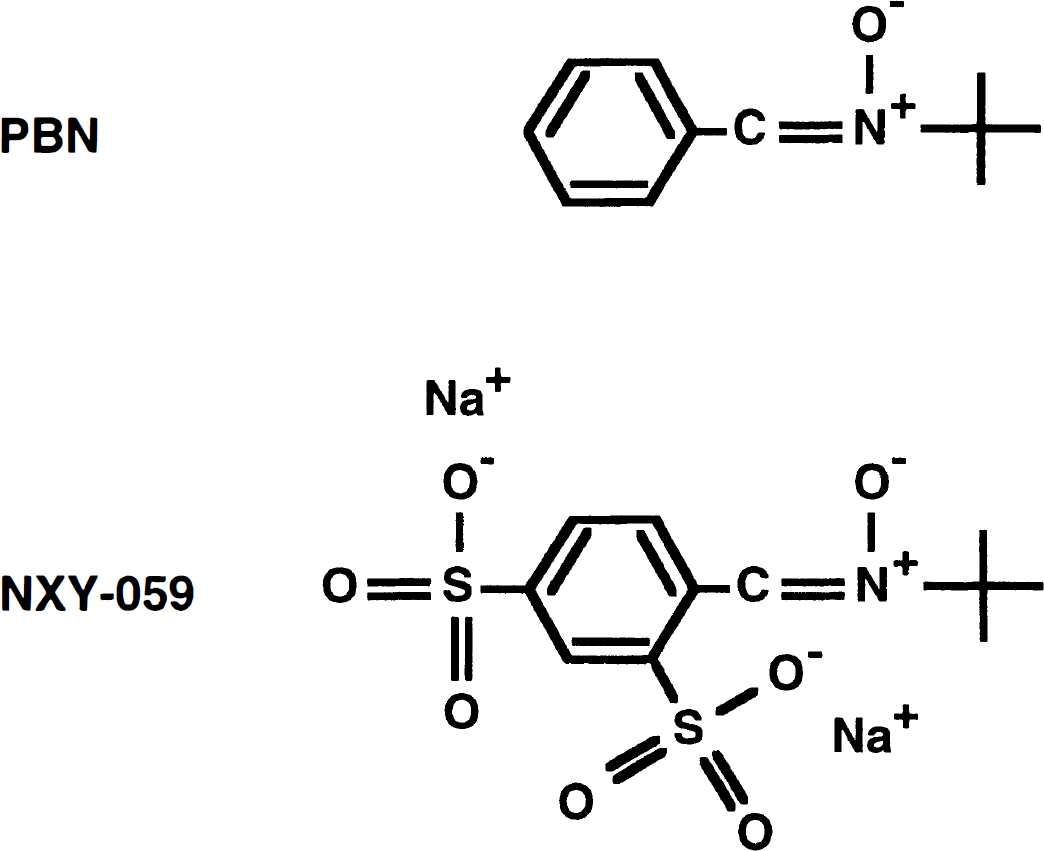
Animal preparation and transient middle cerebral artery occlusion
Our procedures for inducing transient MCA occlusion in anesthetized rats were as described previously (see Zhao et al., 1994; Kuroda et al., 1996) and are summarized here. Male Wistar rats (Mollegaard's Breeding Center, Copenhagen) were fasted overnight but had free access to water (body weight after overnight fast 300 to 345 g). Anesthesia was induced by inhalation of 3.0% halothane in N2O:O2 (70%:30%), and the animals were intubated. They were artificially ventilated with 1.0% to 1.5% halothane in N2O:O2 (70%:30%) during operation. The tail artery was cannulated to monitor arterial blood gases, pH, blood glucose, and blood pressure during surgery. A catheter also was inserted into the right jugular vein for administration of vehicle, PBN, or NXY-059, and then was externalized on the back of the animals at the nape of their neck. Heparin (0.1 mL, 300 units · mL−1) was given just before induction of ischemia to prevent thrombosis distal to the inserted filament. The occlusion was produced using a monofilament fishing line (Stren Supertough, Du Pont, Wilmington, DE, U.S.A.; diameter 0.25 mm). Its tip was rounded by brief heating near an electrical heater under a surgical microscope and coated with silicon rubber (Silascon RTV-8600, Dow Corning K.K., Japan) to make a distal cylinder 0.28 mm in diameter and 5 mm in length. The right common, internal, and external carotid arteries were exposed through a ventral midline neck incision. The external carotid artery was ligated. The common carotid artery was closed by a ligature and was temporarily closed just proximal to the carotid bifurcation by a microvascular clip. A small incision was made in the common carotid artery, and the occluder filament was inserted into the internal carotid artery through the common carotid artery. The filament was advanced until a faint resistance could be felt. Length of the inserted filament was about 19 mm from the carotid bifurcation. A transient (2- to 3-minute) decrease in blood pressure was observed. When the MCA occlusion had been performed, the animals were extubated and allowed to wake up and resume spontaneous breathing. After light reanesthesia with halothane through a face mask, the filament was withdrawn after 2 hours to allow recirculation through the circle of Willis. During the operation, an electrical temperature probe was inserted into the rectum to monitor core temperature, which was regularly maintained at 37.0° to 37.5°C using a Temperature Controller CMA/150 (Carnegie Medicin AB, Stockholm, Sweden). During the 2-hour period of ischemia and for 2 hours after onset of reperfusion (total of 4 hours), the animals were placed in a Styrofoam box in which temperature was kept between 0° and 10°C by flushing the box with air cooled by dry ice. This was performed to avoid the hyperthermia that otherwise would occur from ischemia of hypothalamus, and to keep core temperature close to normal levels during and after MCA occlusion (see Kuroda et al., 1996).
Neurologic assessment
After surgery, the neurologic status of each rat was evaluated according to the neurologic grading scores described by Bederson and colleagues (Bederson et al., 1986b). Thus, rats that extended both forelimbs toward the floor when the animals were held up by the tail and that had no other neurologic deficit were graded O. Rats that showed consistent flexion of the left forelimb and no other abnormality when the animals were held up by the tail were graded 1. Rats that had reduced resistance to lateral push toward the left (paretic) side and gait toward the left side were graded 2. Rats that had same behavior as grade 2 and consistent circling toward the left side were graded 3.
Neurologic examination was carried out in all animals every hour during the 2-hour MCA occlusion. Only rats scoring 0 before surgery and 3 during ischemia were included in the study (Kuroda et al., 1996).
Experimental protocol
Our first series was performed to establish a dose-response curve for NXY-059 (0.30, 3.0, or 30 mg · kg−1 intravenously, followed by continuous infusion of 0.30, 3.0, or 30 mg · kg−1 · h−1 over 24 hours, respectively) and to compare its effect with that of PBN. In one experiment, the treatments were started 1 hour after the start of recirculation, after 2 hours of MCA occlusion. The results showed that infarct volume, assessed after 2 days of recovery, was significantly reduced in a dose-dependent manner and that NXY-059 is much more efficacious than PBN on a molar basis (see later). Therefore, the highest dose (30 mg · kg−1 intravenously and 30 mg · kg−1 · h−1 infusion) of NXY-059 was chosen for the subsequent experiments. In the next series, we evaluated the window of therapeutic opportunity. Treatment with NXY-059 was started 3 or 6 hours after the start of recirculation and was continued for 24 hours. In the third series, we assessed the long-term effects of NXY-059 treatment over 48 hours on ischemic brain damage after 7 days of recovery using histopathologic techniques. In the final experiment, we evaluated the permeability of NXY-059 through the BBB using 14C-labeled drug. Notice that the solubility of this drug is different from that of PBN, which can easily pass through the BBB (Chen et al., 1990; Cheng et al. 1993).
Dose–response study of NXY-059
In this series, the animals were divided into five groups, all being allowed a recovery period of 48 hours after 2 hours of MCA occlusion. In all groups, the drug was given by bolus injection after 1 hour of recirculation and then by continuous infusion over 24 hours. For continuous infusion of the drug in awake and freely moving rats for a long time, a polyethylene tube (0.58 mm internal diameter, 0.96 mm external diameter) was inserted into the right jugular vein and externalized on the back of the animals at the nape of their neck. The drug was infused through the polyethylene tube passing through a stainless steel spring using a CMA Swivel and CMA/100 Microinjection Pump (Carnegie Medicin AB). During continuous infusion of the drug, the animals were allowed to move freely in their cages.
Each drug, NXY-059 or PBN, was dissolved in Krebs solution. The following groups were studied:
Low-dose NXY-059 group (n = 8): 0.30 mg · kg−1 by intravenous bolus injection and 0.30 mg · kg−1 · h−1 by intravenous infusion Middle-dose NXY-059 group (n = 12): 3.0 mg · kg−1 by intravenous bolus injection and 3.0 mg · kg−1 · h-1 by intravenous infusion High-dose NXY-059 group (n = 8): 30 mg · kg−1 by intravenous bolus injection and 30 mg · kg−1 · h−1 by intravenous infusion PBN group (n = 6): 1.4 mg · kg−1 by intravenous bolus injection and 1.4 mg · kg−1 · h−1 by intravenous infusion
This dose of PBN is equimolar to that of the middle-dose NXY-059 group (3.0 mg · kg−1 · h−1). A control group of rats (n = 10) received an equivalent volume of Krebs solution by intravenous bolus and continuous intravenous injections. Neurologic evaluation was performed at 24 and 48 hours of recirculation (see earlier). Infarct volume was assessed using a TTC staining technique (see later).
Window of therapeutic opportunity for NXY-059
The animals were divided into three groups and were allowed a recovery period of 48 hours after 2 hours of MCA occlusion. In all groups, continuous infusion of Krebs solution was started at 1 hour of recirculation. Treated animals received 30 mg · kg−1 of NXY-059 by intravenous bolus injection at 3 (n = 8) or 6 hours (n = 8) after the start of recirculation, and then 30 mg · kg−1 · h−1 of NXY-059 by continuous intravenous infusion for 24 hours. Control animals (n = 5) received only intravenous infusion of Krebs solution for 24 hours. Neurologic evaluations were carried out at 24 and 48 hours of recirculation (see earlier). Infarct volume was assessed after 48 hours of recovery using the TTC staining technique (see later).
Long-term study of NXY-059
The animals were divided into two groups and were allowed a recovery period of 7 days after 2 hours of MCA occlusion. In the treated group (n = 8), 30 mg · kg−1 of NXY-059 was given by intravenous bolus injection at 1 hour of recirculation, and then 30 mg · kg−1 · h−1 of NXY-059 was given by continuous intravenous infusion for 48 hours, as described earlier. A control group of rats (n = 7) received only an equivalent volume of Krebs solution by infusion for 48 hours.
Neurologic evaluations were carried out at 2 and 7 days, respectively, of recirculation (see earlier). Infarct volume was assessed using transcardiac perfusion and conventional histopathologic staining techniques (see later).
TTC staining
The rats were anesthetized by inhalation of 3.0% halothane in N2O:O2 (70%:30%) and killed by decapitation at 48 hours of recirculation, The brain was quickly removed and chilled in ice-cold saline for 10 minutes. Twelve 1-mm coronal slices were cut with a tissue slicer, beginning 1 mm posterior to the anterior pole. The slices were immersed in a saline solution containing 1.0% TTC at 37°C for 30 minutes (Bederson et al., 1986a) and fixed by immersion in 4.0% phosphate-buffered formalin solution. Each brain slice was photographed with black-and-white film (TMAX 100 pro, Kodak, Rochester, NY, U.S.A.), and the unstained area in each photograph was quantified from the developed film using a CCD video camera (SPTM124, Sony, Park Ridge, NJ, U.S.A.) and a video image analyzing system (NIH image, version 1.55). The infarct area in each slice was obtained by subtracting the area of normal tissue of the ipsilateral hemisphere from the total area of the contralateral hemisphere to avoid overestimation of infarct volume resulting from cerebral edema in acute stage of ischemia. The total infarct volume was determined by summing the infarct areas of the 12 slices and was presented as the percentage of the infarct to the total volume of the contralateral hemisphere.
Histopathology
After 7 days of recovery, the animals were reanesthetized and artificially ventilated with 3.0% halothane in N2O:O2 (70%:30%). A thoracotomy was made, and a cannula was inserted into the ascending aorta through the left ventricle. The animals were transcardially perfused with heparinized saline and then with phosphate-buffered 4% formaldehyde (pH 7.4), and the brains were removed. The brain tissues were dehydrated, embedded in paraffin, sectioned coronally at 5-mm thickness, and stained with celestine blue and acid fuchsin for histopathologic evaluation.
Volumes of brain damages were measured according to Osborne and collaborators (Osborne et al., 1987). Coronal sections at 10 levels were examined, and the outlines of infarction and selective neuronal necrosis were drawn on brain maps. The areas of infarction and selective neuronal necrosis at each level were directly analyzed using a video image analyzing system (see earlier) because cerebral edema observed at 2 days of reperfusion had already disappeared. The volume (percentage of the contralateral hemisphere) of infarction, selective neuronal necrosis, and total ischemic damage then was calculated from all sections.
Blood–brain barrier permeability study of [14C]NXY-059
Before experiments, [14C]NXY-059 was mixed with nonradioactive (“cold”) NXY-059 in a 1:1 ratio to reduce its radioactivity. Sixteen rats were used for this study. They were divided into three groups. In the first group, 30 mg · kg−1 of [14C]NXY-059 (86 · Ci · kg−1) was given by intravenous bolus injection to rats subjected to sham operations 3 hours after the operation (n = 5). The other two groups of animals were allowed a recovery period of 1 (n = 5) or 4 hours (n = 6) after 2 hours of MCA occlusion. In the former, 30 mg · kg−1 of [14C]NXY-059 was given by intravenous bolus injection 1 hour after the onset of recirculation. In the latter, 30 mg · kg−1 of cold NXY-059 was given by intravenous bolus injection 1 hour after the onset of recirculation, and then 30 mg · kg−1 · h−1 of cold NXY-059 was continuously infused intravenously. At 4 hours of recovery, 30 mg · kg−1 of [14C]NXY-059 was injected as an intravenous bolus. Uptake of [14C]NXY-059 into the brain was measured in awake animals placed in a small tube-shaped Plexiglas cage, which restricted their movements. Each rat received the [14C]NXY-059 20 minutes before decapitation through the catheter placed into the tail vein. Arterial blood samples (0.05 mL) were collected in 0.4-mL microcentrifuge tubes before and 0.25, 0.5, 1, 2, 5, 10, and 20 minutes after injection of [14C]NXY-059. Just after sampling of the last arterial blood, the animals were decapitated, and the brain was quickly removed. After removal of the arachnoidal membrane and subarachnoidal blood vessels, the brain was immersed in 2-methylbutan (KEBO Lab, Spanga, Sweden), which was cooled to −40°C by liquid nitrogen. The brain was kept in a deep freezer until determination of radioactivity.
Arterial blood samples were centrifuged for 45 to 60 seconds (Sigma 110, Berlin, Germany) to obtain plasma. Twenty microliters of plasma were pipetted into vials containing 0.8 mL of a 1:1 mixture of soluene (Packard, Groningen, The Netherlands) and isopropanol (Fluka Chemie Ab, Buchas, Germany). The following day, 3.5 mL of Ready-Safe (Beckman Instrument, Fullerton, CA, U.S.A.) was added and the vials were mixed and left for at least 5 hours before counting. Subregions from the lateral caudoputamen, somatosensory cortex (“penumbra”), and motor cortex (“focus”) were dissected from coronal sections of 2-mm thickness through the caudoputamen at −15°C. The brain samples were dissolved in soluene over 24 hours in room temperature and then prepared for scintillation counting together with the blood samples. Radioactivity was counted on a liquid scintillation analyzer, TRI-CARB 2100TR (Packard Instrument Company, Meriden, CT, U.S.A.).
A blood-to-brain transfer content (Kin for [14C] NXY-059) was determined according to the equation of Ohno and colleagues (1978) as follows:
Statistics
All values are presented as mean ± SD or the median, the 25th percentile, and the 75th percentile. For comparisons of parametric data, analysis among multiple groups were carried out by one-factor analysis of variance followed by Dunnett's test. Comparison between two groups was carried out using the unpaired t test. For comparisons of nonparametric data, analysis among multiple groups were carried out by Kruskal-Wallis test followed by Mann-Whitney U test. Statistical significance between two groups of nonparametric data was determined using Mann-Whitney U test. Statistical significance was set at P < 0.05.
RESULTS
Physiologic parameters
There were no significant differences in arterial P
Core temperature during recirculation after 2 hours of middle cerebral artery occlusion
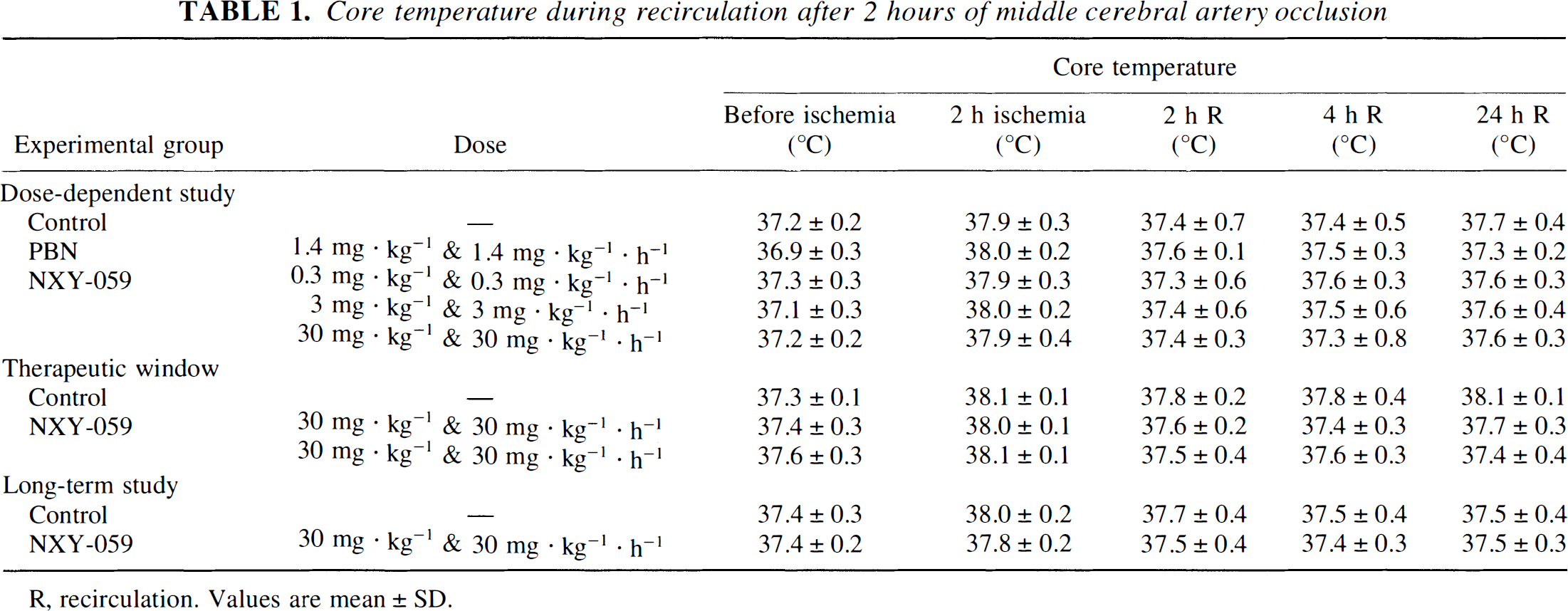
R, recirculation. Values are mean ± SD.
Dose-dependence study of NXY-059: comparison with PBN
Neurologic deficits during recirculation after 2 hours of middle cerebral artery occlusion
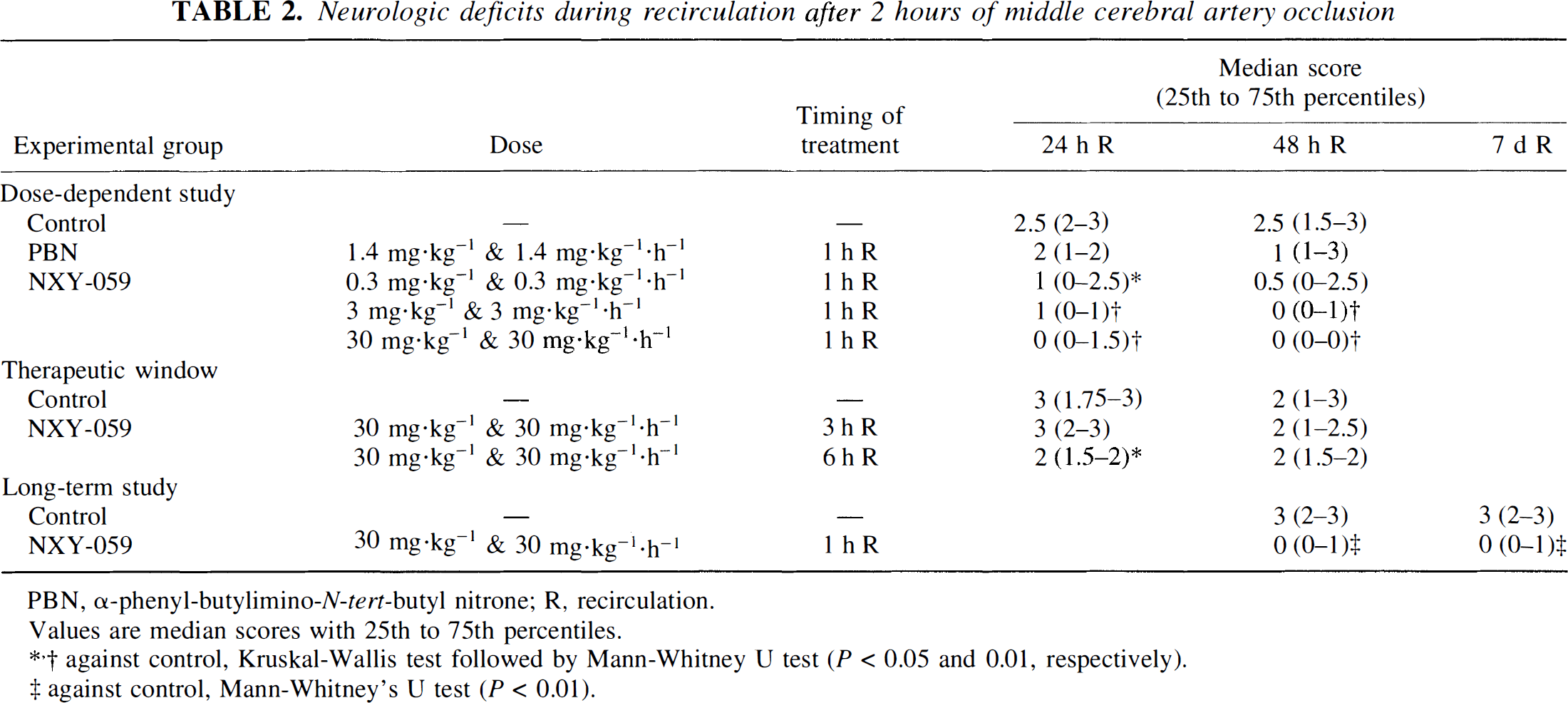
PBN, α-phenyl-butylimino-N-tert-butyl nitrone; R, recirculation.
Values are median scores with 25th to 75th percentiles.
against control, Kruskal-Wallis test followed by Mann-Whitney U test (P < 0.05 and 0.01, respectively).
against control, Mann-Whitney's U test (P < 0.01).
Figure 2 illustrates the effects of treatments on infarct volume at 48 hours of recirculation after 2 hours of MCA occlusion. In the control rats, infarction was observed in the lateral caudoputamen and the cortex fed by the ipsilateral MCA, the percentage of infarct volume varying from 22.9% to 49.1% with a mean value of 35.7 ± 9.0% of the contralateral hemisphere. NXY-059 treatment significantly reduced infarct volume. When the rats were treated with the low, middle, and high doses of NXY-059, infarct volume was attenuated to 17.3 ± 16.1% (P < 0.05), 15.3 ± 12.8% (P < 0.01), and 8.3 ± 3.7% (P < 0.01), respectively. No infarction was detected in 2 (25%) of 8 rats treated with the low dose of NXY-059, in 3 (25%) of 12 rats treated with the middle dose of NXY-059, and in 4 (50%) of 8 rats treated with the high dose of NXY-059. Treatment with PBN in a dose equimolar with the group given NXY-059 in a dose of 3.0 mg · kg−1 failed to affect infarct volume (33.7 ± 4.9%).
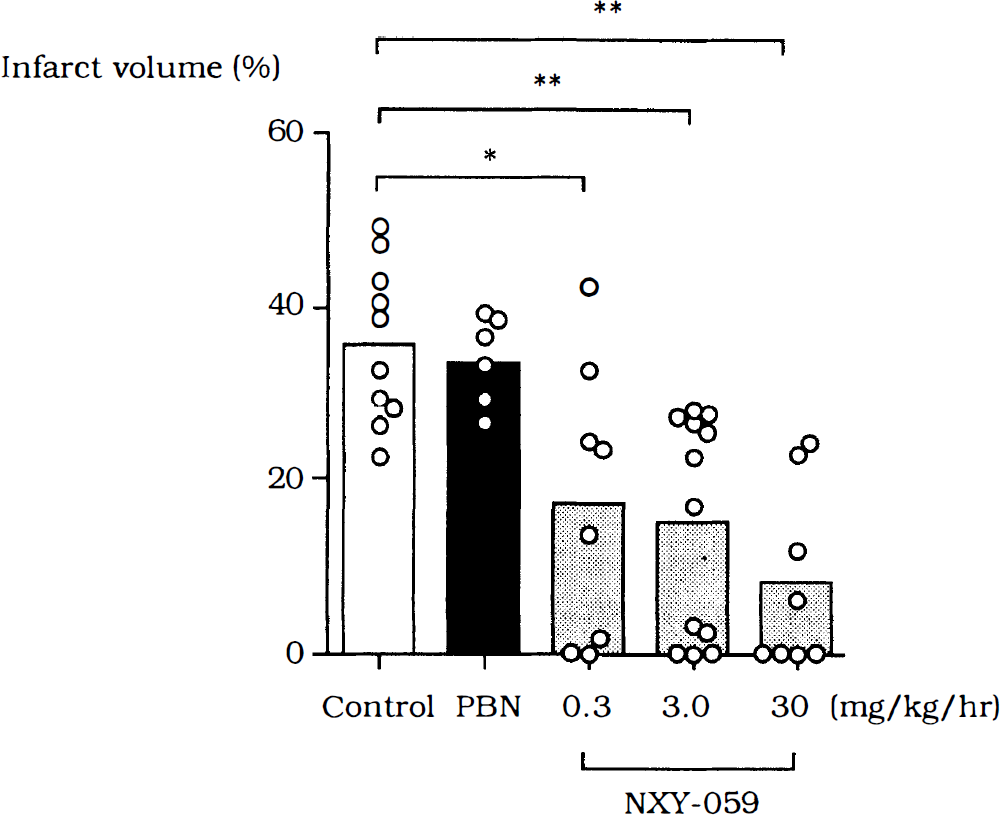
Effects of NXY-059 or PBN (1.4 mg · kg−1) on infarct volume at 48 hours of recirculation after 2-hour MCA occlusion. Each drug was given by intravenous bolus injection at 1 hour after recirculation and then by continuous intravenous infusion for 24 hours. Infarct volume was assessed by staining with 2,3,5-triphenyltetrazolium chloride (TTC) at 48 hours after recirculation. Values are means (columns) and infarct volumes in individual animals (open circles). *, **Against control animals (P < 0.05 and 0.01, respectively), one-factor analysis of variance (ANOVA) followed by Dunnett's test.
Window of therapeutic opportunity for NXY-059
Table 2 shows the effects of NXY-059 on neurologic deficits at 24 and 48 hours of recirculation after 2 hours of MCA occlusion in animals with treatment delays of 3 or 6 hours. The NXY-059 significantly improved neurologic deficits at 24 hours (median value 2, P = 0.0148) when given at 6 hours of reperfusion, but there was no significant improvement at 48 hours of reperfusion in animals with treatment delays of 3 or 6 hours. Figure 3 demonstrates the effects of NXY-059 on infarct volume at 48 hours of recirculation. In the control rats, the percentage of infarct volume varied from 27.4% to 50.7%, with a mean value of 39.7 ± 9.1% of the contralateral hemisphere. The NXY-059 treatment significantly reduced infarct volume when started at 3 hours of recirculation (mean 13.8 ± 15.1%; P < 0.05), although its effects varied widely from 0% to 40.6%. No significant effect of NXY-059 could be observed when the treatment was started at 6 hours of recirculation (31.3 ± 16.4%).
Effects of NXY-059 on infarct volume at 48 hours of recirculation after 2-hour MCA occlusion. The NXY-059 was given by intravenous bolus injection at 3 or 6 hours after recirculation (30 mg · kg−1) and then by continuous intravenous infusion (30 mg · kg−1 · h−1) for 24 hours. Infarct volume was assessed by TTC staining at 48 hours after recirculation. Values are means (columns) and infarct volume in individual animals (open circles). *Against control animals (P < 0.05), one-factor ANOVA followed by Dunnett's test.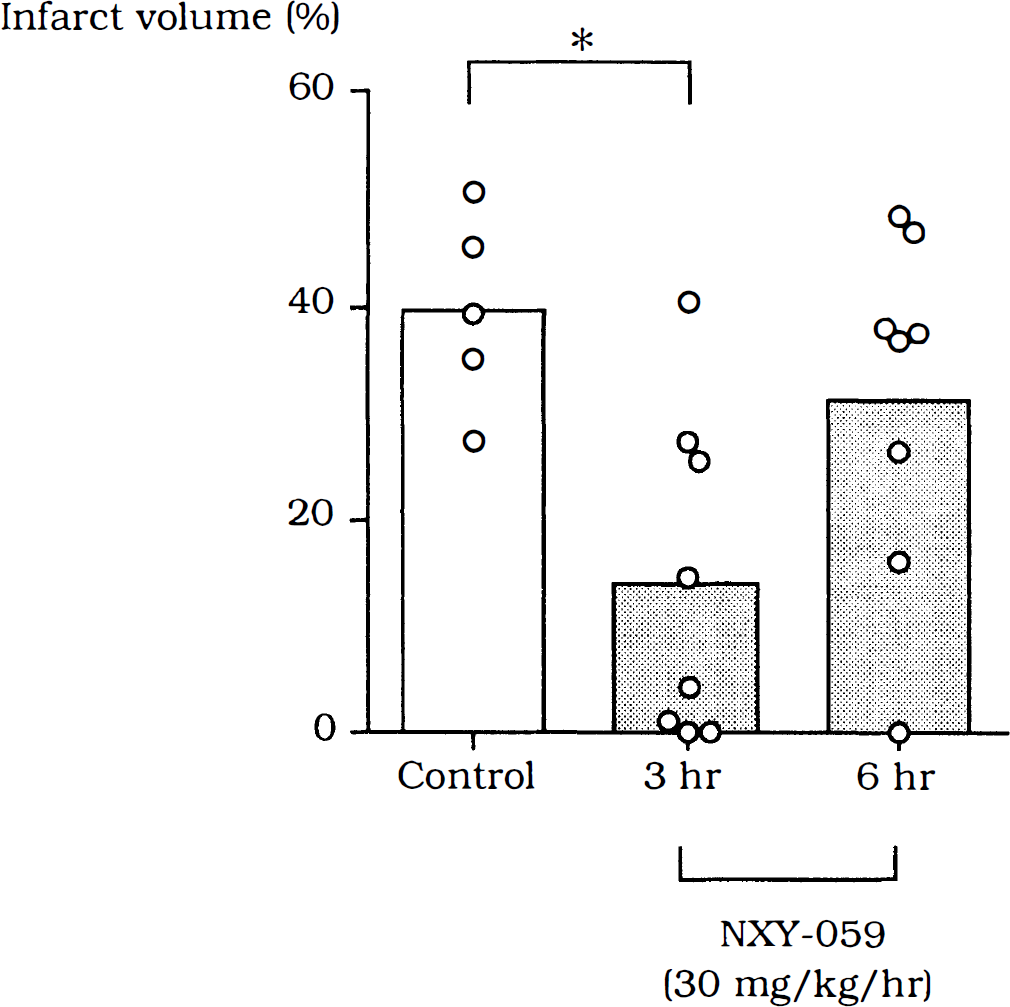
Long-term effects of NXY-059 on brain damage
Table 2 illustrates the effects of treatment with 30 mg · kg−1 · h−1 on neurologic deficits at 2 and 7 days, respectively, of recirculation after 2 hours of MCA occlusion. Control rats showed severe neurologic deficits in both tests at 2 days and 7 days of recirculation, with median scores of 3. The treated rats, though, showed significantly lower scores in both tests at 2 days and 7 days of recirculation with median scores of 0 (P < 0.01).
Figure 4 demonstrates the effects of NXY-059 on brain damage at 7 days of recovery. In all control rats, infarction and a surrounding area of selective neuronal necrosis could be observed in the lateral caudoputamen and the cortex fed by the ipsilateral MCA. Mean volumes of infarct and selective neuronal necrosis were 25.9 ± 5.2% and 3.7 ± 2.0%, respectively. As a result, the mean volume of total damage was 29.6 ± 3.6%. Treatment with NXY-059 for 48 hours significantly ameliorated the brain damage. Thus, mean volumes of infarct and total damage were decreased to 9.2 ± 14.8% (P < 0.05) and 10.9 ± 15.8% (P < 0.01), respectively.
Effects of NXY-059 on infarct volume at 7 days of recirculation after 2-hour MCA occlusion. The NXY-059 was given by intravenous bolus injection at 1 hour after recirculation (30 mg · kg−1) and then by continuous intravenous infusion (30 mg · kg−1 · h−1) for 48 hours. Volumes of infarct and selective neuronal necrosis (SNN) were assessed by transcardiac perfusion and histopathologic technique at 7 days after recirculation. Total volume of damaged tissue (infarct + SNN) also was calculated. Values are means (columns) and infarct volume in individual animals (open circles). *, **Against control animals (P < 0.05 and 0.01, respectively), one-factor ANOVA followed by Dunnett's test.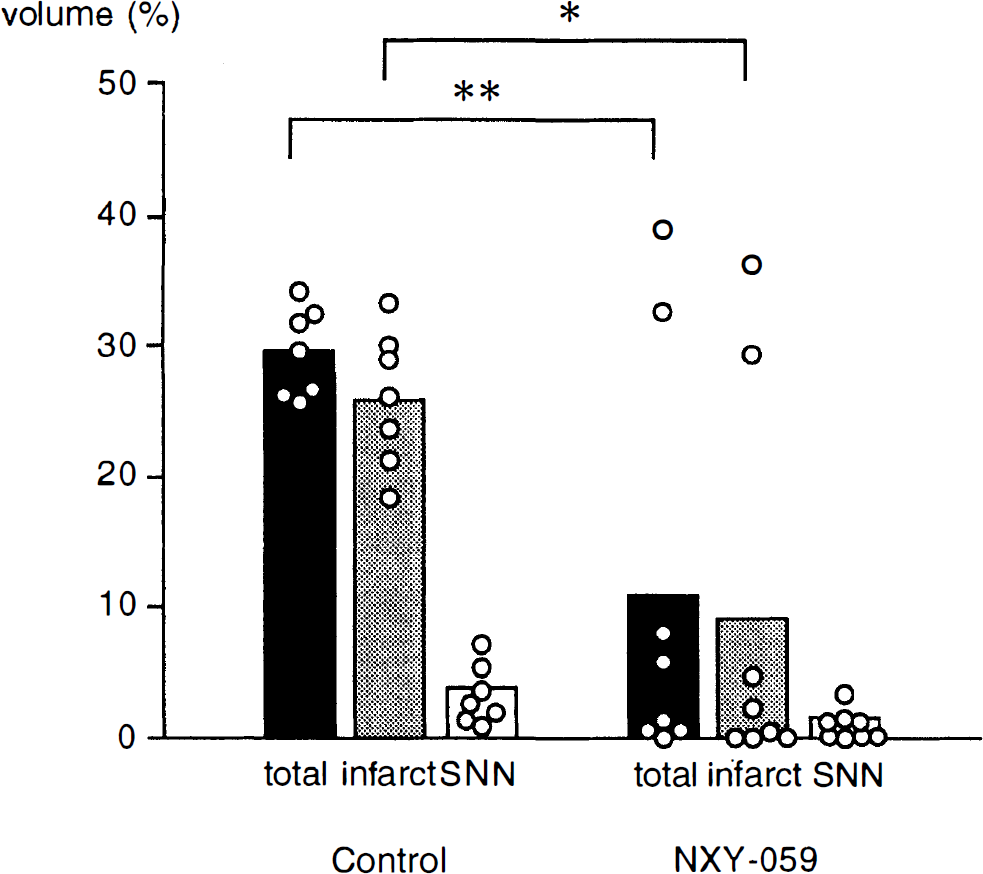
Blood–brain barrier permeability of NXY-059
As discussed earlier, the BBB permeability of NXY-059 was determined by deriving a transfer coefficient (Kin) from the rate of influx of 14C-labeled drug. As shown in Fig. 5, Kin on the contralateral side was low (about 0.1 µL · g−1 · min−1) for all groups. The Kin values for the ipsilateral side and contralateral sides of sham-operated rats were similar (0.06 to 0.09 µL · g−1 · min−1; Fig. 5A). Statistical analysis revealed a significant difference for the caudoputamen Kin value for the ischemic side, compared with the contralateral side, of rats that had a 2-hour MCA occlusion and then were given a 30-mg · kg−1 bolus of [14C]NXY-059 1 hour after the start of recirculation (0.14 ± 0.04 versus 0.07 ± 0.02 µL · g−1 · min−1; P < 0.01). However, there was no significant difference of Kin values in other subregions of animals that had undergone MCA occlusion and were injected with the isotope at either 1 hour (Fig. 5B) or 4 hours (Fig. 5C) after the start of recirculation.
Transfer coefficient (Kin) of NXY-059 in sham-operated rats 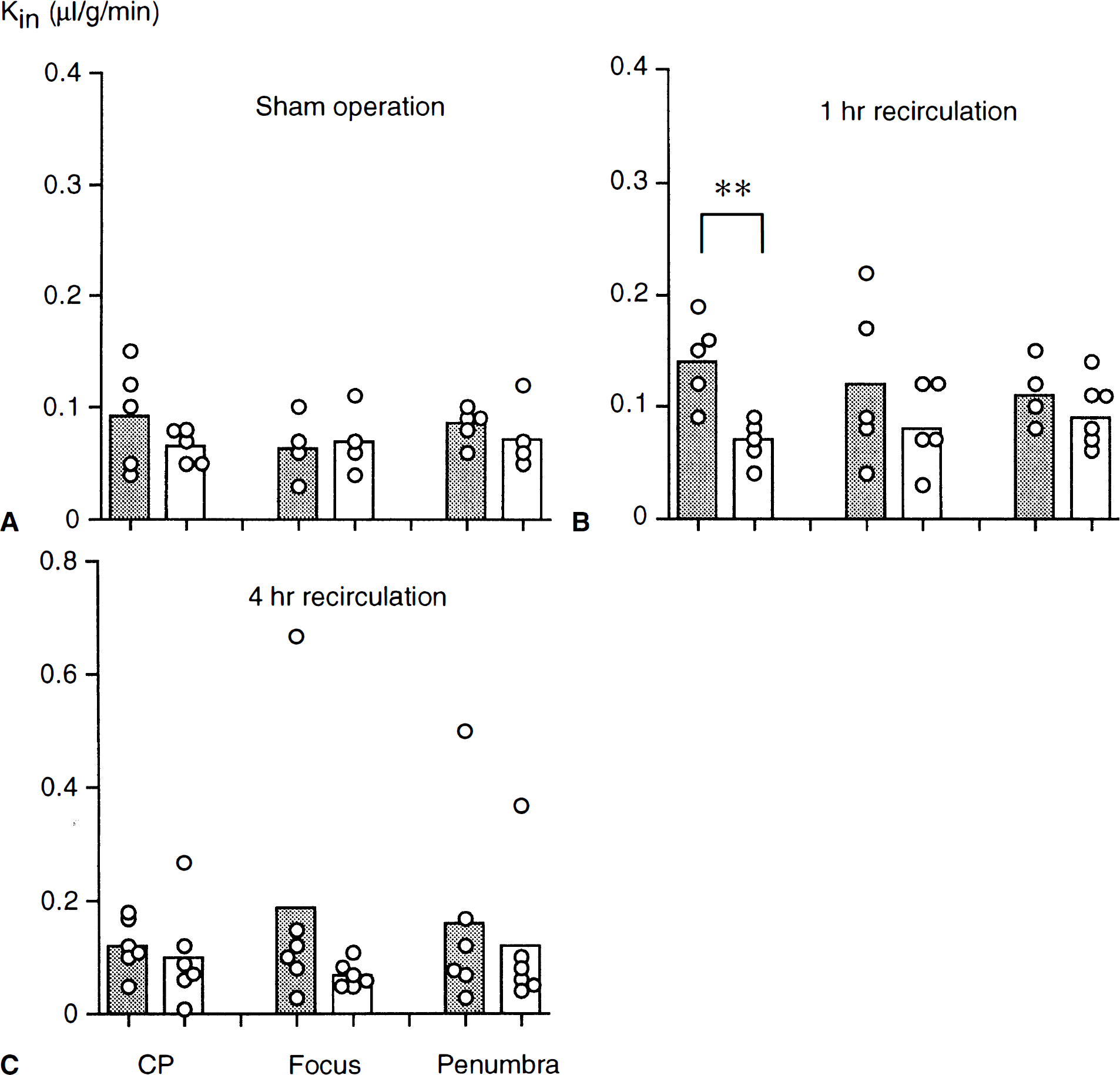
DISCUSSION
Protective effects of PBN and NXY-059
The intraluminal filament technique is widely recognized as a reliable model of focal ischemia, suitable for reperfusion studies. In the current study, transient MCA occlusion induced reproducible damage in the lateral caudoputamen and the overlying cortex in control animals after recovery for 2 days (35.7 ± 9.0% of the contralateral hemisphere) and for 7 days (29.6 ± 3.6%). Previous data from our laboratory showed that 100 mg · kg−1 of PBN markedly reduced infarct volume after 2 days of recovery, even when given by bolus intraperitoneal injection 1 or 3 hours after recirculation (Zhao et al., 1994). Subsequent studies showed that the same dose of PBN prevented secondary deterioration of the bioenergetic state and of mitochondrial dysfunction when given 1 hour after recirculation (Folbergrová et al., 1995; Kuroda et al., 1996).
Whether this reflects an effect of PBN on the adequacy of cerebral circulation remains a matter of debate. Schulz and coworkers (1997), who treated animals with PBN before and during 90 minutes of MCA occlusion, conclude from their Doppler flow and magnetic resonance imaging data that PBN reduces infarct volume by improving postischemic blood flow in the first 1 to 2 hours of reperfusion. Their result is at variance with those obtained with 14C-iodoantipyrine autoradiography (Tsuchidate et al., 1997); besides, it is not consistent with other results from our laboratory in which the laser Doppler flow technique was used to measure erythrocyte velocity, or microelectrodes were used to measure tissue P
These studies suggest that reactive oxygen species (ROS) are important mediators of reperfusion injury and that mitochondria constitute a major target of ROS. α-Phenyl-N-tert-butyl nitrone has been known to rapidly react with free radicals, yielding a relatively stable nitroxide free radical (Floyd, 1990). Clearly, compounds of the class to which PBN belongs are interesting from a clinical perspective. Despite its interesting antiischemic effects, PBN is not an ideal nitrone because three intraperitoneal injections of 100 mg · kg−1 of PBN, the first given at 1 hour after the start of recirculation, postpones rather than prevents damage from 2-hour MCA occlusion. Thus, this dosing gave a marked reduction in infarct size when the latter was evaluated after 2 days of recovery (Zhao et al., 1994) but a less pronounced one when recovery was extended to 7 days (Zhao, Smith, and Siesjö, unpublished data).
Our work with the more soluble nitrone (NXY-059) was inspired by these considerations (and findings). This nitrone could be infused intravenously over 24 or 48 hours in doses of 30 mg · kg−1 · h−1 (or higher) with no untoward effects. Four aspects of the results were noticeable. First, NXY-059 reduced in a dose-dependent manner the infarct volume, as evaluated by TTC staining after 2 days of recovery (Fig. 2). Neurologic symptoms also improved at 24 and 48 hours of recovery in a dose-dependent manner. Second, the therapeutic window for NXY-059 was as wide as that for PBN. Thus, infarct volume decreased to 8.3 ± 3.7% (P <0.01) when treatment was started 1 hour after recirculation, to 13.8 ± 15.1% (P < 0.05) when started at 3 hours, but did not decrease when started at 6 hours (see Fig. 2 and 4). However, neurologic symptoms did not show significant improvements at 48 hours of recovery when treatment was started at 3 or 6 hours of recovery. It is not clear why neurologic symptoms transiently improved at 24 hours, but not at 48 hours, when treated at 6 hours in comparison with controls (Table 2). Third, at equimolar doses (3.0 mg · kg−1 for NXY-059), NXY-059 was more efficacious than PBN (Fig. 2). Fourth, when the compound was infused for 48 hours in a dose of 30 mg · kg−1 · h−1, a marked reduction in infarct size also was observed after 7 days of recovery with a histopathologic endpoint (Fig. 4). Remember that in all experiments within 1 hour after treatment, amelioration of tissue damage was paralleled by an improvement of neurologic status. These findings suggest that NXY-059 has more potent actions on reperfusion injury after focal ischemia than PBN, and that NXY-059 could become clinically important because the drug has a high solubility in water, a wide therapeutic window, and long-lasting effects. However, the results obtained with NXY-059 raise some novel aspects of pathophysiologic events involved in infarct development. Thus, the compound had a marked antiischemic effect, although its penetration through the BBB at 1 or 4 hours was low; in fact, the Kin value obtained is as low as that previously measured for sucrose and molecules considered to be virtually excluded from entering the brain (see Ohno et al., 1978). This finding indicates the possibility that the effects of NXY-059 are exerted at processes occurring at the blood–endothelial cell interface.
It is not known why NXY-059, which has a similar efficacy as PBN in trapping oxygen- and carbon-centered radicals, is more effective than PBN as an antiischemic agent. Data obtained at Centaur Pharmaceuticals in nonischemic rats allow approximations to be made with respect to doses versus steady-state plasma levels. Thus, at NXY-059 doses of 0.3, 3.0, and 30 mg · kg−1 · h−1, the steady-state plasma levels can be approximated to 0.5, 5, and 50 µg · mL−1, or 1.3, 13, and 130 µm, respectively. Infusion of PBN at a dose of 1.4 mg · kg−1 · h−1 is expected to give a steady-state plasma level of 8.6 µg · mL-1 (49 µmol). Thus, although the dose of PBN was equimolar to that of NXY-059, the exposure to PBN was threefold to fourfold greater, probably because PBN has a much slower clearance rate than NXY-059. In view of this, it seems unlikely that PBN is less efficacious than NXY-059 because PBN attains lower concentrations in the vascular bed than NXY-059.
Possible mechanisms of NXY-059
Studies demonstrate that mitochondria are important intracellular sources of ROS under pathologic conditions such as exposure to glutamate or ischemia-reperfusion (Piantadosi and Zhang, 1996). Neuronal nitric oxide synthase is also known to contribute to intracellular production of ROS (Dawson, 1994). As remarked, PBN can easily pass through the BBB and distribute in the brain because of its lipophilic character. Thus, the brain concentration of PBN reaches its peak 20 minutes after injection and then decreases slowly, and is significantly higher than that in blood (Chen et al., 1990; Cheng et al., 1993). Therefore, PBN seems to rapidly react with ROS in the brain and attenuate ROS-mediated tissue damage during reperfusion.
However, the current study reveals that NXY-059 cannot easily pass through the BBB, probably because of its high solubility in water. Other extracellular sources of ROS should, therefore, be considered to explain why NXY-059 showed dramatic protective effects against reperfusion injury. The interactions between endothelial cells and inflammatory cells, such as polymorphonuclear (PMN) leukocytes and macrophages, are believed to be potent extracellular sources of ROS during reperfusion (Betz, 1996; Hallenbeck, 1996). Once activated, these inflammatory cells can generate superoxide anions (O2·−) and hydrogen peroxide (H2O2), which may react to give the highly reactive hydroxyl radical (·OH). These cells also can generate nitric oxide (NO), which reacts with superoxide to yield the peroxynitrite anion through the action of inducible NO synthase. The peroxynitrite anion itself is a highly reactive oxidant that can cause tissue damage but also can decompose to yield -OH (Beckman et al., 1996). It is becoming increasingly clear that the upregulation of adhesion molecules on the surface of the endothelial cells and on the inflammatory cells is essential to the involvement of these cells in reperfusion injury (del Zoppo, 1994; Betz, 1996; Hallenbeck, 1996). Adhesion molecules such as the intercellular adhesion molecule-l can be detected on the endothelial cells during the early phase (1 to 4 hours) of reperfusion after transient MCA occlusion. The PMN leukocytes start to accumulate in the tissue much later, that is, 12 to 24 hours after recirculation. Other studies demonstrate increased activities of myeloperoxidase and inducible NO synthase (Iadecola and Ross, 1997) during this later phase of reperfusion. Matsuo and collaborators directly showed that the circulating PMN leukocytes are a major source of “extracellular” ROS in the early (1- to 2-hour) phase of reperfusion after focal ischemia and that infiltrating PMN leukocytes contribute to ROS production in its late (24-hour) phase (Matsuo et al., 1995). It is thus possible that focal ischemic damage can be ameliorated by drugs that only sparingly pass the BBB (Siesjo and Siesjö, 1996).
Furthermore, it has been shown that endothelial cells have high concentrations of mitochondria, polyunsaturated fatty acids, and xanthine oxidase and that they also are potent generators of ROS. Thus, it has been shown that cultured endothelium can generate ·OH and NO during anoxia-reoxygenation (Kumar et al., 1996). These findings strongly suggest that NXY-059 may attenuate ROS-mediated reperfusion injury by interfering with the interaction between inflammatory cells and the endothelium, or by directly reacting with ROS produced by circulating inflammatory cells or the endothelium.
In summary, we conclude from the current study that the novel nitrone, NXY-059, is a potent neuroprotective drug that has a wide window of therapeutic opportunity and a long-lasting effect on reperfusion injury after focal ischemia. The neuroprotective effect of NXY-059 is possibly related to its ability to yield adducts of ROS or other free radicals that are generated through the inflammatory or immunologic reaction, which is most clearly elicited during reperfusion after focal ischemia (Hallenbeck, 1996).