Abstract
Fluorocitrate, a selective astrocytic toxin, was microinjected into the right striatum of rat brain, and the regional distribution of 14C-acetate was measured using autoradiography. A significant reduction (more than 80%) in 14C-acetate uptake over a 5-minute period was observed in the right striatum, compared with that in the left striatum (saline infused), 4 hours after fluorocitrate (1 nmol/μL) infusion. This effect was transient, and 14C-acetate uptake had almost returned to normal at 24 hours after the fluorocitrate infusion. In contrast, the regional blood flow in the striatum, as determined using 14C-iodoamphetamine, was significantly increased by the fluorocitrate infusion. The present observations indicate that 14C-acetate uptake might be a useful characteristic for examining astrocytic energy metabolism in the intact brain.
Acetate has been used as a selective characteristic for examining astrocytic energy metabolism both in vitro and in vivo (Cerdan et al., 1990; Hassel et al., 1995). 14C-acetate is rapidly (within minutes) incorporated into amino acids derived from the TCA cycle in the brain, producing labeled glutamine with a higher specific activity than that of glutamate, its obligatory precursor; this finding is explained by the “small” (astrocytic) pool of glutamate with a rapid turnover that is segregated from the “large” (neuronal) glutamate pool (Berl, 1973) and by the cellular localization of glutamine synthetase, mainly in astrocytic cells (Martinez Hernandez et al., 1977). Autoradiographic or positron emission tomography (PET) studies using 3H or 11C-acetate have also revealed that acetate is selectively taken up into astrocytes (Muir et al., 1986) and metabolized to 11C-CO2 (Shields et al., 1992).
The selective uptake and metabolism of acetate by cultured astrocytes, compared with synaptosomes and cultured neurons, has been confirmed biochemically and appears to result from the preferential transport, but not metabolism, of acetate into astrocytes (Waniewski and Martin, 1998). The activity of acetyl-CoA synthetase, the first enzymatic step in acetate use, was greater in synaptosomes than in astrocytes, whereas acetate uptake was much more rapid in astrocytes than in synaptosomes. Using NMR, Hassel et al. (1997) also reported that blocking the astrocytic TCA cycle using fluoroacetate, which metabolite fluorocitrate inhibit aconitase in the astrocytic TCA cycle, did not prevent the entry of acetate into mouse brain. We performed an autoradiographic study using the microinjection of fluorocitrate, a selective astrocytic toxin, into the right striatum of rat brain, to determine whether the inhibition of astrocytic metabolism affects acetate uptake in intact brain and found that the fluorocitrate induced a significant reduction in 14C-acetate uptake.
MATERIALS AND METHODS
Animals and chemicals
Male Wistar rats (7 to 8 weeks old) were purchased from Japan SLC (Shizuoka, Japan). The rats were housed under a 12-hour, light-dark cycle and allowed free access to food and water. All experiments on the rats were performed with the permission of the Institutional Animal Care and Use Committee, School of Allied Health Sciences, Osaka University.
DL-fluorocitric acid barium salt was obtained from Sigma-Aldrich Co. (St. Louis, MO, U.S.A.) and prepared as described by Paulsen et al. (1987). 1-14C-acetate (specific activity, 2.0 GBq/mmol) was obtained from Perkin Elmer Life Science Inc (Boston, MA, U.S.A.). N-isopropylmethyl-1,3-14C-p-iodoamphetamine hydrochloride (14C-IMP; specific activity, 2.1 GBq/mmol) was obtained from American Radiolabeled Chemicals Inc. (St. Louis, MO, U.S.A.).
Surgery on rat brains and infusion of fluorocitrate
The rats were anesthetized with pentobarbital (50 mg/kg, i.p.) and placed in a stereotaxic apparatus. Bilateral 26-gauge stainless steel guide cannulas fitted with 33-gauge stainless steel obturators were implanted into the striatum, according to the atlas of Paxinos and Watson (1998); 0.2 mm anterior to the bregma, 3.2 mm lateral to the midline, and 2.0 mm below the cortical surface. The guide cannulae were then fixed to the skull using stainless steel screws and acrylic cement. The rats were then allowed to recover for several days.
Fluorocitrate (0.2 to 2 nmol/μL) was infused through the infusion cannulas (33-gauge, 3.5 mm longer than the guide cannulae) and into the right striatum of each rat while the rat was awake. The infusion was performed for 4 minutes at a flow rate of 0.25 μL/min, and the infusion cannulas were left in place for an additional 3 minutes to reduce the reflux of infused drugs along the cannula track. At the same time, saline solution (1 μL) was infused into the left striatum.
In vivo autoradiography
At 4 or 24 hours after the intrastriatal drug infusion, the animals were given an intravenous bolus injection of 14C-acetate (185 kBq/rat) dissolved in 0.5 mL of saline; 1, 3, 5, or 10 minutes later, the animals were killed by decapitation under a light anesthesia with diethyl ether. To measure the local cerebral blood flow, the rats were intravenously injected with 14C-IMP (185 kBq/rat) dissolved in 0.5 mL of saline over a 30-second period and immediately decapitated. The brains were quickly removed and frozen. Coronal sections (20 μm) were prepared in a cryostat at −20°C and placed in contact with an imaging plate (Fuji Film Co. Ltd., Tokyo, Japan) for several days. The photo-stimulated luminescence (PSL) values in each region of the autoradiograms were then determined using the Bio-Imaging Analyzer System (BAS-1500; Fuji Photo Film). The radioactivity concentrations in regions of interest (ROIs) were expressed as (PSL-background)/area (mm2) [(PSL-BG)/A], and the data were expressed as the proportion of the value on the drug-injected side to that on the saline-injected contralateral side.
Statistical analysis
Values were expressed as the mean ± SD (in each group), and the changes in the values compared using the Student's paired or unpaired t-test. Differences between the saline and fluorocitrate-treated striatum in the time course study were examined using two-way ANOVA.
RESULTS
The kinetics of 14C-acetate uptake in rat striatum is shown in Fig. 1. 14C-acetate was rapidly incorporated in the saline-injected striatum and reached a plateau at 3 to 10 minutes after the injection of the tracer. On the other hand, fluorocitrate (2 nmol) significantly decreased the radioactivity concentrations in the striatum. At 1 minute after the 14C-acetate injection, the radioactivity level in the drug-injected striatum was less than 20% of that observed in the saline-injected striatum. Plasma radioactivity, as counted using a liquid scintillation counter, reached a maximum at 1 minute after the 14C-acetate injection, and the washout at 10 minutes postinjection was less than 40% of its peak values.
The fluorocitrate (1 nmol) caused a significant reduction in 14C-acetate uptake in the rat striatum 4 hours after infusion, with less than 20% of the radioactivity remaining (Figs. 1 and 2). This reduction in uptake had recovered after 24 hours, although some variation among the animals was observed. On the other hand, autoradiograms of 14C-IMP 4 hours after infusion revealed a significant increase in regional blood flow in the right striatum (167 ± 57%, P < 0.05 vs. saline-injected striatum, n = 7) (Fig. 2C).
DISCUSSION
In the present study, a significant decrease in 14C-acetate uptake in the striatum was observed 4 hours after the microinjection of fluorocitrate. The effect of fluorocitrate on 14C-acetate uptake had almost completely disappeared 24 hours after the infusion. On the other hand, the regional blood flow in the right striatum measured with 14C-IMP was significantly increased by the fluorocitrate infusion after 4 hours.
Fluorocitrate has been used as a selective inhibitor of aconitase in the astrocytic TCA cycle. The intrastriatal injection of 1 nmol of fluorocitrate caused a severe reduction in the glutamine level 4 hours after the infusion, and this metabolic inhibitory effect disappeared within 24 hours (Paulsen et al., 1987). It has also been reported that fluorocitrate caused reversible ultrastructual alterations that were selective for glia (Paulsen et al., 1987). Therefore, 14C-acetate uptake in the rat brain appears to occur in parallel with astrocytic energy metabolism and reflects astrocytic conditions.
The rapid uptake of 14C-acetate might be mediated by monocarboxylate transporter (MCT). To date, nine members of the MCT family have been characterized (Halestrap and Price, 1999; Juel and Halestrap, 1999; Price et al., 1998); MCT1 is expressed in astrocytes, whereas MCT2 is expressed in neurons (Broer et al., 1997; Pierre et al., 2002). The time course of the radio-activities in the left striatum in this study is similar to the previous report (Berl and Frigyesi, 1969). The uptake of 14C-acetate in the right (fluorocitrate-infused) striatum was significantly lower than that in the left (saline-infused) striatum at all of the time points after the injection of the tracer in this study (Fig. 1), indicating that delivery process by MCT1 was decreased or reversible transport process was promoted. Previous studies showed that the inhibition of the TCA cycle by fluoroacetate, which is metabolized to fluorocitrate that inhibits aconitase of the astrocytic TCA cycle, did not prevent the entry of acetate into mouse brain, as visualized using NMR (Hassel et al., 1997). Therefore, one possible mechanism for reduction in 14C-acetate uptake in this study is the increment of both delivery and clearance caused by the higher blood flow (Fig. 2C).
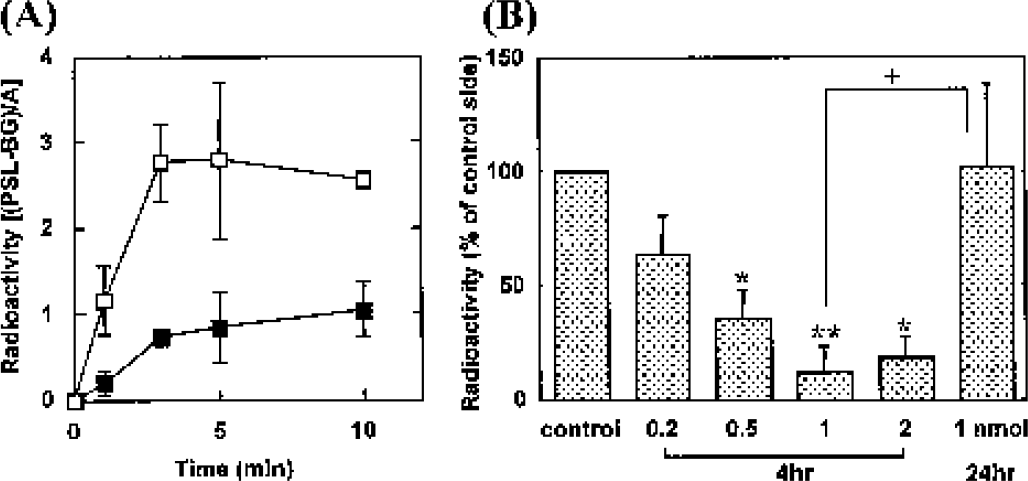
(A) Time course of radioactivity concentrations in the striatum after IV injection of 14C-acetate. Rats were injected with fluorocitrate (2 nmol) in the right striatum (solid square) or saline (open square) 4 hours before the tracer injection. Each value represents the mean ± SD. Significant differences were observed between the saline- and fluorocitrate-injected striatum [F1,20 = 69.60, P < 0.001]. (B) Striatal uptake of 14C-acetate (5 minutes) expressed as the proportion of the value on the drug-injected side to that on the saline-injected contralateral side. Each value represents the mean ± SD of 3 to 6 animals. *P < 0.05, **P < 0.01 vs. saline-injected striatum (paired t-test); +P < 0.05 vs. fluorocitrate-injected striatum (24 hours, unpaired t-test).
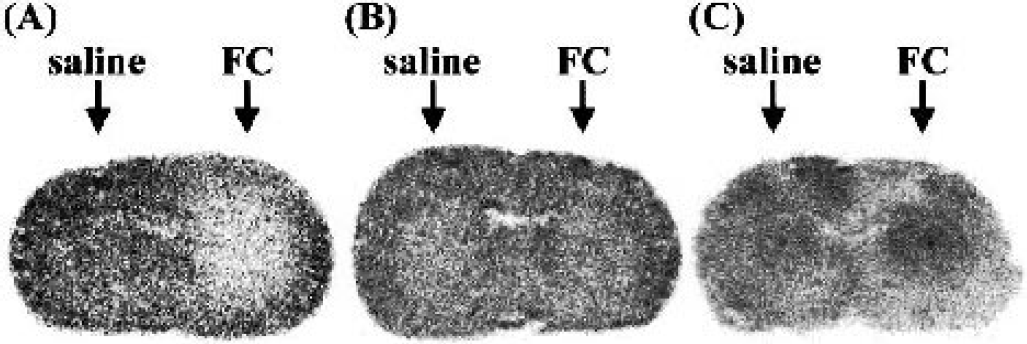
Typical autoradiograms of the rat brain. (A) Incorporation of 14C-acetate over a 5-minute period 4 hours after the intrastriatal infusion of fluorocitrate (FC, 1 nmol). (B) Incorporation of 14C-acetate over a 5-minute period 24 hours after the intrastriatal infusion of fluorocitrate (FC, 1 nmol). (C) Incorporation of 14C-IMP 4 hours after the intrastriatal infusion of fluorocitrate (FC, 1 nmol, 167 ± 57%, P < 0.05 vs. saline-injected striatum, n = 7).
Acetate can also be labeled with 11C, a short half-life positron emitter, enabling astrocytic energy metabolism to be measured in living human brains using PET. To date, the 2-DG and FDG methods have been extensively developed for the study of brain metabolism in vivo. Whereas these procedures enable excellent regional metabolic differentiation, they cannot distinguish between neuronal and astrocytic metabolism. Such differentiation would obviously be useful in gaining a better understanding of the contribution of astrocytic metabolism to brain function and of the metabolic interactions between neurons and astrocytes. This study strongly suggested that 11C-acetate may be an appropriate marker for investigating putative abnormalities in astrocytic energy metabolism in human brain disorders.
