Abstract
The angiogenic role of vascular endothelial growth factor (VEGF) receptors, flk-1 and flt-1, and their downstream signaling pathways, MAPK/ERK and PI-3 kinase, were examined in a fetal rat cortical explant model after exposure to exogenous VEGF. Treatment with VEGF resulted in substantial neovascularization characterized by increased vascular flk-1 receptor expression, whereas flt-1 receptor protein expression was absent. The specific role of flk-1 receptors in the angiogenic process was confirmed by the addition of antisense oligonucleotides (AS-ODNs) to flk-1, which blocked angiogenesis, whereas AS-ODNs to flt-1 had no effect. These results were further supported by the finding that specific chemical inhibition of flk-1 receptors caused disruption of the angiogenic response, whereas inhibition of the flt-1 receptors had no effect. Application of either MAPK/ERK or PI-3 kinase pathway inhibitors disrupted VEGF-induced angiogenesis, thereby indicating that both signaling pathways mediate this process. Thus VEGF binding to the endothelial flk-1 receptor activates the MAPK/ERK and PI-3 kinase pathways, resulting in neoangiogenic events. Of interest is the fact that although VEGF is regarded as a vascular permeability factor, its application to nascent cortical tissue caused an increase in a key physiologic protein of the blood-brain barrier function, glucose transporter-1, suggesting that the cytokine may have a role in blood-brain barrier development.
Keywords
Regulation of vasculogenesis and angiogenesis is of critical importance to the survival and growth of the developing embryo. One such regulator of vascular events is the angiogenic cytokine vascular endothelial growth factor (VEGF). Loss of just one allele coding for VEGF results in embryonic lethality, which is a finding that points to the fundamental function of VEGF in embryonic vascular development (Carmeliet et al., 1996; Ferrara et al., 1996). Moreover, genetic disruption of either of the VEGF receptors, flt-1 (VEGFR-1) or flk-1 (VEGFR-2), results in early embryonic death (between E8.5 and E10.5) because of disruption of normal vascularization and embryonic development (Carmeliet et al., 1996; Fong et al., 1995; Shalaby et al., 1995). Flt-1 and flk-1 are receptor tyrosine kinases that are predominantly expressed in endothelial cells (Klagsbrun and D'Amore, 1996). Activation of their intracellular tyrosine kinase domains results in modulation of particular signaling pathways, resulting in a variety of cellular events (Aiello et al., 1997; Cunningham et al., 1995; Guo et al., 1995; Ku et al., 1993; Seymour et al., 1996). In non-CNS models, the majority of VEGF's actions on endothelial cells in early vasculogenic events such as proliferation and differentiation (Shalaby et al., 1997) have been shown to be mediated by the flk-1 receptor, whereas the biologic role of flt-1 receptors in angiogenic events remains undefined (Zachary and Gliki, 2001).
Binding of VEGF to the flk-1 receptor activates the mitogen-activated protein kinase (MAPK), also known as extracellular signal-regulated kinases (ERKs) pathway (MAPK/ERK), resulting in endothelial cell proliferation (Abedi and Zachary, 1997; Wheeler-Jones et al., 1997). VEGF activation of MAPK/ERK leads to activation of c-Jun N-terminal protein kinase (JNK). PD 98059, a selective inhibitor of ERK activation, and a dominant negative JNK-1 mutant were both shown to inhibit cyclin D1 synthesis and cyclin-dependent kinase 4 activiation, as well as VEGF-induced mitogenesis (Pedram et al., 1998). Expression of the dominant negative Y185F ERK2 mutant blocked JNK activation by VEGF, indicating that the mitogenic response to VEGF might involve cross-talk between the ERK and JNK pathways (Pedram et al., 1998). In addition to its mitogenic functions, VEGF also acts as an endothelial survival factor. VEGF inhibits human umbilical vein endothelial cell (HUVEC) apoptosis in part by activating the antiapoptotic kinase Akt/PKB via a PI-3 kinase-dependent pathway (Gerber et al., 1998; Thakker et al., 1999). Activation of this pathway in the long term results in upregulation of key antiapoptotic proteins such as Bcl-2 and A1 (Gerber et al., 1998), as well as members of the inhibitors of apoptosis (IAP) family, which in turn inhibits terminal effector small prodomain caspases (Tran et al., 1999).
Although the importance of VEGF and its receptors in vasculogenic and early developmental angiogenic events is well illustrated by lethality in knockout animals, a similar determination of the role(s) played by VEGF and its receptors in fetal cerebral angiogenesis is not possible because significant brain vascularization occurs later during fetal development. In E16 to E17 murine brains, VEGF mRNA is detected in choroid plexus epithelium and the ventricular layer (Breier et al., 1992). Temporally, this corresponds to nascent vascularization of the cerebral cortex, where vessels formed at the pial surface grow inward towards the ventricular zone, the potential source of angiogenic factor(s) (Bar, 1983). Transcripts for the VEGF receptors flk-1 and to a lesser extent flt-1 are detected in the endothelium of the in-growing cortical vessels (Dumont et al., 1995; Millauer et al., 1993). Previous studies from our laboratory have shown that organotypic explants of fetal rat neocortex and mesencephalon can be used as a model for studying the effects of VEGF (Rosenstein et al., 1998; Silverman et al., 1999). Those studies showed that VEGF induced robust vascular networks, in which the complexity was dose-dependent (Rosenstein et al., 1998; Silverman et al., 1999). The angiogenic effect was directly attributable to VEGF because the application of a neutralizing antibody to VEGF resulted in almost complete elimination of the angiogenic response. Moreover, the increased vessel/area ratio along with [3H]-thymidine labeling of endothelial cells in the fetal explant culture model strongly suggested that VEGF is a significant mitogen for developing vessels of the CNS to a far greater degree than basic fibroblast growth factor (bFGF) (Rosenstein et al., 1998).
In the present study, we have used several methods to analyze VEGF-induced angiogenesis in fetal cortical explants, including immunohistochemical localization of the receptors to vessels, receptor antisense oligodeoxy-nucletoide treatment, chemical inhibition of the receptors, and inhibition of the MAPK/ERK and PI-3 kinase pathways. The results show that angiogenesis in this model is mediated by the flk-1 receptor because it is blocked both by AS-ODNs and specific chemical inhibitors to flk-1, whereas blockade of the flt-1 receptor by these methods had no effect on VEGF-induced angiogenesis. By using specific inhibitors for the MAPK/ERK and PI-3 kinase pathways, it was determined that the angiogenic process is mediated by both signaling pathways. Finally, exogenous VEGF upregulated the gene product and protein expression of glucose transporter-1 (GLUT-1), which is an important marker for endothelial cell blood-brain barrier (BBB) function, suggesting that the cytokine may play a role in BBB development.
MATERIALS AND METHODS
Pregnant Wistar dams (E18 to E19) were anesthetized with Ketamine (60 mg/kg)/Rompun (6 mg/kg), and the fetal brains were removed under aseptic conditions. The meningeal coverings were removed, and the neocortex was dissected in ice-cold Earle's balanced salt solution (Gibco, Grand Island, NY, U.S.A.). A Stoelting manual tissue slicer was used to produce 400-μm cortical slices from rat fetuses (E18 to E19). Five to six slices were arranged on Millicell-CM culture plate inserts (Millipore, Bedford, MA, U.S.A.), and the inserts were placed in six-well culture dishes. Each well received 1 mL serum-free medium consisting of Neurobasal medium (modified DMEM-F12) with B27 supplement, 0.025mM glutamate, and 0.5mM glutamine (Gibco, Grand Island, NY, U.S.A.). The cultures were incubated under standard culture conditions for 3 days and a 1-week period for some experiments (see below). A 1-μg/mL stock solution of human recombinant VEGF165 (Sigma, St. Louis, MO, U.S.A.) was prepared in 0.1 M PBS, pH 7.4, with 0.1% bovine serum albumin (BSA). VEGF concentrations of 1, 10, 25, 50, and 100 ng/mL were added to the cultures. This range of doses has previously been used in vitro to study various aspects of angiogenesis (Rosenstein et al., 1998; Yoshida et al., 1996). Control explants received vehicle alone. For additional control studies, a nonspecific rat IgG (1, 10, 25, and 50 ng/mL) was added to the cultures instead of VEGF.
Immunocytochemical evaluation of explants was conducted after overnight fixation of whole explants at 4°C in a fixative solution of 4% paraformaldehyde, 0.1M sodium cacodylate buffer, and 3% sucrose, pH 7.2. After rinsing in Tris-buffered saline (TBS), whole explants were processed for immunocytochemistry, which included blocking endogenous peroxidase activity by incubating the explants for 10 minutes in 3% hydrogen peroxide in 10% methanol. This was followed by incubation in the appropriate primary antisera dilutions for 48 hours at 4°C. In general, all antibodies were diluted in 0.05 M TBS containing 1% normal goat serum. After washing with 0.5 M TBS, the explants were incubated with the appropriate secondary antibody for 30 minutes, followed by incubation for 30 minutes, with either rabbit or mouse PAP (Sigma). Reaction products were visualized by diaminobenzidine (DAB) (Sigma, St. Louis, MO, U.S.A.) with or without nickel intensification. Some of the whole explants were subjected to confocal analysis after immunofluorescence. In these specimens, FITC- or Texas Red-conjugated secondary antibodies were used (Vector Labs, Burlingame, CA, U.S.A.). Immunocytochemical analysis of various antigens was repeated at least three times for each antigen and experimental procedure.
Procedures in the control animals included omission of primary antibodies and replacement of the primary antibody with normal serum.
The following antibodies were used: monoclonal anti-laminin IgG (1:50; Developmental Studies Hybridoma Bank, Iowa City, IA, U.S.A.) and polyclonal anti-laminin A & B chains (1:500; SIGMA, St. Louis, MO, U.S.A.) for blood vessel basement membrane detection, monoclonal anti-rat endothelial cell antigen (RECA) (1:50; Developmental Studies Hybridoma Bank, Iowa City, IA, U.S.A.) for endothelial cells, polyclonal glucose transporter (GLUT-1) (1:200, Chemicon, Temecula, CA, U.S.A.); polyclonal flt-1 (1:50; Santa Cruz Biotech, Santa Cruz, CA, U.S.A.), and polyclonal flk-1 (1:50, Santa Cruz Biotech, Santa Cruz, CA, U.S.A.; 1:100, Calbiochem, La Jolla, CA, U.S.A.).
Flk-1 and flt-1 colocalization with vessel-specific markers
Flk-1 and flt-1 expression was studied in whole fetal explants cultured for 3 days with VEGF. Flk-1 and flt-1 polyclonal antibodies (described above) were used for immunofluorescence to visualize colocalization of the receptors with laminin or RECA.
Receptor oligodeoxynucleotide treatment
To determine the angiogenic contributions of flk-1 and flt-1 receptors after VEGF stimulation, explant cultures were treated with 20 mer phosphorothioate oligonucleotides (ODNs) (synthesized by IDT, Coralville, IA). The antisense ODN sequences were as follows (Bernatchez et al., 1999; Robert et al., 2000): flt-1, 5′-GTC GCT CTT GGT GCT ATA-3′; andflk-1, 5′-CCT CCA CTC TTT TCT CAG-3′. The following scrambled phosphorothioate sequences acted as controls: flt-1, 5′-AGC TAG GCA CGA GAG TGA-3′; andflk-1, 5′-TGC TGG CAT GTG CGT TGT-3′. The ODNs (500 nM) were introduced into cultures that had also received VEGF (10 and 25 ng/mL). All cultures were incubated under standard conditions for 3 days; fresh ODNs (500 nM) were added daily. The cultures were fixed in 4% paraformaldehyde and processed for immunofluorescence and confocal analysis.
Chemical inhibition of VEGF receptors
VEGFR Tyrosine Kinase Inhibitor (TKI) (Calbiochem, La Jolla, CA, U.S.A.; cat # 6764751), a potent, dual-selective inhibitor of flt-1 and flk-1 receptors (Hennequin et al., 1999), was used to further delineate the roles played by these receptors in VEGF-induced cortical explant angiogenesis. The inhibitor (flt-1 inhibition, 4 μM; flk-1 inhibition, 200nM) was added to explant cultures that had received 25 ng/mL VEGF, the dosage that produces maximum, significant angiogenesis (Rosenstein et al., 1998). All cultures were incubated under standard conditions for 3 days followed by fixation in 4% paraformaldehyde and processed for immunofluorescence and confocal analysis.
MAPK inhibition
To study the role of MAPK/ERK in the explant cultures, the MAP kinase kinase/ERK kinase (MEK1) inhibitor PD 98059 was introduced into VEGF-treated cultures and examined for neoangiogenesis using laminin immunohistochemistry. PD 98059 is a specific inhibitor of activated MEK1 in vitro and in vivo, which in turn prevents the subsequent phosphorylation, and activation of MAPK/ERK (English and Cobb 2002) (50 μM) (Promega, Madison, WI, U.S.A.) was introduced into cultures that had also received 25 ng/mL VEGF. Control cultures received serum-free medium alone, whereas another set of control cultures was administered PD 98059 (50 μM) only. All cultures were incubated under standard conditions for 3 days followed by fixation in 4% paraformaldehyde and processed for immunofluorescence and confocal analysis.
PI-3 kinase inhibition
The role of PI-3 kinase signaling in cortical explant angiogenesis was studied by introducing the PI-3 kinase inhibitor Wortmannin into VEGF-treated cultures and examining the explants for neoangiogenesis with laminin immunohistochemistry. Wortmannin (100 nM) (Sigma, St. Louis, MO, U.S.A.) was introduced into cultures that had also received 25 ng/mL VEGF. One set of control cultures received serum-free medium alone, whereas another set was administered Wortmannin (100 nM) only. All cultures were incubated under standard conditions for 3 days followed by fixation in 4% paraformaldehyde and processed for immunofluorescence and confocal analysis.
Continuous VEGF treatment
Using standard culture conditions and the VEGF dosages as described above, medium with the appropriate VEGF concentration was replaced on day 3 in culture. Control explants received only fresh medium on day 3 of culture. All explants were cultured for an additional 4 days, after which they were processed for laminin and GLUT-1 immunocytochemistry and subjected to semiquantitative analysis, as described below.
GLUT-1 immunolocalization
Immunocytochemistry for GLUT-1 on whole E18 explants exposed to VEGF was performed to visualize this BBB marker in the newly formed vessels. Whole explants were fixed in 100% methanol for 30 minutes at room temperature followed by washes in 0.01 M cacodylate buffer and TBS. For semiquantitative analyses, GLUT-1 immunocytochemistry was performed and visualized by DAB. Double-label immunofluorescence was performed on 3-day VEGF treated explant cultures to visualize colocalization of RECA and GLUT-1 to determine the extent of GLUT-1 expression in the newly formed vessels.
Semiquantitative analysis of laminin and GLUT-1 immunostained explants
Images of laminin and GLUT-1 stained were digitized using the Image-Pro image analysis system. Images of 6.4″ × 4.8″ were captured at 20× magnification. To measure the relative area occupied by laminin or GLUT-1 immunostained vessels, the threshold color value below which DAB staining would not be counted was determined, and the area above threshold was measured as a ratio to the total area analyzed. Four explants per VEGF dose were used for semiquantitative analysis of angiogenesis (laminin-immunoreactivity) and GLUT-1- immunoreactivity; six regions within each image were sampled. Statistical analysis was performed using Student's t-test and Mann-Whitney test.
Semiquantitative RT-PCR for flt-1, flk-1, and GLUT-1 mRNA
E18 explants treated with VEGF (10 and 100 ng/mL), as well control cultures that did not receive VEGF, were cultured for 1 day under standard conditions. The Purescript RNA isolation kit (Gentra Systems, Minneapolis, MN, U.S.A.) was used to obtain RNA from the explant cultures. The RNA from each sample was reverse transcribed, amplified by PCR, and resolved by 1.2% agarose gel electrophoresis, and the resultant products were stained with ethidium bromide. The following primers were used: GLUT-1 (Wadhwani et al., 1993) forward 5′ GCC TGA GAC CAG TTG AAA GCA C and reverse 5′ CTG CTT AGG TAA AGT TAC AGG AG; flk-1 (Sandner et al., 1997) forward 5′ CAT GCT GTA TCT CGA GGG C 3′ and reverse TTT GTC ATC CCA GGG TAC 3′; flt-1 (Sandner et al., 1997) forward 5′AGG ATA TTG TTC AGG GTC GAA GT and reverse 5′ CAA GTG TAG AGT CCC TTG 3′; and glyceraldehyde-3-phosphate deydrogenase (G3PDH), which served as the loading control forward: 5′ TGA AGG TCG GTG TCA ACG GAT TTG GC 3′ and reverse 5′ CAT GTA GGC CAT GAG GTC CAC CAC 3′. Procedures in control animals included omission of reverse transcriptase during cDNA synthesis. The results shown were obtained from two separate sets of experiments.
RESULTS
Receptor localization
Application of VEGF to the explant cultures always resulted in a robust angiogenic network compared with controls (Rosenstein et al., 1998). Flk-1 colocalization with RECA occurred in neovessels of fetal explants treated with VEGF for 3 days, whereas control cultures (including those receiving IgG alone) did not show immunostaining for the receptor (Figs. 1A and 1B). Significantly, vascular flk-1 immunoreactivity was most extensive at 25 ng/mL VEGF, the dosage that has previously been shown to be most effective at producing angiogenesis (Rosenstein et al., 1998). However, semiquantitative RT-PCR analysis revealed a dose-dependent upregulation of flk-1 mRNA in the fetal cultures with VEGF treatment that was greatest at the 100 ng/mL VEGF dosage (Fig. 1C). Immunoreactivity for flt-1 was not detected in the vasculature of VEGF-treated explants, although RT-PCR analysis showed slight upregulation of flt-1 mRNA at the low dose of VEGF (10 ng/mL), whereas the higher concentration (100 ng/mL) demonstrated a clearer increase in flt-1 mRNA levels (Fig. 1C).
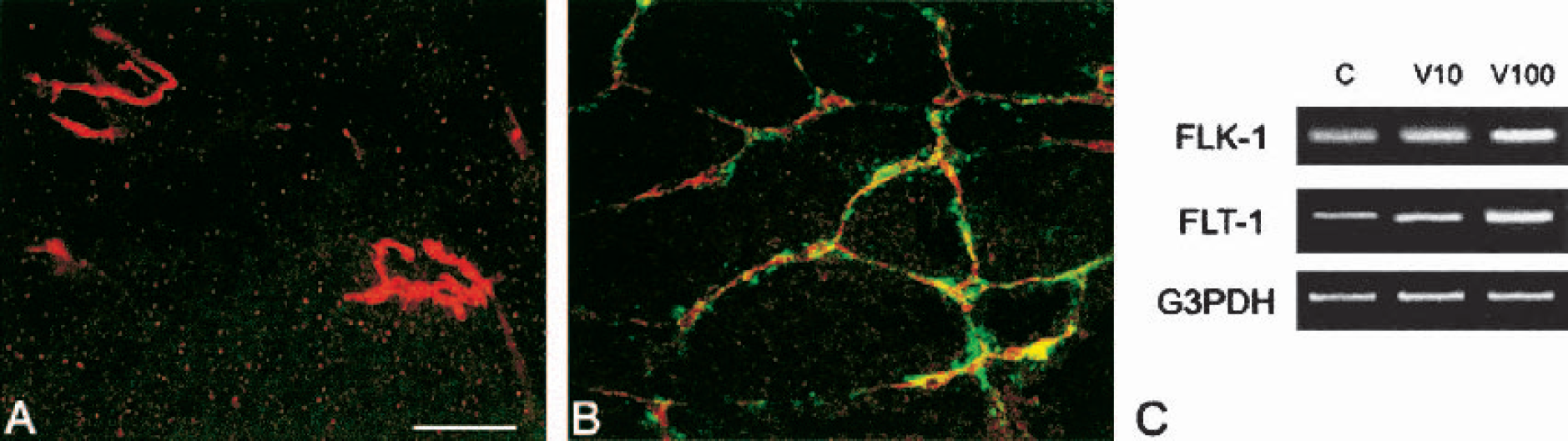
Expression of VEGF receptors in fetal neocortical vessels. Fetal explants were cultured for 3 days with serum-free media in control (A) or with 25 ng/mL VEGF (B) and immunostained for RECA (endothelial cell marker; red) and with flk-1 receptor (green); colocalization (yellow). Note the presence of flk-1 receptors on most RECA-stained vessels in VEGF-treated explant compared with control, which shows no flk-1 staining in the few isolated clusters of vessels. Both images are the same magnification. Bar = 100 μm. (C). Semiquantitative RT-PCR analysis for flk-1, flt-1, and G3PDH mRNA in 1-day fetal explant cultures. C: Control; V10: 10 ng/mL VEGF; V100: 100 ng/mL VEGF. Flk-1 mRNA upregulation is observed with increasing concentrations of VEGF, whereas flt-1 mRNA shows a slight increase at 10 ng/mL VEGF, with a more prominent upregulation at 100 ng/mL VEGF. G3PDH, glyceraldehyde phosphate-3-deydrogenase: loading control. VEGF, vascular endothelial growth factor; RECA, rat endothelial cell antigen.
Receptor inhibition
Treatment with a combination of VEGF (either 10 or 25 ng/mL) and antisense flk-1 obliterated most of the VEGF-induced angiogenesis in explants, resulting in only a few profiles of laminin(+) vessels remaining in the cultures (Figs. 2B and 2C) compared with the robust angiogenic network typically observed in cultures treated with 25 ng/mL VEGF alone (Fig. 2A) (Rosenstein et al., 1998; Silverman et al., 1999). Control cultures that received simultaneous VEGF (25 ng/mL) and sense flk-1 showed angiogenic activity that was entirely comparable with cultures treated with 25 ng/mL VEGF (Fig. 2D).
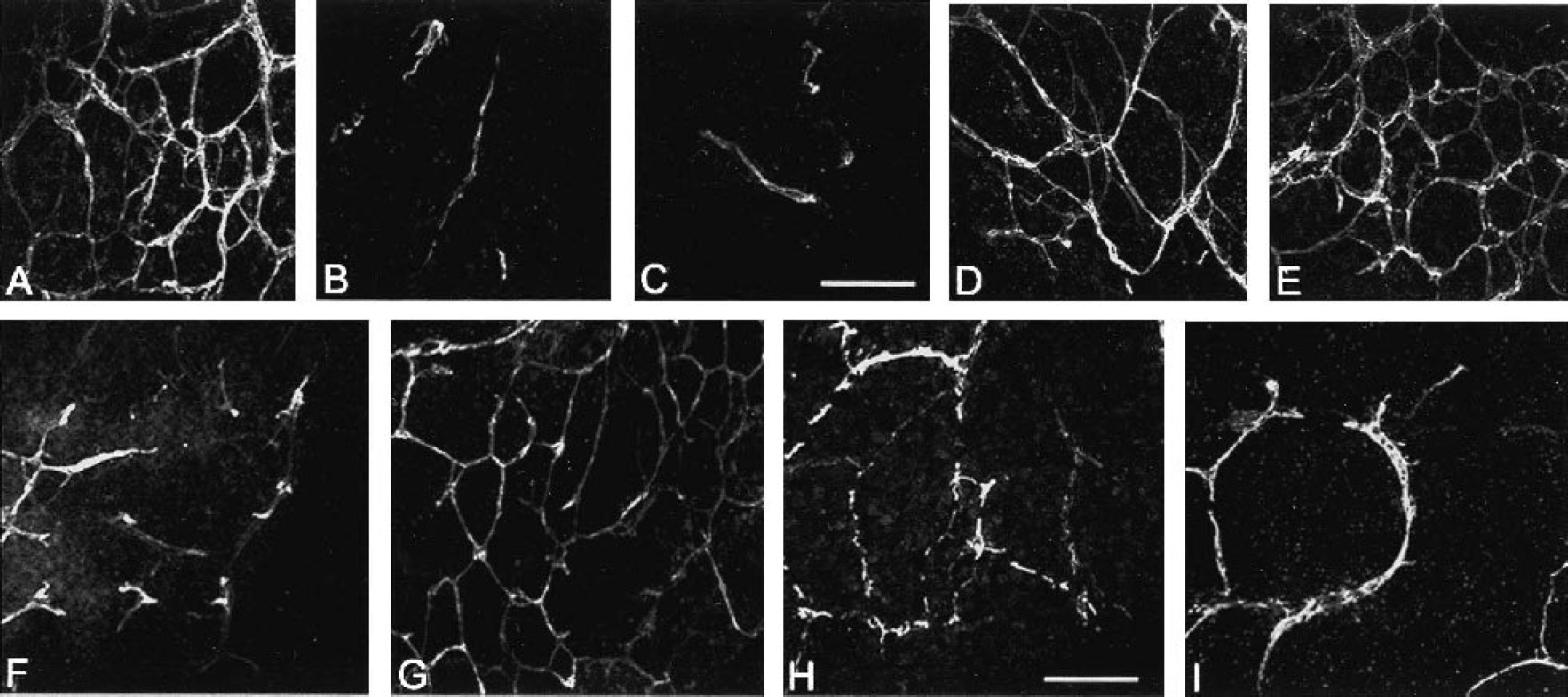
Effect on VEGF-induced angiogenesis after exposure to receptor and signal transduction inhibitors. Fetal neocortical explants were grown in serum-free media supplemented with 25 ng/mL VEGF (A), 10 ng/mL VEGF + 500 nM antisense flk-1(B), 25 ng/mL VEGF + 500 nM antisense flk-1 (C), 25 ng/mL VEGF + 500 nM sense flk-1 (D), or 25 ng/mL VEGF + 500 nM antisense flt-1 (E) and stained with the vessel basement membrane marker laminin. Compared with the robust angiogenic response seen with 25 ng/mL VEGF (A), a near elimination of the VEGF-induced angiogenic response is observed after treatment with the antisense flk-1 (B, C), whereas simultaneous treatment with VEGF and either sense flk-1 (D) or sense flt-1 (E) resulted in angiogenic patterns comparable with treatment with VEGF alone (A). Cultures grown in serum-free media supplemented with 25 ng/mL VEGF + chemical inhibitor TK1 (200 nM: flk-1 inhibition) (F) or 25 ng/mL VEGF + chemical inhibitor TKI (4 μM: flt-1 inhibition) (G) were immunostained for laminin. Only a few remnants of laminin immunoreactive vessels survived in cultures treated for flk-1 inhibition (F), whereas flt-1 inhibition had no effect on angiogenesis (G) and resembled 25 ng/mL VEGF treated cultures (A). Cultures grown in serum-free media supplemented with 25 ng/mL VEGF and either the MAPK inhibitor PD 98059 (50 μM) (H) or the PI-3 kinase inhibitor Wortmanin (100 nM) (I) resulted in a disrupted vascular network with discontinuous laminin stained vessels. All images are the same magnification. Bar = 100 μm. VEGF, vascular endothelial growth factor.
Simultaneous treatment of explant cultures with antisense flt-1 or sense flt-1 together with 25 ng/mL VEGF resulted in robust angiogenesis (Fig. 2E), similar to that observed with 25 ng/mL VEGF alone (Fig. 2A). The result of the antisense treatment experiments indicated that flk-1 receptors play an important role in fetal cortical explant angiogenesis, whereas the role of flt-1 receptors in cortical angiogenesis remains elusive.
The use of a specific chemical inhibitor for flk-1 and flt-1 confirmed the antisense experimental results. Chemical inhibition of flk-1 receptors using TKI (200 nM) in cultures simultaneously receiving 25 ng/mL VEGF resulted in loss of blood vessel network formation, with only a few remnants of laminin(+) vessels remaining in the cultures (Fig. 2F). Flt-1 chemical inhibition (4 μM) in 25 ng/mL VEGF-treated cultures showed robust angiogenesis similar to that observed with 25 ng/mL VEGF alone (Fig. 2G) and thus had no effect upon angiogenesis.
Signal transduction pathways
Treatment with the MEK1 inhibitor PD 98059 together with 25 ng/mL VEGF resulted in the faint appearance of an angiogenic network, but laminin immunoreactive vessels were small and almost entirely discontinuous when compared with cultures that had received 25 ng/mL VEGF alone (Fig. 2H). Cultures that received either PD 98059 or serum-free medium alone had vascular staining for laminin that resembled control non-VEGF treated explants (data not shown).
Similar to what was observed with MAP/ERK inhibition, the PI-3 kinase inhibitor Wortmannin administered together with 25 ng/mL VEGF produced markedly discontinuous networks (Fig. 2I). Cultures treated only with Wortmannin showed angiogenesis patterns similar to control cultures (data not shown).
These results suggest that both the MAPK/ERK and PI-3 kinase pathways play an important role in VEGF-induced fetal cortical explant angiogenesis, and their inhibition prevents formation of vascular networks in this model.
Glucose transporter expression
Confocal fluorescent microcopy used to study GLUT-1 and laminin expression in the vessels showed mainly coincident localization of the two markers in control explants (Fig. 3A). When explants were treated with 25 ng/mL VEGF, the characteristic robust angiogenesis was observed (see Fig. 3B), but not all vessels were double-labeled. Some vessels expressed only laminin, while in others, GLUT-1 expression was incomplete. The presence of some laminin(+)/GLUT-1(−) vessels in the explants (Fig. 3B) suggests that some of the new vessels may not have attained BBB competency. A dose-response relationship for GLUT-1 immunostaining (Fig. 3C), very similar to laminin immunostaining (Rosenstein et al., 1998), was observed with statistically significant increases in GLUT-1 immunostaining compared with controls being observed in E18 explant vasculature in response to VEGF doses in day 3 as well as in day 7 continuous treatment cultures (see below), with maximum GLUT-1-immunoreactivity (IR) observed at 10 ng/mL VEGF (a 4.5-fold increase over control levels) (Fig. 3C). At 100 ng/mL, VEGF GLUT-1 per area values are very similar to control.
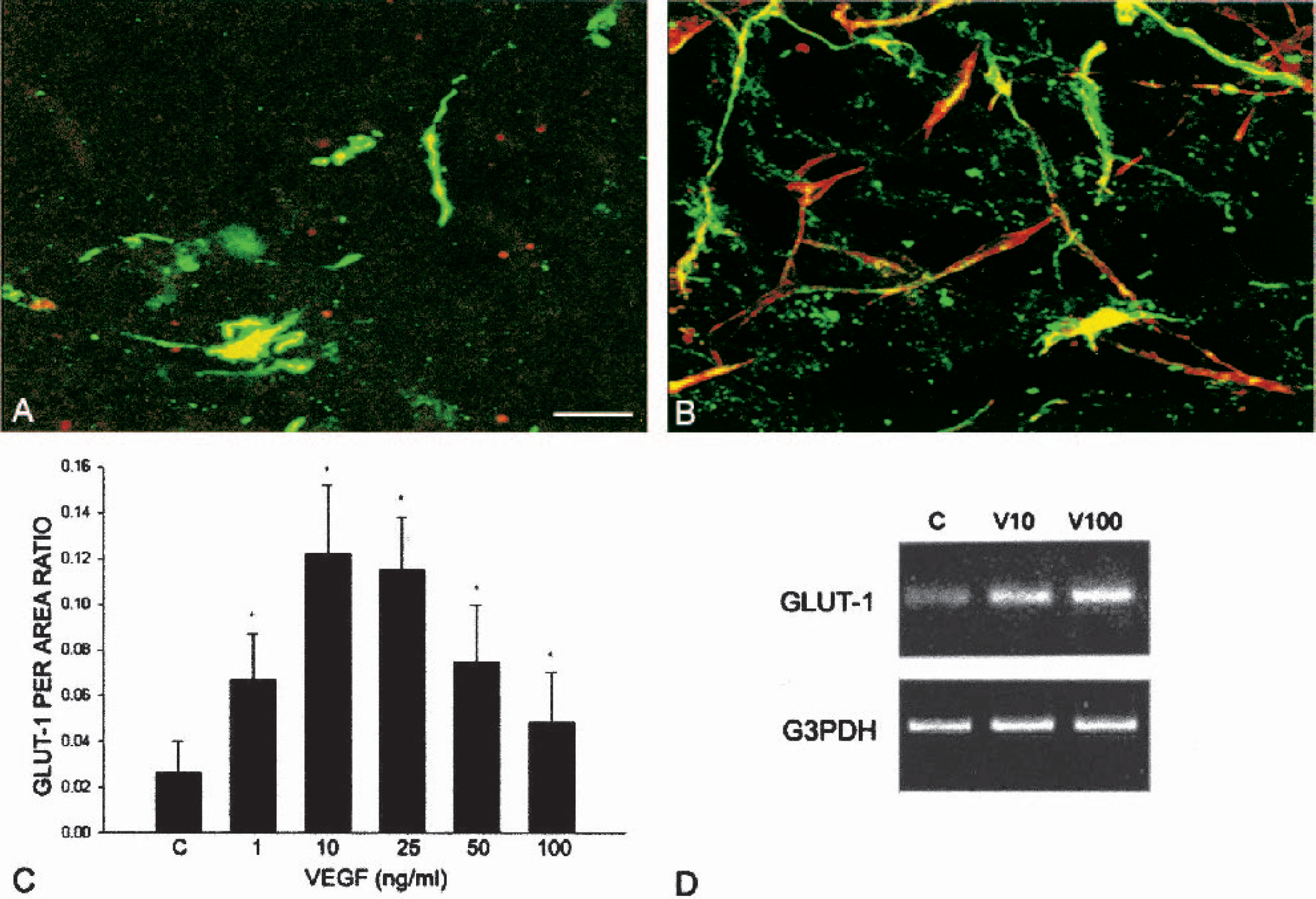
Expression of the BBB marker GLUT-1 in fetal neocortical vessels. Fetal explants were grown in serum-free media in control (A) or with 25 ng/mL VEGF (B) and immunostained for laminin (vascular basement membrane marker; red) and with GLUT-1 (BBB marker; green). Control cultures show more colocalization (yellow) of laminin and GLUT-1 on vessels in 3-day fetal explant cultures compared with 25 ng/mL VEGF-treated cultures where only partial colocalization of the two markers can be seen (Bar = 70 μm). (C) Semiquantitative analysis of GLUT-1-IR in vessels of 3-day, VEGF-treated fetal neocortical explants. Data are means ± SD; *P < 0.05 compared with controls. Graph shows significant GLUT-IR at all doses compared with control, with maximum IR at 10 ng/mL VEGF. (D) Semiquantitative RT-PCR analysis of 1-day fetal explant cultures exposed to various concentrations of VEGF for GLUT-1 mRNA. Increasing concentrations of VEGF result in upregulation of GLUT-1 mRNA. G3PDH mRNA is used as loading control. C: Control; V10: 10 ng/mL VEGF; V100: 100 ng/mL VEGF. BBB, blood-brain barrier; VEGF, vascular endothelial growth factor. IR, immunoreactivity.
Semiquantitative RT-PCR analysis of fetal explants showed an upregulation of GLUT-1 mRNA with increasing VEGF concentrations (Fig. 3D). Although protein expression was highest at the 10 to 25 ng/mL VEGF doses as observed by immunohistochemistry, RT-PCR results indicated that GLUT-1 gene message was greatest at higher VEGF concentrations (50 and 100 ng/mL), suggesting incomplete or restricted translation into protein at higher dosages.
Continuous VEGF treatment
Explants that had been exposed to VEGF (1 to 100 ng/mL) continuously for 1 week exhibited strong vascular laminin immunoreactivity at all doses of VEGF. The vascular pattern observed in the continuous experimental paradigm (Figs. 4A–4D) was more irregular than the reticular pattern of vessels obtained with the 3-day cultures (see Fig. 2A), and the vessels appeared to be more dilated. Continuous VEGF administration resulted in statistically significant angiogenesis at 10, 25, and 100 ng/mL VEGF as measured by laminin per area ratios (Fig. 4E). Maximum angiogenesis was observed at 10 ng/mL (a 182% increase over control), although increased angiogenesis was clearly evident at all VEGF dosages in the long-term cultures, whereas data from 3-day cultures showed that the higher VEGF dosages resulted in a decrease in angiogenesis (Rosenstein et al., 1998). Continuous VEGF (50 and 100 ng/mL) administration for 1 week induced a 3.4-fold increase in angiogenesis, compared with the same doses in 3-day cultures (Rosenstein et al., 1998).
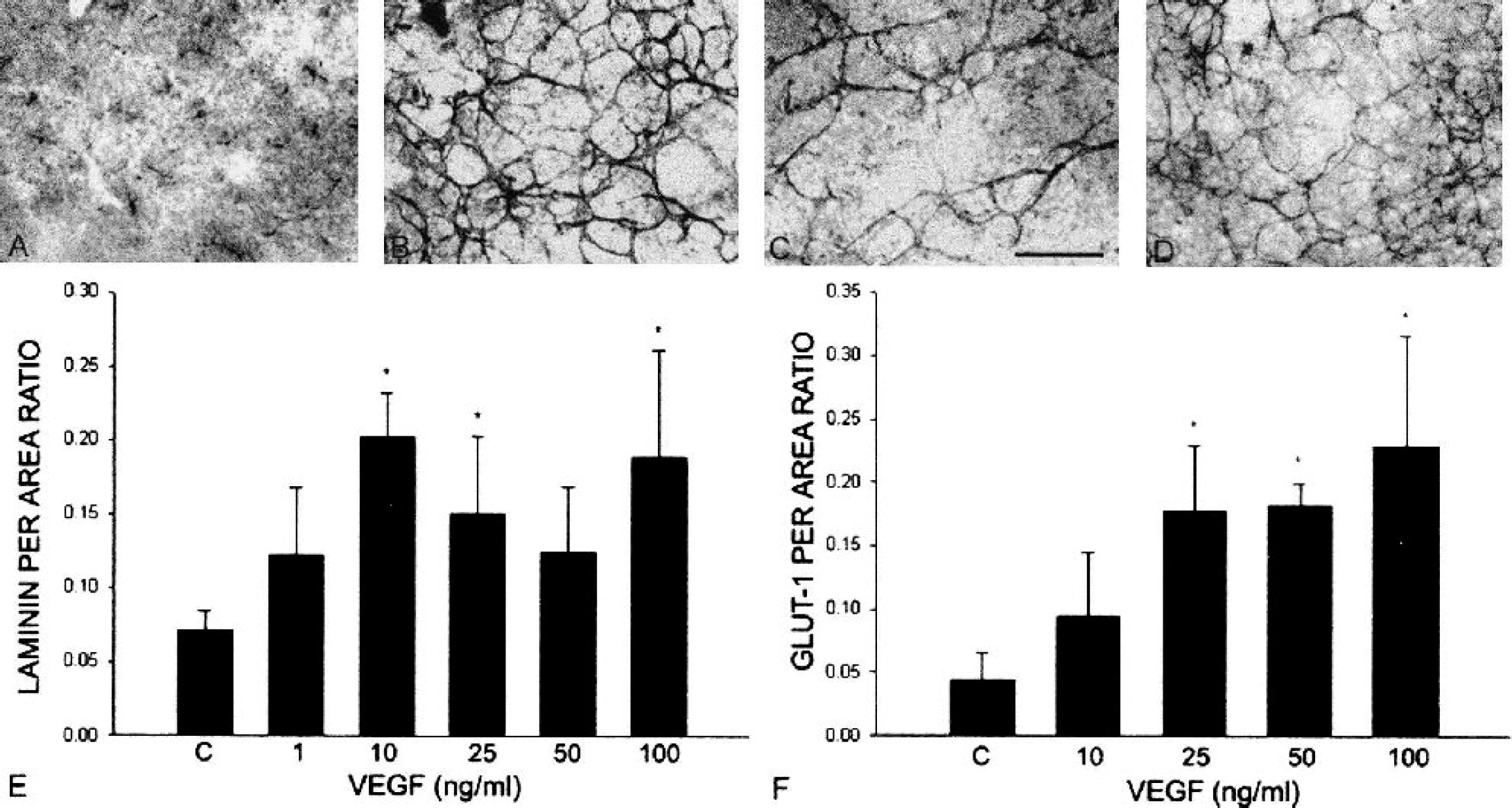
Angiogenic responses after continuous exposure to VEGF in explant cultures for 7 days. E18 fetal explant cultures received control (A) serum-free media, media supplemented with 10 ng/mL VEGF (B), 25 ng/mL VEGF (C), or 100 ng/mL VEGF (D), and they were immunostained with the blood vessel basement membrane marker laminin. Note the increase in laminin(+) vessels at all doses of VEGF compared with control. The vascular networks have a somewhat irregular network, particularly at the highest concentrations of VEGF. All images are the same magnification. Bar = 120 μm. (E) Semiquantitative analysis of laminin-IR in fetal neocortical explants cultured for 7 days in the presence of VEGF. Data are means ± SD; *P < 0.05 compared with controls. Significant angiogenesis is observed at 10, 25, and 100 ng/mL VEGF compared with control, with a trend towards increased angiogenesis, especially at the higher concentrations, compared with 3-day cultures. (F) Semiquantitative analysis of GLUT-1-IR in 7-day, VEGF-treated fetal neocortical explants. Data are means ± SD; *P < 0.05 compared with controls. Note the significant increase in GLUT-1-IR with 50 and 100 ng/mL treatments, compared with control. Compare this response with GLUT-IR in 3-day cultures (Fig. 3C). VEGF, vascular endothelial growth factor; IR, immunoreactivity.
Significant GLUT-1 immunoreactivity was observed in fetal explants after continuous treatment with VEGF (25, 50, and 100 ng/mL) for 7 days (Fig. 4F). Vascular GLUT-1 expression was more uniform in the continuous cultures than in the 3-day cultures, suggesting that the new vessels may have obtained BBB competency or were more mature compared with vessels in the 3-day cultures. Continuous administration of higher VEGF concentrations (50 and 100 ng/mL) for 7 days resulted in a 3.3-fold increase in GLUT-1 expression compared with 3-day cultures (see Fig. 3C).
DISCUSSION
The aim of the present study was to understand the roles played by VEGF and its receptors flk-1 and flt-1 in an explant of model of late fetal cerebral cortical angiogenesis, as well as the signaling pathways involved. Exogenous VEGF application in this model has previously been shown to result in a direct, dose-dependent angiogenic response (Rosenstein et al., 1998). We have now demonstrated that VEGF exposure results in upregulation of flk-1 mRNA in the cultures together with strong expression of the protein in endothelial cells, and, by using two experimental paradigms, we have shown that the angiogenic effects are specifically transduced by the flk-1 receptors. In the first paradigm, antisense flk-1 ODN was introduced into the fetal cortical explant cultures together with VEGF, which resulted in abolition of the angiogenic response. Antisense flk-1 ODN can also disrupt VEGF induced angiogenesis in adult cortical explants (unpublished results, Mani et al, 2003). Further confirmation of flk-1's role in angiogenesis was obtained by using the chemical inhibitor VEGFR Tyrosine Kinase Inhibitor (TKI) at a dose that inhibited the flk-1 receptor (Louissaint et al., 2002), which also resulted in loss of the angiogenic response. On the other hand, both ASODN to flt-1 and VEGFR Tyrosine Kinase Inhibitor (TKI) used at a dose to inhibit flt-1 receptors had no effect on angiogenesis. These data, together with previous results showing an increased vessel/area ratio along with [3H]-thymidine labeling of endothelial cells in 3-day fetal explant cultures (Rosenstein et al., 1998; Silverman et al., 1999), strongly suggest that VEGF-flk-1 interactions are most likely responsible for endothelial cell proliferation in the CNS. Similar to our results with CNS vessels, overexpression studies of flk-1 in HUVEC and NIH3T3 cells in culture (Sawano et al., 1997; Seetharam et al., 1995) and gene targeting of the receptor in mice (Shalaby et al., 1995) have shown it to be the receptor responsible for mediating proliferation of non-CNS endothelial cells.
Of interest is the fact that in contrast to flk-1, flt-1 protein expression was not observed on endothelial cells, although an upregulation of flt-1 mRNA in whole explant cultures was evident. The inability to detect flt-1 immunoexpression in vessels of fetal cortical explant cultures may be related to technical issues in the immunocytochemical procedure and the possibility that flt-1 protein expression may be below the detection limit of the current technique. Evidence from the literature suggests that during developmental vasculogenesis, mRNA for flk-1 receptors are expressed at higher levels than for flt-1 in endothelial cells (Dumont et al., 1995). Flt-1 null mutant mice exhibit embryonic lethality because of abnormal overgrowth of endothelial cells (Fong et al., 1995), whereas mice with deleted flt-1 tyrosine kinase domains show normal embryonic angiogenesis, suggesting that flt-1 functions in the embryo as a negative regulator of VEGF-induced angiogenesis by trapping VEGF in its extracellular domain and does not act as a strong, classical receptor tyrosine kinase (Hiratsuka et al., 1998). It is possible that the relative lack of flt-1 expression in the CNS explant paradigm may have caused an abnormal overgrowth of endothelial cells in response to exposure to VEGF application because the dense reticular vascular pattern observed in the explants is not present in normal brain tissue (Rosenstein et al., 1998). Our studies indicate that the flk-1 receptor mediates proliferation of CNS endothelial cells, and the relative paucity of flt-1 expression could indicate that the normal negative regulatory role of flt-1 is not operational in this model.
Treatment with the MEK1 inhibitor PD98059 combined with VEGF resulted in disruption of the typical angiogenic effects and caused incomplete vessel profiles to be formed. VEGF activation of ERKs is known to cause proliferation in peripheral endothelial cell cultures (Abedi and Zachary, 1997; Wheeler-Jones et al., 1997). VEGF is also able to induce c-Jun N-terminal protein kinase (JNK) via the MAPK/ERK pathway. Inhibition of the MAPK/ERK pathway as well as that of JNK-1 has been shown to inhibit VEGF-induced mitogenesis by affecting various cell-cycle specific events (Pedram et al., 1998). It is intriguing to note that inhibition of the PI-3 kinase pathway by Wortmannin had a similar effect upon VEGF-induced vessel formation in our culture paradigm. PI-3 kinase phosphorylation of AKT promotes survival of peripheral endothelial cells (Dimmeler and Zeiher, 2000; Gerber et al., 1998) as well as migration of brain endothelial cells in vitro (Radisavljevic et al., 2000). The PI-3 kinase pathway is implicated in activating cell protective mechanisms by activating Akt/PKB and the resulting key antiapoptotic proteins, such as Bcl-2, A1, and members of the IAP family (Gerber et al., 1998; Tran et al., 1999). The effect on the PI3 kinase pathway in our cultures suggests that VEGF might use both MAPK/ERK and PI-3 kinase pathways to maintain both the angiogenic response and the integrity of endothelial cells in the cultures. In both peripheral and CNS endothelial cell cultures, VEGF treatment has been shown to increase the expression of adhesion molecules, thereby anchoring cells to the substrate, leading to increased cell survival (Abedi and Zachary, 1997; Radisavljevic et al., 2000; Rousseau et al., 2000; Wu et al., 2000).
With continuous administration of VEGF in E18 explant cultures for a 1-week period, vessel growth was robust, especially at the higher concentrations of the growth factor, but lacked the order and symmetry observed with the 3-day cultures. The irregular vascular pattern obtained was similar to that seen with VEGF injected into early quail embryos where it caused vessel malformations in the cardiovascular system (Drake and Little, 1995). In sharp contrast to the 3-day cultures, angiogenesis was observed for the entire range of VEGF dosages in continuous cultures. The relative lack of dose-dependent responsiveness could be because of sequestration of excess VEGF in the extracellular matrix followed by its slow release by proteases (Larrivee and Karsan, 2000), thereby preventing receptor desensitization and resulting in mitogenic effects at all VEGF concentrations. It may also reflect the ability of VEGF to act as a survival factor for endothelial cells by inducing signal transduction pathways, that is, PI-3 kinase, that prevent endothelial cell apoptosis (Dimmeler and Zeiher, 2000). Studies in human umbilical vascular endothelial cell cultures have shown that the angiogenic action of VEGF involves an antiapoptotic effect that promotes endothelial cell survival mediated via flk-1 receptor and the PI-3 kinase/Akt pathway (Gerber et al., 1998). During development, activation of the PI-3 kinase/Akt pathway by VEGF in endothelial cells has been shown to be crucial in transducing endothelial intracellular survival signals via VE-cadherin/β-catenin (Carmeliet et al., 1999).
GLUT-1 expression had a similar pattern to laminin immunostaining in the 3-day VEGF treated cultures, although an incomplete overlap of the two markers, even on the same vessel profiles, was observed. Semiquantitative RT-PCR analysis for GLUT-1 mRNA showed a marked trend towards mRNA upregulation, which is in agreement with studies in non-CNS endothelial cell cultures (Pekala et al., 1990). The significance of vascular GLUT-1 expression in our culture paradigms raises intriguing questions, especially because this upregulation is observed in the absence of blood-flow, given that VEGF is nominally regarded as a vascular permeability factor. During normal brain development, rat brain endothelial cells begin to express GLUT-1 by embryonic day 14.5 (Dermietzel et al., 1992), whereas endothelial cell proliferation is still ongoing (Robertson et al., 1985). Although GLUT-1 expression has traditionally been linked to maturation of the BBB, explanations for its expression in our fetal organotypic explant cultures after VEGF application include the following possibilities: (1) its expression may reflect a higher metabolic state in the fetal explants, (2) vessels may be extracting or using more glucose from the medium, (3) VEGF is acting as a vessel maintenance factor, or (4) VEGF may intrinsically upregulate GLUT-1 in in vitro paradigms (Pekala et al., 1990; Sone et al., 2000). The finding that control cultures, which received no VEGF, had decreased levels of angiogenesis and GLUT-1 protein compared with cultures that received VEGF supports this last possibility. Also, the significant angiogenesis observed in the cultures suggests that endothelial cells themselves may be the initiators of increased metabolic demand. At higher doses of VEGF, increased immunostaining for developmentally regulated mature neuronal and glial markers has been observed (Mani et al., 2000; Silverman et al., 1999; Sondell et al., 1999). The present study also shows an increase in GLUT-1 mRNA at these doses, supporting the notion of increased metabolic demand in the explants. Of interest is the fact that astrocytes increase in number at higher doses of VEGF (50 to 100 ng/mL) in fetal explant cultures (Mani et al., 2000), whereas VEGF application in vivo has been shown to increase astrocyte mitogenesis (Krum et al., 2002). Hence, in our culture paradigm, it is possible that induction of vascular GLUT-1 expression in vessels occurs subsequent to the higher metabolic demand placed by astrocytes, neurons, and endothelial cells.
As stated previously, early embryonic lethality in the knockout paradigms for VEGF, flk-1, and flt-1 has limited our knowledge of the role played by the growth factor and its receptors during developmental CNS angiogenesis. This study demonstrates that VEGF, acting via the flk-1, MAPK/ERK, and PI-3 kinase pathways, is an important endothelial cell mitogen in fetal cerebral cortical explants grown under normoxic culture conditions and may play the same role during embryonic cortical development. Because VEGF appears to maintain or increase GLUT-1 expression in newly formed vessels, it may play a prominent role in developmental BBB formation and, perhaps, BBB reformation during the repair process after brain injury.
Footnotes
Acknowledgment:
The authors thank Newton Moore, Gilda Kornhauser, and Gauri Tadwalkar for their technical assistance.
