Abstract
The purpose of this study was to determine if the relationship between abnormalities in glucose, lactate, and oxygen metabolism were predictive of neurologic outcome after moderate or severe head injury, relative to other known prognostic factors. Serial assessments of the cerebral metabolic rates for glucose, lactate, and oxygen were performed using a modified Kety-Schmidt method. In total, 31 normal control subjects were studied once, and 49 TBI patients (mean age 36±16 years, median GCS 7) were studied five times median per patient from postinjury days 0 to 9. Univariate and multivariate analyses were performed. Univariate analysis showed that the 6-month postinjury Glasgow Outcome Scale (GOS) was most strongly associated with the mean cerebral metabolic rate of oxygen (CMRO2) (P = 0.0001), mean arterial lactate level (P = 0.0001), mean arterial glucose (P = 0.0008), mean cerebral blood flow (CBF), (P = 0.002), postresuscitation GCS (P = 0.003), and pupillary status (P = 0.004). Brain lactate uptake was observed in 44% of all metabolic studies, and 76% of patients had at least one episode of brain lactate uptake. By dichotomized GOS, patients achieving a favorable outcome (GOS 4-5) were distinguished from those with an unfavorable outcome (GOS1-3) by having a higher CMRO2 (P = 0.003), a higher rate of abnormal brain lactate uptake relative to arterial lactate levels (P = 0.04), and lesser degrees of blood-brain barrier damage based on CT findings (P = 0.03). Conclusions: During the first 6 days after moderate or severe TBI, CMRO2 and arterial lactate levels are the strongest predictors of neurologic outcome. However, the frequent occurrence of abnormal brain lactate uptake despite only moderate elevations in arterial lactate levels in the favorable outcome patients suggests the brain's ability to use lactate as a fuel may be another key outcome predictor. Future studies are needed to determine to what degree nonglycolytic energy production from alternative fuels such as lactate occurs after TBI and whether alternative fuel administration is a viable therapy for TBI patients.
Keywords
Although much is known regarding posttraumatic impairment of oxidative metabolism and its impact on outcome (Jaggi et al., 1990; Robertson et al., 1992; Tabaddor et al., 1972), less is understood about how alterations in glucose and lactate metabolism impact recovery of neurologic function. Cerebral oxidative metabolism generally remains markedly depressed for the first 2 weeks after severe head injury, and the degree of depression has been shown to correlate with poor long-term outcome (Jaggi et al., 1990; Muizelaar et al., 1989; Obrist et al., 1984; Robertson et al., 1992; Tabaddor et al., 1972).
In contrast, alterations in glycolysis and lactate metabolism appear to have a more variable time course and less certain impact upon neurologic recovery. Experimental and clinical studies have shown the existence of an ultra-early posttraumatic activation of glycolysis, also termed “hyperglycolysis,” followed by a subacute depression of glucose metabolism (Bergsneider et al., 1997,2000,2001; Kuroda et al., 1992; Yoshino et al., 1991). The relationship between these two different epochs of glucose metabolism and long-term outcome is unclear. The early hyperglycolytic phase appears to reflect increased energy demands for reversal of ionic imbalances because of excitatory amino acid flux (Katayama et al., 1990; Kawamata et al., 1992, 1995; Nilsson et al., 1990, 1993; Yoshino et al., 1991, 1992) and has also been associated with seizure activity after both experimental (Nilsson et al., 1994) and clinical TBI (Vespa et al., 1998). The subsequent depression of glycolysis may simply indicate decreased energy demands. However, given that mitochondrial function after TBI is severely impaired (Fiskum, 2000; Verweij et al., 2000), it is more likely that energy demands remain high and that injury-related events directly impair glycolysis. Posttraumatic factors such as NAD+ depletion (Ying et al., 2002), high NADH (Bouzier-Sore et al., 2001), and zinc influx (Sheline et al., 2000) may further exacerbate a dearth of ATP and ultimately result in energy failure.
Because lactate is a key intermediary between glycolysis and oxidative metabolism that can be both produced and used by the brain under pathologic conditions, it may play a key role in determining cellular viability and long-term outcome after TBI. In severely head-injured patients with cerebral infarctions, anaerobic glycolysis can result in marked increases in cerebral lactate production (Robertson et al., 1987). Elevated CSF lactate (DeSalles et al., 1986) and extracellular lactate measured by cerebral microdialysis (Goodman et al., 1999) has typically been attributed to ongoing ischemia (Chieregato et al., 2002; DeSalles et al., 1986; Goodman et al., 1999; Robertson et al., 1987). Lactate elevation caused by ischemia leads to lactate output from the brain unless arterial lactate levels are very high. However, there is increasing evidence indicating that lactate uptake also occurs in the injured brain and other injured tissues, which may then be used as an alternative fuel source (Amaral et al., 1986; Inao et al., 1988; Rivers et al., 1991). Several recent studies show that lactate produced from astrocytic glycolysis can be transported to adjacent neurons and used in this manner (Alessandri et al., 1999; Chen et al., 2000a; Magistretti et al., 1999; Ros et al., 2001). Lactate administration also improves cognitive outcome after experimental TBI (Rice et al., 2002) and reduces the need for glucose consumption (Chen et al., 2000a). Finally, lactate may be converted to pyruvate and, instead of entering the Krebs cycle, may act as a free radical scavenger (Cicalese, 2001; Ochiai et al., 2001). In sum, these studies suggest that posttraumatic anaerobic and aerobic metabolism may be more fully understood when carefully considering lactate as both a metabolic by-product and as a source for fuel.
Based upon the emerging concepts related to glucose and lactate metabolism described above, this study was designed to determine how the interrelationships between changes in glucose, lactate, and oxygen metabolism impact long-term outcome. To accomplish this goal, acute serial measurements of glucose, lactate, and oxygen metabolism were analyzed in relation to 6-month Glasgow Outcome Scale, while also considering the outcome predictors of age, Glasgow Coma Scale (GCS), pupillary abnormalities, hypotension, hypoxia, computed tomography findings, and patients' course of ICP and CPP.
METHODS
Patient population
Eligible patients included all mechanically ventilated moderate or severe head-injured patients, aged 14 years and older, who were admitted to UCLA Medical Center within 24 hours of injury. Moderate or severe head injury was defined as closed or penetrating injury, including gunshot and stab wounds, with a postresuscitation GCS less than or equal to 13, or deterioration to a GCS less than or equal to 13 within 24 hours of admission, and required mechanical ventilation and ICP monitoring. Exclusion criteria included the following: (1) terminal illness (e.g., advanced cancer, AIDS); (2) severe neurologic illness (advanced Parkinson's disease, multiple sclerosis, Alzheimer's dementia, disabling cerebrovascular event, mental retardation); and (3) acute complete spinal cord injury. This protocol was approved by UCLA's institutional review board and informed consent was obtained from the patients' legal representatives.
General management protocol
Patients were admitted to the intensive care unit (ICU) after initial stabilization or surgical evacuation of an intracranial hematoma and were treated in accordance with a Level I Trauma Center protocol. Management goals included maintenance of ICP less than 20 mmHg and CPP above 70 mmHg, in accordance with the Guidelines for the management of severe head injury (Bullock et al., 1996). All patients had a jugular bulb catheter inserted as soon as possible following patient admission to allow determination of arterio-venous differences for glucose (AVDglc), lactate (AVDlac), and oxygen (AVDO2).
Jugular bulb catheterization
The dominant jugular vein was selected based upon the dominant jugular foramen visualized on admission CT scan. Using standard techniques a 5 Fr Cordis and a 4 Fr Oxymetric catheter (Baxter Critical care, Baxter Health Care) was inserted to approximately 15 cm until resistance was encountered. Placement of the catheter was confirmed by lateral skull x-ray. The catheter was calibrated in vivo, and repeat calibration was performed every 12 hours. Display of light intensity and oxygen saturation was continuously displayed on the monitor.
Cerebral metabolism and blood flow measurements
Serial measurements of AVDglc, arterial lactate, AVDlac, AVDO2, and CBF were made simultaneously during the first 10 days after injury (postinjury days 0 to 9). Arterial and venous samples were scheduled every 12 hours during postinjury days 0 to 5, 7, and 9; 133Xenon CBF was scheduled for every 12 hours for the first 48 hours after injury (postinjury days 0 and 1), then daily on postinjury days 2, 3, 4, 5, 7, and 9. In this study, to assess the relationship of early brain metabolism to long-term outcome, only metabolic data obtained from postinjury days 0 to 5 was used given that many patients did not have complete data on post-injury days 7 and 9. Because of the patients' clinical status, such as severely elevated ICP (ICP > 30 mmHg), hemodynamic or respiratory instability, or removal of the jugular catheter or extubation following clinical improvement, it was not possible for all scheduled studies to be completed on all patients.
Blood and cerebral spinal fluid sampling and specimen analysis for glucose and lactate determinations
Blood for arterial and venous glucose and lactate levels were collected in the following manner:
In preparation, eight 3-cc syringes were rinsed with heparin, which was then discarded before drawing the blood samples. Before the draws, both the arterial and jugular lines had 5 cc of blood withdrawn as waste. 1 cc was then drawn during the CBF15 study and dispensed into two chilled and labeled 400-μL micro centrifuge tubes coated with heparin and lithium fluoride to prevent coagulation and glycolysis. The tubes were immediately placed on ice. Three additional paired blood draws were collected at 1-min intervals. The chilled tubes then underwent centrifugation at 1,200 g for 3 minutes. Once centrifuged, the supernatant/plasma was transferred to labeled micro centrifuge tubes for storage at −80°C or immediate spectrophotometric analysis. Cerebral spinal fluid (CSF) samples were drawn from the ventriculostomy catheter. Plasma and CSF levels for glucose and lactate were determined via spectrophotometric assay kits (Sigma), which measure the reduction of NAD to NADH at 340 nm (Beckman). The production of NADH is proportional to the concentration of glucose or lactate in the plasma. Samples were run 5 times with a measured error of < 4%. Plasma concentrations of glucose and lactate were corrected for corresponding whole blood values by using the erythrocyte distribution volumes for glucose and lactate, as previously described (Madsen et al., 1995; Simonsen et al., 1994).
For oxygen saturation and AVDO2 measurements, syringes of venous and arterial blood were immediately analyzed using a Corning 178 pH/Blood gas analyzer. For blood gas analysis, 1- to 2-mL blood samples were collected in heparinized syringes, capped, and immediately placed on ice.
Bedside CBF measurement was performed using the intravenous 133Xenon clearance technique (Obrist et al. 1979). Patients were injected with 20 to 30 mCi of 133Xenon dissolved in saline. A Ceretronix Cerebrograph Cortexplorer 16 (Randers, Denmark) measured gamma radiation from 133Xenon using eight detectors positioned over each cerebral hemisphere. End-tidal 133Xenon was measured simultaneously to estimate arterial 133Xenon concentration. Study duration was 11 minutes. Data were collected by a laptop computer, and the mean hemispheric and global CBF15 was calculated. The CBF15 is a CBF parameter calculated using a two-compartment mathematical model and has proven to be very stable in patients with reduced flow (Obrist and Marion, 1996). Pertinent clinical and physiologic data (MABP, ICP, CPP, PaCO2, body temperature, and vasoactive and metabolic suppressive drugs) were recorded at the time of study. All information was then entered into the UCLA CBF Laboratory and Core databases.
Determination of CMRglc, CMRlac, and CMRO2
For each metabolic measurement, the cerebral metabolic rates for glucose, lactate, and oxygen were calculated as the product of the simultaneously measured AVDglc, AVDlac or AVDO2, and the CBF15. Studies were excluded from analysis if at the time of metabolic assessment the patient was receiving metabolic suppressive therapy with high-dose pentobarbital or high-dose propofol, defined as a propofol infusion of 100 μgm/kg/min or greater, given that these two agents are known to significantly suppress both glucose and oxidative metabolism (Cormio et al., 1999; Oertel et al., 2002). The metabolic ratio (MR) was determined by calculating CMRO2/CMRglc in molar units at 12-hour intervals until the jugular catheter was removed.
Global outcome measurement
Neurologic outcome was assessed 6 months after injury by investigators blinded to the patients' metabolic and other clinical data, using the 5-point Glasgow Outcome Scale (GOS) (Jennet and Bond, 1975). In assessing predictors of outcome, the GOS was used as both a linear scale (1 = death to 5 = good recovery) and as a dichotomized outcome score of favorable (good recovery or moderate disability) versus unfavorable (severe disability, vegetative, or death) (Choi et al., 1998).
Predictors of global outcome
The relationships of 6-month GOS to mean levels of AVDglc, arterial glucose and lactate, AVDlac, SjVO2, AVDO2, CBF, CMRglc, CMRlac, CMRO2, the metabolic ratio, and CSF glucose and lactate levels were examined. The impact of increased rates of glucose metabolism was also assessed. Absolute hyperglycolysis (> 6.69 mg/100g/min) was defined as a CMRglc greater than the 97.5th percentile of normal for an awake human (normal mean = 4.46 mg/100g/min±1.16, n = 30), and relative hyperglycolysis (MR < 3.44) was defined as a metabolic ratio (MR = CMRO2/CMRglc in molar units) below the 2.5th percentile of normal (normal MR mean = 5.83±1.41, n = 30).
The associations of outcome to other outcome predictors for TBI were also examined. These clinical predictors included patient age, postresuscitative GCS score, postresuscitative pupillary examination (both normal, one abnormal, both abnormal), hypotension (systolic blood pressure < 90 mmHg), hypoxia (PaO2 < 60 mmHg or apnea) within 24 hours of injury, the mean ICP and CPP, percentage of hours ICP greater than 20 mmHg, and percentage of hours CPP less than 60 mmHg (Marmarou et al., 1991; Marshall et al., 1991a; Vollmer et al., 1991). Previously described CT predictors of outcome seen on patients' first or second CT scans included basilar cistern compression, midline shift greater than 5 mm, acute subdural hematoma requiring evacuation, or the presence of subarachnoid hemorrhage (Marshall et al., 1991b). All patients' first two CT scans were obtained within 12 hours of admission. The degree of blood-brain barrier damage was also assessed by CT for each patient, by determining the total number of the following diagnoses: acute subdural hematomas, intracerebral hematomas, or multiple contusions. Each of these diagnoses has been associated with blood-brain barrier damage in prior experimental or clinical studies (Barzo et al., 1996; Beaumont et al., 2002; Fobben et al., 1989; Okauchi et al., 2002; Xi et al., 2001).
Normal control subjects
A cohort of 31 healthy volunteer subjects was studied to define the normal range of all metabolic parameters, defined as between 2.5th percentile and the 97.5th percentile. For data with an approximately normal distribution, the 2.5th and 97.5th percentiles would correspond to the mean±2 SD. These normal subjects were studied in an awake state. Through use of a femoral vein approach, a jugular bulb catheter was passed under fluoroscopic guidance into proper position in the jugular bulb. A radial arterial line was also placed. Single measurements of AVDglc, AVDlac, and AVDO2 were made by taking simultaneous samples of arterial and venous blood as described above. A CBF measurement was performed using the intravenous 133Xenon clearance technique as described above. Normal ranges for cerebral metabolic rates for glucose, lactate, and oxygen were calculated as the product of the simultaneously measured AVDglc, AVDlac or AVDO2, and the CBF15.
Statistical analysis
Mixed effects linear regression models were used throughout to account for the uneven numbers of studies on patients at different time points (Laird and Ware, 1982). To examine the association of metabolism and 6-month GOS, the measurements of metabolic parameters were modeled as correlated outcomes within patients with different means for each level of the GOS. A variety of covariance structures for the repeated measures were considered; we report results for models with random patient-level means and continuous time AR(1) correlation. For these statistical models, the estimated means by GOS category can be thought of as a data-driven compromise between the means of the studies within each GOS category and the mean of the patient means for each GOS category. We refer to these quantities as the adjusted means in the results section; they are adjusted for the unequal number of studies per patient at different hours postinjury. Similar interpretation can be given to the adjusted standard deviations for each GOS category. Logarithmic transformations of the metabolic measurements were used to obtain more accurate P values where appropriate; however, adjusted means and standard deviations are reported for the original scale data for clarity of interpretation. The univariate mixed effect models were then augmented by controlling for a fixed set of clinical predictors including patient age, ISS, pupillary findings, CT findings, mean ICP, mean CPP, and best GCS. Comparisons of TBI patients to normal volunteers were performed in a similar manner. In several instances, we consider repeated measures of a dichotomous indicator (e.g., lactate uptake above a certain threshold). Generalized Estimating Equations methods were used to analyze these data (Liang and Zeger, 1986; Zeger and Liang, 1986). Comparison of TBI patients with normal volunteers was performed in a similar manner. Statistical significance was defined at P < 0.05, but in some instances, attention is drawn to suggestive results given the small sample sizes.
RESULTS
Head injury cohort
The 49 TBI patients were enrolled in the study between July 1998 and March 2002. The mean patient age was 36±16 years (range 16 to 81 years) and 13 (27%) were female. (Table 1) The median postresuscitation GCS score was 7 with a range of 3 to 14; 86% of patients had an initial GCS less than or equal to 8. Mechanisms of injury included 16 motor vehicle accidents, 11 pedestrian versus automobile accidents, 11 falls, 4 assaults, 3 gunshot wounds, 2 motorcycle accidents, and 2 bicycle accidents. In total, 28 patients (57.1%) required a craniotomy for evacuation of an intracranial hematoma, including 11 with intracerebral hematoma/contusions, 8 with combination lesions, typically subdural hematomas in combination with cerebral contusions, 6 with subdural hematomas, and 3 with epidural hematomas.
Head injury cohort demographics and outcome
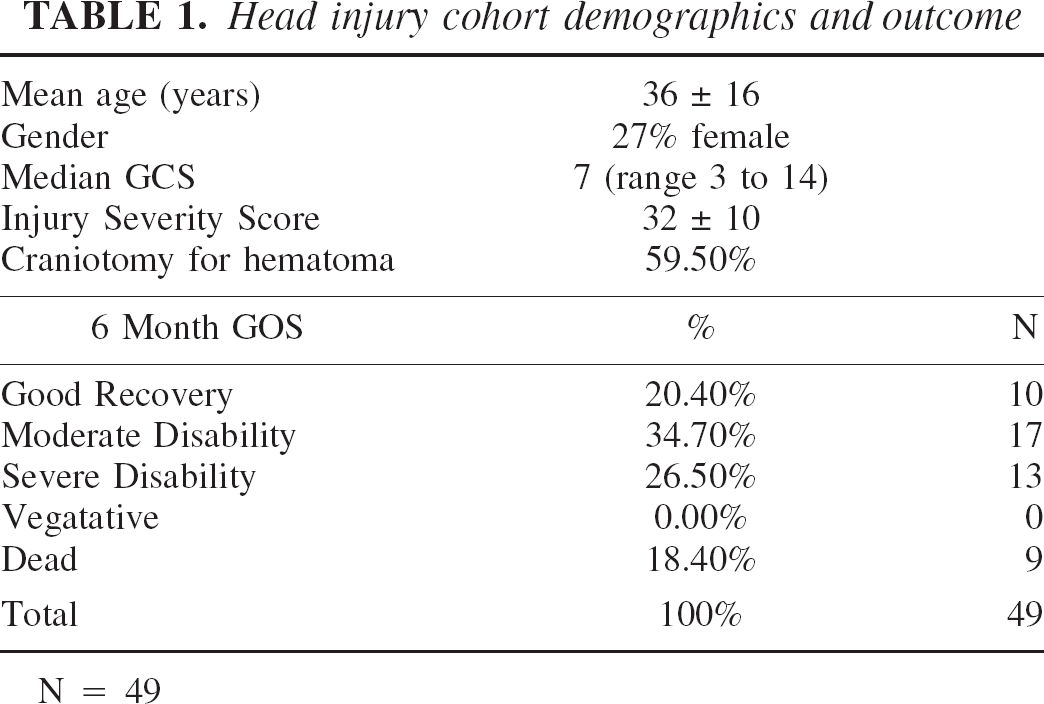
N = 49
At 6 months after injury, global neurologic outcome as measured by the GOS was good recovery in 10 patients (20.4%), moderate disability in 17 (34.7%), severe disability in 13 (26.5%), and death in 9 (18.4%); no patients remained in a vegetative state. Overall, 55% of the patients achieved a favorable outcome, and 45% achieved a poor outcome (Table 1).
Metabolic rates of TBI subjects versus normal control subjects
Thirty-one normal volunteers (mean age 33 ± 8 years, 26% female) had a single determination of metabolic rates in an awake state. The 49 TBI patients had a total of 185 CMRO2, 185 CMRglc, 179 CMRlac, and 174 Metabolic Ratio determinations (median 5 per patient, range 1 to 13) over postinjury days 0 to 5. As can be seen in Table 2, the mean value of each parameter over postinjury days 0 to 5 for the 49 TBI patients was significantly different from those of the control subjects. The parameters most severely depressed relative to the normal subjects' values were CMRO2 (45% of normal), AVDO2 (55% of normal), and CMRglc (76% of normal), whereas the mean arterial lactate level for TBI patients was most elevated relative to normal (210% of normal). Notably, global cerebral ischemia as defined by an AVDO2 greater than or equal to 9.6 mL/dl was not seen in any TBI patients.
Comparison of metabolic parameters for normal subjects and trauma patients
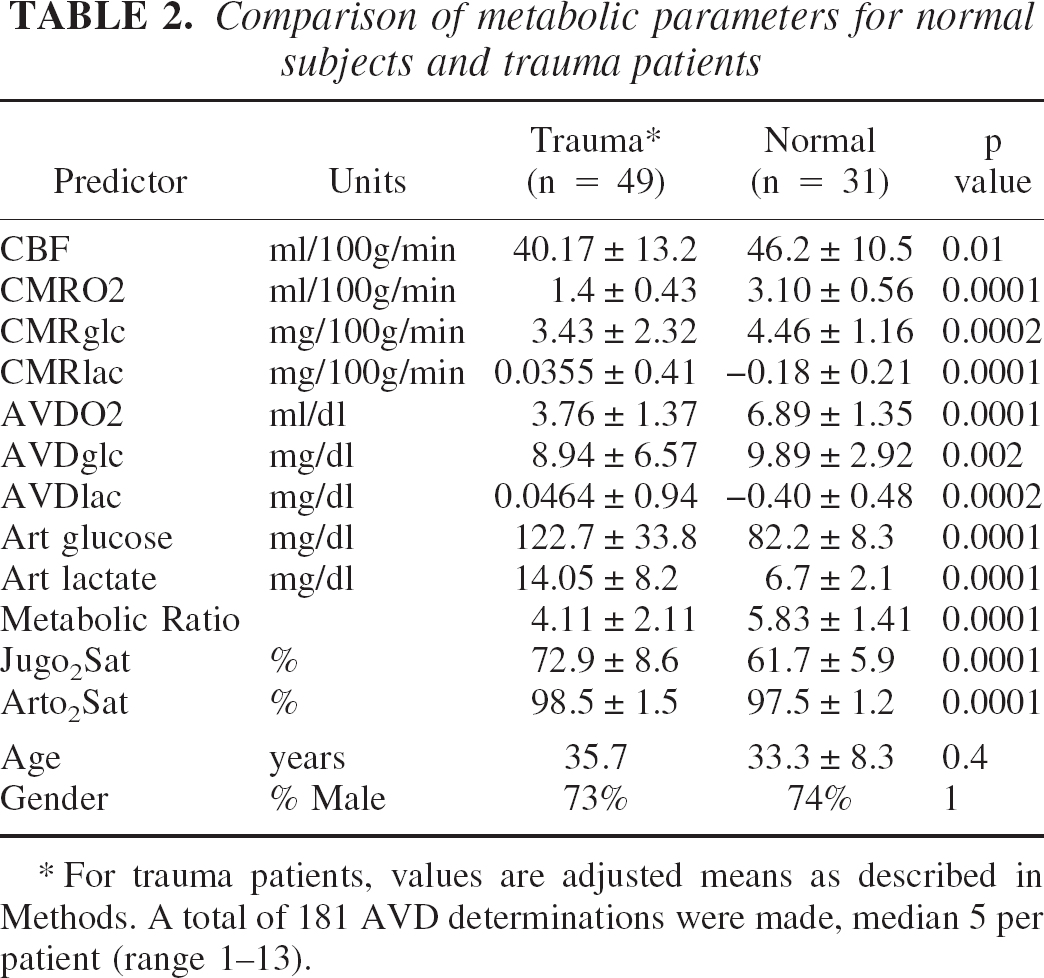
For trauma patients, values are adjusted means as described in Methods. A total of 181 AVD determinations were made, median 5 per patient (range 1–13).
Predictors of global outcome: Univariate analysis
The mixed effect linear regression models yielded the following associations of metabolic and other prognostic parameters with 6-month GOS (Table 3). For predicting 6-month GOS category, the strongest factors were mean CMRO2 (P = 0.0001), mean arterial lactate level (P = 0.0001), mean arterial glucose (P = 0.0008), mean CBF (P = 0.002), postresuscitation GCS (P = 0.003), and pupillary status (P = 0.004). For predicting a favorable outcome (GOS 4 or 5) versus an unfavorable outcome (GOS 1 or 3), the strongest factors were mean CMRO2, mean arterial lactate level, mean arterial glucose level, mean CBF, CSF lactate concentration, postresuscitation GCS, pupillary status, and number of CT diagnoses associated with blood-brain barrier damage.
Association of 6-month outcome to metabolic and clinical parameters
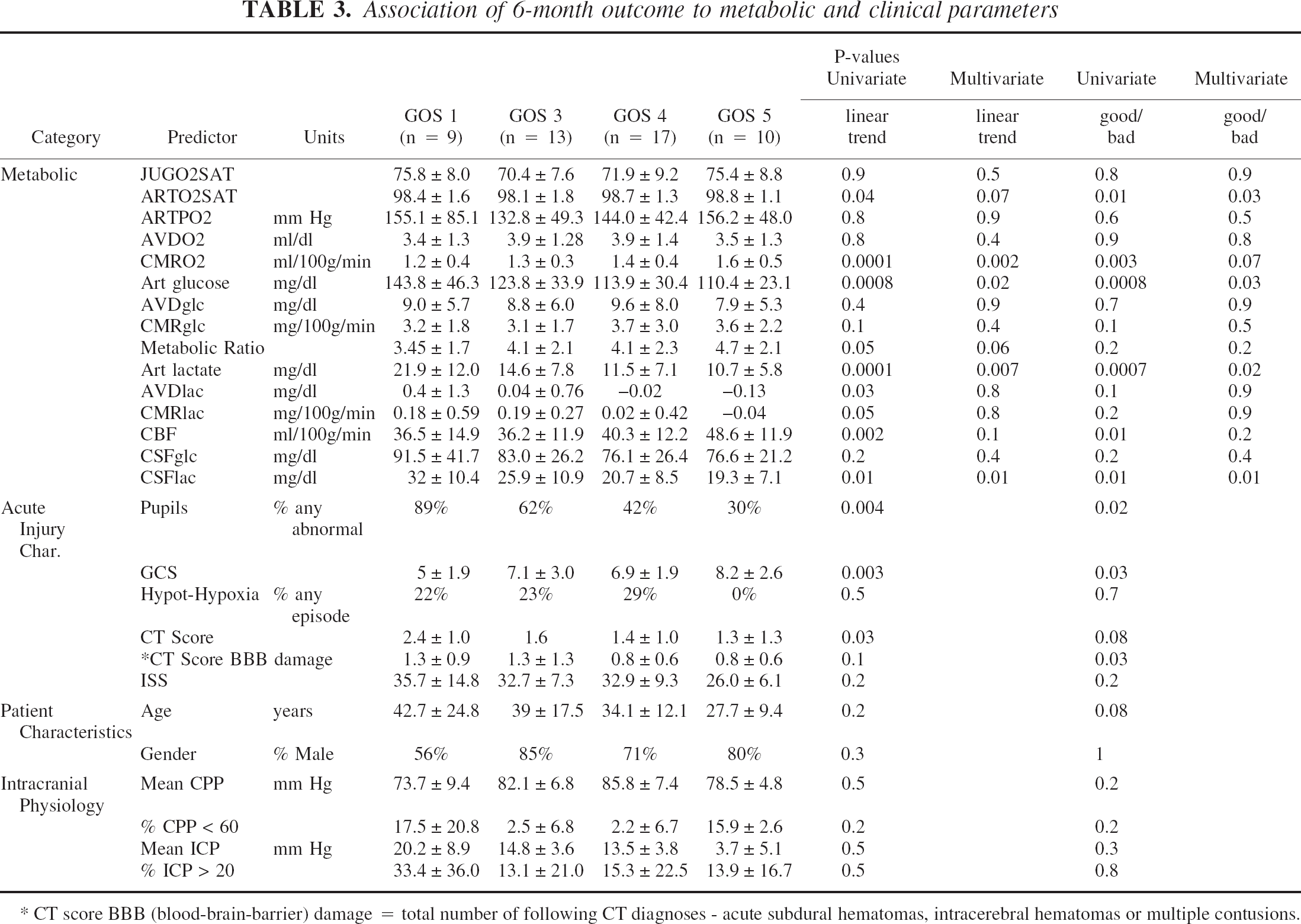
CT score BBB (blood-brain-barrier) damage = total number of following CT diagnoses - acute subdural hematomas, intracerebral hematomas or multiple contusions.
Multivariate analysis
After controlling for injury characteristics, age, and parameters of ICP and CPP, the associations between GOS category and the following factors remained strong: CMRO2 (P = 0.002), arterial lactate (P = 0.007), CSF lactate (P = 0.01), and arterial glucose (P = 0.02) (Table 3). A statistical trend existed for CBF (P = 0.1) and metabolic ratio (P = 0.06).
Oxygen metabolism and outcome
CMRO2 was significantly and ubiquitously depressed with 97% (179 of 185 studies) of measurements below the 2.5th percentile (2.23 mL/100g/min) of the normal cohort. Overall, the mean CMRO2 was lowest in patients who died. Patients with a good recovery had a mean CMRO2, which was 44% greater than those patients who died.
Lactate metabolism and outcome
In the TBI cohort, 80 of 181 (44%) AVDlac measurements were positive, indicating brain uptake of lactate, and 32 of 42 (76%) patients had at least one episode of brain uptake of lactate. Positive AVDlac values occurred across all injury days although they tended to occur earlier after injury. As seen in Fig. 1, the greatest departure from normal CMRlac values was in the positive direction (i.e., brain lactate uptake) and occurred most frequently in the nine patients who died, in which 20 of 30 (67%) measurements were above zero. Patients who died and those who remained severely disabled also had the highest systemic arterial lactate levels, as seen in Table 3, and arterial lactate levels overall had a strong inverse correlation with 6-month GOS (P = 0.0001). As seen in Fig. 2, abnormal brain lactate production (CMRlac below the 2.5th percentile of normal, i.e., < −0.71 mg/100g/min) was uncommon, occurring in only 4 (2%) of 179 measurements and in four (8%) patients, whereas abnormal brain lactate uptake (CMRlaclac above the 97.5th percentile of normal, i.e., > 0.11 mg/100g/min) was observed in 51 (28%) of 179 measurements. Despite significantly lower levels of systemic arterial lactate in the favorable outcome patients (GOS 4 or 5), abnormal brain lactate uptake still occurred relatively frequently, being observed in 26% and 33% of the measurements in these patient groups, respectively (Fig. 3). Overall, a higher rate of brain lactate uptake relative to the simultaneous arterial lactate level was observed in the favorable outcome group (GOS 4 and 5) compared with the unfavorable outcome group (GOS 1 and 3) (P = .04).
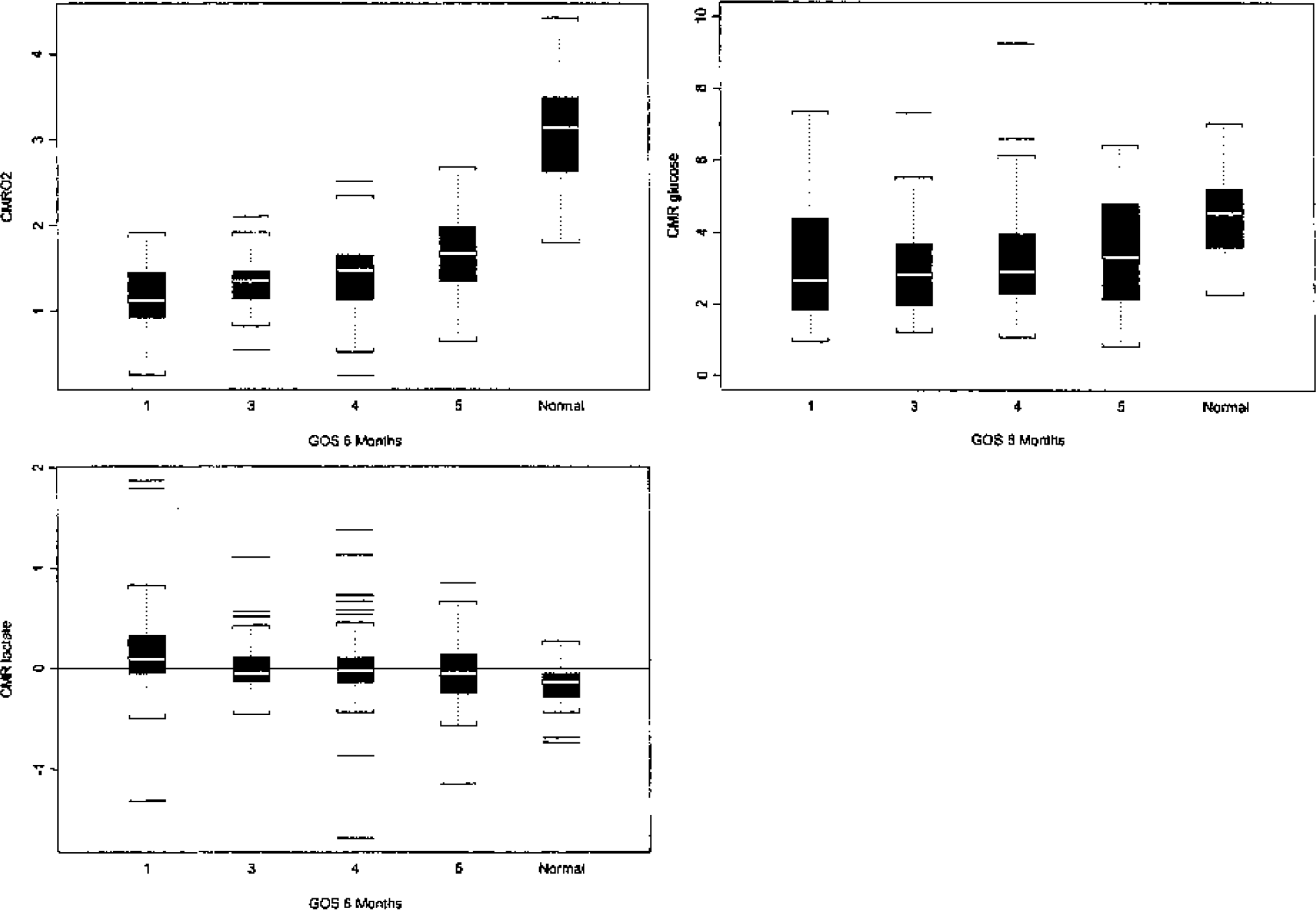
Boxplots of CMRO2, CMRglc, and CMRlac by 6-month Glasgow Outcome Scale. Summary of values in 49 patients collected from postinjury days 0 to 5, stratified by 6-month GOS and compared with values of 31 normal control subjects. Lines within the box indicate median, the box interquartile range, whiskers bracket 95% of the data, and straight bars outliers. Overall CMRO2 is higher in patients with a good outcome but is depressed in all groups compared with control patients. CMRglc is also depressed after injury but to a lesser degree than CMRO2. CMRlac was positive in almost half of all studies; note that the most positive values are in the patients who died (GOS 1).
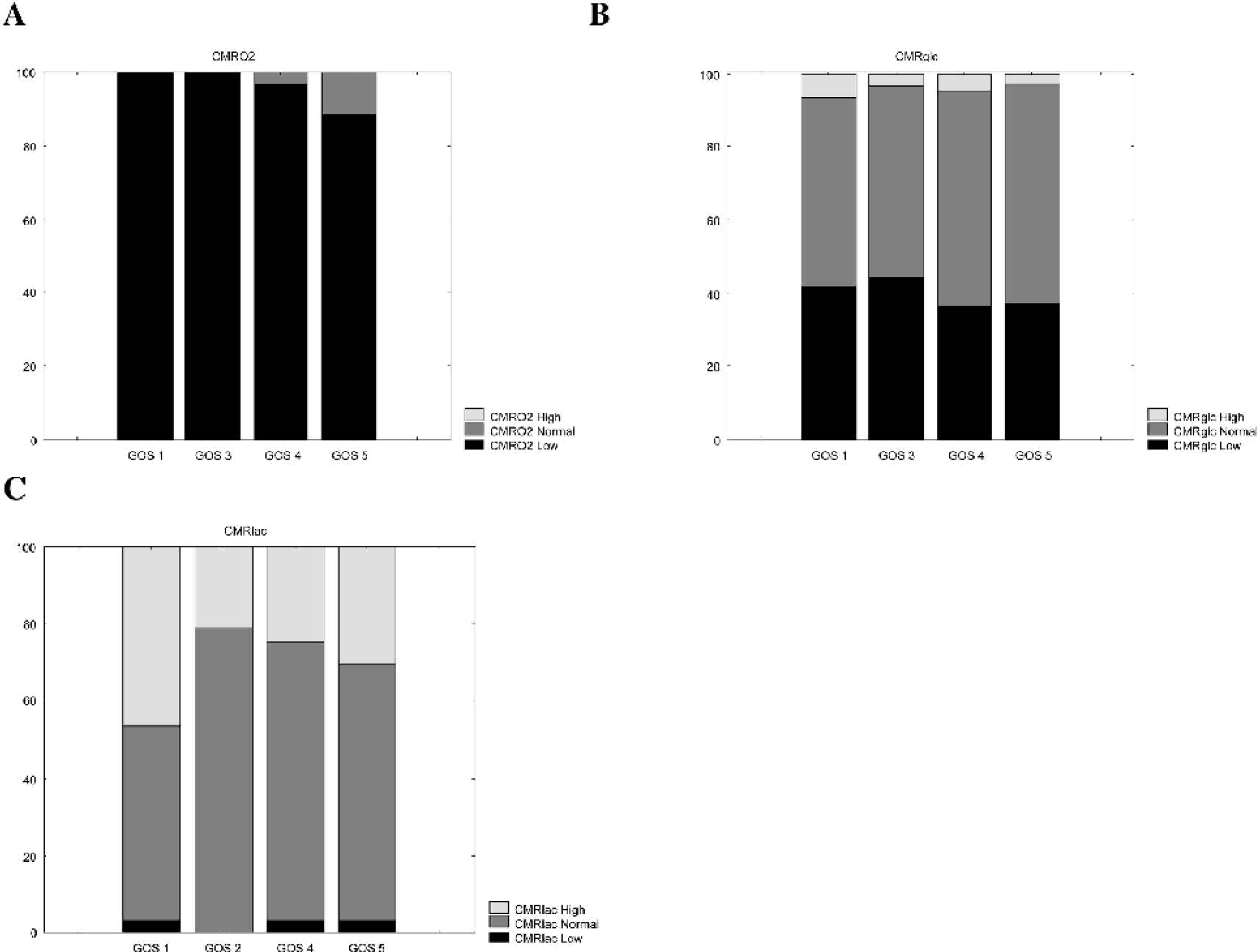
Graph showing the percent of CMR measurements that were either below the 2.5th percentile, above the 97.5th percentile, or within normal range of control subjects, stratified by 6-month GOS score. Note the relatively uncommon finding (2%) of abnormally negative CMRlactate
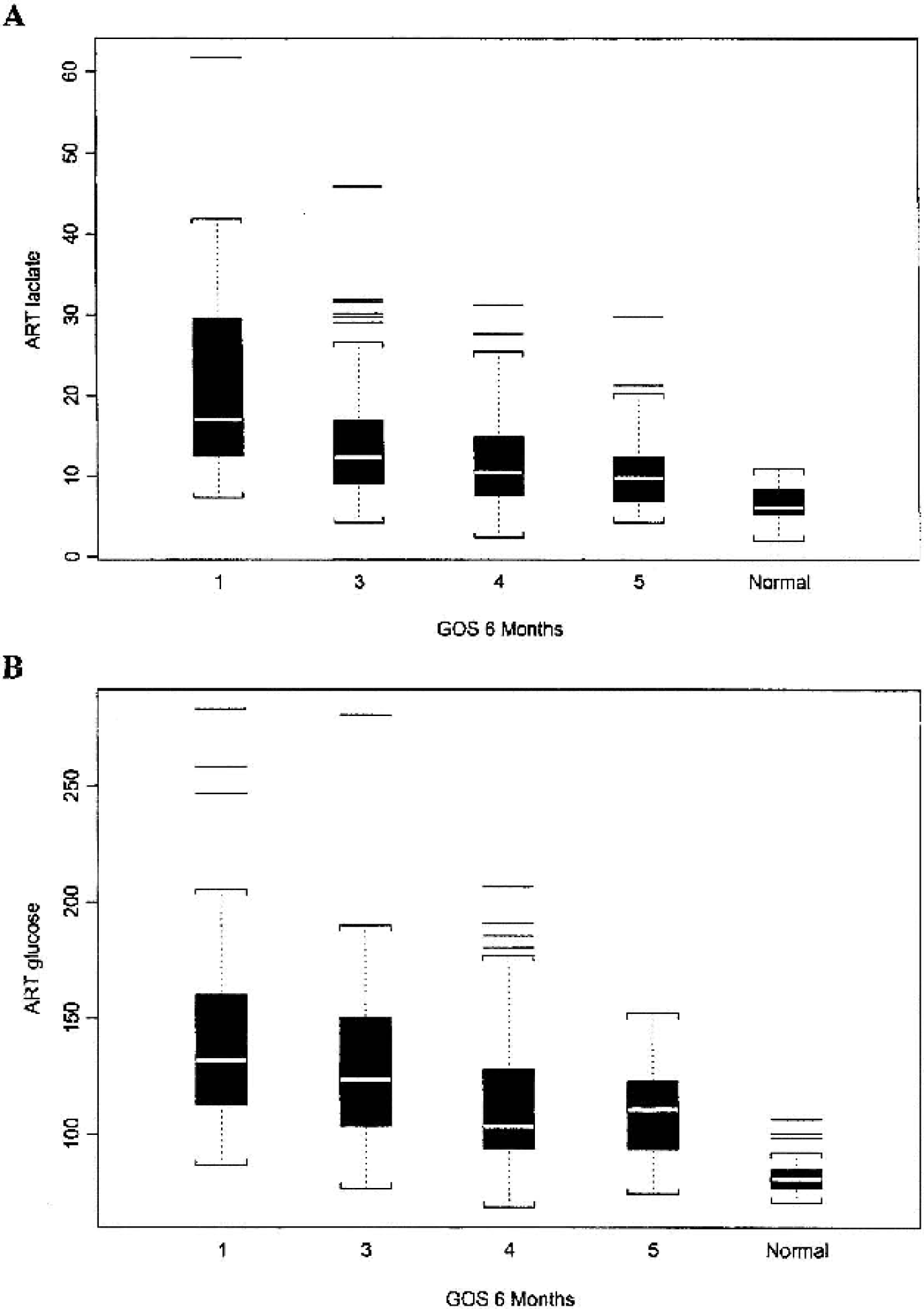
Mean arterial lactate and glucose levels. Mean arterial plasma glucose and lactate concentrations were lower in patients with higher GOS (4 and 5), but both were abnormally elevated compared with control volunteers.
Glucose, hyperglycolysis, and outcome
High plasma glucose levels were associated with poor outcome (Fig. 3). The mean plasma glucose was 23% greater in patients who died compared with patients with a GOS of 5. Relative hyperglycolysis (metabolic ratio < 3.44) was observed in 44% of studies, and at least one episode of relative hyperglycolysis was seen in 39 of 49 (80%) patients. Nine episodes of absolute hyperglycolysis (5% of 185 measurements) were documented in nine patients. In these nine patients with absolute hyperglycolysis, five achieved a favorable outcome, and four achieved a poor outcome by 6-month GOS. Relative hyperglycolysis appeared to be more frequent in patients who died with 48% (19 of 40) studies showing a metabolic ratio less than 3.44, whereas patients with a good outcome had only 29% (10 of 35) studies in the relative hyperglycolysis range. However, neither episodes of absolute or relative hyperglycolysis were strongly associated with 6-month GOS.
Cerebral blood flow and outcome
Using univariate analysis, CBF was a strong predictor of 6-month GOS but using multivariate analysis, CBF was a relatively weaker outcome predictor (P = .1 for linear trend and P = .2 for good/bad) (Table 3). CBF in the ischemic range rarely occurred as it was observed in only four instances. However, three ischemic CBF values (< 20 mL/100g/min) were observed in patients who died. Mean CBF was lower in patients with GOS 1 and 3, compared with the GOS 4 and 5 outcome groups.
DISCUSSION
Overview of findings
This study of patients with moderate and severe head injuries reconfirms previous works on the prognostic importance of acute changes in cerebral oxidative metabolism and blood flow (Jaggi et al., 1990; Kelly et al., 1997; Obrist et al., 1984; Robertson et al., 1992). This analysis also brings new insights as to how posttraumatic interactions between glycolysis, lactate metabolism, and oxidative metabolism impact long-term outcome. The major findings of this study are as follows:
Moderate or severe TBI resulted in an acute reduction of CMRO2 and, to a lesser extent, CMRglc on average, to 45% and 76% of normal, respectively. Relative hyperglycolysis was common early after TBI, being observed at least once in 83% of patients. The best clinical predictors of 6-month GOS were CMRO2, arterial lactate level, arterial glucose level, CBF, and postresuscitation GCS and pupillary status. Brain lactate uptake was observed in 44% of all metabolic studies, and 76% of patients had at least one episode of brain lactate uptake. Abnormal uptake of lactate by the brain (CMRlac > 97.5th percentile of normal) occurred in 28% of studies, whereas abnormal lactate production by the brain occurred in only 2% of studies. Patients achieving a favorable outcome had a higher rate of abnormal brain lactate uptake relative to arterial lactate levels in comparison with patients achieving an unfavorable outcome.
These findings are discussed below in relation to previous studies on metabolic changes after TBI and implications for future TBI therapies.
Oxygen metabolism
Consistent with the present study, it is well-established that CMRO2 is reduced by approximately 50% from normal in comatose head injury patients (Bouma et al., 1991; Martin et al., 1997; Obrist et al., 1984; Robertson et al., 1992). This reduction in CMRO2 occurs both in areas affected by focal traumatic lesions and in remote areas, which appear normal on computerized tomographic scans (Tenjin et al., 1990). The depression in CMRO2 is seen when it is first measured after admission to the hospital, is proportional to the depth of coma, and appears to be stable at this level (Bouma et al., 1991; Jaggi et al., 1990; Obrist et al., 1984, 1993). The low CMRO2 returns at least partially toward normal in patients who recover, indicating this depression is not simply caused by irreversible destruction of brain cells (Tenjin et al., 1990).
Glucose metabolism
Unlike the depression of oxidative metabolism, an increase in cerebral glucose use has been a consistent finding across animal brain-injury models, including concussive brain injury (Kawamata, 1992; Sunami et al., 1989; Yoshino et al., 1991), cortical contusions (Sunami et al., 1989), and experimental subdural hematoma (Inglis et al., 1992). These studies have revealed that injured cells are exposed to a massive ionic flux, in part because of the release of excitatory amino acids (Katayama et al., 1989, 1990). This ionic imbalance transiently increases the use of cerebral glucose, which activates Na+/K+ pumps in an effort to restore ionic homeostasis (Hovda, 1996; Hovda et al., 1992; Yoshino et al., 1991).
The existence of an increased cerebral metabolic rate of glucose (CMRglc) in the acute posttraumatic human is supported by several independent methods. Using [18F]-fluorodeoxyglucose positron emission tomography (FDG-PET), we confirmed experimental autoradiographic studies by demonstrating that regional CMRglc is elevated in the majority of patients with severe head injuries studied within 1 week of injury (Bergsneider et al., 1997). A similar finding has been reported using FDG-PET combined with an (15O) oxygen PET study (Yamaki et al., 1996). Given the relative depression of oxidative metabolism after TBI, one might predict marked compensatory increases in cerebral glycolysis to maximize ATP production. However, as seen in this study, glucose metabolism was also globally depressed but not to the same degree as oxidative metabolism, resulting in the common finding of “relative hyperglycolysis.” Several factors are likely responsible for this absolute reduction in glycolysis. Lactate uptake by the brain likely impedes glycolysis if it is converted to pyruvate (Amaral et al., 1986). Additional posttraumatic factors such as NAD+ depletion (Ying et al., 2002), high NADH (Bouzier-Sore et al., 2001), and zinc influx (Sheline et al., 2000) may also acutely suppress glycolysis. It is also likely that episodes of “absolute hyperglycolysis” were rarely captured because of the few measurements taken within hours of injury when absolute hyperglycolysis is typically observed (Kuroda et al., 1992; Yoshino et al., 1991). Overall, “absolute hyperglycolysis” was seen in only 5% of measurements in this study. Such episodes of hyperglycolysis are generally expected to result in brain lactate production, but of interest is the fact that in five of nine such episodes, abnormal lactate uptake by the brain was observed. The simultaneous presence of absolute hyperglycolysis, marked brain lactate uptake, and low CMRO2 is likely indicative of a cerebral energy crises, as evidenced by the fact that all three patients in this metabolic state had poor outcomes. Perhaps an important caveat to the interpretation of global measurements of glucose metabolism is that the metabolic fate of glucose, that is, glycolysis or biosynthesis of other compounds, cannot currently be ascertained using these techniques.
Absence of ischemia and rare brain lactate production
This study is in contrast to previous reports that have stressed cerebral production of lactate as a marker of brain ischemia (Goodman et al., 1999; Robertson et al., 1987). In the present study, extreme degrees of cerebral lactate production were seen in only 2% of studies, and AVDO2 values in the ischemic range were never observed. Previous microdialysis studies (Goodman et al., 1999; Meixensberger et al., 2001; Reinert et al., 2000) and our own ongoing microdialysis studies showing elevated microdialysate levels of lactate may in part be measuring lactate that is diffusing into or being transported into the interstitial space from the extracranial circulation (Chen et al., 2000b; Nemoto and Severinghaus, 1974; Rivers et al., 1991). Admittedly, the global technique used in this study cannot detect areas of regional ischemia where high lactate production may occur.
Brain lactate uptake: Two scenarios
The findings regarding lactate metabolism raise two key questions. First, given that the majority of patients are in a state of relative hyperglycolysis acutely after injury in which CMRO2 is decreased to a greater degree than CMRglc, why does abnormal uptake of lactate by the brain occur so much more frequently than brain lactate production? Second, why is brain uptake of lactate associated with both favorable and unfavorable clinical outcomes? To answer these questions, we propose that two very different clinical scenarios exist after TBI. In a favorable outcome scenario (in patients who achieve a GOS of 4 or 5), uptake of lactate by the brain occurs primarily by facilitated transport through a largely intact blood-brain barrier. Given a less depressed CMRO2 in these patients, the lactate can be effectively used as a fuel source. In an unfavorable outcome scenario (in patients who achieve a GOS of 1 to 3), large amounts of lactate passively enter the brain as a result of high systemic arterial lactate levels and a damaged blood-brain barrier. Given a severely depressed CMRO2 in these patients, the abundant lactate cannot be effectively used.
Several findings in this study support this two-scenario concept of posttraumatic lactate metabolism. Despite the fact that the favorable outcome patients had lower arterial lactate levels than the poor outcome patients, these patients had a higher rate of abnormal brain lactate uptake relative to arterial lactate levels. This finding of a high rate of brain lactate uptake despite lower arterial lactate levels suggests the lactate was being actively taken into the brain. The favorable outcome group was further distinguished from the unfavorable outcome group by having higher values for CMRO2 and CMRglc and lesser degrees of blood-brain barrier damage based upon CT findings. The lesser degree of blood-brain barrier damage in the favorable outcome patients also argues against the possibility that the brain lactate uptake in these patients was simply a result of diffusion into the brain.
Previous studies confirm that under certain conditions, the brain and other tissues, such as heart, kidney, liver, and muscle, can use lactate through so-called “lactate shuttles” and facilitated transport mechanisms (Amaral et al., 1986; Brandt et al., 1984; Brooks, 2002; Rivers et al., 1991). Others have shown brain uptake of lactate and its conversion to pyruvate to enter the Kreb's cycle for ATP production (Chen et al., 2000a; Magistretti et al., 1999; Ros et al., 2001). Lactate may also be used to scavenge free radicals (Cicalese, 2001; Ochiai et al., 2001). In contrast, under conditions of high arterial lactate levels and a damaged blood-brain barrier, lactate may passively enter the brain but not be used because the neurons and glia have severely depressed metabolic capacity (Clausen et al., 2001; Fiskum, 2000; Nemoto and Severinghaus, 1974; Verweij et al., 2000).
Ways to improve energy production
The correlation of severely depressed cerebral metabolism with poor outcome (Jaggi et al., 1990; Obrist et al., 1984; Robertson et al., 1992) raises the question of whether increased energy production after TBI will enhance outcome. Several possible therapeutic avenues have been previously proposed, including use of hyperbaric oxygen (Rockswold et al., 2001), increased inspired oxygen concentration (Menzel et al., 1999), and cyclosporin A (Alessandri et al., 2002). Given the increasing evidence that the injured brain can use alternative substrates such as lactate and that lactate administration itself can improve outcome after experimental TBI (Rice et al., 2002), future studies aimed at identifying safe and effective fuel sources may prove fruitful. Pyruvate, which can directly enter the Kreb's cycle or be used as a free radical scavenger, may be one such compound (Cicalese, 2001; Ochiai et al., 2001). Two additional compounds, beta-hydroxybutyrate, a ketone body known to be used by the brain under starvation conditions, and acetyl-L-carnitine also have potential as alternative fuel sources after TBI (Rosenthal et al., 1992; Suzuki et al., 2001).
Methodologic issues
The modified Kety-Schmidt method used in this study is a well-established technique that has been used extensively in TBI patients (Jaggi et al., 1990; Muizelaar et al., 1989; Obrist et al., 1984). Regarding the timing of studies, data from only the first 6 days (postinjury days 0 to 5) after injury were assessed in part because the first several days after injury have generally been considered the most dynamic in terms of metabolic and blood flow changes (Bouma et al., 1991; Jaggi et al., 1990). In the present study, a median of five metabolic and CBF determinations were performed during these first 6 days after injury. For practical reasons and patient safety issues, it was not possible to obtain all planned studies over this time period. Clinical factors such as high ICP or hemodynamic instability precluded a minority of studies. Patients being treated for intractable intracranial hypertension with high-dose pentobarbital or propofol were also excluded from analysis, given that these agents significantly reduce both oxidative and glucose metabolism (Cormio et al., 1999; Kelly et al., 1999; Oertel et al., 2002). After postinjury day 5, neurologic improvement and extubation and removal of the jugular bulb catheter were the most common reasons for patients leaving the study.
The statistical analyses used in this study specifically accounted for the variable numbers of studies nested within subjects and the relatively small sample size of TBI subjects. Although it was not feasible to perform regression analyses of outcome on metabolic and other predictors, the longitudinal nature of the study minimized the problem of a small sample size by allowing for regression of metabolic measurements on outcome and other predictors.
CONCLUSION
This study of acute posttraumatic cerebral metabolic changes suggests that the interplay between energy supply and demand ultimately dictates global neurologic outcome. It again shows the prognostic importance of acute measurements of cerebral oxidative metabolism and blood flow. However, it also suggests that brain lactate metabolism may be another key outcome determinant. We propose that patients with relatively less severe injuries as characterized by a higher CMRO2, lesser degrees of blood-brain barrier damage, and lower systemic lactate and glucose levels can use lactate as an additional fuel source and ultimately have a favorable long-term outcome. In contrast, patients with more severe injuries as characterized by a lower CMRO2, higher degrees of blood-brain barrier damage, and higher systemic lactate levels are unable to use the lactate and ultimately have a poor outcome. Thus a distinguishing feature between patients who make a good recovery and those who do not may be their ability to meet energy demands by whatever means possible, including the use of alternative fuel sources such as lactate. Future studies are needed to better understand alternative fuel use after head injury and to develop strategies that augment posttraumatic cerebral metabolic capacity.
Footnotes
Acknowledgment
We thank Maria Etchepare, Jill Hutchinson, and Kathy Langlois for their help in data collection. We also thank Brenda Rinsky, Oscar Barcenas, and Chris Hanuscin from the UCLA Cerebral Blood Flow Laboratory for their technical support.
