Abstract
HDJ-2, a member of the HSP40 family, functions as a cochaperone to promote protein folding both by binding to unfolded polypeptides and by regulating the activity of HSP70. This study tested whether HDJ-2 overexpression alone could provide significant protection from ischemia-like injury. Primary mouse astrocyte cultures were infected with an HDJ-2 encoding retroviral vector or control retrovirus lacking HDJ-2. Expression of HDJ-2 was confirmed by immunohistochemical staining and immunoblotting. Injury paradigms to mimic ischemia were glucose deprivation (GD) for 24 hours and oxygen-glucose deprivation (OGD) for 8 hours. Cell death was determined by trypan blue exclusion and cell counting. Overexpression of HDJ-2 alone significantly reduced astrocyte injury after both GD and OGD, independent of an elevation in HSP70. To further search for the mechanism of HDJ-2 protection, cultured astrocytes allowed to recover 16 hours after 8 hours GD were labeled with a monoclonal antiubiquitin antibody that recognizes both free ubiquitin and ubiquitinated proteins. The immunolabeling pattern changed from a relatively even distribution in both nuclei and cytoplasm in control cells to heterogeneous aggregates and marked reduction of nuclear staining in most of the cells after GD. When HDJ-2 was overexpressed, the number of cells with evidence of protein aggregation was significantly reduced. Thus, blocking protein aggregation may be an important mechanism by which HDJ-2 protects cells from damage.
Heat shock proteins (HSPs) play important roles in normal cellular function and survival. They are molecular chaperones, which facilitate de novo protein folding and recognize and recover misfolded proteins under normal and stressed conditions. In addition to their role in protein folding, chaperones facilitate translocation of proteins across membranes, assembly, and disassembly of protein complexes, presentation of substrates for degradation, and suppression of protein aggregation (Hartl, 1996;Ohtsuka and Hata, 2000). The HSP40 family constitutes a major group of HSP70 cochaperone proteins. HDJ-2, a human DnaJ member of the HSP40 family is highly homologous to the bacterial DnaJ protein from Escherichia coli. HDJ-2 interacts with HSP70 through its J domain (Gebauer et al., 1997;Minami et al., 1996;Tang et al., 1997), targeting HSP70 to specific intracellular tasks and accelerating HSP70 ATPase activity.
Proteins must be correctly folded to be functional. Unfolded or misfolded proteins expose their hydrophobic segments and are prone to aggregate. Protein aggregates are thought to be toxic to the cell, so to avoid aggregation, abnormal proteins are either kept soluble by molecular chaperones or quickly degraded by the ubiquitin/proteasome system (Hershko and Ciechanover, 1998). Under pathologic conditions, the level of abnormal proteins in the cells may exceed the ability to maintain them in a soluble form or promptly degrade then, resulting in aggregation (Cohen, 1999). Protein aggregates commonly contain ubiquitin immunoreactivity, suggesting that ubiquitinated proteins are components of protein aggregates (Alves-Rodrigues et al., 1998). Protein aggregates have been found in most neurodegenerative diseases (Kakizuka, 1998), forebrain and focal ischemia (Hu et al., 2000, 2001), and hypoglycemic coma (Ouyang and Hu, 2001).
HDJ-2 has been shown to decrease injury in models of degenerative brain disease involving protein aggregation. Cotransfection of HDJ-2 with mutant ataxin-1 resulted in a significant reduction in aggregate formation (Stenoien et al., 1999). In a model of the polyglutamine disease Huntington's disease, overexpression of HDJ-2 suppressed aggregate formation and was associated with decreased toxicity (Jana et al., 2000). No data has yet been presented on the ability of this cochaperone to protect brain cells from ischemia-like injury. In this study, primary cultures of murine cerebral cortical astrocytes were used to study the effect of overexpression of HDJ-2 on cell damage and protein aggregation under experimental conditions simulating ischemia and reperfusion.
MATERIALS AND METHODS
Primary astrocyte cultures were prepared from postnatal day 1 to 3 Swiss Webster mice (Simonsen, Gilroy, CA) as previously described (Dugan et al., 1995;Ouyang et al., 2002). The retroviral vector construct was made by inserting FLAG-HDJ-2, a gift from Dr. Don DeFranco, into the LXSH backbone that also encodes the hygromycin resistance gene for selection of transfected cells. For control infection LXSH without an insert was used. Viral vectors were produced using Ψ-2 packaging cells. Cultures were infected 24 hours after plating by exposure to medium collected from the Ψ-2 packaging cells after filtration and containing 8 μg/ml polybrene (Sigma, St. Louis, MO, U.S.A.) for 12 hours. Astrocytes expressing HDJ-2 or control vector were selected in 125 μg/ml hygromycin (Sigma) for 4 days. The infected astrocytes grew for 2 weeks, during which time they were fed every 5 days with plating medium containing 62.5 μg/ml hygromycin. Cells were used for experiments between 21 to 35 days in vitro.
Expression of HDJ-2 and efficiency of selection were verified by immunoblotting and immunostaining as described previously (Papadopoulos et al., 1996;Ouyang et al., 2000). Briefly, after separation by 12.5% SDS-polyacrylamide (Bio-Rad, Hercules, CA, U.S.A.) gel electrophoresis, proteins were transferred to Immobilon PVDF membranes (Millipore, Bedford, MA, U.S.A.). The gels and membranes were stained with 0.15% Coomassie blue and Ponceau S red solutions, respectively, to ensure equal loading and transfer of protein. The membranes were then blocked overnight, incubated for 2 hours with a 1:1000 dilution of mouse monoclonal anti-HDJ-2 antibody (NeoMarker, Fremont, CA, U.S.A.) or mouse monoclonal anti-FLAG antibody (Sigma) or mouse anti-HSP70 (StressGen Biotechnologies Corp., Victoria, Canada) antibody, washed with 1% bovine serum albumin (BSA) in PBS, and incubated with a horseradish peroxidase-linked anti-mouse antibody (1:2000 dilution; Cell Signaling, Beverly, MA, U.S.A.) for 1 hour. Immunoreactive bands were visualized with the enhanced chemiluminescence detection system (Amersham, Piscataway, NJ, U.S.A.) using Kodak X-AR film (Eastman Kodak, Rochester, NY, U.S.A.). Cultures were immunostained after fixing in methanol: acetone (1:1) at −20°C for 20 minutes, air-drying, PBS rinse, and incubation in a monoclonal mouse anti-FLAG antibody (Sigma) diluted 1:200 for 2 hours at room temperature. The cells were then rinsed and incubated with horseradish peroxidase-linked anti-mouse antibody (Cell Signaling) and diluted 1:250 for 1 hour. After rinsing in PBS, antibody binding was visualized by exposure to 3,3′-diaminobenzidine (DAB) (0.7 mg/ml) (Sigma) and H2O2 (0.2 mg/ml) with urea (Sigma) for 5 minutes. No staining was observed when the primary antibody was omitted.
Fluorescence immunocytochemistry was performed on cell cultures on coverslips as previously described (Ouyang and Giffard, 2003). The cells on the coverslips were fixed with 4% paraformaldehyde for 1 hour and washed twice in phosphate-buffered saline (PBS) for 5 minutes at room temperature (RT) and then in PBS containing 0.2% Triton-X100 (TX100) for 30 minutes. Nonspecific binding sites were blocked in 3% bovine serum albumin (BSA) in PBS/0.2% TX100 for 30 minutes. A monoclonal antiubiquitin antibody that recognizes both free ubiquitin and ubiquitinated proteins was from Chemicon (Temecula, CA, U.S.A.). The cells were incubated with primary antibody (1:500 dilution in PBS/0.1% TX100 and 1% BSA) overnight at 4°C then washed in PBS containing 0.1% TX100 three times at RT. The primary antibody was visualized with fluorescein-labeled anti-mouse secondary antibody. Coverslips were washed several times in PBS/0.1% TX100, mounted on glass slides using Fluoromount-G (Southern Biotechnology Associates, INC., Birmingham, AL, U.S.A.) and observed with an epifluorescence microscope (Nikon Diaphot, Nikon Corporation, Tokyo, Japan).
Injury paradigms of glucose deprivation (GD) or oxygen glucose deprivation (OGD) were carried out in balanced salt solution as previously described (Papadopoulos et al., 1996;Xu and Giffard, 1997). Astrocyte injury was evaluated morphologically using phase-contrast light microscopy. Cell survival was quantitated after Trypan blue staining by cell counting. Trypan blue (Sigma) (5%) was added to culture wells, and stained (dead) and unstained (live) cells were counted in five high-power (200×) fields per culture. The extent of cell death was expressed as the number of Trypan blue-positive cells as a percent of the total number of cells counted. At least 200 cells were counted per culture.
Differences between groups were analyzed by ANOVA followed by Student-Newman-Keuls for multiple comparisons using Sigmastat (SPSS, Chicago, IL, U.S.A.). Data are presented as mean + SD, with n = 9 cultures. Significance was established at the P < 0.05 level.
RESULTS AND DISCUSSION
HDJ-2 expression was verified by immunostaining, which allowed assessment of individual cellular expression of HDJ-2 and demonstrated that after selection essentially all of the cells express the gene of interest (Fig. 1A). Expression of HDJ-2 was further confirmed by western blot, using antibody against HDJ-2. The HDJ-2 protein band, from HDJ-2 infected astrocytes, appeared at the expected 45 kDa position on the membrane (Fig. 1B). We investigated the possibility that HDJ-2 overexpression might affect the level of HSP70 in the cell by western and found that HSP70 levels were not changed by HDJ-2 overexpression (Fig. 1C).
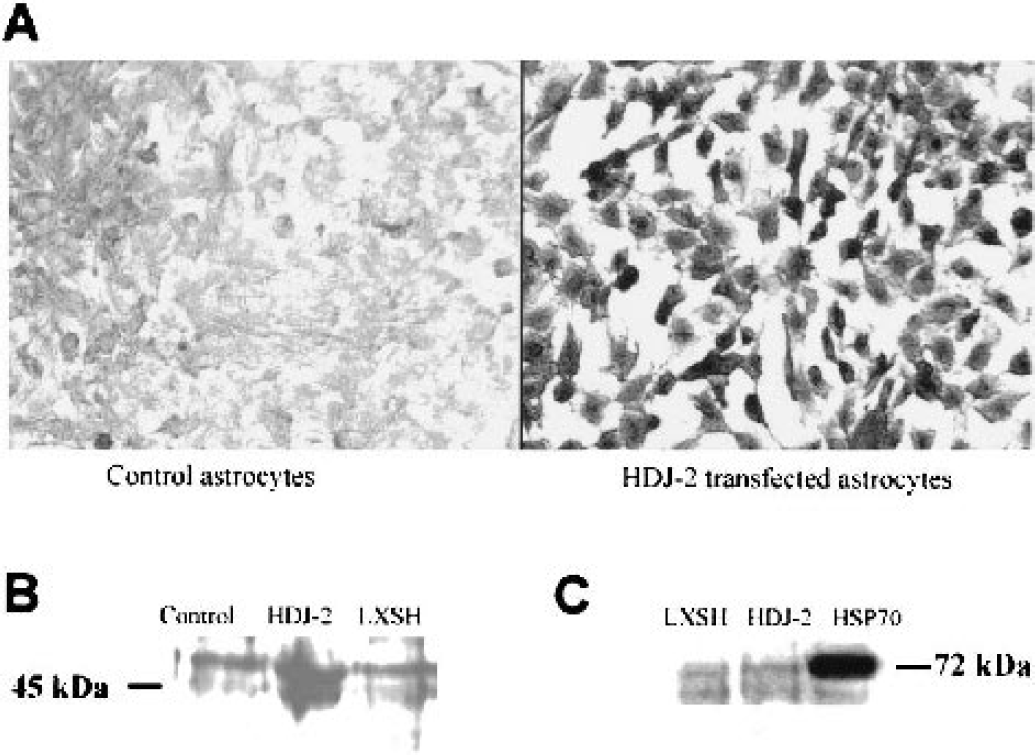
Expression of HDJ-2 in astrocytes by immunohistochemical staining and immunoblotting.
The effect of HDJ-2 overexpression on astrocyte survival in response to ischemia-like injuries was then tested. Astrocytes overexpressing HDJ-2 showed significant protection from 8 hours OGD injury (25.3 ± 2.9% cell death) compared with astrocytes expressing LXSH vector (70.7 ± 5.5%) or uninfected astrocytes (93.5 ± 1.1%) assessed by cell counting (n = 9, P < 0.05) (Fig. 2). Similarly, after 24 hours GD astrocytes overexpressing HDJ-2 showed significantly less cell death (21.5 ± 1.7%) than astrocytes expressing control LXSH vector (67.4 ± 7.1%) or uninfected astrocytes (73.0 ± 6.2%) when assessed by Trypan blue staining and cell counting (n = 9, P < 0.05) (Fig. 2). Morphologically after 24 hours GD cell shrinkage was evident, and this was inhibited by HDJ-2 overexpression (data not shown).
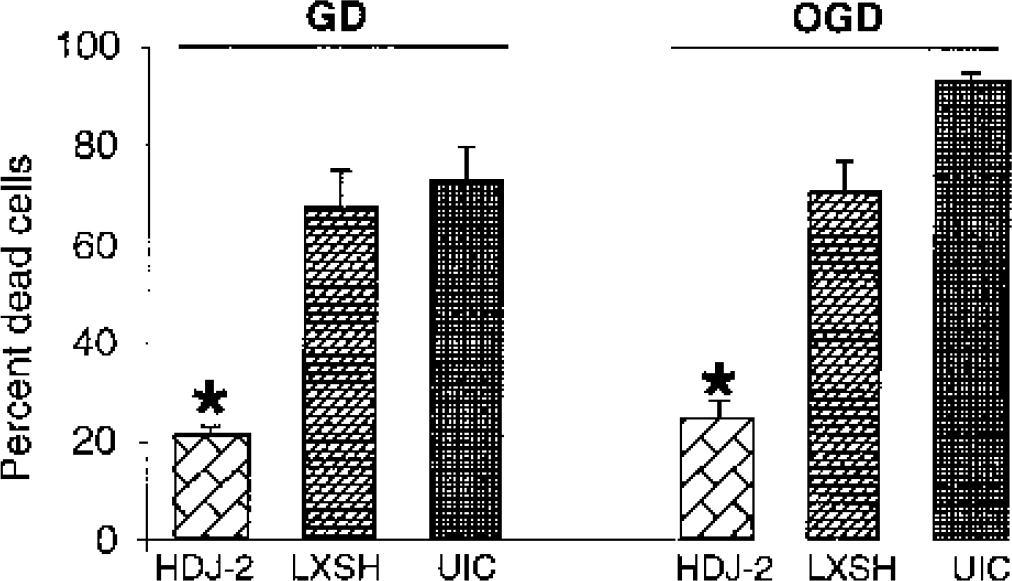
The effect of HDJ-2 overexpression on astrocyte injury. The first three bars show the extent of injury of cultures subjected to glucose deprivation (GD) for 24 hours. HDJ-2 provides significant reduction in cell death. The second three bars show injury due to 8-hour oxygen-glucose deprivation (OGD) followed by reoxygenation and reperfusion for 16 hours. HDJ-2 overexpression again markedly reduces injury. ∗ P < 0.05 compared with LXSH-expressing and uninfected controls (UIC).
Astrocytes from controls and cells allowed to recover for 16 hours after 8 hours of GD were labeled with a monoclonal antiubiquitin antibody (Fig. 3). Ubiquitin immunolabeling was evenly distributed in both the nucleus and cytoplasm of control astrocytes (Fig. 3A). After GD, however, the immunolabeling pattern clearly changed from a relatively even distribution to one of heterogeneous, clumped aggregates. The antiubiquitin positive aggregates of various sizes were observed scattered around the nucleus and a marked reduction of nuclear staining was seen in most of the cells (Fig. 3B). To quantify this effect, we counted cells that showed a reduction in nuclear staining compared with cytosolic staining and the presence of ubiquitin stained clumps as a percent of the total number of cells. When HDJ-2 was overexpressed, the number of cells with evidence of protein aggregation was significantly reduced (Fig. 3C).
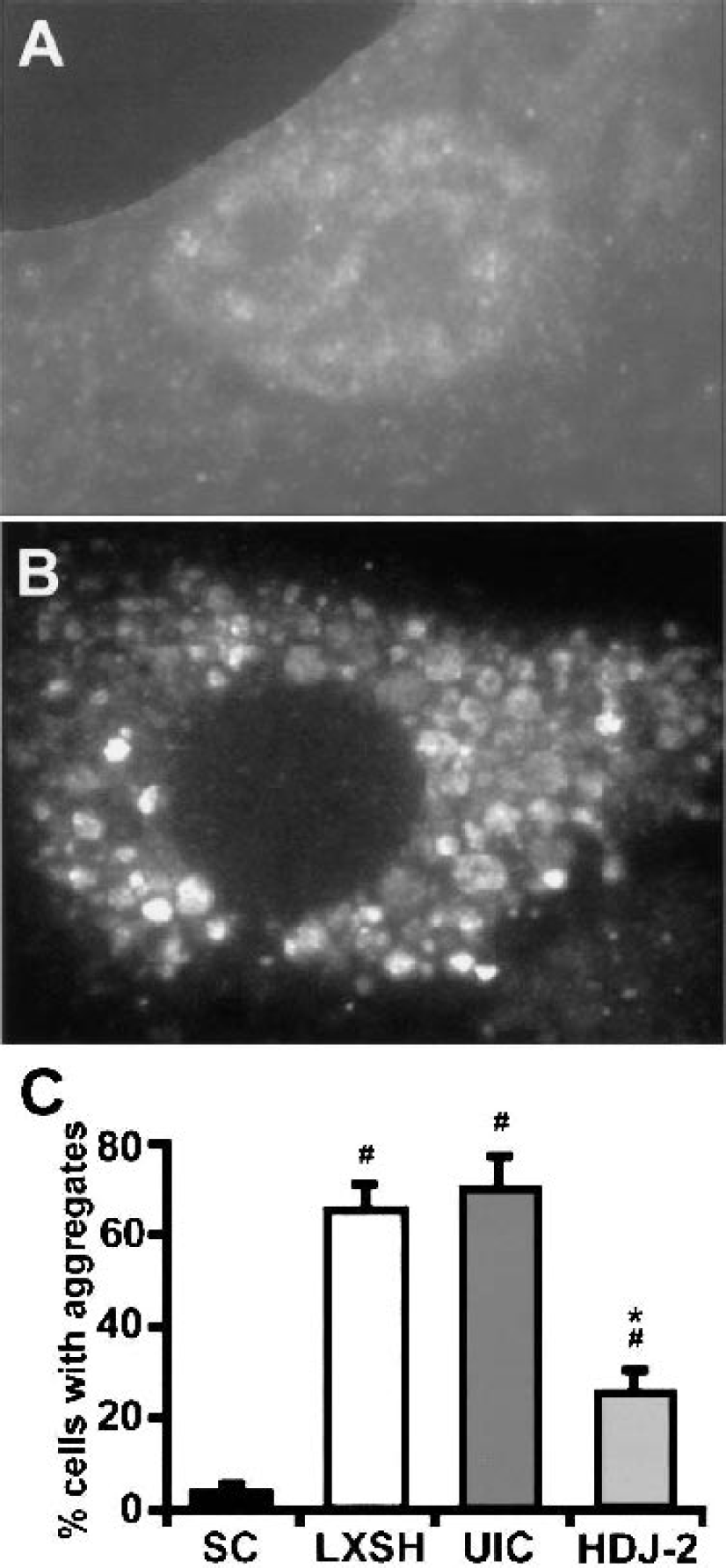
Effect of HDJ-2 overexpression on protein aggregation. Photomicrographs of astrocytes labeled with anti-ubiquitin from untreated control
It has been shown that overexpression of HSP70 by either viral or transgenic overexpression (Plumier et al., 1997;Yenari et al., 1998) before ischemia in vivo protects neurons from injury. The protection may be mediated by refolding denatured proteins, by preventing unfolded and damaged proteins from aggregating, or by a direct antiapoptotic mechanism. HDJ-2 is a cochaperone that regulates the activity of members of the DnaK/HSP70 family and has been shown to protect against several models of neurodegenerative diseases in which protein aggregation is thought to play a role (Chai et al., 1999;Jana et al., 2000). The range of functions of HDJ-2 described to date is more limited than those known for HSP70. Results obtained from this study now demonstrate for the first time that overexpression of HDJ-2 in cultured astrocytes provides marked protection from ischemia-like injuries, reducing injury by two-thirds, and providing some evidence that blocking protein aggregation may be an important mechanism for this protection. This helps us narrow down the functions that may be involved in this kind of brain cell protection.
The J domain of HDJ-2 is the region of the protein responsible for mediating the binding of HDJ-2 to HSP70 (Hartl, 1996). HDJ-2 binds to and stimulates the ATPase activity of HSP70, thereby enhancing the chaperone function of HSP70 (Hartl, 1996). The fact that HDJ-2 is still able to suppress aggregation when the J domain is deleted suggests that this suppression occurs independent of interactions with HSP70 (Chai et al., 1999). It has been suggested that HDJ-2 alone can bind misfolded proteins, suppress aggregation, and facilitate delivery of misfolded polypeptides to the cellular machinery for proteolytic degradation (Stenoien et al., 1999). It has been speculated that the chaperone function of HDJ-2 in the brain may be one of the factors responsible for the relative resistance of brain cells to damage (Miller et al., 1990;Jana et al., 2000). Because HSP70 levels are not increased in parallel with HDJ-2, the protection observed in this study is likely to reflect the direct effects of HDJ-2, possibly by inhibiting protein aggregation. However, we cannot rule out a contribution from modulation of the function of the HSP70, which is present in the cells. It will be interesting to test deletion mutants of HDJ-2 in future experiments to identify the domains required for protection from ischemia-like injury. Overexpression of HDJ-2 should now be tested against in vivo cerebral ischemia. HDJ-2 represents a new approach for brain protection from ischemia as well as other neurodegenerative diseases.
Footnotes
Acknowledgment
Full-length human FLAG-HDJ-2 was a gift from Dr. Don DeFranco, University of Pittsburgh, Pennsylvania, U.S.A. The authors thank Beth Hoyte for help with preparation of the figures.
