Abstract
This study examined whether neuronal hemoglobin (Hb) is present in rats. It then examined whether cerebral ischemia or ischemic preconditioning (IPC) affects neuronal Hb levels in vivo and in vitro. In vivo, male Sprague-Dawley rats were subjected to either 15 mins of transient middle cerebral artery occlusion (MCAO) with 24 h of reperfusion, an IPC stimulus, or 24 h of permanent MCAO (pMCAO), or IPC followed 3 days later by 24 h of pMCAO. In vitro, primary cultured neurons were exposed to 2 h of oxygen—glucose deprivation (OGD) with 22 h of reoxygenation. Results showed that Hb is widely expressed in rat cerebral neurons but not astrocytes. Hemoglobin expression was significantly upregulated in the ipsilateral caudate and the cortical core of the middle cerebral artery territory after IPC. Hemoglobin levels also increased more in the penumbral cortex and the contralateral hemisphere 24 h after pMCAO, but expressions in the ipsilateral caudate and the cortical core area were decreased. Ischemic preconditioning modified pMCAO-induced brain Hb changes. Neuronal Hb levels in vitro were increased by 2 h of OGD and 22 h of reoxygenation. These results indicate that Hb is synthesized in neurons and can be upregulated by ischemia.
Introduction
Hemoglobins (Hbs) are heme proteins widely distributed in diverse organisms, including bacteria, fungi, protists, plants and animals. They carry out many different functions, including transport of oxygen between tissues, intracellular oxygen transport and catalysis of redox reactions (Hardison, 1998). In vertebrates, Hb is a heterotetramer of two α-globin and two β-globin subunits. Each subunit contains a hydrophobic pocket in which a heme molecule binds tightly and allows for the reversible binding of oxygen (Poyart et al, 1992). Vertebrate Hb is usually considered as an important oxygen transport protein mainly present in erythrocytes circulating in the blood. However, recent studies have showed that Hb is expressed in neurons of embryonic and adult brain (Ohyagi et al, 1994). Hemoglobin expression has also been found in the hippocampus and in the cerebellum (Blalock et al, 2003; Slemmon et al, 1994; Wu et al, 2004).
The presence of neuronal Hb and its high affinity for oxygen indicate it may have an important role in regulating oxygen hemostasis in neurons, which may be important under conditions of cerebral ischemia or neuronal hypoxia. Conversely, it has been known that free Hb is deleterious to the brain after intracerebral hemorrhage and brain trauma (Xi et al, 2006). Exogenous Hb can induce cell death in rat cerebral cortical neurons (Wang et al, 2002). Our earlier studies have shown that intracerebral infusion of Hb and its degradation products cause brain damage (Hua et al, 2003; Huang et al, 2002). To date, little is known about the response of endogenous neuronal Hb to cerebral ischemia.
In this study, Hb expression was examined after 15 mins of transient middle cerebral artery occlusion (MCAO), an ischemic preconditioning (IPC) stimulus (Masada et al, 2001), permanent MCAO (pMCAO) and IPC followed by pMCAO in the rat. The effects of oxygen—glucose deprivation (OGD) on neuronal Hb levels were also examined in vitro.
Materials and methods
Animal Preparation and Middle Cerebral Artery Occlusion
The protocols for these animal studies were approved by the Committee of University Michigan in the Animals Use and Care. Adult male Sprague-Dawley rats (275 to 325 g; Charles River Laboratories, Portage, MI, USA) were used. Rats underwent surgery after an overnight fast with free access to water. Anesthesia was induced with 5% isoflurane. After the animals were intubated, anesthesia was maintained by 2.25% isoflurane through mechanical ventilation with a rodent ventilator model 683 (Harvard Apparatus Inc., St Natick, MA, USA). A polyethylene catheter (PE-50) was then inserted into the femoral artery to monitor arterial blood pressure and to obtain blood samples for analysis of pH, PaO2, PaCO2, hematocrit and blood glucose concentrations before cerebral ischemia. Body temperature was maintained at 37.5°C by using a feedback-controlled heating pad.
Middle cerebral artery occlusion was induced by insertion of a 3-0 monofilament nylon suture via the left common carotid artery as described earlier (Karabiyikoglu et al, 2004). The suture was gently advanced into the internal carotid approximately 19 or 20 mm from the bifurcation to occlude the middle cerebral artery. Cortical blood flow of rats was measured in the core area of middle cerebral artery territory by a laser-Doppler flowmeter (Laserflo BMP2; Vasamedics Inc., Little Canada, MN, USA) before the onset of ischemia and during the ischemia to confirm that blood flow reached the ischemic level. The sham operation was identical except that the intraluminal suture was not inserted.
Experimental Groups
The in vivo study was divided into three parts. In the first part, rats were subjected to 15 mins of transient MCAO, an IPC stimulus or sham operation on the left side (n=6, each group) and were killed 24 h later for western blot analysis and real-time polymerase chain reaction (PCR). This duration of transient MCAO has been shown to induce IPC in rats (Masada et al, 2001). In the second part, rats underwent a pMCAO to induce brain injury or a sham operation and were killed 24 h later for immunohistochemistry, Western blot analysis and real-time PCR (n=10, each group). In the third part, rats underwent a further pMCAO 3 days after IPC or sham operation (n=7, each group). The rats were killed 24 h later for western blotting and real-time PCR. In addition, three rats were used for brain Hb determination.
Cell Culture
Primary neuronal cultures were obtained from embryonic day-17 Sprague-Dawley rats (Charles River Laboratories). Cultures were prepared according to an earlier described procedure with some modifications (Jiang et al, 2002). Briefly, cerebral cortex was dissected, stripped of meninges and dissociated by a combination of 0.5% trypsin digestion and mechanical trituration. The dissociated cell suspensions were seeded into poly-
Astrocyte cultures were prepared from the brains of 1- to 3-day-old Sprague-Dawley rat pups (McCarthy and de Vellis, 1980) with some modifications. Cerebral cortex was isolated, meninges were removed and the tissue was incubated in 0.5% trypsin for 20 mins at 37°C. After digestion, the tissue was rinsed twice in Hank's buffered salt solution, followed by a mechanical dissociation in Dulbecco's modified Eagle's medium (DMEM, 10% fetal calf serum, 0.5 mmol/L glutamine and 2% Antibiotic—Antimycotic). The cells were plated at a density of 6 × 100,000/cm2 on precoated poly-
Oxygen—Glucose Deprivation
Primary cultured neurons were deprived of oxygen and glucose for 2 h for ischemic injury in vitro as described earlier (Andjelkovic et al, 2003). The culture medium was removed. Cells were washed twice with phosphate-buffered saline (PBS) (pH 7.4) and then placed in OGD medium (DMEM without glucose, gassed with 85% N2, 10% H2 and 5% CO2 for 30 mins). Cells were maintained in an anaerobic chamber (Coy Laboratory, Grass Lake, MI, USA) containing a gas mixture composed of 85% N2, 10% H2 and 5% CO2 at 37°C for 2 h. Anaerobic conditions in the chamber were monitored using an oxygen sensor (Teledyne Analytical Instruments, Indusery, CA, USA). Oxygen—glucose deprivation was terminated by removing cells from the chamber, followed by replenishing with serum-free DMEM solution and placing them back into the normoxic incubator for 22 h. Control cell cultures were maintained in the normoxic incubator for 24 h with serum-free DMEM solution.
Western Blot Analysis
Rats were anesthetized with pentobarbital (60 mg/kg, intraperitoneally) and underwent intracardiac perfusion with 0.1 mol/L PBS (pH 7.4) until the outflow fluid from the right atrium was colorless. The brains were removed quickly from the skull and divided into two hemispheres. The caudate was sampled from the ipsilateral and contralateral hemispheres. The cortices were then sampled using 5 and 7mm cork borers (Masada et al, 2001). A cortical core sample was taken from the tissue underlying the middle cerebral artery immediately distal to the region of occlusion using the 5-mm borer. Later, a ring of tissue surrounding the core was taken with the 7-mm borer. The tissue was called the ‘intermediate’ sample and it represents, in the ipsilateral hemisphere, less densely ischemic tissue than the ‘core’ sample. Samples were frozen in liquid nitrogen.
In vitro, cell medium was removed and the plates were washed three times with chilled PBS. The cells were quickly scraped and collected by centrifugation at 4°C and then stored at −80°C.
Western blot analysis was performed as described earlier (Xi et al, 1999). Brain and cell samples were sonicated with Western blot lysis buffer. Protein concentration was determined using a Bio-Rad protein assay kit (Hercules, CA, USA). Equal amounts of protein from each sample were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a Hybond-C pure nitrocellulose membrane (Amersham, Piscataway, NJ, USA). Membranes were blocked in Carnation nonfat milk and probed with the primary and secondary antibodies. The primary antibodies were polyclonal rabbit anti-hemoglobin antibody (1:1,000 dilution; Cappel, MP Biomedics Inc., Solon, OH, USA). The second antibody was goat anti-rabbit IgG (1:2,500 dilution; Bio-Rad). The antigen—antibody complexes were observed with the ECL system (Amersham) and exposed to Kodak X-OMAT film. The membranes were then stripped and reprobed with antibody against β-actin (1:6,000 dilution; Sigma, St Louis, MO, USA). The relative densities of bands were analyzed with the NIH Image J.
Immunohistochemistry
For the in vivo models, rats were reanesthetized with pentobarbital (60 mg/kg, intraperitoneally) and underwent intracardiac perfusion with 4% paraformaldehyde in 0.1 mol/L PBS (pH 7.4). The brains were removed and kept in 4% paraformaldehyde overnight, and then immersed in 30% sucrose for 3 to 4 days at 4°C. Specimens were then placed in embedding compound and sectioned (18 μm thickness) on a cryostat. For the in vitro studies, the culture medium was removed and cells were washed three times with chilled PBS. The cells were fixed with 4% paraformaldehyde in 0.1 mol/L PBS (pH 7.4) for 30 mins. Immunostaining for brain sections and cultured cells was performed according to an earlier described method (Xi et al, 1999). The primary antibody was polyclonal rabbit anti-hemoglobin antibody (1:400 dilution; Cappel, MP Biomedics Inc.). Rhodamine-conjugated goat anti-rabbit antibody (1:600 dilution; Chemicon International Inc., Temecula, CA, USA) was used as a second antibody. Normal rabbit immunoglobulin G was used as a negative control.
For immunofluorescent double labeling, the primary antibodies were rabbit anti-hemoglobin (1:100 dilution; Cappel, MP Biomedics Inc.), mouse anti-glial fibrillary acid protein (1:100 dilution; Chemicon International Inc.) and mouse anti-rat neuronal nuclei (1:100 dilution; Chemicon International Inc.). Rhodamine-conjugated goat anti-rabbit antibody (1:100 dilution; Chemicon International Inc.) and fluorescein isothiocyanate-labeled horse anti-mouse antibody (1:100 dilution; Vector Laboratories, Burlingame, CA, USA) were used as second antibodies. The double labeling was analyzed using fluorescence microscope.
Real-Time Quantitative Polymerase Chain Reaction
For the in vivo studies, rats were anesthetized and decapitated. The brain tissue was sampled as described for western blot analysis. For the cells, the medium was removed and plates were washed three times with chilled PBS. The cells were quickly scraped and collected by centrifugation at 4°C and then stored at −80°C. Total RNA was extracted from the frozen brain tissue and cultured cells with Trizol reagent (Gibco BRL, Grand Island, NY, USA), and 1 μg RNA was digested with deoxyribonuclease I (amplification grade; Gibco BRL). Complimentary DNA was synthesized by reverse transcription using the digested 1-μg RNA (11 μL) with 14 μL reaction buffer (Perkin Elmer, Foster City, CA, USA) containing dNTP (dATP, dCTP, dGTP and dTTP), 25 mmol/L MgCl2, 10 × polymerase chain reaction buffer II, Random Hexamer Primer, RNase inhibitor and murine leukemia virus (MuL V) reverse transcriptase. The reaction was performed at 42°C for 30 mins and terminated at 99°C after 5 mins. Diethyl pyrocarbonate water (75 μL) was added to dilute the complimentary DNA to 100 μL and stored at −20°C for later use.
Real-time quantitative PCR was performed with SYBR green as a double-strand DNA-specific dye in Eppendorf Mastercycler ep realplex (Eppendorf North America Inc., Westbury, NY, USA). The primers for rat hemoglobin α-chain (HbA) and hemoglobin β-chain (HbB) were designed from known sequences of rat HbA mRNA (GenBank no. U29528) and HbB mRNA (GenBank No. NM_033234) searched by PrimerQuest (Integrated DNA Technologies Inc., Coralville, IA, USA). The primers were rat HbA 5′-TGATCCACTTCCTTCTCTGCCCAA-3′ (forward primer) and 5′-ATCAGTTGCCCAAGTGCTTCTTGC-3′ (reverse primer) and HbB 5′-ATGGCCTGAAACACTTGGACAACC-3′ (forward primer) and 5′-TGGTGGCCCAAC-ACAATCACAATC-3′ (reverse primer). Rat GAPDH primers were 5′-CCGTGCCAAGATGAAATTGGCTGT-3′ (forward) and 5′-TGTGCATATGTGCGTGTGTGTGTG-3′ (reverse) as a housekeeping gene served as a control. Polymerase chain reaction was run in triplicate on 96-well plate with a total volume of 20 μL per well using 2.5 × SYBR® Green universal master mix. Cycling conditions were 2 mins at 95°C, 30 secs at 95°C, 30 secs at 60°C, 1 min at 72°C, 40 cycles and a melting curve program (60°C to 95°C with warming of 1.75°C per minute). The relative quantification analysis module was used to compare expression levels of a target gene. The expression levels were calculated by using the ΔΔCT method (Livak and Schmittgen, 2001). Using this method, we obtained value equal to 1 when no change in relative expression occurred between untreated and treated samples. We defined overexpression when 2−ΔΔCT >1 and underexpression when 2−ΔΔCT <1.
Sequencing of the Polymerase Chain Reaction Products
Polymerase chain reaction products were isolated by 2% agarose gel electrophoresis. Gels were observed with ethidium bromide staining and ultraviolet transillumination. The DNA fragments of interest were excised and then purified with an Agarose Gel DNA Extraction kit (Roche, Diagnostics GmbH, Mannheim, Germany). Purified DNA fragments were sequenced. All sequencing was performed on Applied Biosystems DNA Sequencers (Model 3730 XL, Foster City, CA, USA). Sequence data were analyzed using the nucleotide blast at National Center for Biotechnology Information.
Spectrophotometric Assay of Hemoglobin
Brain Hb content was quantified by spectrophotometric assay (Choudhri et al, 1997). Normal rats were perfused transcardially with 0.1 mol/L PBS under deep anesthesia until the outflow fluid from the right atrium was colorless. Brains were rapidly removed and dissected into two parts, the cortex and the basal ganglia. The tissues were weighed on an electronic balance and then homogenized in 0.1 mol/L PBS, followed by 30-min centrifugation (13,000 g). Later, 200 μL reagent (QuantiChrom™ Hemoglobin Assay Kit, BioAssay Systems, Hayward, CA, USA) was added to 50 μL supernatant. After 15 mins, optical density was measured at 400 nm with a spectrophotometer. Hemoglobin content was expressed as microgram per gram of wet brain tissue.
Statistical Analysis
All data in this study are presented as mean±s.d. Data were analyzed with Student's t-test and analysis of variance, or Mann—Whitney U-rank test. Statistical significance was set at 0.05.
Results
Physiologic variables including blood pH, PaO2, PaCO2, glucose, hematocrit and blood pressure were measured before the onset of MCAO and controlled in normal range. The combined mean physiologic variables are shown in Table 1. Hemoglobin levels in normal rat brains were 161±16 μg/g in the cortex and 240±19 μg/g in the basal ganglia.
Physiological parameters
IPC, ischemic preconditioning; MABP, monitor arterial blood pressure; MCAO, middle cerebral artery occlusion.
Values are mean±s.d.
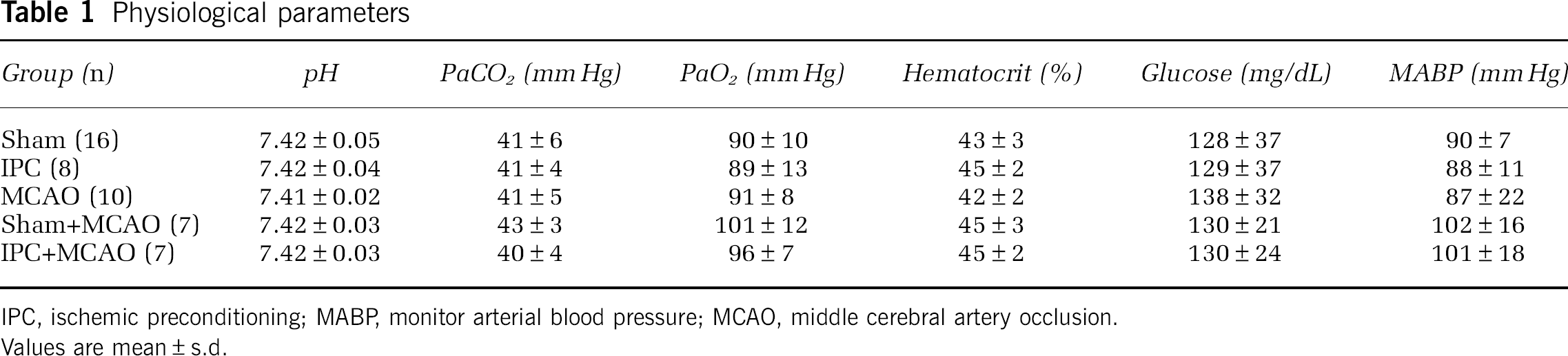
Hemoglobin Expression in Neurons
Immunohistochemistry revealed that Hb was widely expressed in cerebral cortex, with lower levels in the caudate. Most Hb-positive cells appeared to be neurons and Hb immunoreactivity was primarily localized within the cytoplasm and dendrites. Negative control sections showed no staining. Double labeling showed that Hb-positive cells were colocalized with neuronal nuclei-positive cells, but not glial fibrillary acid protein-positive cells (Figure 1A). Western blot analysis of primary cultured cerebral neurons showed one Hb band around 10 kDa and another band around 30 kDa. Hemoglobin protein levels were significantly higher in primary cultured neurons than those in the primary cultured astrocytes (Figure 1B).
(
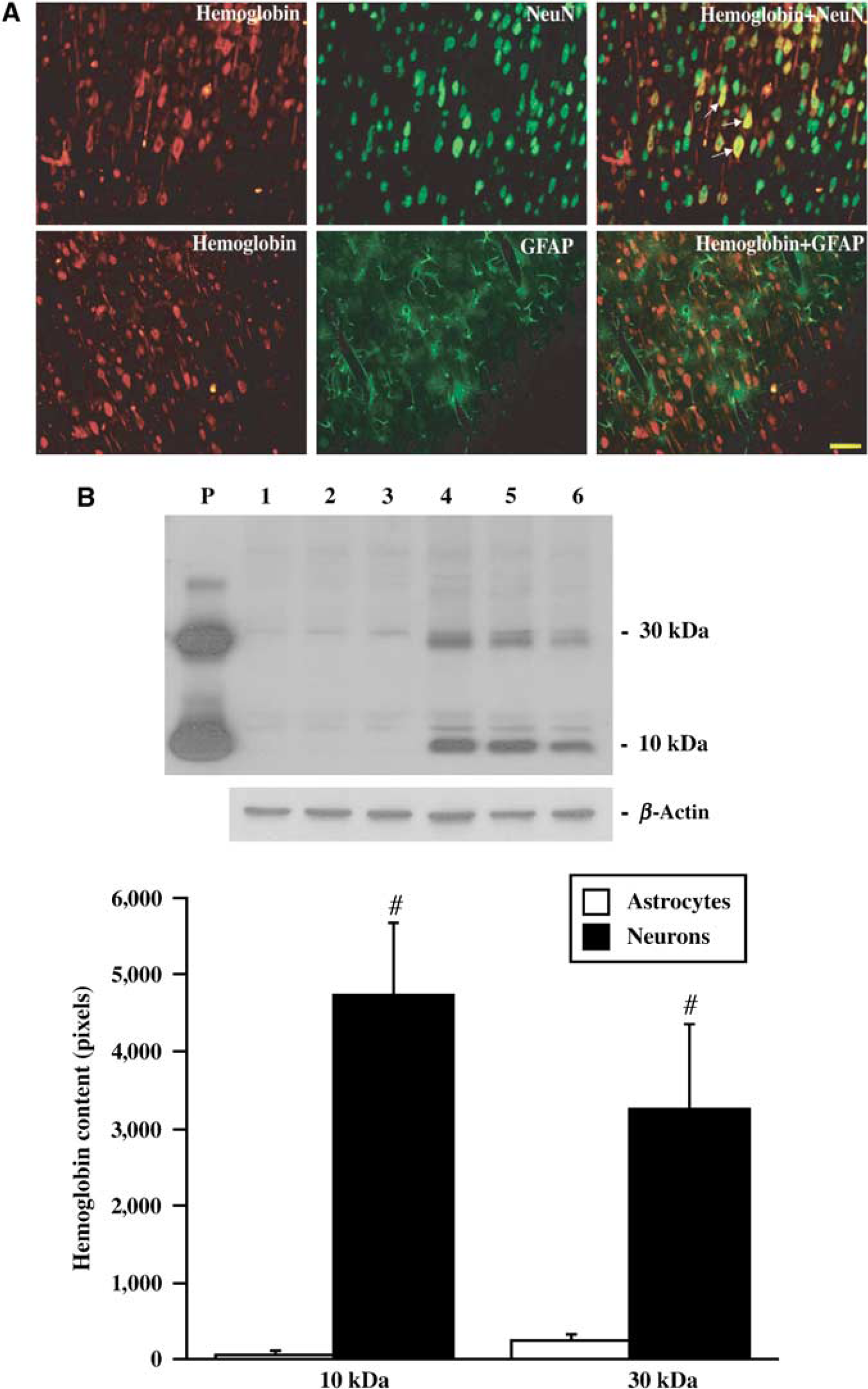
Ischemic Preconditioning-Induced Hemoglobin mRNA and Protein Upregulation
Western blot analysis showed that the Hb protein levels were higher in the ipsilateral caudate and the ipsilateral cortical core sample 24 h after 15 mins of IPC compared with both the contralateral side and the ipsilateral side of sham-operated rats (Figure 2). To further confirm the synthesis of Hb within the brain, quantitative real-time PCR was performed on rat brains subjected to IPC or sham operation. There was a 1.8±0.9-fold increase in HbA mRNA in the ipsilateral caudate and 2.6±1.2-fold increase in the ipsilateral cortical core sample in IPC-treated rats compared with shams (P<0.05). Hemoglobin β-chain mRNA was also upregulated by 27.9±8.4 times in the ipsilateral caudate and 4.8±2.3 times in the ipsilateral cortical core compared with shams (P<0.05).
Western blot analysis showing Hb levels in the caudate (
Permanent Middle Cerebral Artery Occlusion-Induced Hemoglobin Expression
To test the effect of ischemic injury on Hb expression, rats were subjected to 24 h of pMCAO. Immunohistochemistry staining was performed on brain sections and Hb-positive cells were counted in six sites, the ipsilateral and contralateral caudate, and cortical core and intermediate zones. The number of Hb-positive cells in the ipsilateral intermediate zone of rats that underwent pMCAO was increased compared with shams (P<0.05, Figure 3A). There were more Hb-positive cells in the contralateral caudate, and cortical core and intermediate zones in pMCAO rats than in shams. However, Hb-positive cells in the ipsilateral caudate and cortical core were less in pMCAO rats than those in shams (Figure 3A).
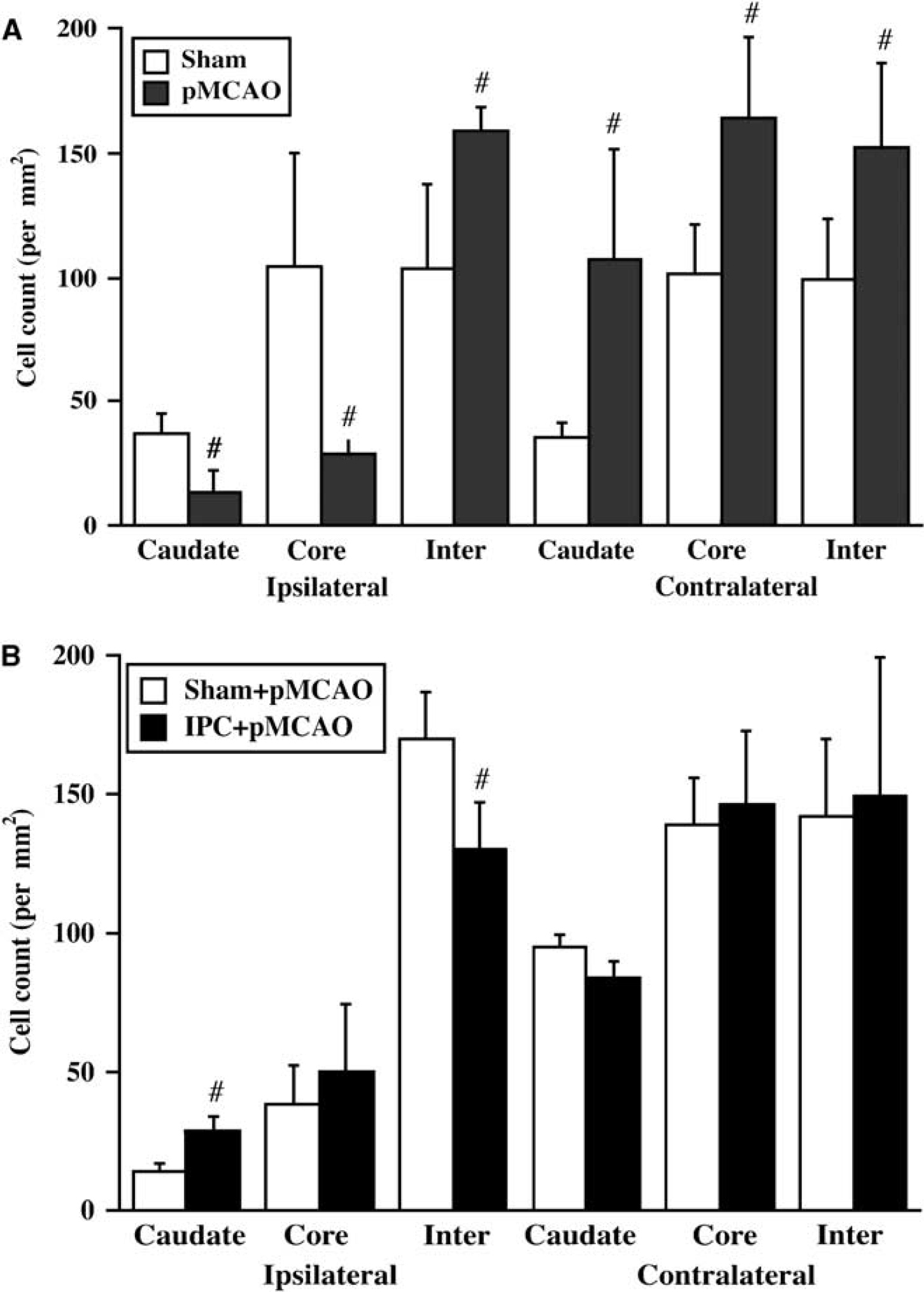
(
By quantitative real-time PCR, there was a 3.2±2.4-fold increase in HbA mRNA in the ipsilateral intermediate zone of MCAO-subjected rats compared with shams (P<0.05, Table 2). In contrast, a decreased expression of HbA mRNA was observed in the ipsilateral caudate and cortical core 24 h after MCAO (P<0.05, Table 2). There were no significant differences in HbA mRNA levels in the contralateral caudate, cortical core and intermediate zones between MCAO-subjected and shams. Changes in HbB mRNA expression 24 h after pMCAO were similar to those for HbA (Table 2). To confirm the PCR products were HbA and HbB, the products were sequenced. Polymerase chain reaction products were HbA and HbB rather than neuroglobin.
Fold changes in HbA and HbB mRNA after 24 h of permanent MCAO
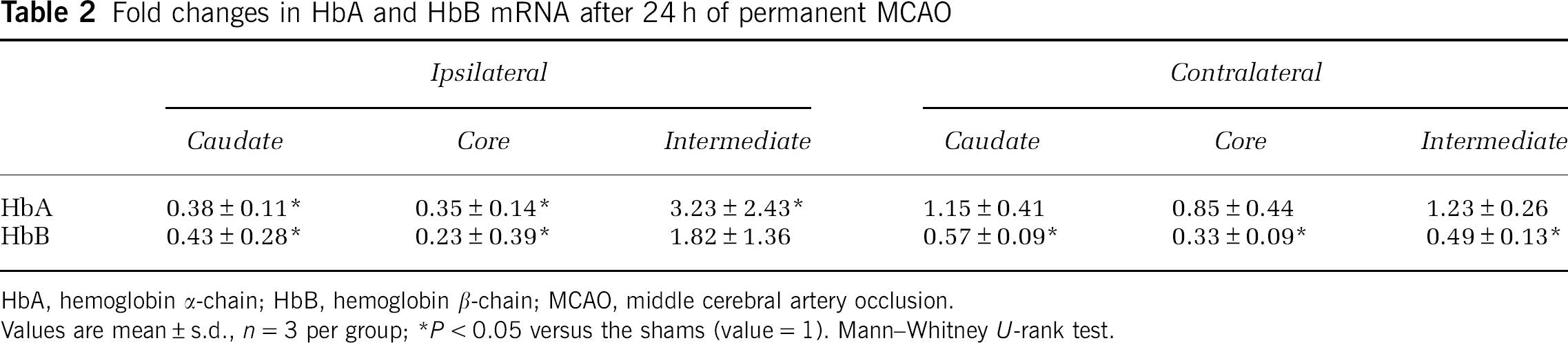
HbA, hemoglobin α-chain; HbB, hemoglobin β-chain; MCAO, middle cerebral artery occlusion.
Values are mean±s.d., n=3 per group; ∗P<0.05 versus the shams (value=1). Mann—Whitney U-rank test.
Effects of Ischemic Preconditioning on Permanent Middle Cerebral Artery Occlusion-Induced Hemoglobin Expression
To investigate the effects of IPC on Hb expression after pMCAO, rats underwent pMCAO 3 days after 15 mins of IPC or sham operation and were killed 24 h after pMCAO. The rat brains were used for immunohistochemistry analysis and quantitative real-time PCR. The number of Hb-positive cells in the ipsilateral caudate was significantly increased in IPC-treated rats (P<0.05, Figure 3B). In contrast, IPC-treated rat had fewer Hb-positive cells in the ipsilateral intermediate zone than those in shams (P<0.05, Figure 3B). There was similar number of Hb-positive cells in the contralateral side in these two groups (Figure 3B).
HbA mRNA levels in the ipsilateral caudate in IPC-treated rats were 2.4±1.4 folds higher than in sham-operated rats undergoing pMCAO (P<0.05, Table 3). Hemoglobin β-chain mRNA expressions in the ipsilateral caudate and cortical intermediate zone were also increased by 1.9±0.6 and 47.3±2.9-fold, respectively (P<0.05, Table 3). However, IPC-treated rats had lower HbB mRNA levels in the contralateral caudate and cortex core than those in shams (P<0.05, Table 3).
Fold changes of HbA and HbB mRNA after 24 h of permanent MCAO in IPC-treated rats
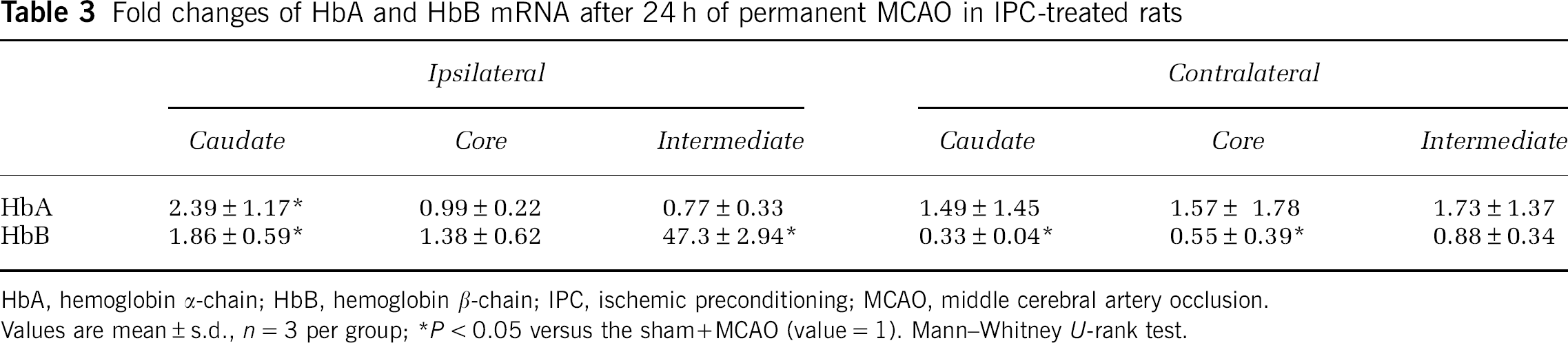
HbA, hemoglobin α-chain; HbB, hemoglobin β-chain; IPC, ischemic preconditioning; MCAO, middle cerebral artery occlusion.
Values are mean±s.d., n=3 per group; ∗P<0.05 versus the sham+MCAO (value=1). Mann—Whitney U-rank test.
Oxygen—Glucose Deprivation-Induced Hemoglobin Expression in Cultured Neurons
To investigate whether Hb expression is induced by ischemic-like conditions in vitro, primary cultured neurons were deprived of oxygen and glucose for 2 h followed by 22 h reoxygenation. Western blot analysis showed that OGD-treated cells had markedly higher Hb protein levels than those in controls (Figure 4A). Immunohistochemistry indicated that Hb immunoreactivity was localized in the cytoplasm and dendrites of cultured neurons (Figures 4B and 4C). Quantitative real-time PCR showed a 1.9±0.4-fold increase in HbA mRNA in OGD-treated cells compared with controls (P<0.05), whereas HbB mRNA expression was decreased to 0.3±0.1 times in the control (P<0.05).
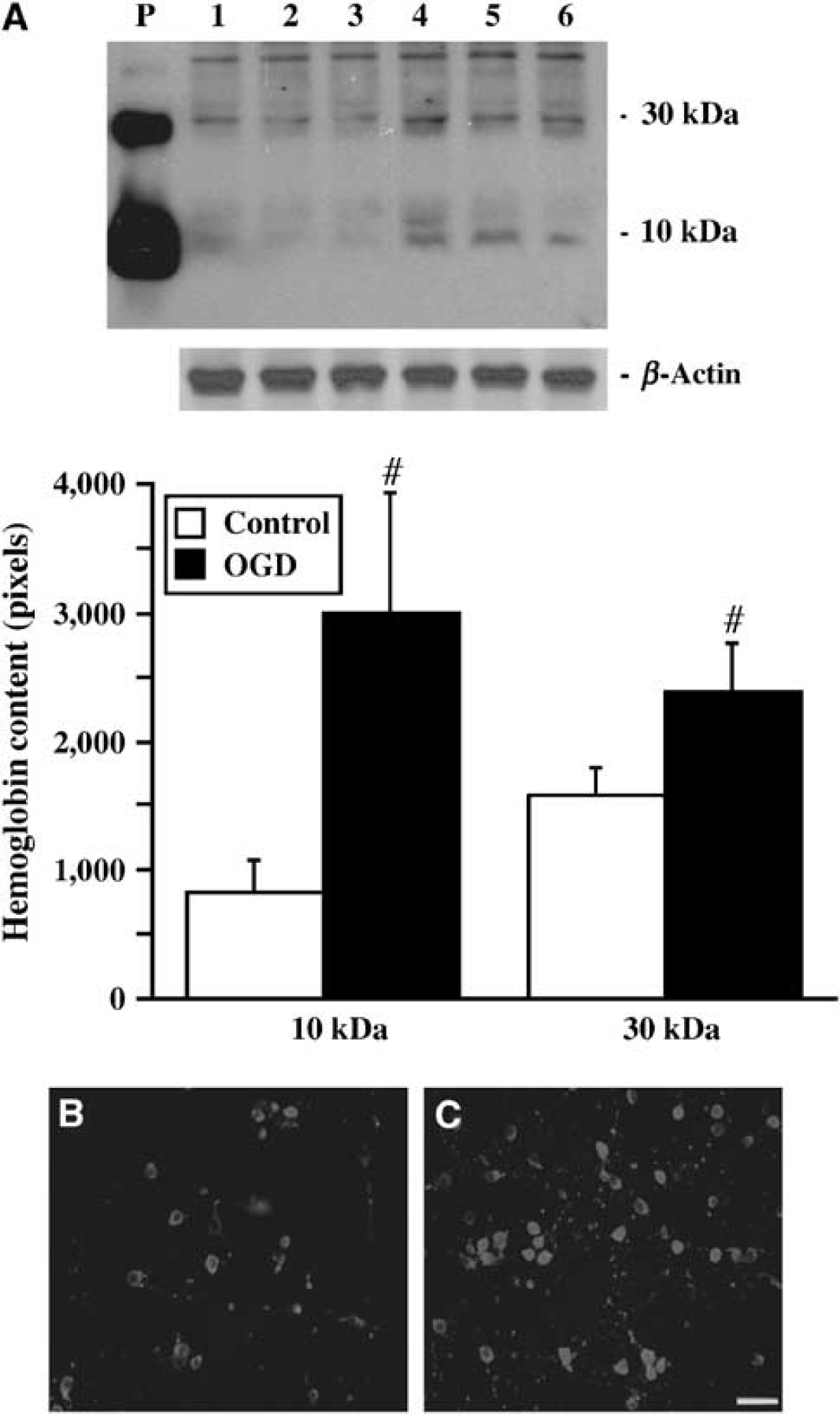
(
Discussion
The main findings of our study were that (1) Hb was mainly expressed in cerebral neurons in vivo and in vitro; (2) Hb protein and mRNA levels were significantly upregulated in the ipsilateral caudate and cortical core samples after IPC; (3) Hb was increased in the ischemic penumbra (intermediate zone) and nonischemic hemisphere after 24 h of pMCAO, but it was decreased in ischemic core areas; (4) Hb was increased in neurons after exposed to OGD for 2 h with 22 h of reoxygenation; and (5) IPC reduced pMCAO-induced Hb downregulation. These results indicate that neurons can synthesize Hb, which can be upregulated by ischemia. Ischemic preconditioning-induced Hb upregulation may contribute to IPC-induced neuroprotection.
Hemoglobin has been studied for over a century and has become clear that it is widely distributed in diverse organisms including those which do not possess a bloodstream (Bogusz et al, 1988; Poyart et al, 1992; Wakabayashi et al, 1986; Zhu and Riggs, 1992). In vertebrates, Hb is usually considered as an important oxygen transport protein mainly present in erythrocytes circulating in the blood. In this study, we provide the evidence that Hb is also present in neurons. Immunohistochemistry of adult rat brains revealed that Hb is preferentially expressed in cerebral neurons, with very low expression in glia. Western blotting of primary cultured cerebral neurons and astrocytes showed that neurons have much higher levels of Hb than astrocytes, consistent with the in vivo results. Ohyagi et al (1994) also found Hb in neurons of embryonic and adult mice brain, with low or no expression in the glia cells. The expressions of Hb and its mRNA have also been found in neurons in hippocampus and granule cells in the cerebellum (Blalock et al, 2003; Slemmon et al, 1994; Wu et al, 2004). Taken together, these data suggest that Hb is widely expressed in brain and is primarily neuronal.
The function of neuronal Hb is still unclear. The concentration of Hb in brain is only about one thousandth of that in blood, indicating that it does not represent a major repository of oxygen. It may be that neuronal Hb acts to facilitate the movement of O2 from the cell membrane to mitochondria, in a manner analogous to myoglobin in slow-twitch muscle fibers that rely on oxidative phosphorylation to produce ATP. It is also possible that Hb has functions in brain that are not directly related to O2; for example, Hb could be a substrate for degradation by heme oxygenases resulting in the production of carbon monoxide, Fe and bilirubin.
As Hb is an oxygen-binding protein preferentially expressed in neurons and it is of interest to understand whether cerebral ischemia can regulate neuronal Hb levels. In this study, we found that Hb protein is significantly increased in the ipsilateral caudate and cortical core after IPC, which can induce ischemic tolerance in rats (Masada et al, 2001). Quantitative real-time PCR indicated that such IPC also significantly upregulates the expression of HbA and HbB mRNA. An important point to be discussed here involves the opposite results obtained by an earlier study in mice, where HbB mRNA was significantly decreased in the ischemic cortex compared with the nonischemic cortex 24 h after IPC (Stenzel-Poore et al, 2003). Several factors including species, brain region and severity of ischemia may account for such discrepancies.
Upregulation of Hb after IPC may be related to the activation of hypoxia inducible factor-1, which is an important regulator of the response to hypoxia and oxygen homeostasis (Sharp and Bernaudin, 2004). Hypoxia inducible factor-1 modulates β-globin gene expression during vertebrate development (Bichet et al, 1999). There is evidence that hypoxia inducible factor-1α is activated in response to IPC (Bergeron et al, 2000; Gidday et al, 1994). Further studies will be required to investigate the mechanisms of induction of Hb by IPC.
In this study, we found that the number of Hb-positive cells in the cortical intermediate (penumbral) zone and nonischemic hemisphere of rats undergoing 24 h of pMCAO was markedly increased compared with those in shams, whereas Hb-positive cells in the ischemic core were significantly less than in shams. To confirm the expression of Hb in the brain, we performed quantitative real-time PCR after 24 h of pMCAO. Real-time PCR revealed that Hb mRNA expression was increased in the cortex intermediate zone compared with shams, with markedly lower levels in the ischemic core. The reductions in Hb protein and mRNA within the core of the infarct probably reflect the severe reductions in blood flow within that region with concomitant decrease in mRNA levels and protein synthesis.
Oxygen—glucose deprivation is used as an in vitro model for ischemia—reperfusion injury (Goldberg and Choi, 1993). In this study, we showed that 2 h of OGD followed by 22 h of recovery significantly increased Hb protein levels in neurons similar to the effects of IPC in vivo. However, whereas PCR revealed that HbA mRNA expression was upregulated by 2 h of OGD, HbB mRNA expression was downregulated, suggesting that α-globin and β-globin may have a different expression pattern.
There is growing evidence showing that IPC provides robust protection or tolerance against a subsequent prolonged ischemic event (Dirnagl et al, 2003; Kirino, 2002; Kitagawa et al, 1990; Murry et al, 1986). Our earlier studies have also shown that IPC attenuates brain edema formation and cerebrovascular injury after pMCAO (Masada et al, 2001). The precise mechanisms of neuroprotection that lead to ischemic tolerance are not well understood. As mentioned above, Hb is upregulated after IPC in rats. We then tested the effects of IPC on Hb expression after pMCAO. Immunostaining showed that the number of Hb-positive cells in the ipsilateral caudate was increased after pMCAO in IPC-treated rats compared with the sham-treated rats, whereas the number of Hb-positive cells in the ipsilateral intermediate zone after pMCAO was less in IPC-treated rats. Quantitative real-time PCR revealed that IPC-treated rats had higher Hb mRNA levels in the ipsilateral caudate than those the shams. An increased expression of Hb in the ischemic core area may help neurons to survive under cerebral ischemia. There is evidence showing that enhanced expression of neuroglobin, a respiratory protein of the nervous system, promotes neuronal survival under brain hypoxia and ischemia (Khan et al, 2006; Sun et al, 2001, 2003).
It is well known that exogenous Hb released from red blood cells after intracerebral hemorrhage is neurotoxic (Xi et al, 2006). Recent studies have shown that exogenous Hb can induce cell death in the rat cerebral cortical neurons through the activation of caspase cascades and oxidative stress (Wang et al, 2002). However, the function of endogenous neuronal Hb, which is present in much lower concentrations than found in blood, is still not clear. Our earlier study found that ICH-induced brain injury in the iron-deficient rats is greater than in normal control rats (Shao et al, 2005). It is not clear whether iron deficiency results in lower neuronal Hb levels and anemia. Future studies should determine whether endogenous neuronal Hb is neuroprotective.
In conclusion, this study shows that Hb is expressed in cerebral neurons, and that neuronal Hb is inducible after cerebral ischemia. An understanding of the mechanisms of induction of neuronal Hb after ischemia and the function of neuronal Hb should be helpful in seeking effective new treatment for stroke.
Footnotes
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
