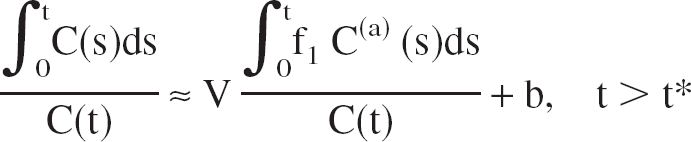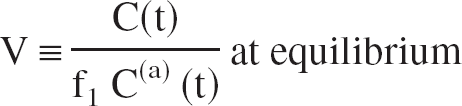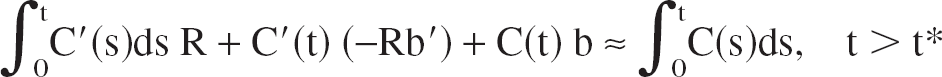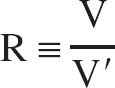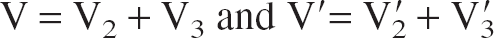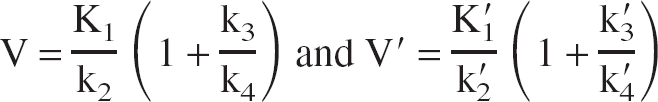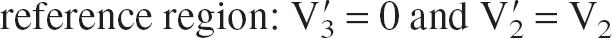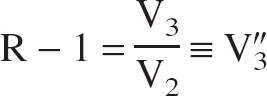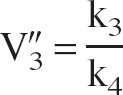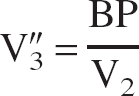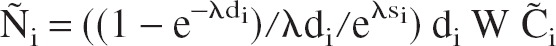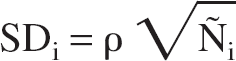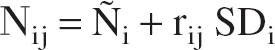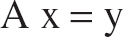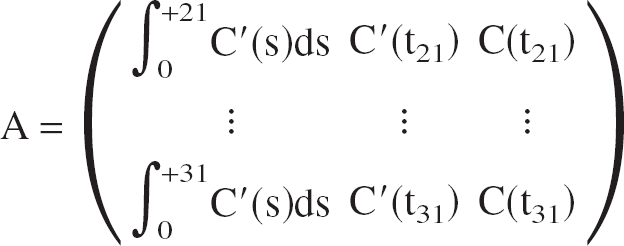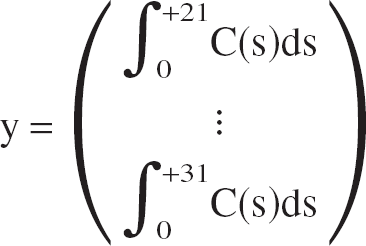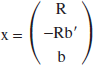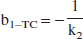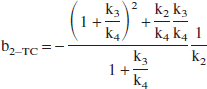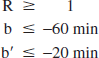Abstract
A multilinear reference tissue approach has been widely used recently for the assessment of neuroreceptor–ligand interactions with positron emission tomography. The authors analyzed this “multilinear method” with respect to its sensitivity to statistical noise, and propose regularization procedures that reduce the effects of statistical noise. Computer simulations and singular value decomposition of its operational equation were used to investigate the sensitivity of the multilinear method to statistical noise. Regularization was performed by truncated singular value decomposition, Tikhonov-Phillips regularization, and by imposing boundary constraints on the rate constants. There was a significant underestimation of distribution volume ratios. Singular value decomposition showed that the bias was caused by statistical noise. The regularization procedures significantly increased the test–retest stability. The bias could be reduced by applying linear constraints on the rate constants based on their normal range. Underestimation of distribution volume ratios by the multilinear method is caused by its sensitivity to statistical noise. Statistical power in the discrimination of different groups of subjects can be significantly improved by regularization procedures without introducing additional bias. Correct distribution volume ratios can be obtained by imposing physiologic constraints on the rate constants.
Linear regression methods, also called graphical methods, are widely used for the quantification of reversible binding of radioligands to neuroreceptors in positron emission tomography (PET) and single-photon emission computed tomography (SPECT) imaging. Linear methods are appreciated because they can be easily applied on a voxel-by-voxel basis to generate parametric images of the brain. The most widely used linear method, the Logan plot (Logan et al., 1990), is an “invasive” approach in that it requires arterial blood sampling, which is an additional source of statistical and systematical errors and involves significant discomfort for most patients.
A new class of techniques suggests the replacement of the arterial input function by the time–activity curve (TAC) of a receptor-free reference region. These reference tissue methods significantly improve compliance of subjects. A reference tissue method characterized by a multilinear operational equation, and thus combining the advantages of reference tissue approaches and linear regression, was developed by Ichise et al. (1996, 2001). This method is subsequently referred to as the multilinear method.
The multilinear method has been increasingly used recently. For example, the method was used to determine serotonin transporter occupancy of (unlabeled) serotonin reuptake inhibitors during treatment in humans (Meyer et al., 2001, 2002). However, the sensitivity of the multilinear method to statistical noise has not yet been evaluated in detail.
Therefore, we analyzed the sensitivity of the multilinear method to statistical noise via singular value decomposition (SVD) of its operational equation in a typical PET patient study using the serotonin transporter ligand [11C](+)McN5652 (Suehiro et al., 1993). Taking into account the rather low in vivo specific-to-nonspecific binding ratios in humans for most receptor ligands, reliable detection of small changes in receptor availability is of great importance. Therefore, stabilization of the multilinear method by different regularization procedures was also investigated.
MATERIALS AND METHODS
Multilinear method
The multilinear method (Ichise et al., 1996) can be derived from the well-known Logan plot for the graphical analysis of reversible radioligand binding (Logan et al., 1990, 2000; Patlak et al., 1983):
where C(t) is the TAC of the volume of interest (VOI), f1 C(a) (t) is the free fraction of the unmetabolized tracer in arterial plasma (input function), V is the total equilibrium DV of the tracer in the VOI, and b is (almost) constant at late times t > t*. The distribution volume V is defined as the equilibrium ratio of the tracer concentration in the VOI to the free arterial concentration:
Combining the Logan plot formula (Eq. 1) for two different tissue TACs, C(t) and C′(t), by solving the C′(t)-formula for the plasma integral, inserting the result in the C(t)-formula, and multiplying both sides by C(t) yields
where the equilibrium DV ratio R is defined as the ratio of the total DVs in the two regions with concentrations C to C′:
Equation 3 is the operational equation of the multilinear method. It is linear in R, b, and b′.
Within the two-tissue (2T) compartment model consisting of a nondisplaceable compartment, C2, and a specific compartment, C3, the total distribution volume V is the sum of the nondisplaceable equilibrium distribution volume, V2, and the specific equilibrium distribution volume, V3:
Fractional blood volume has been neglected. In terms of the rate constants:
where K1 f1 C(a) is the tracer flux from arterial plasma to C2, k2 is the unidirectional rate constant for the loss of tracer from the tissue through C2, and k3 and k4 are the rate constants for the tracer transfer from C2 to C3 and vice versa.
If C′ is assumed to represent a proper reference region for the receptor-rich region C:
one obtains from Eqs. 4 and 5:
where R-1 is the so-called specific-to-nonspecific equilibrium partition coefficient, usually denoted by V″3 (Laruelle et al., 1994). Within the 2T-compartment model, V″3 is given by:
V″3 is related to the binding potential (BP), defined as the ratio of the concentration of available receptors, Bmax, to the equilibrium dissociation constant, KD, according to (Laruelle et al., 1994):
Note that for the DV ratio R to reflect the receptor density Bmax, both Kd and V2 must be constant between individuals and diseases (Ichise et al., 2001).
Data
[11C](+)McN5652 has been previously shown to be one of the most appropriate tracers to quantify availability of the presynaptic serotonin transporter in vivo in the human brain (Buck et al., 2000; McCann et al., 1998; Parsey et al., 2000; Szabo et al., 1995, 1999). Time-activity curves from a typical [11C](+)McN5652 PET patient study were provided by the commercial PMOD software package (Version 2.35 for Windows NT; Medical Imaging Software, Zurich, Switzerland; Mikolajczyk et al., 1998). Details on PET imaging and blood sampling have been described previously (Buck et al., 2000). From these data, metabolite-corrected tracer concentration in arterial plasma, Ci and TACs of a receptor-rich region (left thalamus), Ci, and a reference region (gray matter of left cerebellum), C′i, were used (Fig. 1). The subscript i = 1, …, 31 refers to the individual time frames. The free fraction f1 of [11C](+)McN5652 in arterial plasma was set to 1 in the following analyses, which is of no concern for the present investigation.
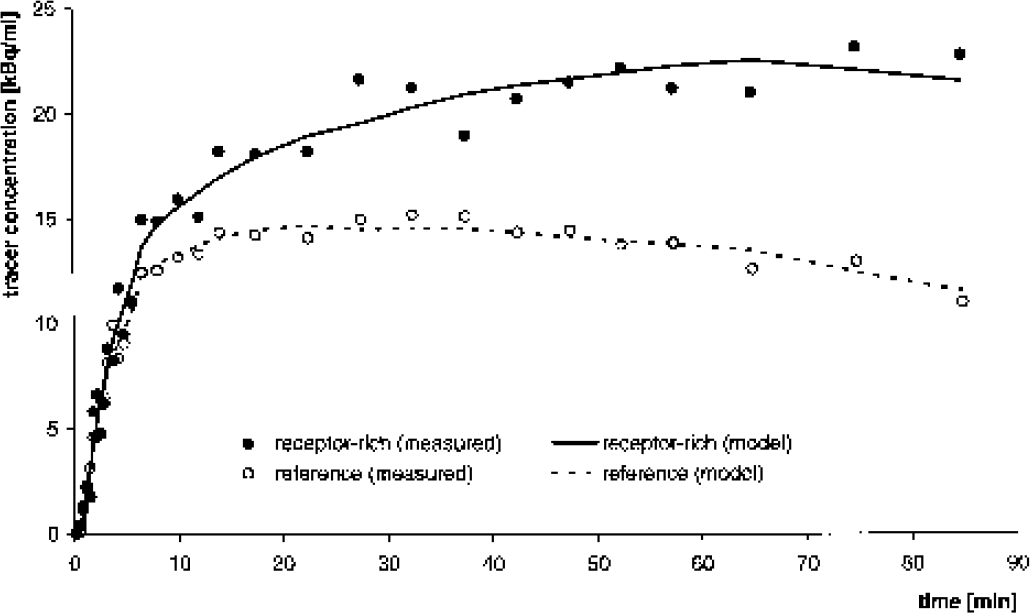
Positron emission tomography patient study using the serotonin transporter ligand [11C](+)McN5652 showing time– activity curves (TACs) of a receptor-rich region (thalamus) and a receptor-free reference region (cerebellum). Measured TACs were taken from the [11C](+)McN5652 example in the commercial software package PMOD. Model TACs were obtained from measured TACs by invasive quantification using the 2T-compartment model.
Data were first analyzed by the invasive 2T-compartment model, neglecting fractional blood volume, to yield model curves C͂i and C͂′i for the receptor-rich region and the reference region, respectively (Fig. 1). The multilinear method was then applied using both the measured TACs Ci, C′i and the model TACs C͂i, C͂′i.
The operational Eq. 3 of the multilinear method was solved in the least-squares sense (Moore-Penrose solution) by SVD (Appendix). t* = 20 minutes p.i. was used (i.e. frames 21–31 were included in the fit).
Statistical noise
To simulate statistical noise, the number of radioactive decays within the receptor-rich region during frame i, Ñi, was computed according to the following equation:
where W is the volume of the receptor-rich region (taken to be 3 mL), λ is the decay constant of 11C, and si and di the start and the duration of the ith frame, respectively. Then, Ñi was assumed to be a sample from a normal distribution with mean Ñi and standard deviation:
The number of decays during frame i of the jth dynamic scan, Nij, was simulated according to the following equation:
where rij is a random matrix whose elements are normally distributed with mean 0 and variance 1. For a typical patient study, ρ = 100 is adequate (see below in this section). The radioactivity concentration Cij corresponding to Nij was obtained by inversion of Eq. 11. This procedure was applied to the model TAC of the reference region, C͂′i, analogously (volume of the reference region W = 20 mL). Independent random matrices were generated for the receptor-rich region and the reference region. Figure 2 shows relative standard deviations of C͂i and C͂′i. Comparing the relative standard deviations of the simulated noisy TACs with the differences between measured TACs and modeled TACs (Fig. 1), the simulated noise levels appear realistic. A total of 104 dynamic scans were simulated. The multilinear method was applied to each data set.
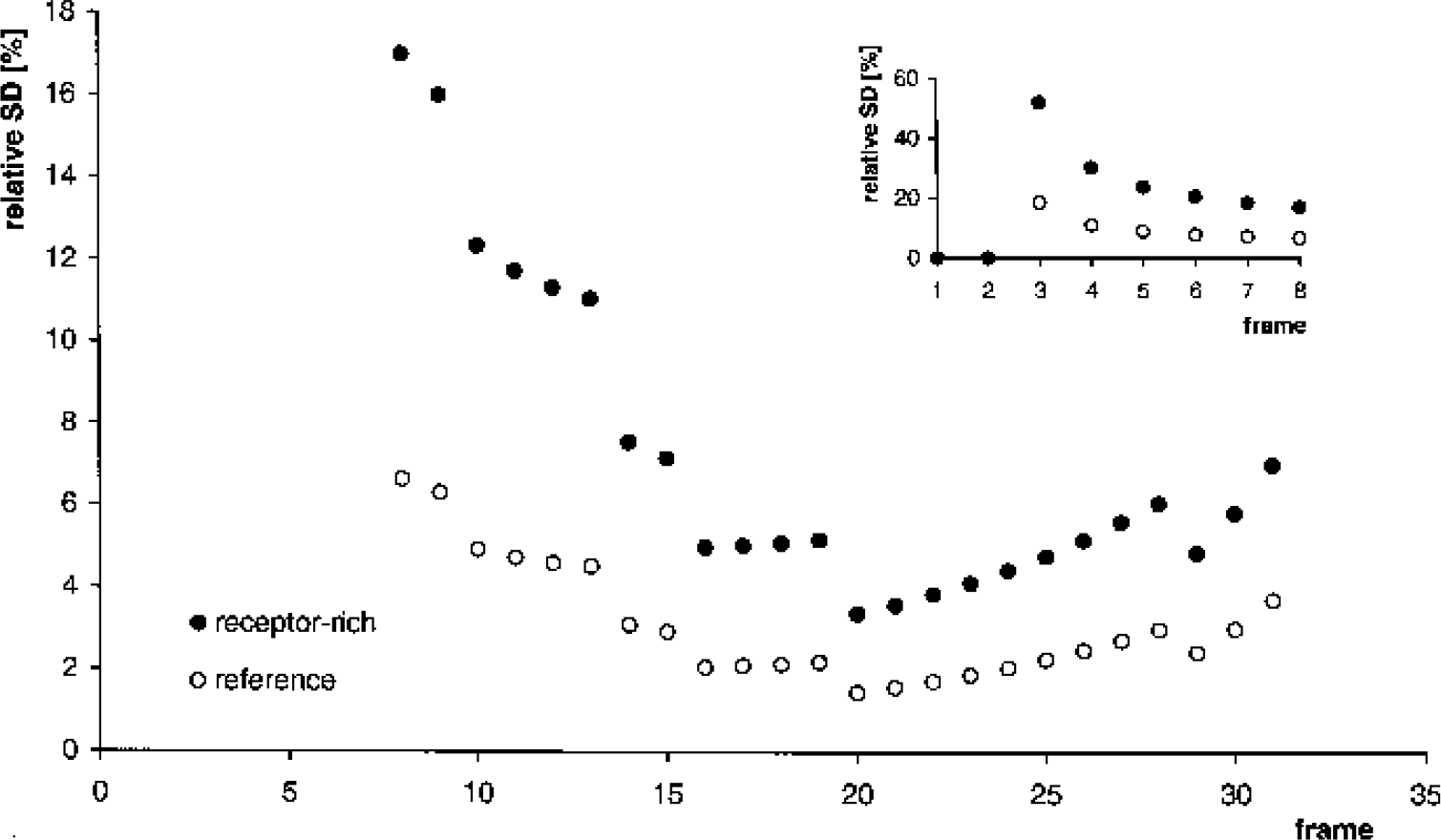
Simulation of statistical noise showing the standard deviation of tracer concentration relative to the mean tracer concentration (%). The scan protocol included 31 frames with a total acquisition time of 90 minutes using frame durations of 9 × 20, 4 × 30, 2 × 60, 4 × 120, 9 × 300, and 3 × 600 seconds (Buck et al., 2000). The standard deviation of the tracer concentration in the first two frames was set to 0 because the model time–activity curves vanished in these frames.
Singular value decomposition
Using frames 21 to 31 of the TACs, the operational Eq. 3 reduces to a system of 11 linear equations with 3 unknowns, which can be written in matrix notation as:
where A is the 11 × 3 coefficient matrix given by
y is the 11-dimensional right-side vector
and × is the 3-dimensional unknown vector
(ti is the frame midtime of the ith frame).
The SVD of the coefficient matrix Ã, computed from the model TACs C͂i and C͂′i, was analyzed and compared with the SVD of the coefficient matrix A, computed from the measured (noisy) TACs Ci and C′i.
Regularization by truncated singular value decomposition
The operational Eq. 3 of the multilinear method was solved by truncated singular value decomposition (TSVD; Eq. A7). According to the results of the analysis of the SVD, p = 2 was chosen (i.e., contributions of the third, the smallest singular value, were omitted). A total of 104 dynamic scans were simulated.
Regularization by Tikhonov-Phillips regularization
The operational Eq. 3 of the multilinear method was solved using Tikhonov-Phillips regularization (Eq. A8). The regularization parameter γ was chosen to lie between the second and the third, the smallest singular value (γ = 10). A total of 104 dynamic scans were simulated.
Smoothing of time–activity curves
The effect of simply smoothing noisy TACs before data evaluation was also analyzed. Noisy TACs were first smoothed by the method of cubic smoothing splines of Schoenberg and Reinsch (sum of squared error weighted differences < number of data points; De Boor, 1987) and than evaluated by the multilinear method without further regularization or constraints. A total of 104 dynamic scans were simulated.
Constraints
The intercept b in the Logan plot formula (1) is always negative. For example, in the one-tissue (1T) and 2T-compartment models:
However, we have observed that the multilinear method applied to noisy data tends to yield positive results for both b and b′. Therefore, the operational Eq. 3 of the multilinear method was solved (least squares) with b and b′ constrained to negative values (b, b′≤0). A total of 104 dynamic scans were simulated.
Parsey et al. (2000) performed [11C](+)McN5652 PET in six healthy volunteers. Application of the invasive 1T-compartment model yielded k2 = 0.009 ± 0.001 min−1 and k′2 = 0.014 ± 0.002 min−1 in a receptor-rich region (thalamus) and a reference region (cerebellum), respectively (mean ± 1 SD). Using Eq. 18, this corresponds to b = −110 ± 10 minutes and b′ = −70 ± 10 minutes. Assuming that the values for each individual subject lie in the range mean ± 5 SD, one may formulate the physiologic constraints:
The operational Eq. 3 of the multilinear method was solved (least squares) using these constraints. A total of 104 dynamic scans were simulated.
Regularization by Tikhonov-Phillips regularization with nonpositivity constraints
To evaluate the combined effect of filtered SVD and nonpositivity constraints, the operational Eq. 3 of the multilinear method was solved using Tikhonov-Phillips regularization with nonpositivity constraints imposed on b and b′ (Appendix). γ = 10 was chosen, and 104 dynamic scans were simulated.
RESULTS
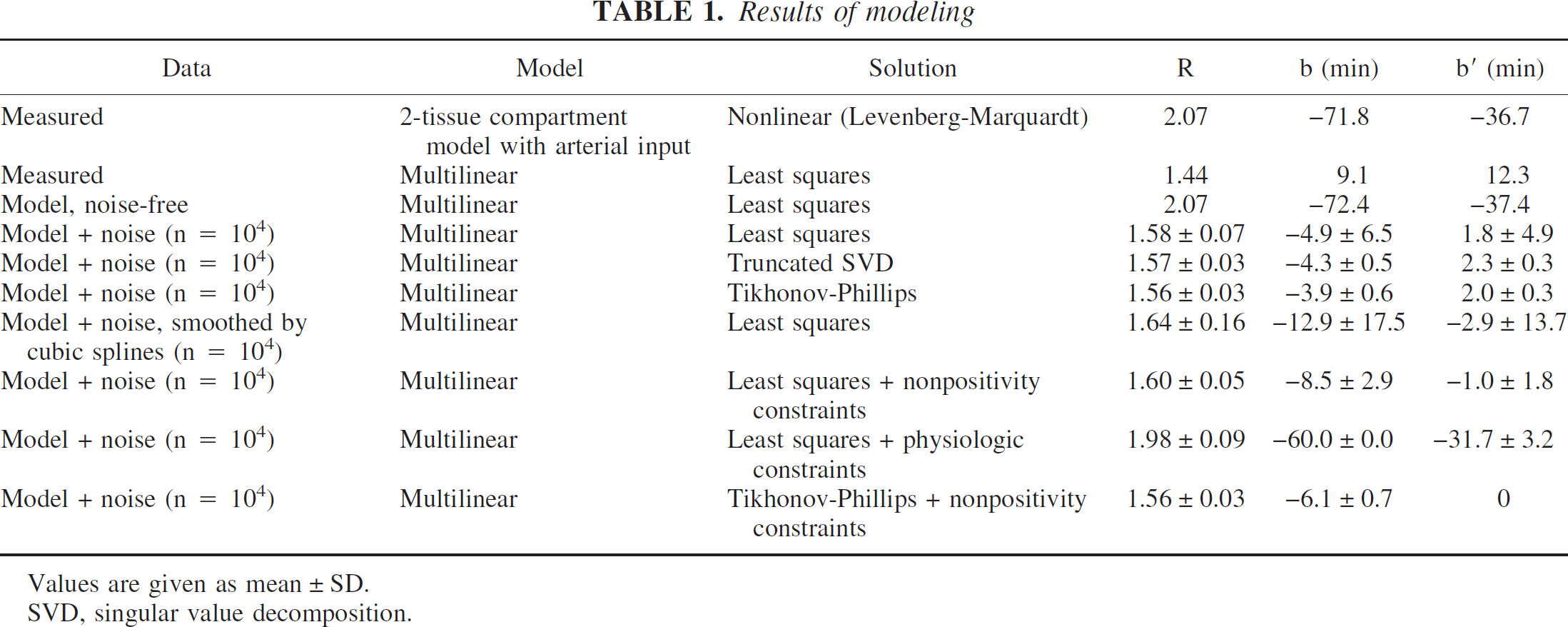
The results of the modeling are summarized in Table 1. Using the invasive 2T-compartment model (and Eqs. 4 and 6), the equilibrium DV ratio R was 2.07. This was the reference value. The multilinear method applied to the measured TACs, Ci and C′i, yielded R = 1.44 (i.e., R was underestimated by about 30%). In addition, estimates for both b and b′ were positive, in contradiction to the compartment model interpretation. In contrast, application of the multilinear method to the model data, C͂i and C͂′i, reproduced the reference values of R, b, and b′ very accurately.
Results of modeling
Values are given as mean ± SD.
SVD, singular value decomposition.
Application of the multilinear method to 104 noisy data sets obtained from the model data by adding Gaussian noise yielded R = 1.58 ± 0.07 (mean ± 1 SD); that is, R was underestimated by almost 25%. With increasing noise (increasing ρ, Eq. 12), the mean value of R showed a slight decrease while the standard deviation increased (data not shown).
The singular values of the coefficient matrix Ã, computed from the model TACs C͂i and C͂′i, were σ1 = 1.4 * 105, σ2 = 31, σ3 = 0.5. Thus the condition number of à was very large, in the order of 105. This implies instability of the multilinear method with respect to noise (Eq. A6). The left singular vectors of à are shown in Fig. 3 together with the left singular vectors of the coefficient matrix A, computed from the measured (noisy) TACs Ci and C′i. The first two left singular vectors, u1, u2 corresponding to the singular values σ1, σ2 are very similar for the modeled à and the measured A. However, there is no similarity at all between the left singular vectors u3 corresponding to the smallest singular value, σ3, of à and A. The left singular vector u3 of the measured coefficient matrix A is very noisy; it oscillates heavily around 0 (Fig. 3C). This causes the corresponding coefficient uT3y of the v3-contribution to the least-squares solution of the multilinear method to be too small (Eq. A3). The components of v3, the right singular vector to the smallest singular value σ3, are shown in Fig. 4. The R-component of v3 is positive, its (−R b′)-component is also positive, and its b-component is negative. This explains why underestimation of the coefficient uT3y, caused by oscillations of u3 due to statistical noise, leads to systematic underestimation of R, and overestimation of both, b and b′.
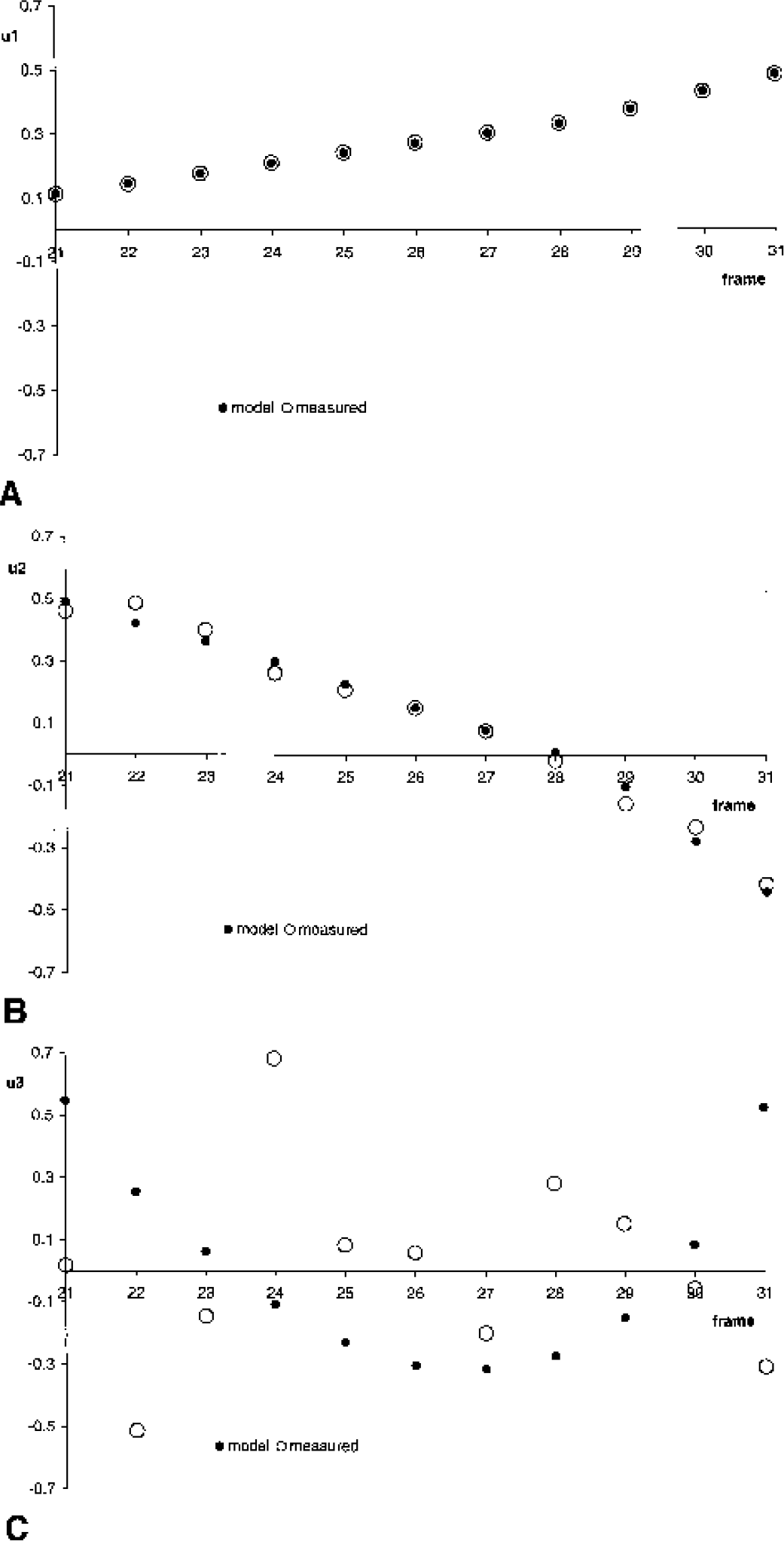
Singular value decomposition (SVD) of the coefficient matrix of the operational equation of the multilinear method: left singular vectors to the singular values σ1
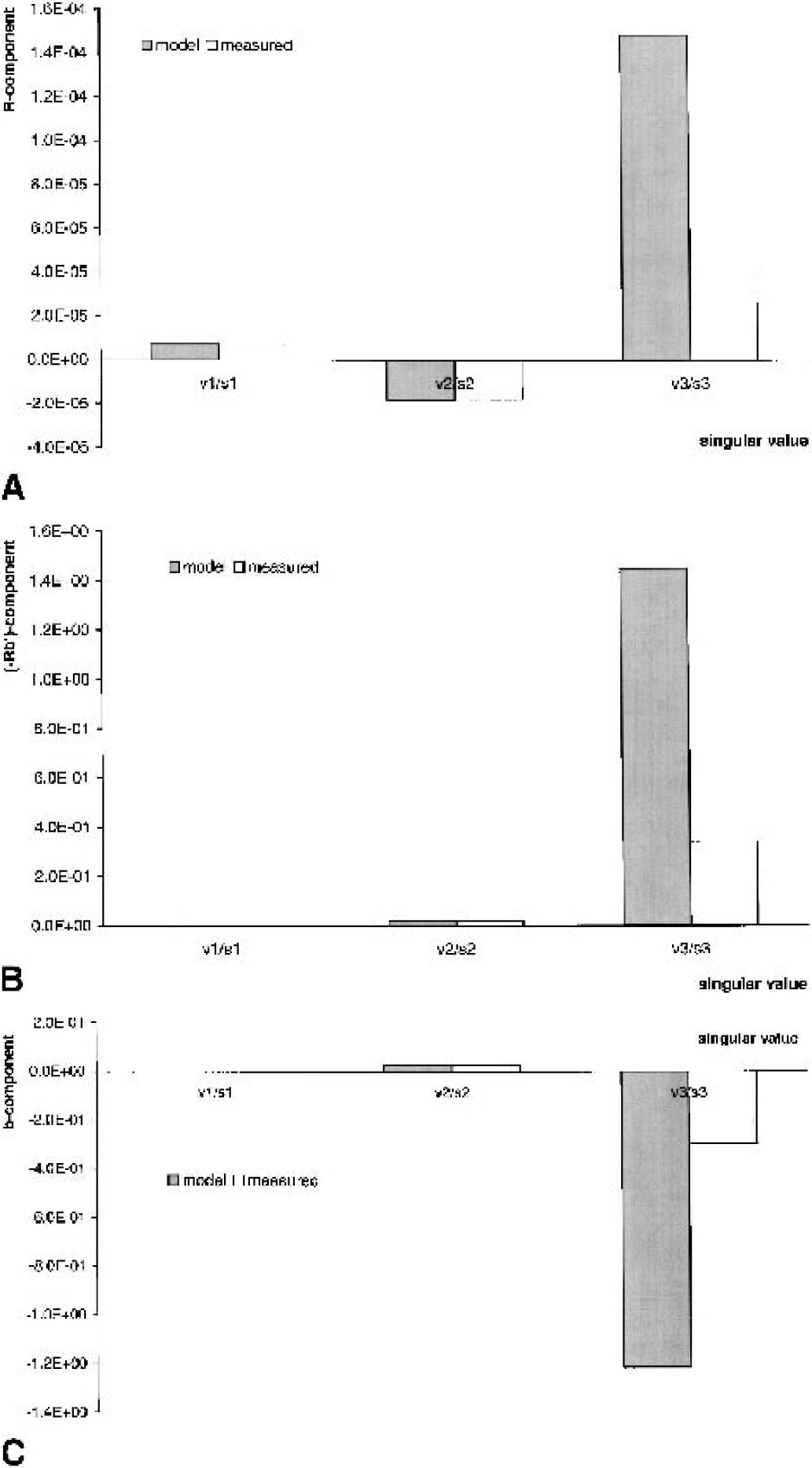
Singular value decomposition (SVD) of the coefficient matrix of the operational equation of the multilinear method: components of right singular vectors to the singular values σ1
Since the contribution of u3 to the solution of the multilinear method in case of noisy data was (almost) pure noise, it seemed reasonable to omit it. This is precisely the effect of TSVD with P = 2. As was to be expected, TSVD left mean values unchanged, but significantly reduced standard deviations. The standard deviation of R was reduced from 0.07 to 0.03 (i.e., by a factor of about 2).
Tikhonov-Philipps regularization (TP) yielded very similar results as TSVD. Nonpositivity constraints on b and b′ also provided stabilization, although somewhat less than TSVD and TP. The underestimation of R was slightly less severe than with TP and TSVD.
Application of physiologic constraints on b and b′ resulted in almost correct results for the DV ratios R. Underestimation of R was reduced to about 4%. The constraint on the parameter b (b≤–60 minutes = mean + 5 SD; Eq. 20) was consistently active (b = −60.0 ± 0.0 minutes; Table 1). Therefore, in order to analyze the sensitivity of R to the particular choice of the physiologic constraints, the upper bound of b was varied (keeping the upper bound of b′ fixed). This resulted in R = 2.28 ± 0.14 (b≤–100 minutes = mean + 1 SD), R = 2.13 ± 0.11 (b≤–80 minutes = mean + 3 SD), R = 1.98 ± 0.09 (b≤–60 minutes = mean + 5 SD), R = 1.84 ± 0.07 (b≤–40 minutes = mean + 7 SD), and R = 1.71 ± 0.06 (b≤–20 minutes = mean + 9 SD) (true value: R = 2.07). Thus, the estimates of R decreased with loosening the physiologic constraints and approached the value obtained with the nonpositivity constraints as the upper bound of b approached zero. However, the variation of the estimate of R was restricted to about ± 10% (relative to its value resulting when using b≤–60 minutes = mean + 5 SD) when the upper bound of b was varied between [mean + 1 SD] and [mean + 7 SD]. Moreover, the error (bias) of the estimate amounted to at most 10% for this range of constraints (error without regularization was approximately 25%).
The combination of TP and nonpositivity constraints did not provide further improvement, and the results obtained were almost the same as TSVD and TP without constraints.
DISCUSSION
The multilinear method applied to noisy TACs from a patient study underestimated the DV ratio R by about 30% as compared with quantification using the 2T-compartment model together with the arterial input function (kinetic modeling). However, application of the multilinear method to the model TACs, obtained from the full kinetic modeling, reproduced the reference value very accurately. This shows that the multilinear method is “correct,” in that it yields the correct result in a case of noiseless data.
In a second step the multilinear method was applied to 104 noisy data sets obtained from the model data by adding Gaussian noise. R decreased with increasing noise levels. For realistic noise levels, R was underestimated by about 25%. This supports the hypothesis that the underestimation of R by the multilinear method is caused by its sensitivity with respect to statistical noise.
This finding was further confirmed by the SVD of the coefficient matrix of the operational equation of the multilinear method. The condition number was found to be extremely large, in that the multilinear method is rather ill conditioned (i.e., it is quite sensitive to statistical noise). In case of noisy data, the left singular vector corresponding to the smallest singular value was very noisy, its components oscillating heavily around zero. This caused a systematic underestimation of the contributions of the right singular vector corresponding to the smallest singular value. Since the contribution of this right singular vector to R was positive, this explained the systematic underestimation of R.
The multilinear method has been reported to provide increased test-retest reproducibility compared to invasive techniques (Acton et al., 2001). However, in order to further stabilize the algorithm by omission or suppression of the contributions of the smallest singular value, TSVD and the method of Tikhonov-Philipps was applied. Mean values of R were unchanged by these procedures, but the standard deviations were significantly reduced. Therefore, the test-retest stability of the multilinear method may be further improved by regularization. Taking into account that intersubject variability of the [11C](+)McN5652 DV ratio obtained by the multilinear method is below 10% relative standard deviation (Buchert et al., 2003) the amount of the improvement of the test-retest stability by regularization is of practical relevance. It should be noted that improvement of test-retest stability is of practical relevance despite the fact that the error (bias) in the estimation of DV ratios is still large. Assuming that there is a stable one-to-one correspondence between the true values and the biased estimators, the power of a method to separate different groups of subjects (e.g., drug users versus drug naïves) or, in clinical applications, a single patient from a group of normal controls (e.g., by voxel-based analysis using SPM) strongly depends on the variance of the parameter of interest. In order to detect alterations of the receptor status, the examined parameter need not be unbiased. Thus, the ability to reduce the variance of a biased estimator is definitely desirable.
Imposing nonpositivity constraints on the rate constants provided stabilization, although somewhat less than TSVD or TP.
Simply smoothing noisy TACs before data evaluation resulted in even larger standard deviation of the DV ratio R as without smoothing and without regularization. This indicates that simple smoothing of TACs is not appropriate for the stabilization of the multilinear method because modeling itself is some kind of smoothing in time. Additional smoothing of TACs before modeling is redundant and should not be applied. Regularization cannot be achieved by simple smoothing.
The multilinear method yielded correct DV ratios when supplemented by linear boundary constraints on the combinations b and b′ of rate constants based on their normal range. The sensitivity of the estimate of R to the particular choice of the physiologic constraints was rather small over a wide range of constraints. Therefore, these constraints allow bias-free estimates of DV ratios. However, the formulation of physiologic constraints requires some knowledge about mean values and variances of rate constants in each population to be studied. Since the sensitivity of the method to the particular choice of the physiologic constraints is rather small, the physiologic constraints for normal volunteers used in the present paper should also be applicable to cases with mild pathology. In clinical applications with severe pathology, the formulation of physiologic constraints may be rather difficult so that the use of nonpositivity constraints might be preferred.
In the present paper, TACs of a single patient study were used to derive model data, to which simulated noise was added. However, the results presented here were confirmed by further simulation studies. TACs corresponding to different sets of rate constants were generated using the invasive 1T-compartment model and a simulated arterial input function after a bolus injection of 400 MBq [11C](+)McN5652 (data not shown).
The Logan plot is also known to introduce a bias that results in the underestimation of distribution volumes (DVs) (Carson, 1993). The bias depends not only on the level of statistical noise but also on the magnitude of the DV, and it is more pronounced at high level of statistical noise and at large DV (Slifstein and Laruelle, 2000). The underestimation of DVs by the Logan plot is related to the fact that simple linear regression takes into account errors in the y variable but not errors in the × variable (Slifstein and Laruelle, 2000). The regularization methods discussed here account for errors on both sides of the operational equation (Appendix). However, regularization alone could not avoid the underestimation of the DV ratio.
Different strategies have been described to avoid the underestimation of DVs by the Logan plot. Logan et al. (2001) proposed a modification of the generalized linear least squares method developed by Feng et al. (1993), which essentially is an iterative approach to the solution of the operational equation. Varga and Szabo (2002) applied a linear regression model that minimizes the sum of squared perpendicular rather than vertical distances between data points and the fitted straight line. This regression model accounts for errors in both the × and y variables of the straight line. Recently, Ichise et al. (2002) reported attempts to improve neuroreceptor DV estimates based on mathematical rearrangement of the operational equation. The main drawbacks of these techniques are that the compensation is only partial, and they result in increased test-retest variability. Therefore, it might be useful to combine these methods with the regularization procedures that we have described here.
CONCLUSIONS
Underestimation of equilibrium DV ratios by the multilinear method is caused by its sensitivity to statistical noise. The bias can be reduced by imposing boundary constraints. Regularization procedures do significantly improve the stability of the multilinear method, which results in increased statistical power.