Abstract
Nitric oxide (NO) has important regulatory functions within the central nervous system. NO is oxidized in vivo to nitrate and nitrite (NOx). Measurement of these products gives an index of NO production. The purpose of this study was to examine the relation between the brain extracellular concentration of NO metabolites and cerebral blood flow (CBF) after severe traumatic brain injury. Using a chemiluminescence method, NOx concentrations were measured in 6,701 microdialysate samples obtained from 60 patients during the first 5 d after severe head injury. Regional and global values of CBF obtained by xenon-enhanced computed tomography were used for analyses. Dialysate NOx values were the highest within the first 24 h after brain trauma and gradually decreased over the 5 postinjury d (time effect, P < 0.001). Mean dialysate concentration of NOx was 15.5 ± 17.6 μmol/L (minimum 0.3, maximum 461 μmol/L) and 65% of samples were between 5 and 20 μmol/L. There was a significant relation between regional CBF and dialysate NOx levels (r2 = 0.316, P < 0.001). Dialysate NOx levels (9.5 ± 2.2 μmol/L) in patients with critical reduction of regional CBF (<18 mL · 100 g−1 · min−1) were significantly lower than in patients with normal CBF (18.6 ± 8.1 μmol/L; P < 0.001). This relation between the dialysate concentration of NOx and regional CBF suggests some role for NO in the abnormalities of CBF that occur after traumatic brain injury.
Nitric oxide (NO) is a cell membrane–permeable free radical gas and is the smallest known biologically active molecule. The known functions of NO are very diverse and include maintenance of basal vasomotor tone, inhibition of platelet aggregation and leukocyte aggregation, macrophage-mediated cytotoxicity, neurotransmission, selective neuroprotection, apoptosis, synaptogenesis, intercellular signaling, N-methyl-D-aspartate-mediated neuronal excitotoxicity and free radical generation. As a consequence of these diverse biologic roles, the regulation of enzymes responsible for producing NO is very complex.
Nitric oxide is a potent vasodilator. In the central nervous system, NO participates in the regulation of basal cerebral blood flow (CBF) and in the cerebrovascular responses to metabolic activity and to hypercapnia (Goadsby et al., 1992; Iadocola and Zhang, 1996). Some studies have suggested a role for NO in pressure autoregulation (White et al., 2000). Under pathologic conditions, both excesses and deficiencies of NO may have deleterious effects, potentially leading to hypoperfusion and to direct neurotoxicity and cellular injury (Faraci and Brian, 1994; Chao et al., 1994; Merrill et al., 1993; Albina et al., 1993).
Nitric oxide is derived from arginine in two steps catalyzed by nitric oxide synthase (NOS). The catalytic activity of NOS is related to the monooxygenase activity of cytochrome P450. There are three isoforms of NOS: a neuronal type called nNOS, an endothelial type called eNOS, and an inducible form called iNOS. The neuronal and endothelial NOS isoforms are constitutively expressed and regulated by calcium concentration via calmodulin interaction. Inducible NOS is only expressed under certain pathologic conditions such as cytokine regulation or pathologic induction in the presence of endotoxins or cytotoxins.
Because NO has important regulatory functions within the central nervous system, there is interest in examining changes in NO metabolism that might occur after traumatic brain injury (TBI). Low levels of NO might be associated with vasoconstriction of cerebral vessels, resulting in decreased CBF or even ischemia. Increased production of NO could result in cytotoxicity in the brain as NO is metabolized to peroxynitrite. Despite this interest, relatively few studies have been published regarding NO metabolism after human TBI (Clark et al., 1994; Uzan et al., 2001; Gahm et al., 2002).
Part of the reason for this paucity of human studies may be the difficulty in measuring NO. Nitric oxide is difficult to measure in vivo because of its short half-life. In experimental models NO can be measured directly with an NO electrode or by using a method for trapping NO in a stable form (Malinski et al., 1993; Balcioglu and Maher, 1993). In humans, direct measurement is not practical and some indirect method must be used. One method is to measure the end products of NO metabolism, nitrate and nitrite (NOx), as an index of NO production. In experimental studies, simultaneous measurement of NO after TBI by both NO electrode and by microdialysis NOx assays shows similar changes (Cherian et al., 2000).
The purpose of this study was to measure the concentration of NO in the brain after TBI in severely head-injured patients. To accomplish this, extracellular concentrations of NOx were measured in samples collected from a microdialysis probe placed in the brain after injury.
MATERIALS AND METHODS
Patient management
The research protocol was approved by the Baylor Institutional Review Board for Human Subject Research, and informed consent was obtained from each patient's nearest relative for participation in the study. When relatives were unavailable to give informed consent, the patients were enrolled in the study using an approved emergency consent procedure. The demographic characteristics of the patients are summarized in Table 1.
Demographic characteristics of 60 patients following severe head injury
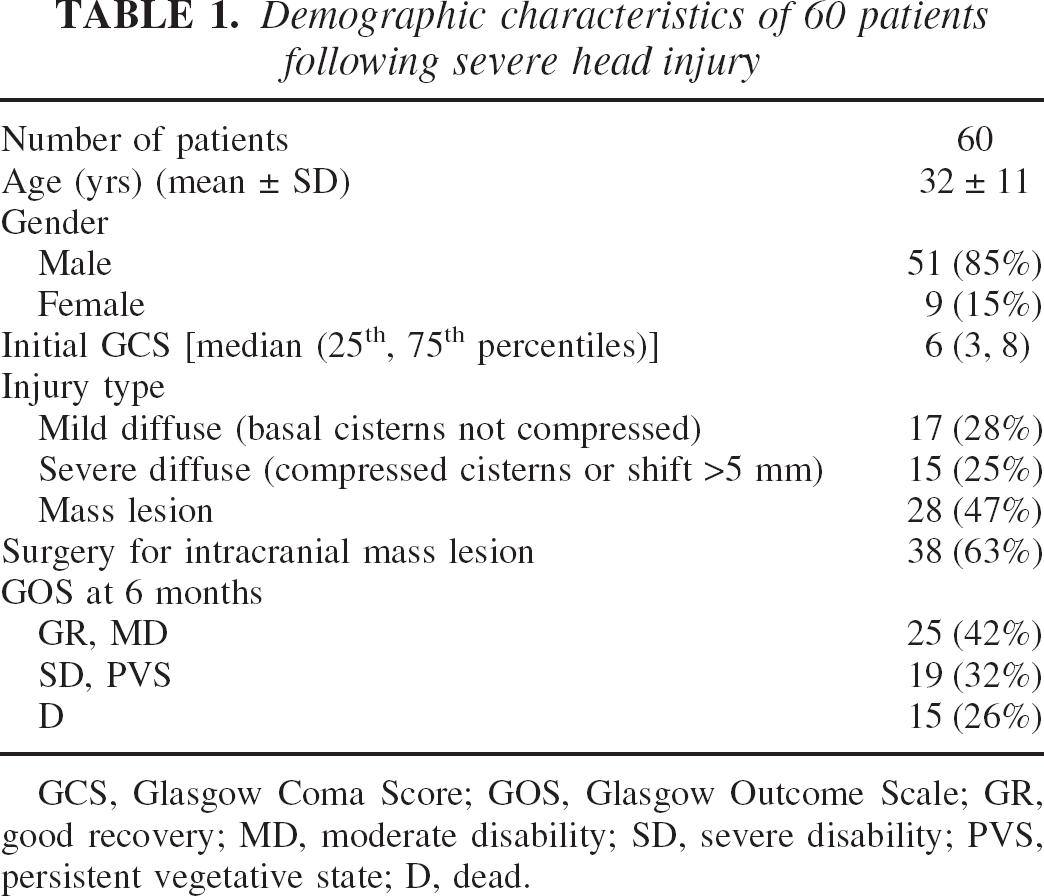
GCS, Glasgow Coma Score; GOS, Glasgow Outcome Scale; GR, good recovery; MD, moderate disability; SD, severe disability; PVS, persistent vegetative state; D, dead.
The eligibility criteria for the study included following: (1) TBI, (2) age ≥15 years; and (3) motor Glasgow Coma Score of ≤5. All patients admitted to Ben Taub General Hospital with a severe TBI were resuscitated per Advanced Trauma Life Support guidelines and evaluated for systemic injuries as well as for the severity and nature of the brain injury. Those patients with an intracranial mass lesion on the admission computed tomography (CT) scan were taken immediately to the operating room for evacuation of the hematoma. A microdialysis probe was placed in the brain at the end of the surgical procedure. Patients with a diffuse brain injury were taken to the intensive care unit for further stabilization and for placement of monitors, including blood pressure, intracranial pressure, jugular venous oxygen saturation (SjvO2), and brain tissue PO2 (PbtO2). A microdialysis probe was placed at the time of the ventriculostomy catheter and tissue PO2 probe placement.
All patients were managed by a standard protocol that emphasized prompt evacuation of intracranial mass lesions and prevention of secondary insults to the brain. Intracranial pressure values >20 mm Hg were treated by an algorithm based on the Guidelines for the Management of Severe Traumatic Brain Injury (Bullock et al., 2000). Systemic factors that exacerbate intracranial hypertension, including hypoxia, hypercapnia, fever, and hypotension, were corrected. Treatment goals were defined as MABP of at least 80 mm Hg and cerebral perfusion pressure of at least 60 mm Hg, unless the initial xenon-enhanced computed tomography (XeCT) or other measures of cerebral perfusion indicated that a higher cerebral perfusion pressure was required.
Measurement of cerebral blood flow
All CBF studies were performed on a CT scanner (CTI; General Electrics Medical System, Waukesha, WI, U.S.A.) equipped with a stable xenon gas delivery system. For the calculation of CBF, the XeCT system 2 (Diversified Diagnostic Products, Houston, TX, U.S.A.) was used. For evaluation of mixed cortical CBF, the CT scans were performed in four axial planes with a thickness of 5 mm, each 20 mm apart. After completion of two baseline scans at each level, a mixture of 32% stable xenon gas and 68% oxygen was administered through the ventilator for 4.5 minutes, during which four additional scans were performed at each level. Analysis of the XeCT CBF was performed by outlining 20 cortical regions of interest in each of four axial planes. The mixed cortical CBF in all cortical regions of interest from all four axial planes were averaged to estimate a global value for CBF. This value was used as the global average CBF in subsequent comparisons.
During XeCT measurement, the ventilatory settings of the patients were adjusted to obtain an end-tidal CO2 of 33 to 38 and, when possible, an arterial tension of CO2 (PaCO2) between 35 and 40 mm Hg. Fifty-seven measurements of CBF by using XeCT were performed in the patients studied.
To calculate regional CBF in an area of the brain around the tip of microdialysis probe, a region of interest of approximately 2 cm2 was drawn. The regional CBF was averaged in the selected area. Dialysate NOx values in the samples collected during the period of XeCT examination were used in subsequent comparisons.
Measurement of nitrate and nitrite
A microdialysis probe (CMA-70; CMA Microdialysis, Stockholm, Sweden) was placed usually for 5 d in an area of brain that appeared to be injured but potentially viable either by direct inspection at surgery or by CT scan if surgery was not performed. The position of the probe and the relations to any surrounding brain injury and to the tissue PO2 probe were noted on follow-up CT scan. Using this approach, the microdialysis probe typically was observed to be located in the frontotemporal region of the brain, and near the tissue PO2 probe. There were no cases where the two probes were positioned so close together that there were concern about interference. The microdialysis probe was perfused at 2 μL/min with normal saline, and samples were collected every 30 minutes.
Analysis of dialysate nitrate and nitrite concentration was performed using NO chemiluminescence (NO Analysis System; Antek Instruments, Houston, TX, U.S.A.) after vanadium stripping (reduction of nitrate and nitrite back to NO in a heated vanadium chloride solution) from 10-μL samples of microdialysate, as described in detail in a previous publication (Goodman et al., 2002). The results were expressed as the sum of nitrate and NOx contained in each sample.
RESULTS
Microdialysate nitrate–nitrite values
Six thousand seven hundred one microdialysis samples in 60 patients after severe head injury were analyzed for levels of NOx. The mean concentration of NOx in these samples was 15.5 μmol/L with SD ± 17.6 μmol/L. In 65% of the samples, the concentration of NOx was between 5 and 20 μmol/L, but the individual values ranged from a low of 0.3 μmol/L to a high of 461 μmol/L. Figure 1 shows the distribution of the dialysate NOx values pooled from all 60 severely head-injured patients.
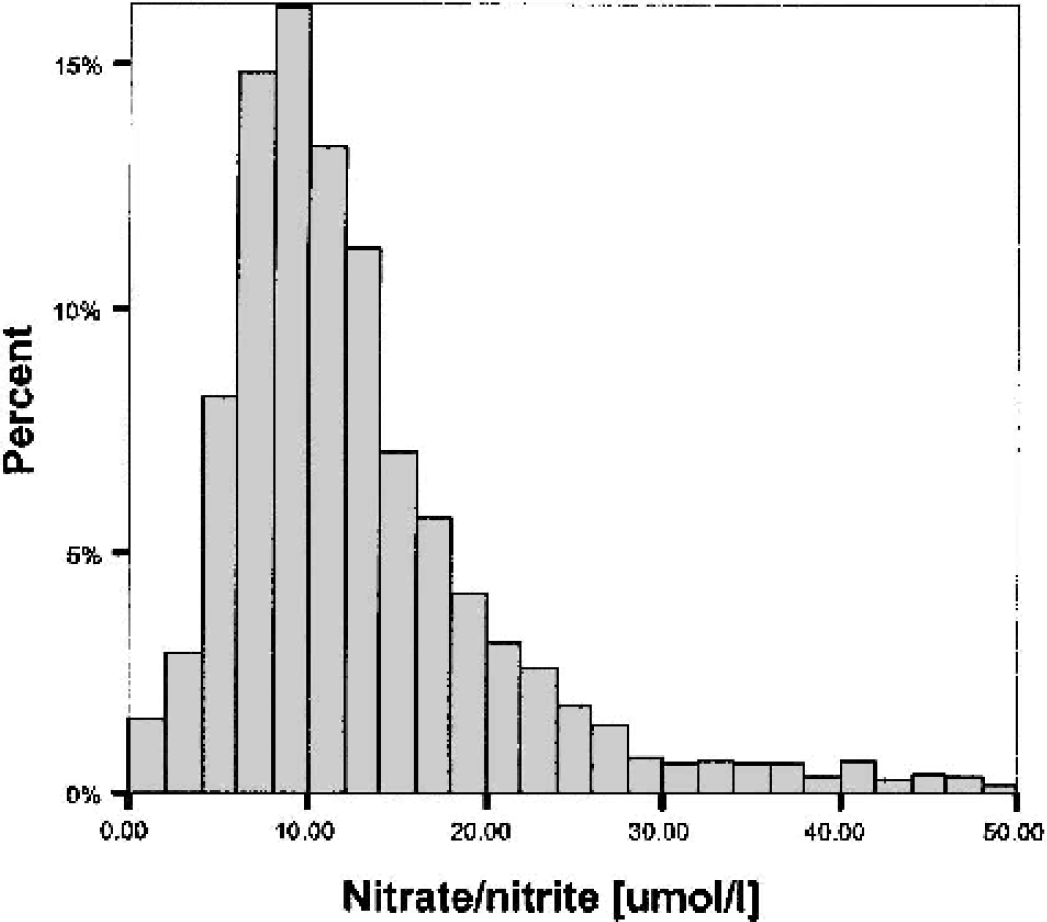
Frequency histogram of nitrate–nitrite values in 6,701 microdialysis samples in 60 patients after severe head injury.
The demographic characteristics of this group of patients were typical for a head-injured population (Table 1). None of these demographic characteristics were closely related to the dialysate NOx levels. Injury severity measures such as Glasgow Coma Score or CT scan category were also not closely related to dialysate NOx values. No significant relation was found between neurologic outcome measures and dialysate NOx levels.
Changes in nitrate and nitrite over the time
Figure 2 shows the average dialysate NOx concentrations on days 1 to 5 after injury in 6-h blocks. When the whole group of 60 patients was examined together, the mean value for dialysate NOx was highest on day 1 and gradually decreased over the 5-d monitoring period (time effect, P < 0.001). On day 1 after injury, the dialysate NOx concentration averaged 29.5 ± 42.7 μmol/L, compared with 12.6 ± 9.3 μmol/L on day 5 after head injury. There was a wide distribution of values, especially on day 1 (minimum 7.9 μmol/L, maximum 296.6 μmol/L), indicating considerable individual variability.
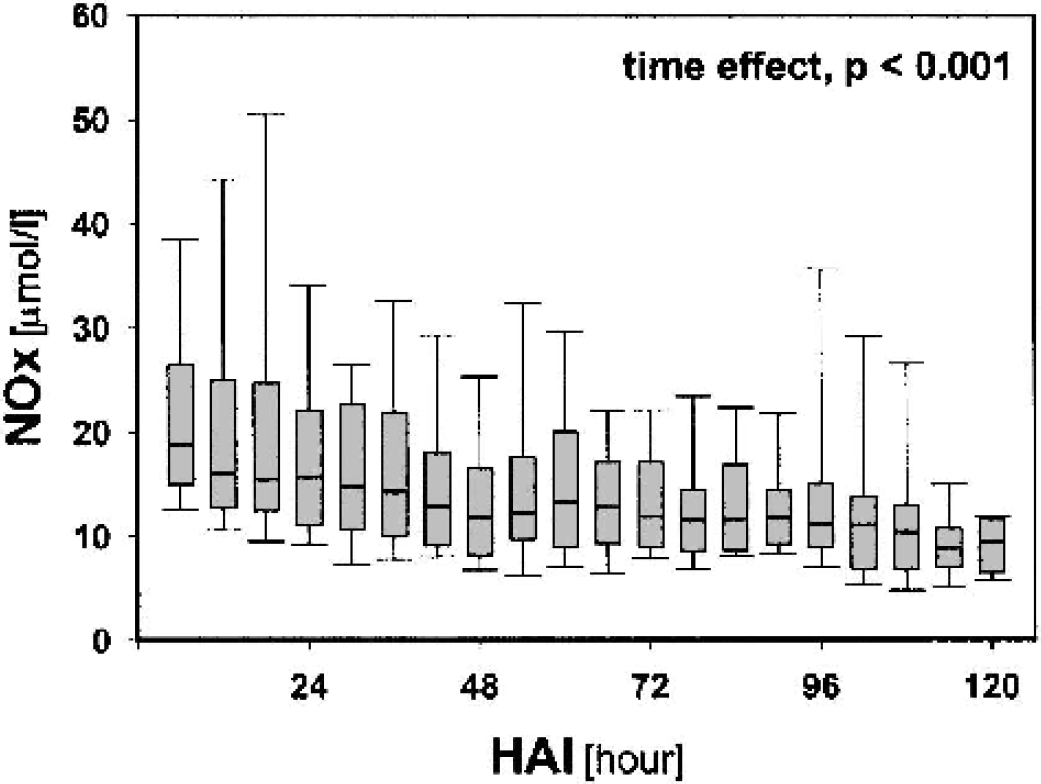
Time course of nitrate–nitrite (NOx) values during 5 d of monitoring in 60 patients after severe head injury. HAI, hour after admission to intensive care unit.
Cerebral blood flow and nitrate-nitrite values
XeCT measurement of CBF was available in 44 of the 60 patients during intracerebral microdialysis monitoring, at a median time of 31 h after injury (range 1–195 h). Average global CBF in this group of patients was 43 ± 16 mL · 100 g−1 · min−1, ranging from a low value of 14 mL · 100 g−1 · min−1 to a high value of 82 mL · 100 g−1 · min−1. Regional CBF values were obtained by averaging CBF in a region of interest in the area of brain around the microdialysis probe (as illustrated in Fig. 3). Average regional CBF at the probe site in this group of patients was 31 ± 15 mL · 100 g−1 · min−1 and the values varied from 6 mL · 100 g−1· min−1 to 74 mL · 100 g−1 · min−1.
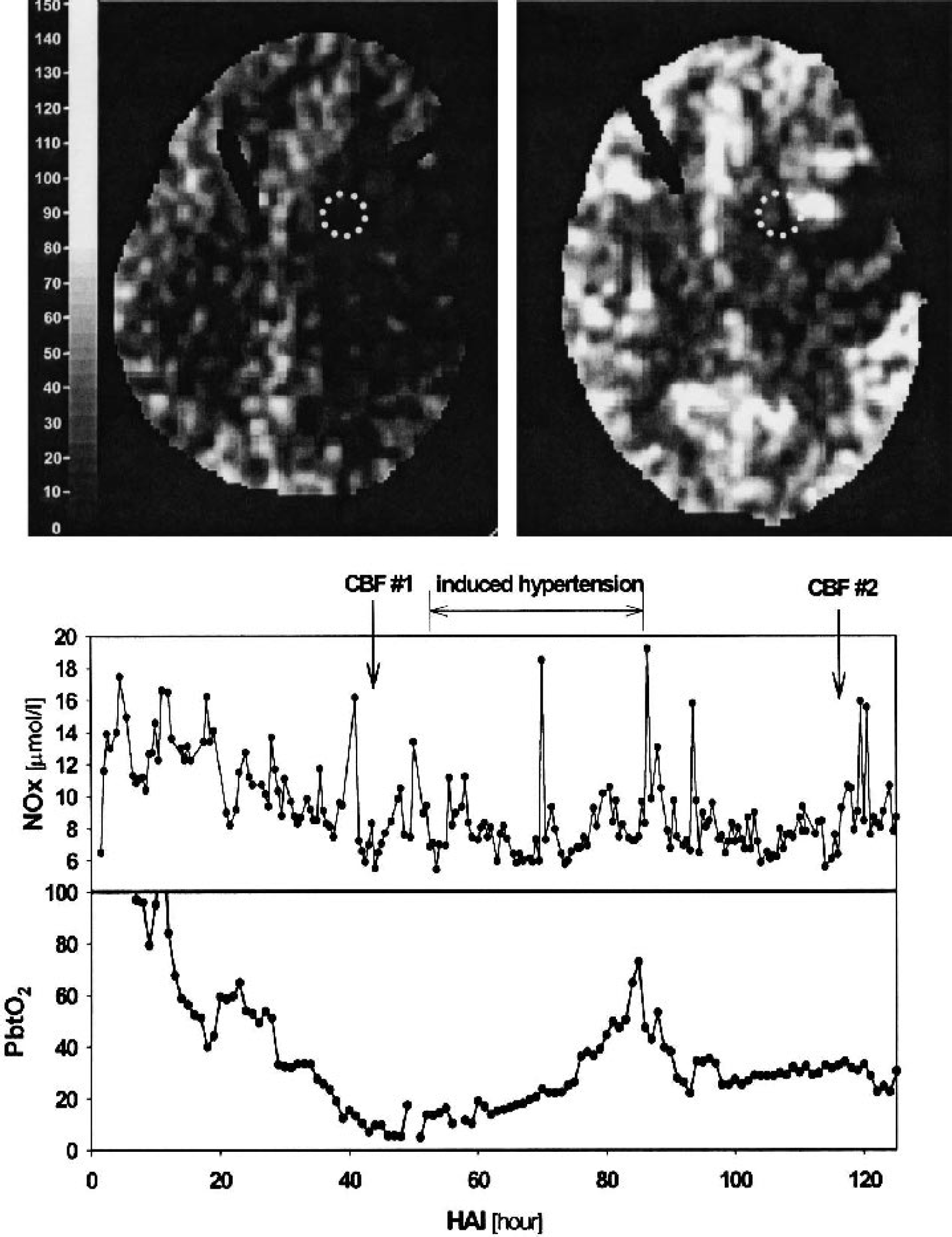
Graph of brain tissue PO2 (PbtO2) and dialysate nitrate–nitrite values in a patient who developed regional hypoperfusion in contused brain after evacuation of a subdural hematoma. Global cerebral blood flow (CBF) was 28 and 54 mL · 100 g−1 · min−1 in scans 1 and 2, respectively. Regional CBF at the microdialysis and PbtO2 probe site (dotted circle) was 6 and 19 mL · 100 g−1 · min−1 in scans 1 and 2, respectively. HAI, hour after admission to intensive care unit.
The average dialysate NOx concentration in all patients at the time of the CBF measurements was 13.5 ± 5.7 μmol/L (minimum 4.5 μmol/L, maximum 32 μmol/L). Regional CBF at the probe site was closely related to these dialysate NOx values obtained during the measurement of CBF (r2 = 0.316) (Fig. 4—top), whereas global CBF was not (r2 = 0.051) (Fig. 4—bottom).
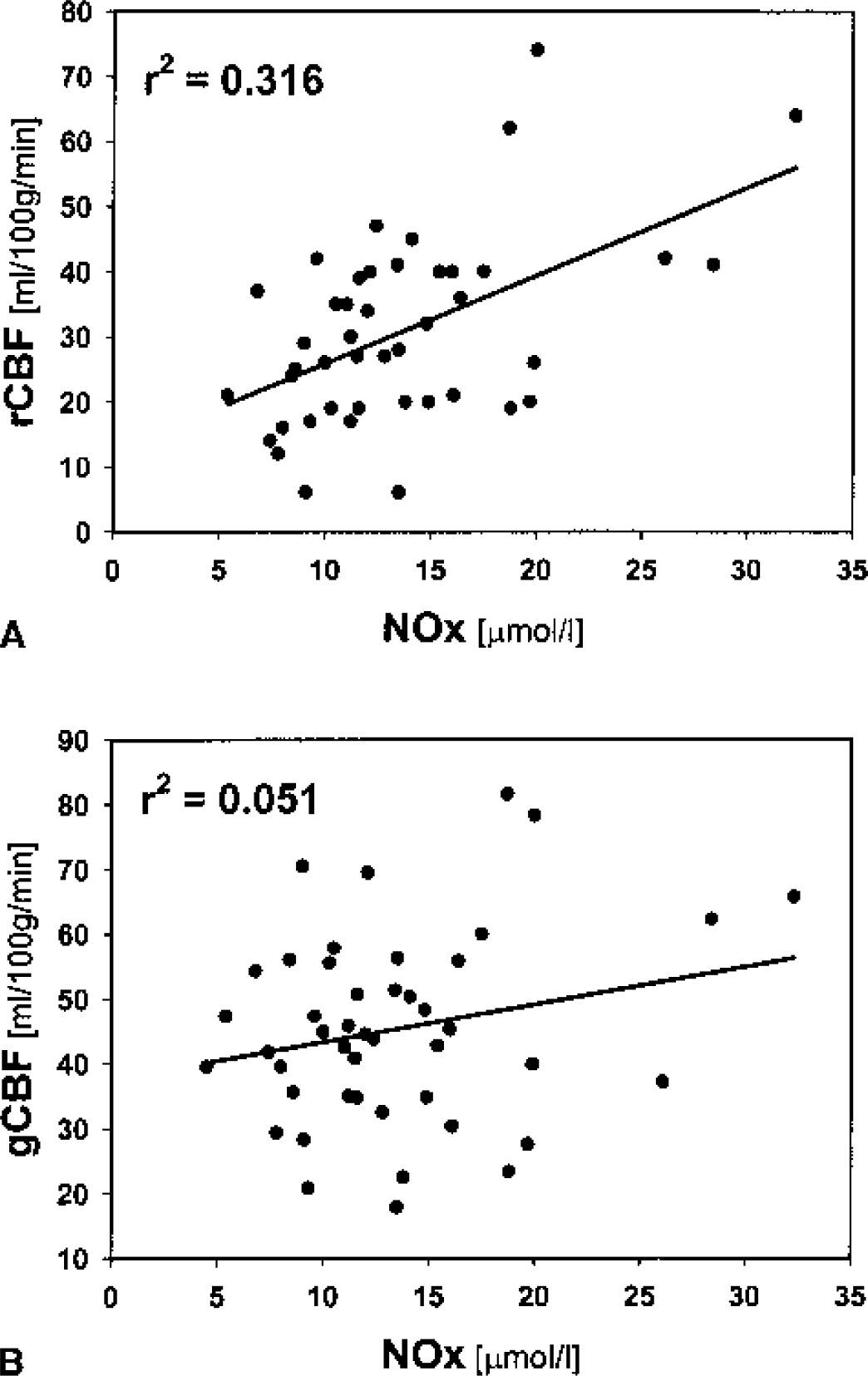
Correlation between regional cerebral blood flow (rCBF) and nitrate–nitrite values (NOx;
When regional CBF was >40 mL · 100 g−1 · min−1, the dialysate NOx values averaged 19.4 ± 7.9 μmol/L (mean + SD), compared with 9.1 + 2.2 μmol/L when regional CBF was <18 mL · 100 g−1 · min−1 (P < 0.05, Mann-Whitney rank sum test, followed by Dunn test). Dialysate NOx values were intermediate, averaging 12.7 ± 3.7 μmol/L when regional CBF was between 18 and 40 mL · 100 g−1 · min−1.
Other physiologic parameters and dialysate nitrate–nitrite levels
Monitored physiologic variables (intracranial pressure, cerebral perfusion pressure, SjvO2, PbtO2) did not have a close relation to dialysate NOx levels when the whole group was examined. However, in individual patients, changes in dialysate NOx concentrations did parallel other physiologic parameters during physiologic events.
The patient illustrated in Fig. 3 had a subdural hematoma evacuated on admission to the hospital. Brain tissue PO2, which had been normal or even elevated immediately postoperatively, gradually over a period of 36 h decreased to values <10 mm Hg. As the brain tissue PO2 decreased, the concentration of dialysate NOx also decreased from 14.1 μmol/L to 6.5 μmol/L at the time of the lowest PbtO2. An XeCT study at the time (Fig. 3—left scan) showed that the regional CBF in the area of the microdialysis and PbtO2 probes was 6.3 mL · 100 g−1 · min−1. Two days later when the PbtO2 had returned to normal values, regional CBF in the area of the microdialysis probe averaged 19.2 mL · 100 g−1 · min−1 (Fig. 3—right scan) and the dialysate NOx returned levels of approximately 12 μmol/L.
Two patients in the group developed refractory intracranial hypertension with severe global ischemia (Fig. 5). Both patients had a progressive drop in the dialysate NOx values to approximately 5 μmol/L as the cerebral perfusion pressure and PbtO2 decreased to zero.
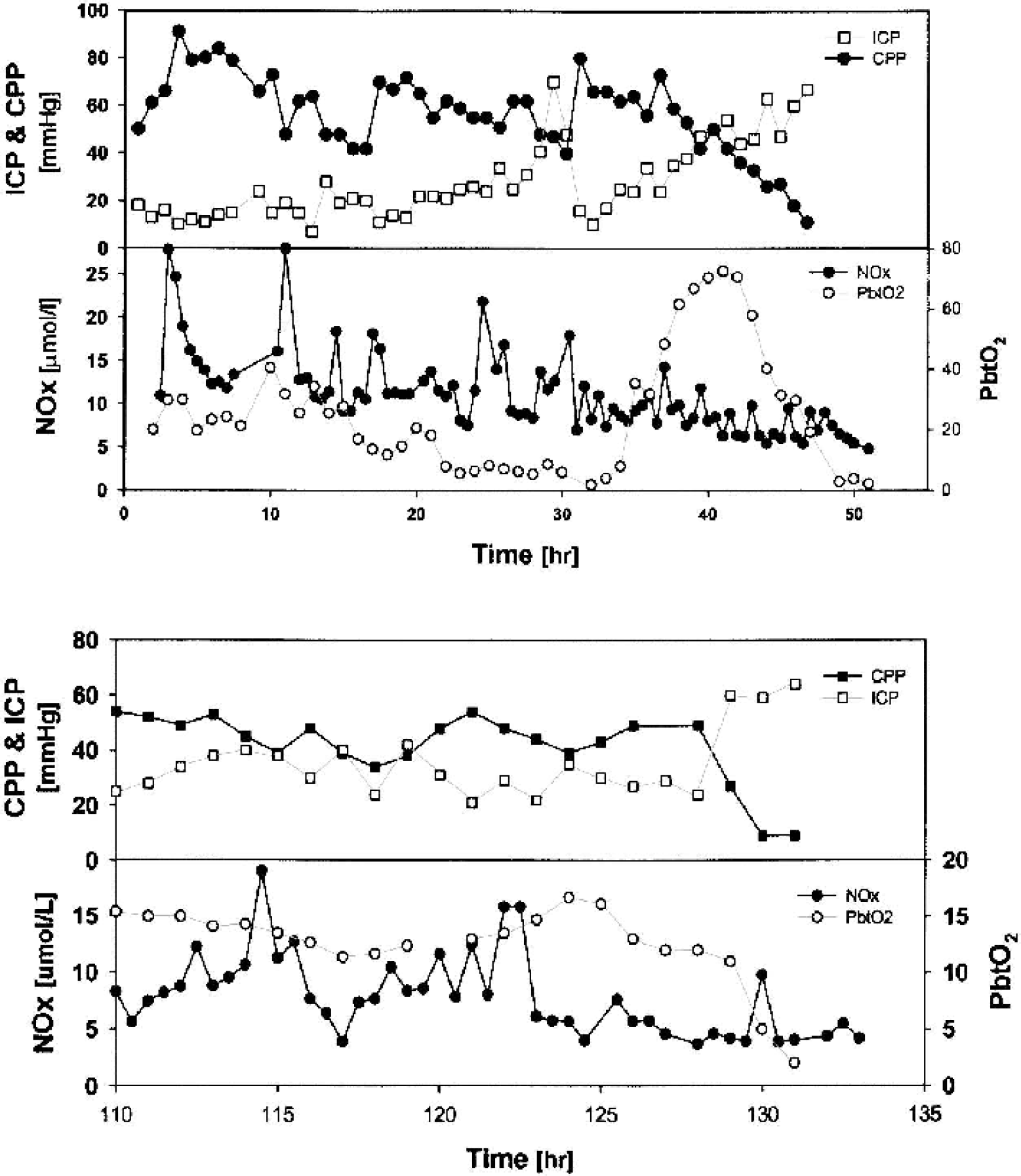
Example of individual patterns in nitrate–nitrite levels (NOx; closed circles in bottom graphs) in two patients who developed refractory intracranial hypertension. ICP, intracranial pressure; CPP, cerebral perfusion; PbtO2, brain tissue PO2.
DISCUSSION
In experimental brain injury models, a characteristic pattern of change in tissue NO levels and in tissue NOS activity has been observed. Immediately after the injury, there is a marked release in NO in the injured brain. Within a few minutes, NO levels fall to levels that are significantly lower than baseline, and remain that way for at least 3 h after injury (Cherian et al., 2000). Constitutive NOS activity has been found to be increased at 5 minutes after injury, but significantly reduced between 1 and 7 d after injury (Wada et al., 1998a). In contrast, inducible NOS activity has been observed to increase 3 d after injury and peak on day 7 after injury (Wada et al., 1998b).
The changes in dialysate NOx concentrations observed over time in the present study are similar to the changes that have been reported after human subarachnoid hemorrhage. Sakowitz et al. (2002) reported in 10 patients with aneurysmal subarachnoid hemorrhage that dialysate concentrations of NOx decreased from a high of 46.2 ± 34.8 μmol/L on day 1 to 23.5 ± 9.0 μmol/L on days 6 to 7 after hemorrhage. In this small group of patients, there was no relation between the level of NOx and the development of delayed ischemic neurologic deficits. However, Khaldi et al. (2001) reported a strong correlation between PbtO2 and dialysate NOx concentrations in patients with subarachnoid hemorrhage and suggested that NO may play a role in ischemia after subarachnoid hemorrhage.
In previous studies in TBI, the time course of changes in CSF NOx levels seems to be somewhat different than observed in the present study for dialysate concentrations of NOx. In one study in adults, CSF NOx concentrations were reported to peak at 20 h after injury at a concentration of 25 ± 6.2 μmol/L (Uzan et al., 2001). In pediatric TBI, CSF NOx concentrations have been reported to peak between 30 and 42 h after injury at a level of 26.4 ± 3.3 μmol/L (Clark et al., 1994). These investigators found no consistent relation between global CBF and the CSF level of NOx; however, patients who died of their brain injury had a significantly higher CSF level of NOx than patients who survived.
One important limitation of measuring the end-products of NO metabolism by the microdialysis technique is that no information is obtained about the source of the NO. It seems most likely that the dialysate values of NOx represent the sum of all sources of NO production. However, since the majority of the samples were collected during the first few days after injury, the constitutive forms of NOS would be expected to be the predominant source of NO. The pattern of dialysate NOx concentrations over time observed in the present human study is similar to course of constitutive NOS activity after experimental TBI. The highest values were observed initially after injury with a gradual decrease over the 5 d of monitoring. In addition, the major physiologic parameter that seemed to correlate with dialysate NOx concentrations in the present study was the regional CBF at the dialysis probe site. These findings may suggest that the dialysate NOx values are reflecting NO produced by endothelial NOS.
The dialysate NOx values could also have been influenced by systemic sources of NO production. In patients with polytrauma, plasma levels of NOx have been observed to be elevated particularly during the first 2 h after injury, and to return to normal by 24 h after injury (Gebhard et al., 1998). Since plasma levels of NOx were not obtained in the present study, the possibility of some influence of systemic sources of NO production to the early increase in dialysate NOx concentration is possible.
The source of NO production has important implications regarding the interpretation of these microdialysate findings. Nitric oxide produced by endothelial NOS has two potentially beneficial effects: maintenance of CBF by vasodilation and the inhibition of postischemic microvascular plugging by platelets and leukocytes (Gahm et al., 2000). In contrast, NO produced by neuronal NOS may have predominantly detrimental effects after TBI. Inhibiting neuronal NOS activity with the selective inhibitor 7-nitroindizole consistently has protective effects after TBI (Mesenge et al., 1996; Wada et al., 1998b). The effect of NO produced by inducible NOS is more controversial in models of TBI. Some studies suggest that treatment with aminoguanidine is protective (Wada et al., 1998b), whereas others have observed detrimental effects (Sinz et al., 1999).
Another limitation of measuring NOx concentrations using microdialysis is that the values obtained are not absolute concentrations of NOx in the brain tissue and are dependent on several factors including flow rate of the dialysate, and tortuosity of the tissue being monitored. For this reason, it is difficult to assess the values obtained relative to normal values, which in humans are limited. There are several studies in the literature where NOx concentrations have been measured in the CSF of healthy subjects (Zecca et al., 1998). Levels of approximately 10 to 15 μmol/L appear to be normally present in CSF. One study, using a lower dialysate flow rate than in the present study, has reported that NOx concentrations in normal brain average 36.7 ± 9.6 μmol/L (Sakowitz et al., 2002). In the present study, the levels of dialysate NOx that were associated with a relatively normal regional CBF were intermediate to these two values obtained from the literature, averaging 19.4 ± 7.8 μmol/L.
One final limitation of microdialysis measurements that is important to acknowledge is that the findings represent the levels of NOx only in a small volume of tissue surrounding the probe. In disorders such as TBI, where the injury can be very heterogeneous within the brain, it is important to understand where the probe is in relation to injured tissue. This characteristic of microdialysis monitoring probably explains why the dialysate NOx levels in the present study correlated only with the regional CBF measured at the probe site and not with global CBF values.
CONCLUSIONS
The measurement of NO metabolites in microdialysis samples is technically feasible and may provide valuable insights into both normal neurochemistry and neurochemical derangements in disease. Dialysate levels of NOx are highest initially after TBI and gradually decrease over time. A close relation was found between NOx extracellular levels and regional CBF at the microdialysis probe site, suggesting a role for NO in the abnormalities in CBF that occur after trauma.
Footnotes
Acknowledgments:
The efforts of the research fellows, residents, technical staff and nurses of the Ben Taub General Hospital are gratefully acknowledged.
