Abstract
The relative importance of hemodynamic factors in the pathogenesis of thrombotic or embolic stroke is unclear. Of particular therapeutic interest are those substances that facilitate vasodilation and the clearance of platelet aggregates in the compromised microvasculature. A likely contributor to these functions is nitric oxide because it is known to inhibit platelet aggregability and promote vascular relaxation. To investigate the involvement of nitric oxide in the hemodynamic changes after experimental ischemia, photochemically induced nonocclusive common carotid artery thrombosis (CCAT) was studied. CCAT is a rat model of unilateral carotid artery stenosis and platelet embolization to the brain. This study characterized the acute hemodynamic consequences of CCAT and the resultant pattern of platelet deposits with and without nitric oxide synthase inhibition by nitro-
It has been estimated that 85% of all strokes in humans are of thromboembolic origin (Camarata et al., 1994). Focal ischemic damage often results from the obstruction of a cerebral vessel by a downstream-lodged embolus or thrombus. The hemodynamic perturbations after an ischemic insult are important contributors to the final pathologic state of the stroke patient. Many substances act on the endothelium to regulate the cerebral circulation and contribute to the normalization of local cerebral blood flow (1CBF) after a perfusion deficit. Among these are a balance of endogenous vasoconstrictors such as thromboxane and serotonin, and vasodilators such as adenine nucleotides and nitric oxide (NO) (Vanhoutte and Houston, 1985; Faraci and Heistad, 1991).
Although many experimental ischemia models exist, few can examine the response of the brain to a platelet shower in the middle cerebral artery (MCA) territory with respect to a variety of endpoints such as the location of platelet deposits and the corresponding cerebral blood flow (CBF) changes. Nonocclusive common carotid artery thrombosis (CCAT) in the rat is a model of unilateral carotid stenosis caused by the adherence and aggregation of platelets to the vascular endothelium, which has been damaged by a photochemical reaction (Watson et al., 1987). The initial injury to the carotid endothelium results in rapid platelet embolization to the brain and downstream microvascular and neuronal changes (Futrell et al., 1988; Dietrich et al., 1993a,b).
Because NO has a role in maintaining normal vascular tone and in inhibiting platelet and leukocyte aggregability (Lancaster, 1992; Faraci and Brain, 1994; Iadecola et al., 1994) and because its role in cerebral ischemia is still controversial, we were interested in studying its involvement in this model. Since it is generally accepted that both the duration and extent of the blood flow reductions seen by the ischemic brain determine the long-term viability of the tissue (Heiss, 1993), the vascular actions of NO can potentially be important in brain injury. Also, because most ischemia models use mechanical occlusion of a vessel, the effects of platelet emboli that are superimposed on the CBF reductions have not been studied. Specifically, it is not known to what degree platelet deposits contribute to perfusion or functional deficits and what the acute hemodynamic and subsequent pathologic outcomes after platelet-derived thrombosis are in the rat.
It has been demonstrated that nitric oxide synthase (NOS) inhibition with nitro-
METHODS
Animal groups
Male Wistar rats weighing between 300 and 350 g were randomly assigned to five groups: 1) CCAT, 2) CCAT +
Surgery
Rats were food deprived 12 hours before surgery. Common carotid artery thrombosis was produced using the photochemical technique (Watson et al., 1987, Wester et al., 1992). Briefly, the animals were anesthetized with halothane, intubated, and artificially ventilated using a rodent respirator. Rats were maintained on a mixture of 70% nitrous oxide, 30% oxygen, and 1% halothane. Femoral venous and arterial catheters were inserted to administer drugs and measure blood gases and blood pressure. Muscle relaxation was achieved by injecting pancuronium bromide. Under a Zeiss operating microscope, the right common carotid artery was exposed by blunt dissection and retraction of the surrounding musculature. The vagus nerve was carefully separated from the common carotid artery, and the artery was freely suspended in a saline bath.
A tunable argon dye laser (wavelength 562 nm, peak power 325 mW) was focused onto the right common carotid artery for 10 minutes. Simultaneously, the photosensitizing dye rose bengal (15 mg/mL in 0.9% saline) was injected intravenously into the circulation over a period of 90 seconds at a dose of 15 mg/kg. This irradiation procedure has been shown to produce 50% to 75% stenosis (Wester et al., 1992). Depending on the group to which the rat was assigned, the drug (15 mg/kg
Indium platelet labeling
The method for 111In labeling of platelets has been described previously (Dietrich et al., 1993a). To summarize, blood (30 mL) was collected from the femoral arteries of two donor rats and placed in a syringe containing acid citrate dextrose (ACD) solution. After centrifuging the donor blood for 10 minutes at 200g, the platelet-enriched supernatant was again centrifuged for 10 minutes at 1600g. The platelet pellet was resuspended in 1 mL ACD/saline. Tropolone, 25 μg, was added to 200 μCi 111In chloride and vortexed. The platelets then were added to the 111In–tropolone and allowed to incubate at room temperature for 30 minutes in an additional 1 mL of ACD/saline. The platelets were again centrifuged for 10 minutes at 1600g and were resuspended in 2 mL ACD/saline solution. From this solution, 1 mL of labeled platelets were injected into the recipient rat and allowed to circulate for 30 minutes before irradiation or sham procedures.
[14C]iodoantipyrine autoradiography
Thirty minutes or 24 hours after the irradiation was completed, blood flow measurements were performed using the [14C]iodoantipyrine technique (Sakurada et al., 1978). 14C-iodoantipyrine (20 μCi/1 mL saline; specific activity 40 mCi/mmol; New England Nuclear) was delivered intravenously at a constant rate for a period of 45 seconds as arterial blood samples were collected from the femoral arterial catheter. A computer interface recorded the timing of sample collection. On completion of the isotope infusion, the animals were decapitated. The brains were quickly removed, frozen in liquid nitrogen, and stored at −4°C until they were processed.
Visualization of labeled platelets and cerebral blood flow
Frozen sections (20 μm) were cut the following day on a cryostat and exposed to Kodak SB-5 x-ray film. The 111In-labeled platelets were visualized after exposing the film for 5 days. After 1 month, the 111In had decayed and a second exposure for 10 days detected the appearance of [14C]iodoantipyrine blood flow changes.
Image processing
Coronal autoradiographic films from individual animals were digitized with eight-bit precision using a CCD camera (Xillix Technologies Corp., Vancouver, Canada), and eight coronal sections (2.2 and 0.7 mm anterior and 0.3, 1.3, 2.3, 3.3, 5.3, and 6.3 mm posterior to bregma) were chosen from each film. Films of both blood flow and platelets were digitized in the same manner. Images were acquired at 70-μm resolution, and 14[C]-methylmethacrylate standards placed on each film also were digitized. Image files were subsequently transferred to a computer cluster consisting of a DEC 3600 minicomputer and 3200 VaxStations (Digital Equipment Corp.) for registration and averaging of corresponding coronal sections in each experimental group. A 250 AlphaStation (Digital Equipment Corp.) was used for image display and photography.
The theories of image registration and averaging have been derived (Zhao et al., 1993) and validated (Zhao et al., 1995). In brief, we mapped the corresponding coronal sections into a preselected template. In our study, we digitized a published functional–anatomic atlas of the coronally sectioned rat brain (Zilles, 1985) at corresponding levels and implemented software to map digitized autoradiographic images into these templates (Alexis et al., 1996). After the mapping process, group mean and SD images for each coronal level were produced by simple algebraic manipulation. The atlas was used to specify and standardize regions of interest for analysis across animals.
Labeled platelet composite images were produced by summing the platelets from the animals in each group. We designed the following procedure for counting pixel numbers of platelets. First, a threshold value for each brain section was obtained so that only labeled platelets could be visualized. This threshold level was verified by examining the original autoradiograms over a light box for the presence of platelets. Second, using software that counted the pixels falling within the given threshold range, we obtained the number of pixels representing platelets for each half of the section. The final step was to separately calculate, on two hemispheres, the number of pixels for all coronal levels to produce the total number for each hemisphere.
Data analysis and statistics
For each individual animal, regions of interest were analyzed for platelet counts (represented as the number of pixels) and mean CBF. The following regions of interest were selected: frontal cortex (rostral and caudal), parietal cortex (medial and lateral), hippocampus, entorhinal cortex, gustatory cortex, occipital cortex, corpus callosum, and internal capsule. In addition to being common locations of histopathologic damage in this model, these areas are of functional significance in the rat (Alexis et al., 1995; Stagliano et al., 1997). Each hemisphere was analyzed separately. We also determined on a pixel-by-pixel basis relations between blood flow reductions and focal platelet accumulation. For this purpose, a circular cursor with a diameter of 800 μm was placed over or immediately adjacent to a platelet focus. Thus, 1CBF readings could be directly compared in regions with and without indium-labeled platelets.
Statistical analyses were performed using repeated measures analysis of variance (ANOVA) across the five groups. The ANOVA was followed by Bonferroni post hoc comparison. P < 0.05 was taken to be significant. Values are presented as mean ± SD.
RESULTS
Cerebral blood flow
At 1 hour after CCAT, the thrombosed hemisphere (right) in both treated and nontreated experimental groups was significantly hypoperfused compared with sham controls (P < 0.01). These flow deficits were present in all cortical regions analyzed and also were evident in the hippocampus and white matter tracts. In addition, 1CBF was significantly depressed in many regions of the contralateral hemisphere (see Table 1 for individual regions of interest). Figure 1 shows mean CBF images for all coronal levels analyzed for the three groups. Although no between-group differences were significant, a strong trend existed for reduced perfusion in the
Mean CBF values at 1 hour after CCAT (mL/g/min)
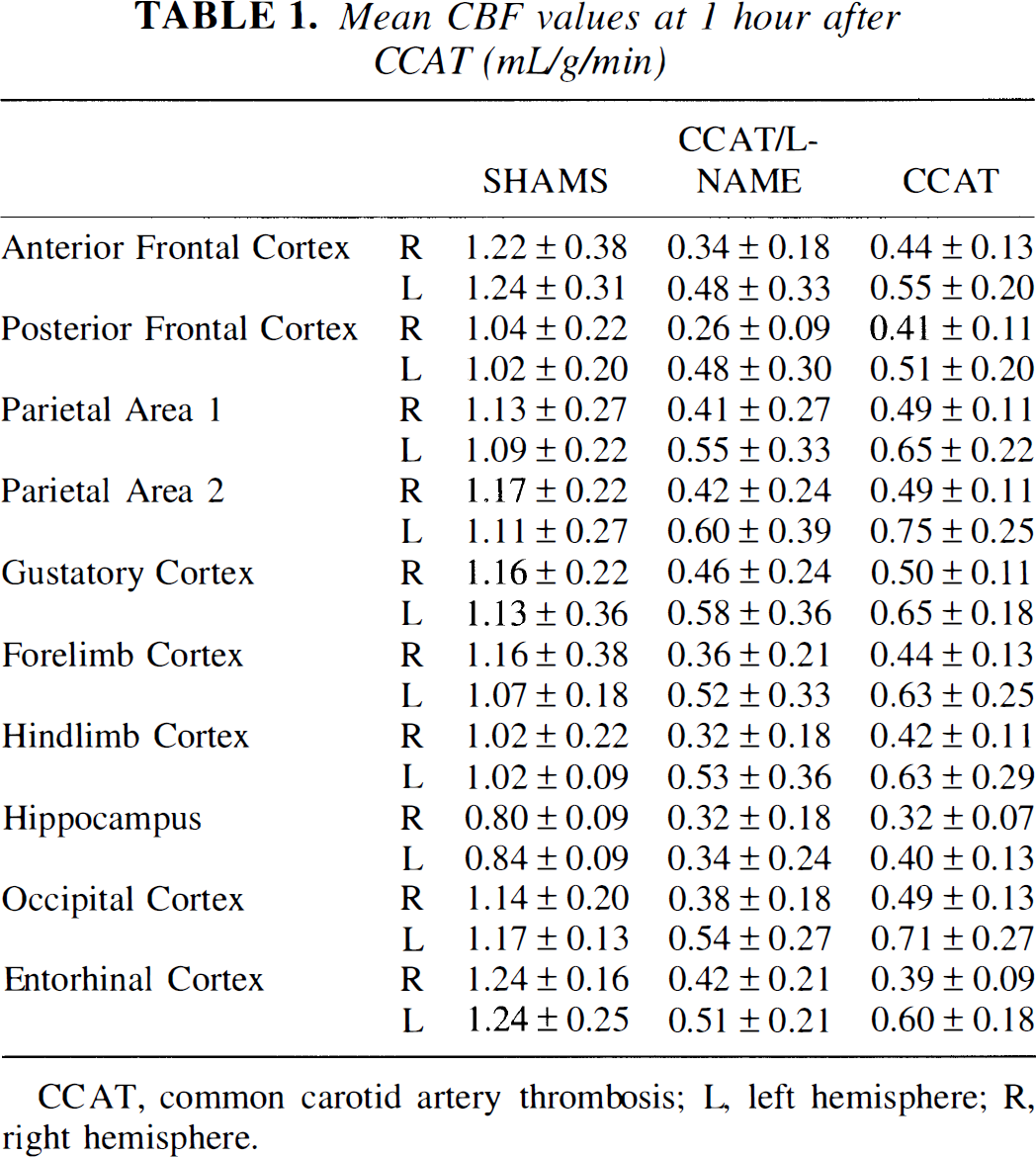
CCAT, common carotid artery thrombosis; L, left hemisphere; R, right hemisphere.
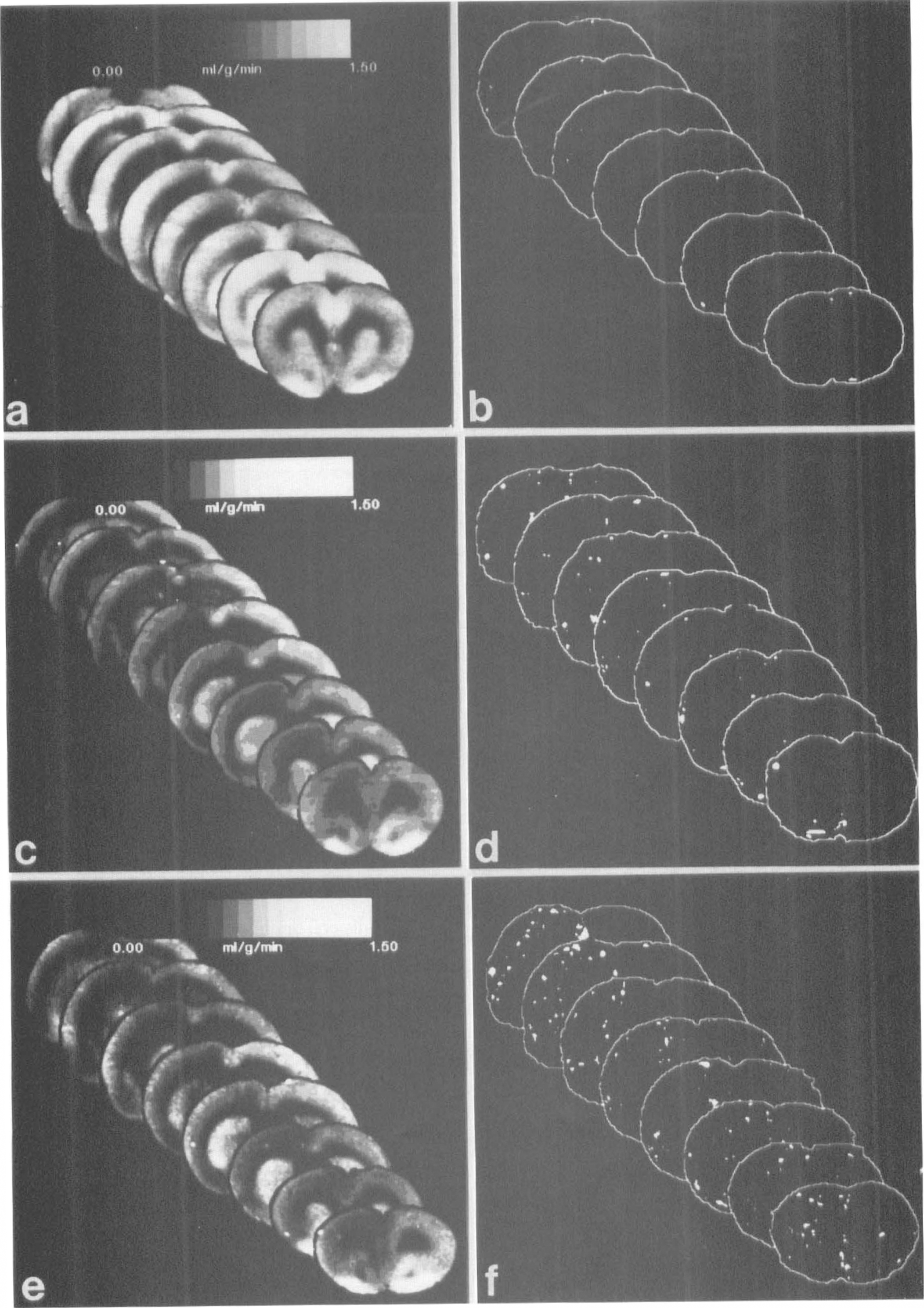
Group CBF averages for eight coronal levels (left column) and labeled platelet composite images (right column) of sham
At 24 hours after thrombosis, CBF in untreated CCAT rats returned to sham levels (Table 2) whereas both
Mean CBF values at 24 hours (mL/g/min)
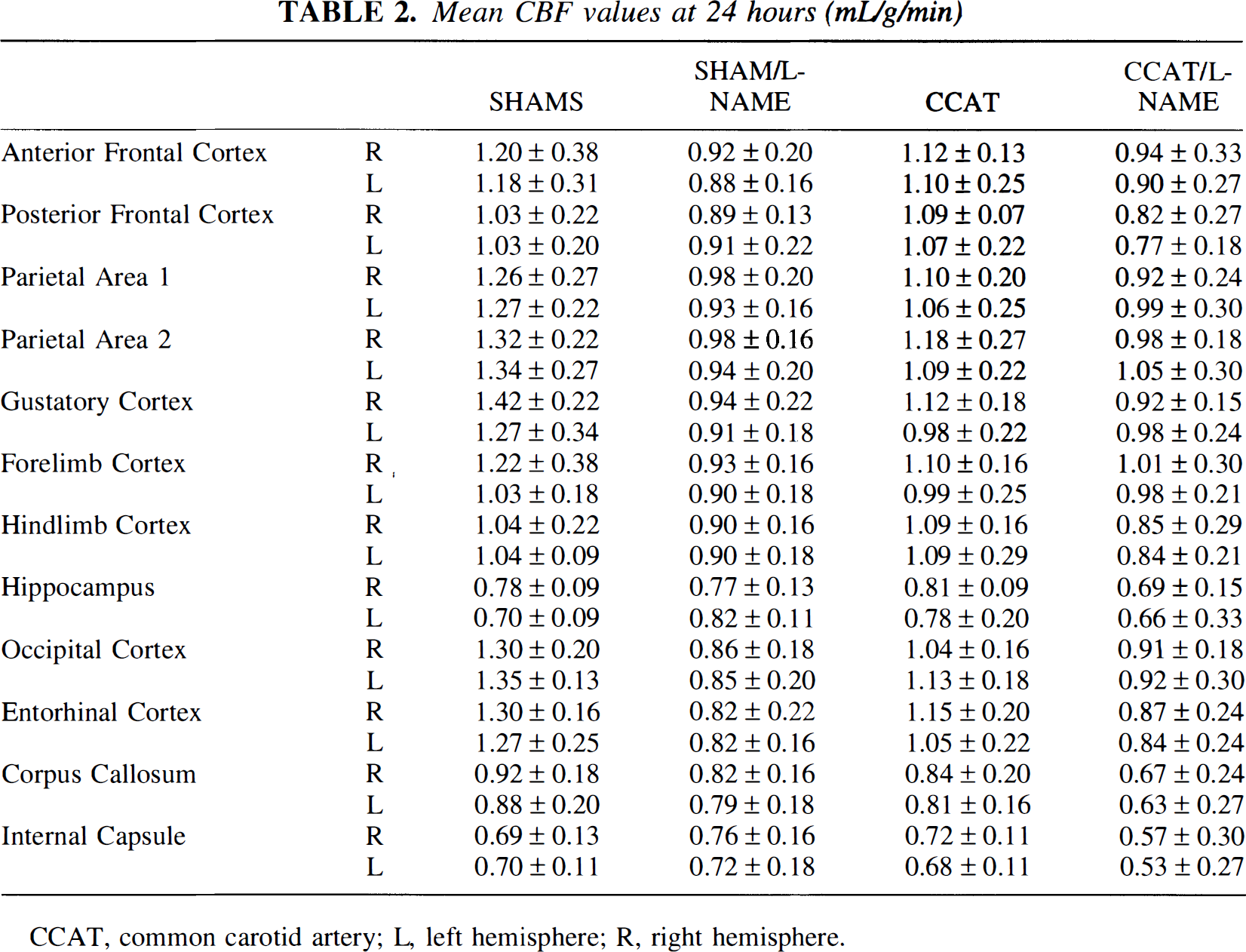
CCAT, common carotid artery; L, left hemisphere; R, right hemisphere.

Group CBF averages for eight coronal levels at 24 hours after thrombosis for sham-operated
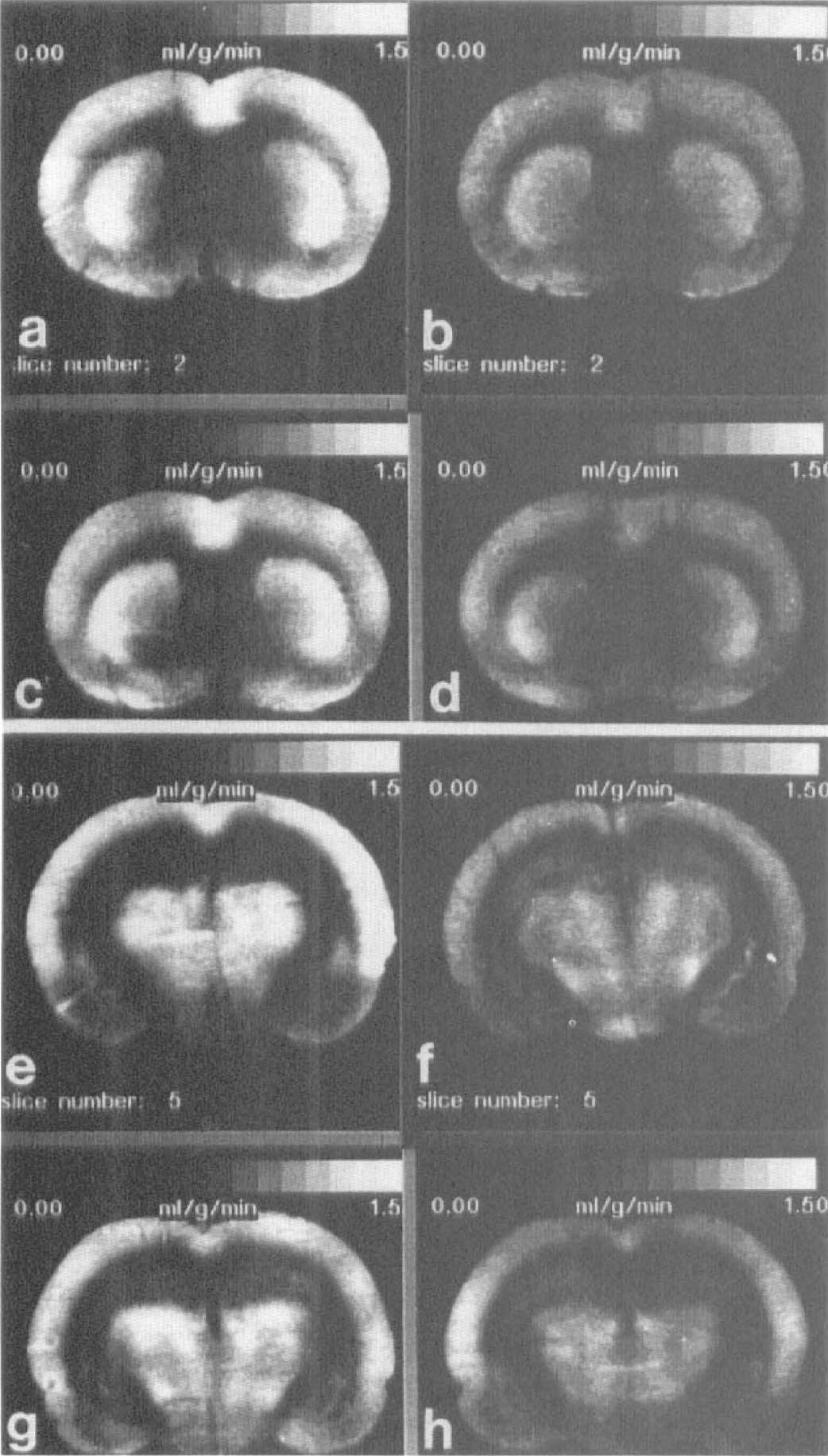
Mean CBF images for four groups at 24 hours after CCAT at two coronal levels, 0.3 and 3.3 mm posterior to bregma, where white matter was significantly affected.
Labeled platelets
Treatment with
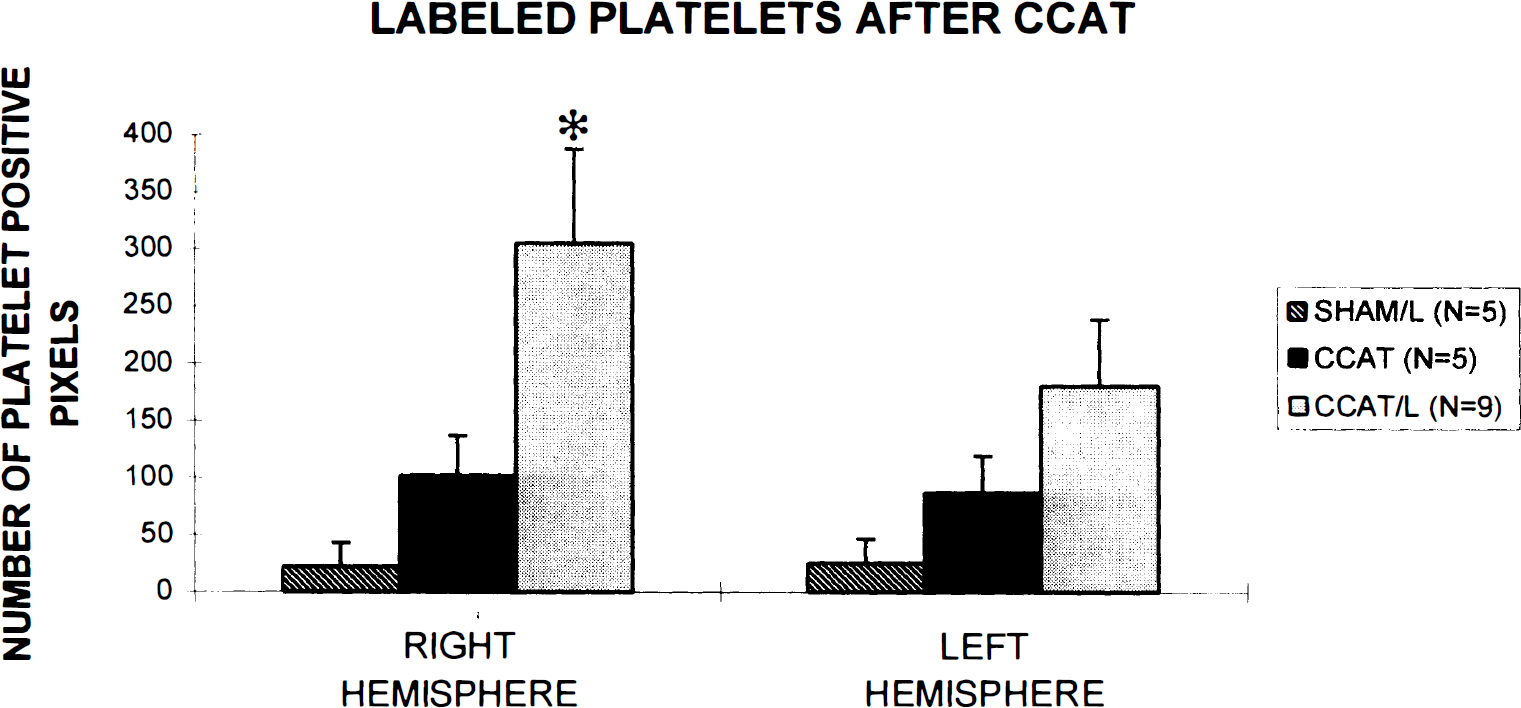
Total platelet counts in the thrombosed hemisphere (right) and contralateral hemisphere (left) at 1 hour after CCAT. *Indicates significantly different from CCAT at shams.
Regional analyses yielded two areas significantly affected by NOS inhibition: the anterior frontal (P < 0.05; ANOVA; compared with all groups) and occipital cortices (P < 0.05; ANOVA; compared with shams) in the hemisphere ipsilateral to thrombosis. Figure 1 shows labeled platelet composites for the three groups.
Correlation of platelet deposits with cerebral blood flow changes
Using the same regions of interest listed in Tables 1 and 2, platelet pixel counts and mean CBF were measured for each animal at each level. The two values were correlated using a Spearman's ranking correlation coefficient. No correlation was observed between platelets and 1CBF in any of the groups tested (R = 0.0069). In addition, no significant differences were demonstrated when we correlated (on a pixel-by-pixel basis) relations between blood flow reductions and focal platelet accumulation. For example, in the 1-hour CCAT/
DISCUSSION
This study determined the effect of nonspecific NOS inhibition on regional patterns of acute platelet accumulation and acute and subacute cerebral blood flow changes after thromboembolic stroke in the rat. These experiments make use of a double label autoradiographic strategy to visualize platelets and 1CBF in the same brain section. Our data indicate that at approximately 1 hour after a unilateral thromboembolic insult, labeled platelets are distributed in both brain hemispheres and that bilateral moderate reductions in 1CBF are present. However, no direct correlation appears to exist between the quantity or size of platelet deposits and the degree of flow reductions. In the more chronic setting (24 hours), untreated thrombosed animals recovered cerebral blood flow to normal levels, whereas
Common carotid artery thrombosis is a model of mild stroke and transient ischemic attack (Dietrich et al., 1994). Many experimental stroke models result in robust, sustained histopathologic, hemodynamic, and metabolic consequences (Back et al., 1995). It was of interest to us to determine whether a mild platelet-mediated stroke model (which has been shown to produce transient functional deficits, blood–brain barrier permeability increases, and heterogeneous hemodynamic disturbances) (Dietrich et al., 1991, 1993a,b; Alexis et al., 1995) resulted in lasting blood flow changes and if the generation of NO was critical to the restoration of normal flow. Previous work with this model has shown that the dysfunction caused by CCAT can be modified by pharmacologic manipulation of the NO pathway (Stagliano et al., 1997).
The acute consequences of CCAT on labeled platelet deposition and changes in CBF are widespread. The data indicate that after unilateral carotid stenosis and subsequent platelet embolization to the brain, labeled platelets and moderate reductions in CBF are present bilaterally. This is consistent with data from patients with unilateral carotid stenosis where bilateral embolic signals were detected using transcranial Doppler ultrasound (Georgiadis et al., 1994). When
Our original hypotheses were that there would be a positive correlation between local perfusion deficits and platelet deposits after CCAT, and that additional vascular constriction would increase platelet accumulation and perfusion in a regionally specific manner. However, with or without drug administration, there did not appear to be a spatial relationship between platelet deposits and the severity of the blood flow reduction. Other researchers have attempted to link platelet activation to stroke severity (Joseph et al., 1992) or platelet aggregation to neurologic outcome (Galante et al., 1993), but no correlations were found. In addition to local occlusion of a vessel by a platelet embolus, humoral factors might be generated that can cause bilateral circulatory disturbances. In an experiment in which blood collected downstream from the forming carotid thrombus was injected into a naive rat, widespread, rapidly resolving blood flow changes were reported (Dietrich et al., 1991). Subsequent neurochemical studies documented elevated plasma levels of serotonin immediately after CCAT (Wester et al., 1992). Therefore, vasoactive substances in the blood, possibly released from activated platelets or resulting from endothelial disruption, are potent modifiers of global CBF and deserve further study.
There were no statistically significant structural or regional differences in 1CBF in the untreated or the
Flow changes after CCAT normalized within 24 hours. Rapid recovery of profound flow deficits have been shown in patients to result from migration of an embolus and subsequent arterial recanalization (Baird et al., 1995). In these patients, minimal neurologic deficits and tissue damage are detectable. The transient nature of platelet accumulation in the brain also has been reported after CCAT (Dietrich et al., 1993a). It may not be surprising that a relatively mild insult such CCAT does not cause long-term flow changes; what cannot be elucidated from the regional analyses performed in this work are the low-flow zones at sites of focal infarction and irreversible damage. Because the location of infarcts is unpredictable in this model, computer averaging and statistical analysis are not likely to detect these no-flow regions that may correlate with sites of irreversible damage.
The chronic 1CBF data in this report indicate that even in sham animals treated with
The finding that
Take together, the results of this study imply that nonspecific NOS inhibition with
