Abstract
Regional cerebral blood flow (rCBF) was studied in type III nitric oxide (NO) synthase (endothelial, eNOS) mutant and wild type mice during mechanical whisker stimulation before and after nitro-L-arginine (L-NA) superfusion using the closed cranial window technique. rCBF increased equally in cortical barrel fields in both strains during stimulation, as measured by laser Doppler-flowmetry, and was inhibited by L-NA superfusion (1 mM) in both groups. Hence, coupling of blood flow and metabolism appears neuronal NOS- (nNOS) but not eNOS-dependent in cortical barrel fields of the mouse.
Nitric oxide (NO) is an attractive candidate for coupling of blood flow and metabolism due to its rapid synthesis and release from neurons, short half-life, and anisotropic as well as vasoactive properties (Edelman and Gaily, 1992; for review, see Iadecola, 1993). In rats and mice NO synthase (NOS) inhibitors, such as NG-nitro-L-nitro-arginine methyl ester (L-NAME) or nitro-L-arginine (L-NA) attenuate regional cerebral blood flow (rCBF) augmentation during somatosensory stimulation in most studies (Irikura et al., 1994; Dirnagl et al., 1993; Northington et al., 1992), although the relative importance of the two constitutively expressed isoforms could not be determined using these nonselective inhibitors.
Here, we present evidence that type I NOS (nNOS), but not type III NOS, participates in rCBF coupling following mechanical whisker stimulation. In these experiments, we document that L-NA superfusion blocks the augmented rCBF response in eNOS mutant mice. Because only NOS type I is expressed in eNOS mutant mice, this neuronal isoform modulates rCBF coupling in barrel field cortex.
METHODS
Male, 20–28 g SV-129 wild-type mice (n = 11) and mice lacking the eNOS gene (mutant mice) (n = 8) were housed under diurnal lighting conditions and allowed food and tap water ad libitum. Mice were initially anesthetized with 2% halothane and intubated transorally. Halothane anesthesia was then discontinued and urethane administered (750 mg/kg in wild-type and 850 mg/kg in eNOS knockout mice, i.p.); eNOS knockout mice required a higher dose to stabilize anesthesia. Mice were paralyzed (pancuronium bromide, 0.4 mg/kg, i.p., q 45 min) and mechanically ventilated (70% N2O and 30% O2, vol/vol). Mean arterial blood pressure (MABP) (ETH 400 transducer amplifier, ADInstruments, Milford, MA, U.S.A.), endtidal CO2, and rectal temperature were monitored, as described (Dalkara et al., 1995).
A closed cranial window was placed, and artificial cerebrospinal fluid (aCSF) was continuously superfused (0.5 ml/min, pH 7.35–7.45) as described before (Ma et al., 1996). The window was sealed by cover glass (12 mm diameter) using dental acrylic. Intracranial pressure was maintained at 5–8 mm Hg by adjusting the outlet tube to an appropriate height.
rCBF was measured by a laser-doppler flowmeter (LDF) (LaserFlo, BPM403A, Vasamedic) and averaged over 5 s, using a flow probe of 0.8 mm diameter placed 3 mm posterior and 4 mm lateral to the bregma. The probe was placed on the glass cover (∼0.15 mm thick) and kept away from the large pial vessels. The specificity of the rCBF response was tested by stimulating the ipsilateral vibrissae.
Contralateral vibrissae were cut to an equal length of 0.5 cm and stimulated manually using a cotton swab at 2–3 Hz for 60 s. Consecutive 60-s stimulations were spaced 4–5 min apart and repeated two-to-three times to provide an average. We repeated whisker stimulations two-to-three times, before and 60 min after L-NA superfusion. Data are presented as an average for each period.
Mice were stabilized for 30 min after window placement. No significant blood pressure changes were recorded during tail pinch. (L-NA, 1 mM), or nitro-D-arginine (D-NA) was then superfused for 20 min. The L-NA-containing superfusate was kept under the window for an additional 40 min, after which stimulation was repeated. Concentration and duration of L-NA application were sufficient to inhibit acetylcholine (ACh)-induced vasodilation by ∼80% and NOS activity by >70% in subjacent cortices in both groups (Ma et al., 1996; Meng, unpublished observations).
Data are expressed as mean ± SD rCBF increase during 60 s whisker stimulation. Wild-type and eNOS mutant mice groups were compared by Student's t-test. rCBF responses of control and 60 min L-NA groups were compared by the paired t-test. Correlation analysis was performed for changes in resting rCBF and the whisker response inhibition. Probability values of less than 1/20 were considered significant.
RESULTS
Physiological parameters were not different between groups under baseline conditions (Table 1).
Physiological parameters
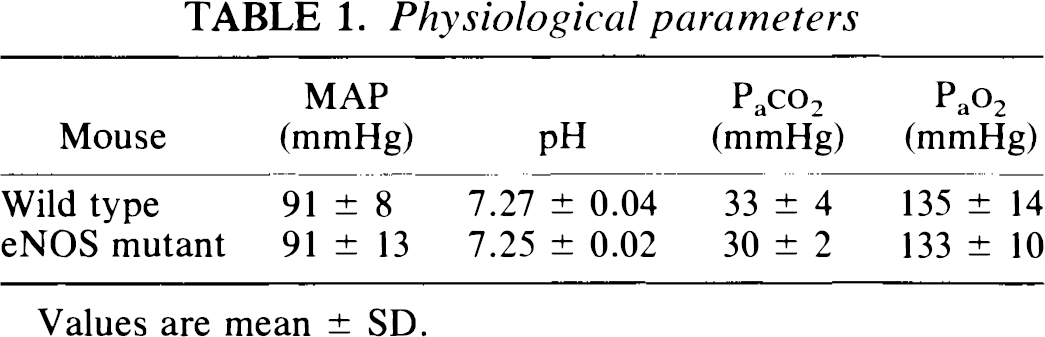
Values are mean ± SD.
rCBF increased by 24 ± 6 and 25 ± 4% in wild-type and eNOS mutant mice, respectively, during whisker stimulation (Fig. 1). L-NA superfusion attenuated the increase by ∼40% (wild type) and ∼50% (eNOS knockout) (Fig. 1). Maximum inhibition was achieved 60 min after superfusion.
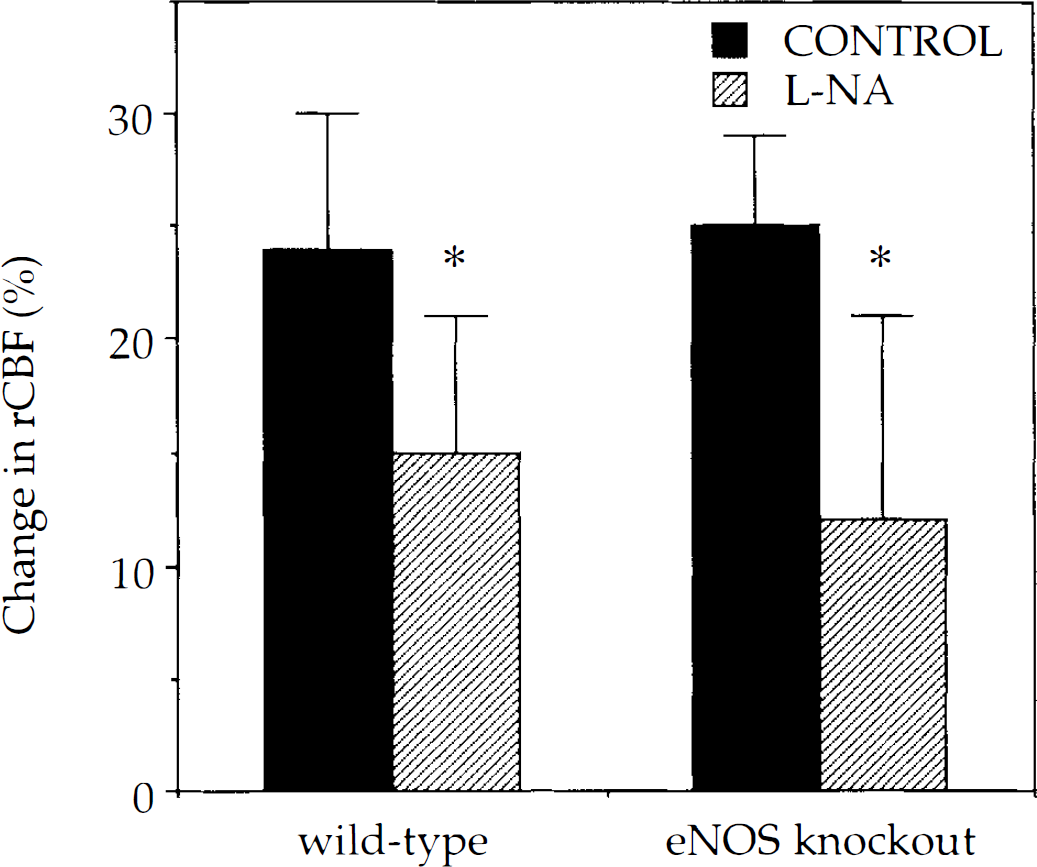
L-NA inhibited the rCBF response to mechanical vibrissal stimulation in both wild-type (n = 11) and type III NOS (n = 8) mutant mice. L-NA, 1 mM, was superfused for 60 min in a closed cranial window placed over the barrel cortex. *, p < 0.05 versus control, paired t-test. Values are mean ± SD.
Baseline rCBF decreased by 17 ± 23% after L-NA in wild type mice (p < 0.05 versus baseline); however, it increased by 16 ± 34% in eNOS mutant mice (p > 0.05 versus baseline). There was no correlation between change in baseline and degree of rCBF inhibition during whisker stimulation (r2 = 0.20, p = 0.26) in eNOS mutant mice. The maximum change in resting rCBF was reached within 30 min after L-NA superfusion in all strains. L-NA superfusion did not change MABP.
D-NA, 1 mM, (n = 3) superfusion did not affect resting rCBF nor the magnitude of the response to vibrissal stimulation (data not shown).
DISCUSSION
We used genetically engineered mice to establish the importance of the neuronal versus endothelial isoform in the coupling between blood flow and brain metabolism during vibrissal stimulation.
The eNOS mutant mouse was developed by homologous recombination and replacement of a gene segment encoding the NADPH ribose and adenine binding sites by a neomycin resistance gene (Huang et al., 1995). Proper disruption of the eNOS gene was documented by Southern and Western blotting techniques and by functional studies showing that aortic rings were resistant to ACh-induced dilation. Mutants are hypertensive when compared to wild-type mice, although under deeper anesthesia, the blood pressure is comparable to wild-type (Table 1). nNOS expression however, is robust, as documented by NOS assay—[3H] L-arginine to [3H] L-citrulline conversion—and by binding of [3H]L-NA in autoradiographic studies (Huang et al., 1995; Hara, unpublished observations).
rCBF increased similarly in wild-type and eNOS mutant mice and was inhibitable to the same extent by L-NA, but not D-NA, superfusion during mechanical whisker stimulation. We presume that nNOS inhibition decreases the rCBF response in the eNOS mutant mouse. The findings are consistent with recent results showing that nNOS mutant mice were resistant to the effects of L-NA during whisker stimulation (Ma et al., 1996). Apparently, nNOS mutants develop non-NO-dependent mechanisms mediating flow-metabolism coupling in the presence of normal eNOS expression. Hence, these findings mitigate against an important role for eNOS in the coupling response in nNOS mutants as well. Together, the findings strongly suggest a primary role for nNOS in metabolism/flow coupling during cortical barrel field activation. A potential role for NOS in cortical activation was suggested by a report that topical L-NA suppresses cortical-evoked potentials in response to sciatic nerve stimulation (Ngai et al., 1995). The contribution of this action to the rCBF inhibition however, remains to be determined.
L-NA decreased resting rCBF in wild-type mice (Ma et al., 1996) as reported in other species (Irikura et al., 1994), whereas it consistently increased resting rCBF and dilated pial vessels in eNOS knockout mice (Ayata, unpublished observations). These and previously published data (Kovach et al., 1992) suggest that basal production of NO influences cerebral vascular tone in wild-type mice and that the endothelium is an important source (Huang et al., 1995). nNOS may also contribute to resting CBF since pial arteriolar constriction was significantly weaker in nNOS mutant than in wild-type after L-NA superfusion (Ma et al, 1996).
Gene deletion strategies provide an essential alternative approach to drugs when pharmacological tools are unavailable, nonselective or have multiple actions that limit their use. By applying this technology, we have now shown that nNOS is the predominant isoform that either couples brain metabolism to blood flow or acts as an important modulator of this response.
Footnotes
Acknowledgment:
Supported by Massachusetts General Hospital Interdepartmental Stroke Program Projects NS10828 NS33335 (P.L.H.) and an Unrestricted Research Award in Neuroscience (M.A.M.).
