Abstract
The stage of neurogenesis can be divided into three steps: proliferation, migration, and differentiation. To elucidate their detailed relations after ischemia, the three steps were comprehensively evaluated, in the subventricular zone (SVZ) through the rostral migratory stream (RMS) to the olfactory bulb (OB), in adult gerbil brain after 5 minutes of transient forebrain ischemia. Bromodeoxyuridine (BrdU), highly polysialylated neural cell adhesion molecule (PSA-NCAM), neuronal nuclear antigen (NeuN), and glial fibrillary acidic protein (GFAP) were used as markers for proliferation, migration, and differentiation, respectively. The number of BrdU-labeled cells that coexpressed PSA-NCAM and the size of PSA-NCAM–positive cell colony increased in the SVZ with a peak at 10 d after transient ischemia. In the RMS, the number of BrdU-labeled cells that coexpressed PSA-NCAM increased, with a delayed peak at 30 d, when the size of RMS itself became larger and the number of surrounding GFAP-positive cells increased. In the OB, BrdU + NeuN double positive cells were detected at 30 and 60 d. NeuN staining and terminal deoxynucleotidyl dUTP nick-end labeling staining showed no neuronal cell loss around the SVZ, and in the RMS and the OB after transient ischemia. These findings indicate that transient forebrain ischemia enhances neural stem cell proliferation in the SVZ without evident neuronal cell loss, and has potential neuronal precursor migration with activation of GFAP-positive cells through the RMS to the OB.
Keywords
Persistent neurogenesis occurs in discrete regions of the adult brain, including the subventricular zone (SVZ) of the lateral ventricle (Altman, 1969; Doetsch et al., 1997; Kornack and Rakic, 2001a; Pencea et al., 2001) and the subgranular zone of the hippocampal dentate gyrus (Altman and Das, 1965; Kaplan and Hinds, 1977; Bayer et al., 1982). In the SVZ of adult rodent brain, new neuronal progenitor cells are normally born, and migrate through the rostral migratory stream (RMS) to the olfactory bulb (OB). In the RMS, these young neurons form elongated chain aggregates through glial tunnels formed by processes of astrocytes (Lois et al., 1996). After reaching the core of the OB, they move radially into the granular and the periglomerular layer to differentiate into mature neurons (Luskin, 1993; Lois and Alvarez-Buylla, 1994). Such a cell proliferation in the SVZ can be stimulated by addition of exogenous substances including epidermal growth factor, tumor growth factor-α, and fibroblast growth factor-2 (Morshead et al., 1994; Craig et al., 1996; Kuhn et al., 1997; Tropepe et al., 1997). Because the SVZ provides progenitor cells in normal adult brain, which can be activated by pharmaceutical reagents, neurogenesis is now expected to have a potential for compensation and recovery of neural functions that were lost by central nervous system disorders.
Because ischemic stroke decreases neural functions due to brain cell loss, neurogenesis after ischemia should be important for compensation and recovery of those functions in patients who have had a stroke (Abe, 2000). In hippocampal dentate gyrus, neurogenesis increased after transient forebrain ischemia (Liu et al., 1998; Yagita et al., 2001) or focal cerebral ischemia (Jin et al., 2001). Our previous report showed changes of three steps of neural stem cell development (proliferation, migration, and differentiation) in the gerbil dentate gyrus after transient ischemia (Iwai et al., 2002). Simple increase of neural stem cells in the SVZ was reported after transient focal ischemia (Jin et al., 2001; Zhang et al., 2001). A comprehensive study, however, on such three steps of stem cell development from the SVZ to the OB has never been reported after transient forebrain ischemia.
A thymidine analog, 5-bromodeoxyuridine (BrdU), can be used as an early marker of cell proliferation in normal adult rodent brain (Kuhn et al., 1996). Highly polysialylated neural cell adhesion molecule (PSA-NCAM) is a glycoprotein that mediates cell–cell adhesion and recognition, which is present on the surface of the migrating cell in the RMS (Bonfanti and Theodosis, 1994; Doetsch et al., 1997). Doublecortin (DCX), a microtubule-associated protein, is also important for neural cell migration in normal development, which is expressed strongly in migrating neural precursors within the RMS (Francis et al., 1999; Gleeson et al., 1999). Neuronal nuclear protein (NeuN) and glial fibrillary acidic protein (GFAP) are used as markers of differentiated mature neuron and astrocytes, respectively. In the present study, therefore, we evaluated the three steps of neurogenesis from the SVZ through the RMS to the OB in gerbil brain after transient forebrain ischemia using BrdU, PSA-NCAM, DCX, NeuN, and GFAP as markers for proliferation, migration, and differentiation, respectively.
MATERIALS AND METHODS
Surgical preparations
The experimental protocol and procedures were approved by the animal committee of Graduate School of Medicine and Dentistry, Okayama University. Adult male Mongolian gerbils (11 weeks, 64 to 72 g; SLC, Shizuoka, Japan) were used for the experiments. The animals were lightly anesthetized by inhalation of a nitrous oxide–oxygen–halothane (69%:30%:1%) mixture through a closely fitting face mask. After a neck incision, both common carotid arteries (CCAs) were exposed. When the animals began to be awake after the discontinuation of the anesthesia, both CCAs were occluded with aneurysm clips for 5 minutes, and the clips were then removed to restore cerebral blood flow according to our previous report (Abe et al., 1987). The rectal temperature was monitored and maintained at 37.0 ± 0.5°C with a heating pad (Bio Research Center, Aichi, Japan) during the operation. The surgical incision was then closed and animals were allowed free access to water and food at ambient temperature. Sham-operated control animals were treated identically, except for the occlusion of CCAs.
Bromodeoxyuridine labeling
A cell proliferation marker BrdU (Sigma, St. Louis, MO, U.S.A.) was dissolved in saline and was intraperitoneally injected (50 mg/kg) three times at 8-h intervals during the day. To investigate the quantitative change of newly proliferated cells in the SVZ, and the spatial and chronological change of migrating newborn cells, four patterns of BrdU injection were performed in relation to the time the animals were killed after transient forebrain ischemia (Fig. 1A). BrdU was injected at 9 days in the sham-operated (n = 3) and 10-d (n = 5) groups; at 9 and 19 days in the 20-d group (n = 5); at 9, 19, and 29 days in the 30-d group (n = 5); and at 9, 19, 29, and 59 days in the 60-d group (n = 5). In each group, gerbils were killed after 24 h of the last BrdU injection.
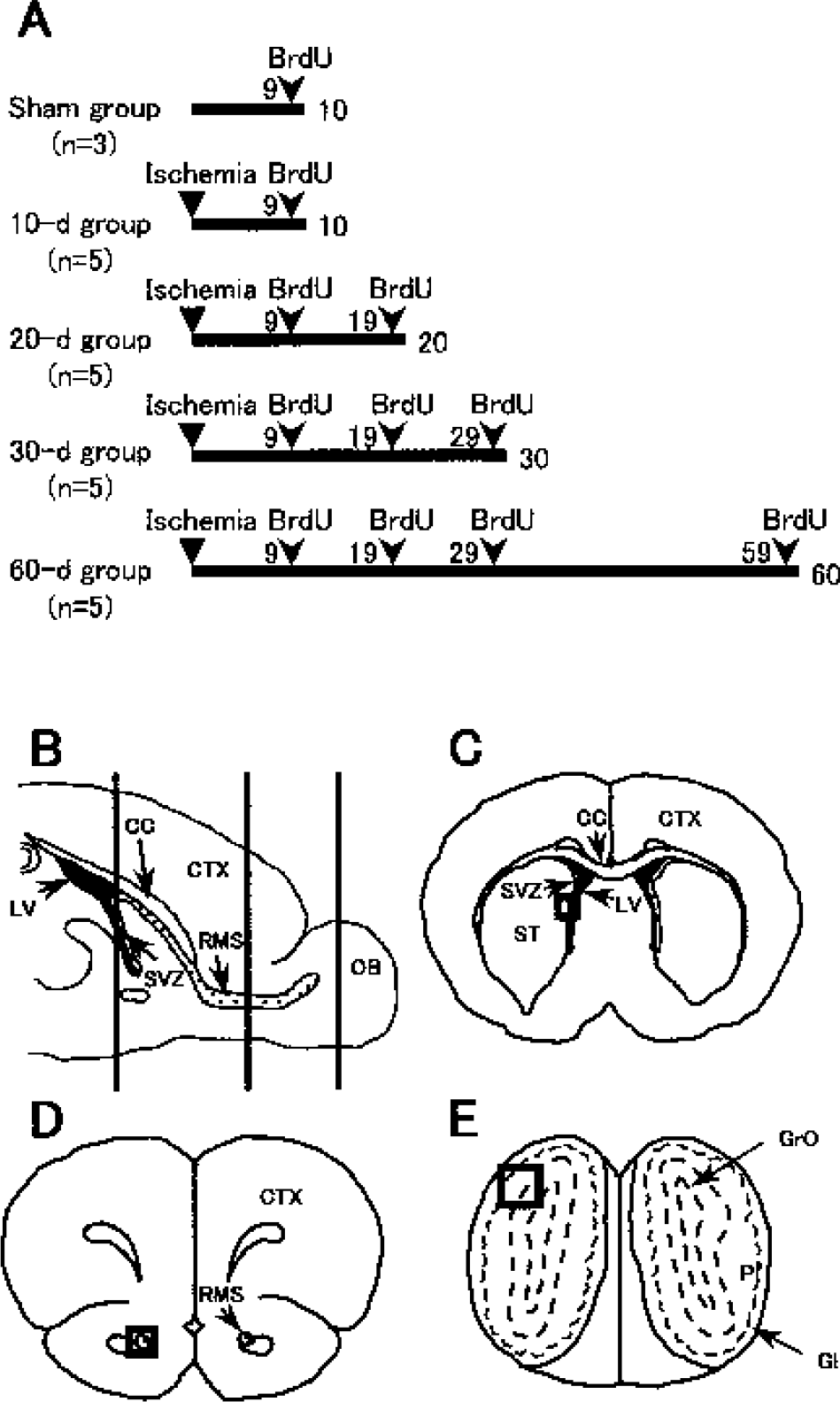
Experimental groups with bromodeoxyuridine (BrdU) injections
Tissue preparation and single immunohistochemistry
All gerbils were killed with deep anesthesia by diethyl ether. The brains were quickly removed and frozen in 2-methylbutane chilled by liquid nitrogen. On a cryostat at −20°C, the left side of the cerebral hemisphere was serially cut in 10-μm-thick sagittal sections for double immunofluorescence staining, and the right brain was serially cut in 10-μm-thick coronal sections at the SVZ, RMS, and OB levels (Fig. 1B) for single immunohistochemical staining and double immunofluorescence staining. The slices were mounted on glass slides coated with polylysine, and then were stored at −80°C until use.
All brain sections were fixed in 4% paraformaldehyde (PFA) for 20 minutes at room temperature and were incubated in 0.3% H2O2 in methanol for 30 minutes and 5% bovine serum albumin for 1 h before incubation with first antibodies diluted in phosphate-buffered saline (PBS) with 0.1% bovine serum albumin and 0.3% Triton X-100.
For immunohistochemical detection of BrdU labeling, brain sections were pretreated to denatured DNA in 50% formamide 2× saline-sodium citrate buffer at 65°C for 2 h and then incubated at 37°C for 30 minutes in 2N HCl. Finally, the sections were rinsed for 10 minutes at 25°C in 0.1 mol/L boric acid, pH 8.5. Sections were incubated overnight with mouse monoclonal immunoglobulin G (IgG) anti-BrdU (1:200; Oncogene Research Products, Boston, MA, U.S.A.) and were then incubated with biotinylated horse anti–mouse IgG (1:200; Vector Laboratories, Burlingame, CA, U.S.A.) for 1 h at room temperature.
For immunohistochemical detection of PSA-NCAM, brain sections were incubated 24 h at 4°C with mouse monoclonal immunoglobulin M (IgM) anti–PSA-NCAM (1:1,000; kindly provided by Dr. T. Seki, Tokyo, Japan [Seki and Arai, 1993]) and were then incubated in biotinylated goat anti–mouse IgM (1:200; Vector Laboratories) for 1 h at room temperature.
For immunohistochemical detection of NeuN, brain sections were incubated 24 h at 4°C with mouse monoclonal IgG anti-NeuN (1:500; Chemicon, Temecula, CA, U.S.A.) and were then incubated in biotinylated horse anti–mouse IgG for 1 h at room temperature.
For immunohistochemical detection of GFAP, brain sections were incubated 24 h at 4°C with goat polyclonal IgG anti-GFAP (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) and were then incubated in biotinylated rabbit anti–goat IgG (1:200; Vector Laboratories) for 1 h at room temperature.
Immunoreactivities were developed in horseradish peroxidase–streptavidin–biotin complex solution (Vectastain ABC Kit; Vector Laboratories) for 30 minutes and were incubated for 2 minutes in a peroxidase reaction solution (0.02% diaminobenzidine, 0.02% H2O2). Peroxidase staining was examined using Olympus microscope (BX-51; Olympus, Tokyo, Japan).
In situ detection of DNA fragmentation
For detection of DNA fragmentation, terminal deoxynucleotidyl dUTP nick-end labeling (TUNEL) was performed with a kit (NeuroTACSII in situ apoptosis detection kit model 4823–30-K; Trevigen, Gaithersburg, MD, U.S.A.). In brief, after the fixation with 3.7% PFA, a set of brain sections was washed 3 times with cold PBS and made permeable with Neuropore (kit component) for 30 minutes. After blocking endogenous peroxidase with 3% H2O2 methanol for 5 minutes, double-strand breaks in genomic DNA were detected with the mixture of terminal deoxynucleotidyl transferase (TdT) dNTP Mix, TdT enzyme, and TdT Mn2+ (kit components) in 1× TdT-labeling buffer for 1 h at 37°C. The reaction stopped with 1× Stop buffer, followed by incubation for 30 minutes with streptavidin–horseradish peroxidase, and the staining was developed with 2,3′-diaminobenzidine tetrahydrochloride.
Double immunofluorescence
For the immunofluorescence detection of BrdU and PSA-NCAM, sections were first incubated with mouse monoclonal IgM anti–PSA-NCAM (1:1,000) for 24 h at 4°C, followed by rhodamine-labeled goat anti–mouse IgM (1:500; Kierkegaard & Perry Laboratories, Gaithersburg, MD, U.S.A.) for 1 h. After washing in PBS, the sections were pretreated to denatured DNA, as mentioned previously. Sections were incubated with mouse monoclonal IgG anti-BrdU for 12 h at 4°C followed by fluorescein–isothiocyanate (FITC)-labeled horse anti–mouse IgG (1:500; Vector) for 1 h.
For the immunofluorescence detection of BrdU and doublecortin (DCX), sections were first incubated with goat polyclonal IgG anti-DCX (1:800; Santa Cruz) for 24 h at 4°C, followed by Alexa Fluor 546–labeled donkey anti–goat IgG (1:500; Molecular Probes, Eugene, OR, U.S.A.) for 1 h. After denaturing DNA, sections were incubated with mouse monoclonal IgG anti-BrdU for 12 h at 4°C followed by FITC-labeled horse anti–mouse IgG for 1 h.
For the double immunofluorescence detection of BrdU and NeuN, sections were pretreated to denatured DNA and were first incubated both with sheep polyclonal IgG anti-BrdU (1:500; Biodesign, Saco, MA, U.S.A.) and mouse monoclonal IgG anti-NeuN for 24 h at 4°C, followed by FITC-labeled donkey anti–sheep IgG (1:500; Molecular Probes) and rhodamine-labeled goat anti–mouse IgG (1:500; Chemicon) for 1 h. For the double immunofluorescence detection of BrdU and GFAP, sections were pretreated to denatured DNA and were first incubated both with mouse monoclonal IgG anti-BrdU and goat polyclonal IgG anti-GFAP for 24 h at 4°C, followed by FITC-labeled horse anti–mouse IgG and Alexa Fluor 546–labeled donkey anti–goat IgG (Molecular Probes) for 1 h.
For the double immunofluorescence detection of PSA-NCAM and DCX, NeuN, or GFAP, sections were first incubated with mouse monoclonal IgM anti–PSA-NCAM and rabbit polyclonal IgG anti-DCX, mouse monoclonal IgG anti-NeuN, or goat polyclonal IgG anti-GFAP for 24 h at 4°C, followed by FITC-labeled rabbit anti–mouse IgM (1:500; Rockland, Gilbertsville, PA, U.S.A.) and Alexa Fluor 546–labeled donkey anti–goat IgG or rhodamine-labeled goat anti–mouse IgG for 1 h.
These sections were scanned with confocal microscope equipped with argon and HeNe1 laser (LSM-510; Zeiss, Jena, Germany). Sets of fluorescent images were acquired sequentially for the red and green channels to prevent crossover of signals from green to red or red to green channels.
Cell counting
With single immunohistochemical staining and double immunofluorescence staining, the number of positive cells was counted and averaged in 4 to 7 coronal sections (10 μm, spaced 50 μm apart) per animal for the right SVZ, RMS, and OB in the sham-operated control (n = 3), and 10, 20, 30, and 60 d of ischemic groups (each group, n = 5). The sections of the SVZ were begun at 0.1 mm rostral to bregma (Fig. 1C), the sections of the RMS were begun at 3.5 mm rostral to bregma (Fig. 1D), and the sections of the OB were begun at 6.0 mm rostral to bregma (Fig. 1E). Each microscope image was digitized using Hamamatsu Acuacosmos System (Hamamatsu Photonicus, Hamamatsu, Japan), and positive cells were counted on a computer monitor.
The number of BrdU-labeled cells was counted in the SVZ (Fig. 1C), the RMS (Fig. 1D), and the square area (field size, 660 × 660 μm) of the OB that contained both the plexiform layer (Pl) and the granular cell layer of OB (GrO; Fig. 1E), respectively. The number of PSA-NCAM–positive cells and the number of BrdU-labeled cells that coexpressed PSA-NCAM were counted in the SVZ (Fig. 1C) and the RMS (Fig. 1D). The number of GFAP-positive cells was counted in the square areas (field size, 660 × 660 μm) of the lateral ventricle (Fig. 1C), the RMS (Fig. 1D), and the OB that contained both the Pl and the GrO (Fig. 1E), respectively.
The cell numbers were presented as mean ± SD within the investigated region. In the numbers of BrdU-single-labeled cells and BrdU-labeled cells that coexpressed PSA-NCAM, statistical analyses were performed only between sham-operated control and 10-d groups because of using the same BrdU administration protocol. Student's t -test was performed, with P < 0.01 as significant. For positive cell numbers with PSA-NCAM and GFAP, statistical analyses were performed between sham-operated control and each postischemic group. Kruskal-Wallis H-test, followed by post hoc test using the Dunnett test, was performed, with P < 0.01 as significant.
Estimation of the area of rostral migratory stream
To examine a possible influence of transient ischemia, the size of RMS area was measured in five coronal sections and was averaged per animal in each group beginning at 3.0 mm rostral to bregma with GFAP staining. The area of crowded GFAP-positive cells was regarded as a rostral migratory stream because the migrating cells go through glial tunnels in the RMS (Lim and Alvarez-Buylla, 1999). The size of RMS area was measured with a computer-based imaging system (UTHSCSA Image Tool version 2.0 Alpha 3; Microsoft, Redmond, WA, U.S.A.), and statistical analyses were performed for the size of RMS area between sham-operated control and each postischemic group. Kruskal-Wallis H-test, followed by post hoc test using the Dunnett test, was performed, with P < 0.01 as significant.
RESULTS
Change of bromodeoxyuridine-labeled cell
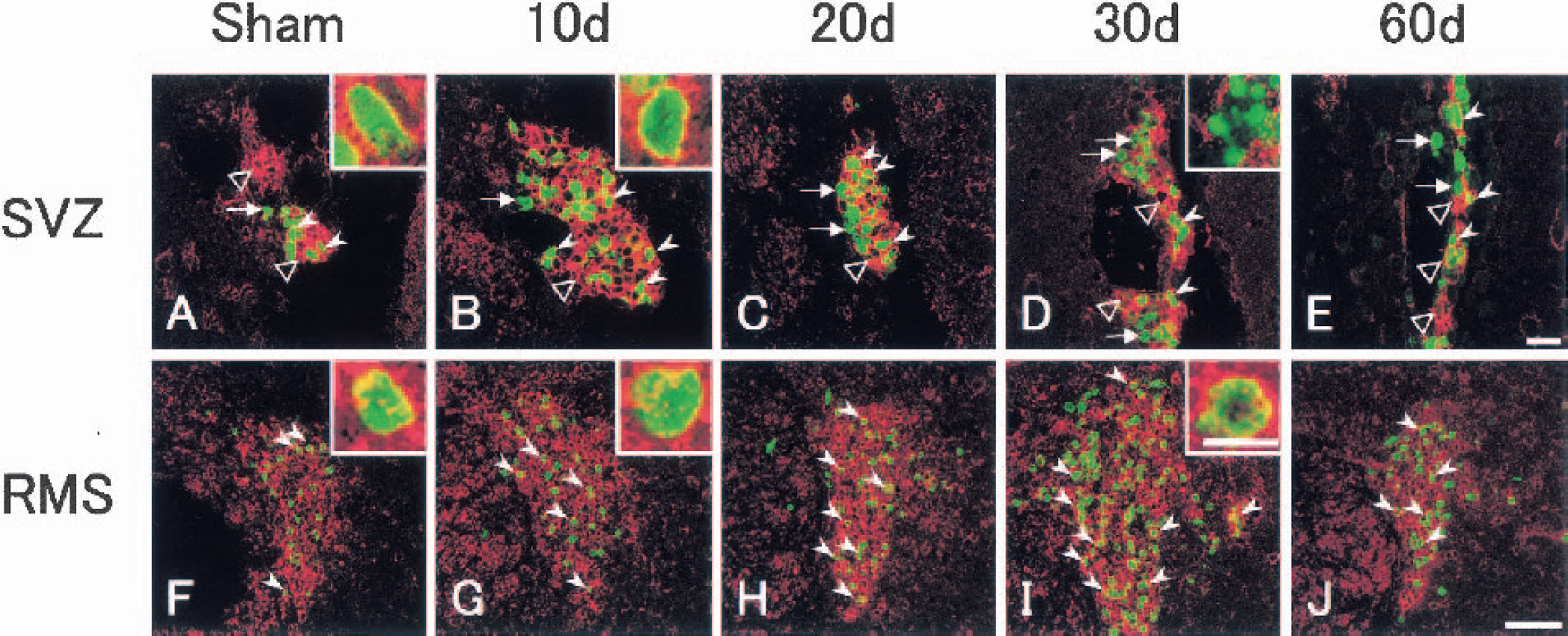
In sham-operated control brain, basal level of BrdU-labeled cells was found in the SVZ (Fig. 2A, arrowheads) and the RMS (Fig. 2F), but not in the OB (Fig. 2K).
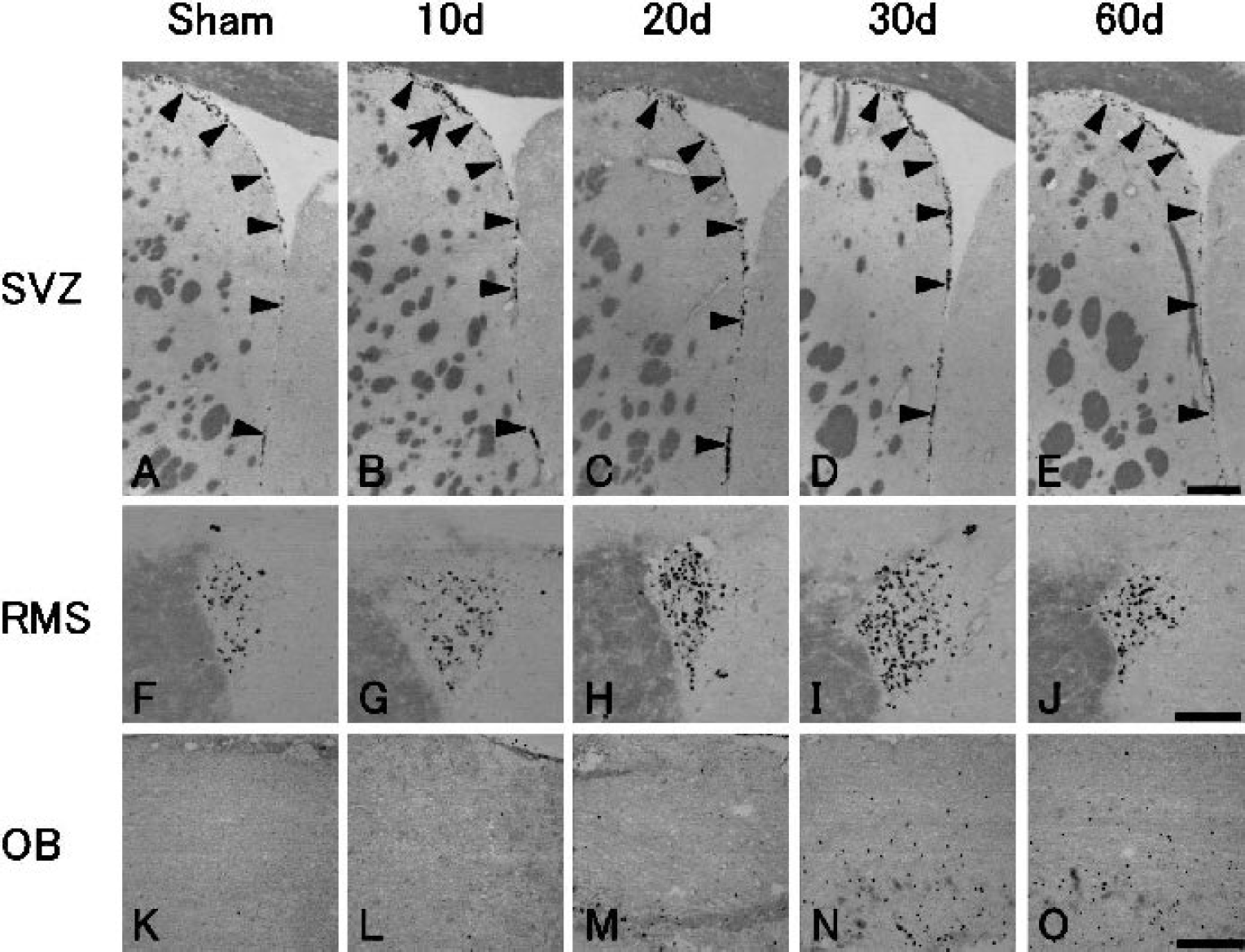
Immunohistochemistry for bromodeoxyuridine (BrdU) of the subventricular zone (SVZ;
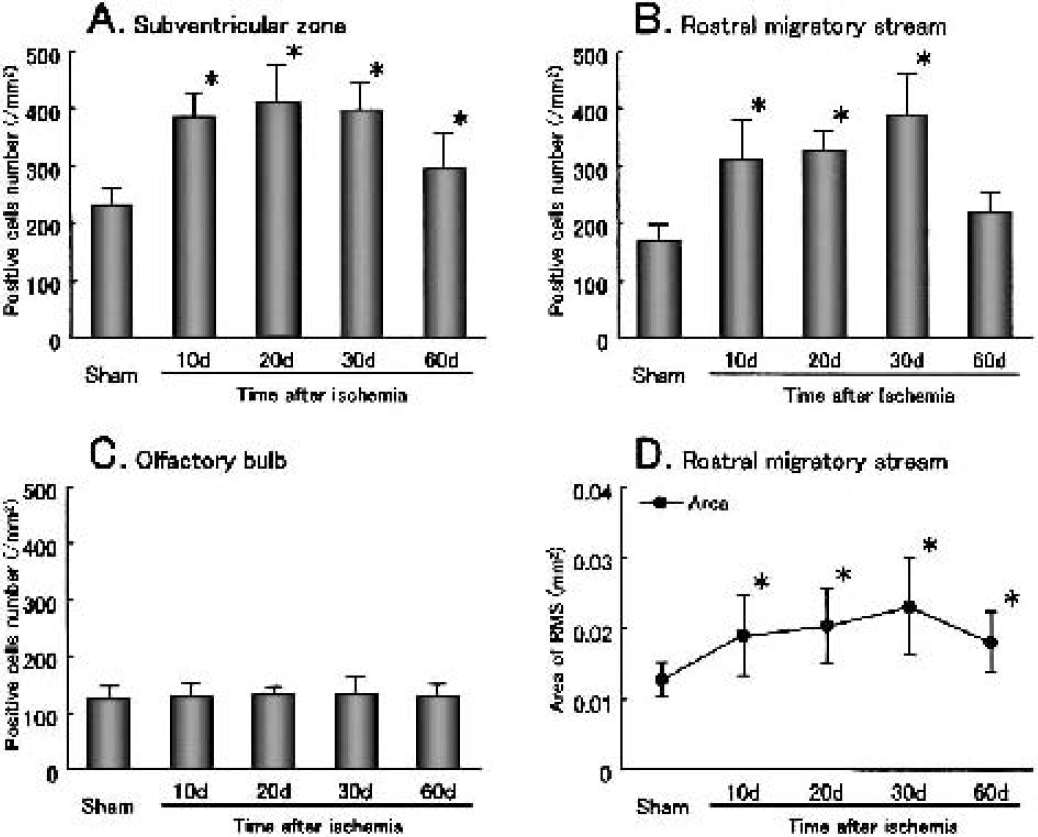
After transient ischemia, the number of BrdU-labeled cells significantly increased at 10 d, 1.5-fold in the SVZ (Fig. 2B, arrowheads) and 1.9-fold in the RMS (Fig. 2G). It gradually increased until 30 d in the SVZ (Figs. 2C and 2D, arrowheads) and the RMS (Figs. 2H and 2I), and returned to the sham-operated control level by 60 d in the SVZ (Figs. 2E and (3A, arrowheads) with prolonged elevation, 1.6-fold, in the RMS (Figs. 2J and 3B). In contrast, in the OB it increased slightly at 20 d (Fig. 2M) and greatly, with widespread distribution, at 30 d (Fig. 2N), and the increase lasted until 60 d (Figs. 2O and 3C). Some BrdU-labeled cells were present within the caudate putamen outside SVZ at 10 d after transient ischemia (Fig. 2B, arrow).
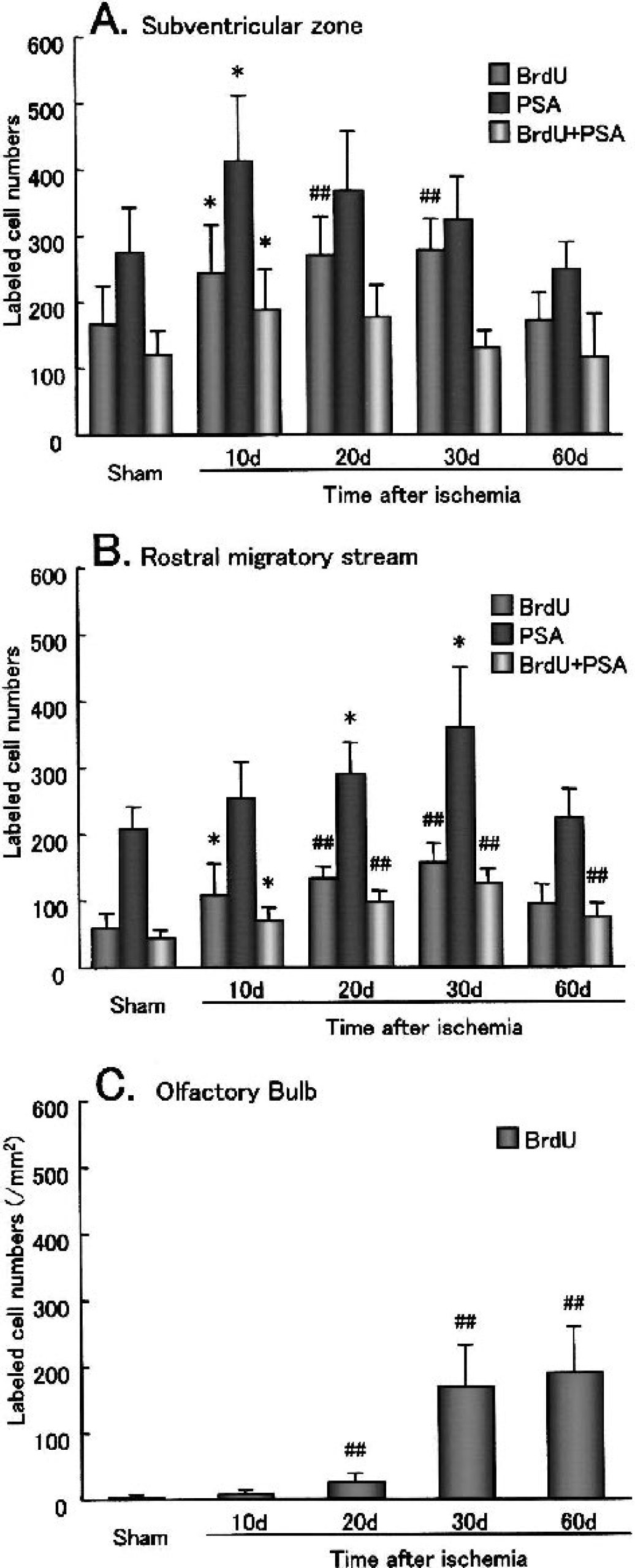
Changes of the number of bromodeoxyuridine (BrdU) single-labeled cells, polysialylated neural cell adhesion molecule (PSA-NCAM) single positive cells, and BrdU-labeled cells that coexpressed PSA-NCAM in the subventricular zone (SVZ;
Change of PSA-NCAM–positive cell
In sham-operated control brain, PSA-NCAM–positive cells were presented in the SVZ (Fig. 4A, arrowheads), the RMS (Fig. 4C) and all over the OB (Fig. 4E, inset).
Immunohistochemistry for highly polysialylated neural cell adhesion molecule (PSA-NCAM) of the subventricular zone (SVZ;
After transient ischemia, the number of PSA-NCAM–positive cells significantly increased in the SVZ, with a peak at 10 d (1.5-fold, Figs. 3A and 4B, arrowheads), and in the RMS with a delayed peak at 30 d (1.7-fold, Figs. 3B and 4D, arrowheads). The number of PSA-NCAM–positive cells returned to the sham-operated control level by 60 d in both the SVZ and the RMS (Figs. 3A and 3B). In the OB, the number and location of PSA-NCAM–positive cells did not change after transient ischemia (Fig. 4F, inset).
Cell proliferation and migration from the subventricular zone to the rostral migratory stream
In sham-operated control brain, BrdU-labeled cells (green) that coexpressed PSA-NCAM (red) were located in the SVZ (Fig. 5A, arrowheads, inset). In the SVZ, PSA-NCAM–positive cells formed colonies (Fig. 5A, open arrowheads), and a BrdU single-labeled cell was located out of the colony (Fig. 5A, arrow). In the RMS, BrdU-labeled cells (green) that coexpressed PSA-NCAM (red) were always located within the RMS (Fig. 5F, arrowheads, inset).
Double immunofluorescence analysis for bromodeoxyuridine (BrdU; green) + polysialylated neural cell adhesion molecule (PSA-NCAM; red) of the subventricular zone (SVZ;
After transient ischemia, the number of BrdU-labeled cells (green) that coexpressed PSA-NCAM (red) increased in the SVZ with a peak at 10 d (1.6-fold; Fig. 5B, arrowheads, inset). The colony of PSA-NCAM–positive cells (Fig. 5B, open arrowhead) became larger, and about three fourths of BrdU-labeled cells coexpressed PSA-NCAM at 10 d (Figs. 3A and 5B, arrowheads). Although the number of BrdU-labeled cells that coexpressed PSA-NCAM began to decrease at 20 d and returned to the sham-operated control level by 60 d in the SVZ, about half of the BrdU-labeled cells did not coexpress PSA-NCAM, and some of these nuclei became speckled at 30 d in the SVZ (Figs. 3A and 5D, arrows, inset). After 20 d, the colony size of PSA-NCAM–positive cells diminished gradually until 60 d in the SVZ (Figs. 5C to 5E, open arrowheads). In contrast, the number of BrdU-labeled cells that coexpressed PSA-NCAM increased in the RMS, with a delayed peak (2.9-fold) at 30 d, which was still elevated 1.7-fold at 60 d (Figs. 3B and 5H to 5J). The ratio of these cells among total BrdU-labeled cells was not different between sham-operated control and each postischemic brain. BrdU-labeled cells that coexpressed PSA-NCAM were always located within the RMS (Figs. 5G to 5J, arrowheads, insets).
No neuronal loss around the subventricular zone, and in the rostral migratory stream and the olfactory bulb
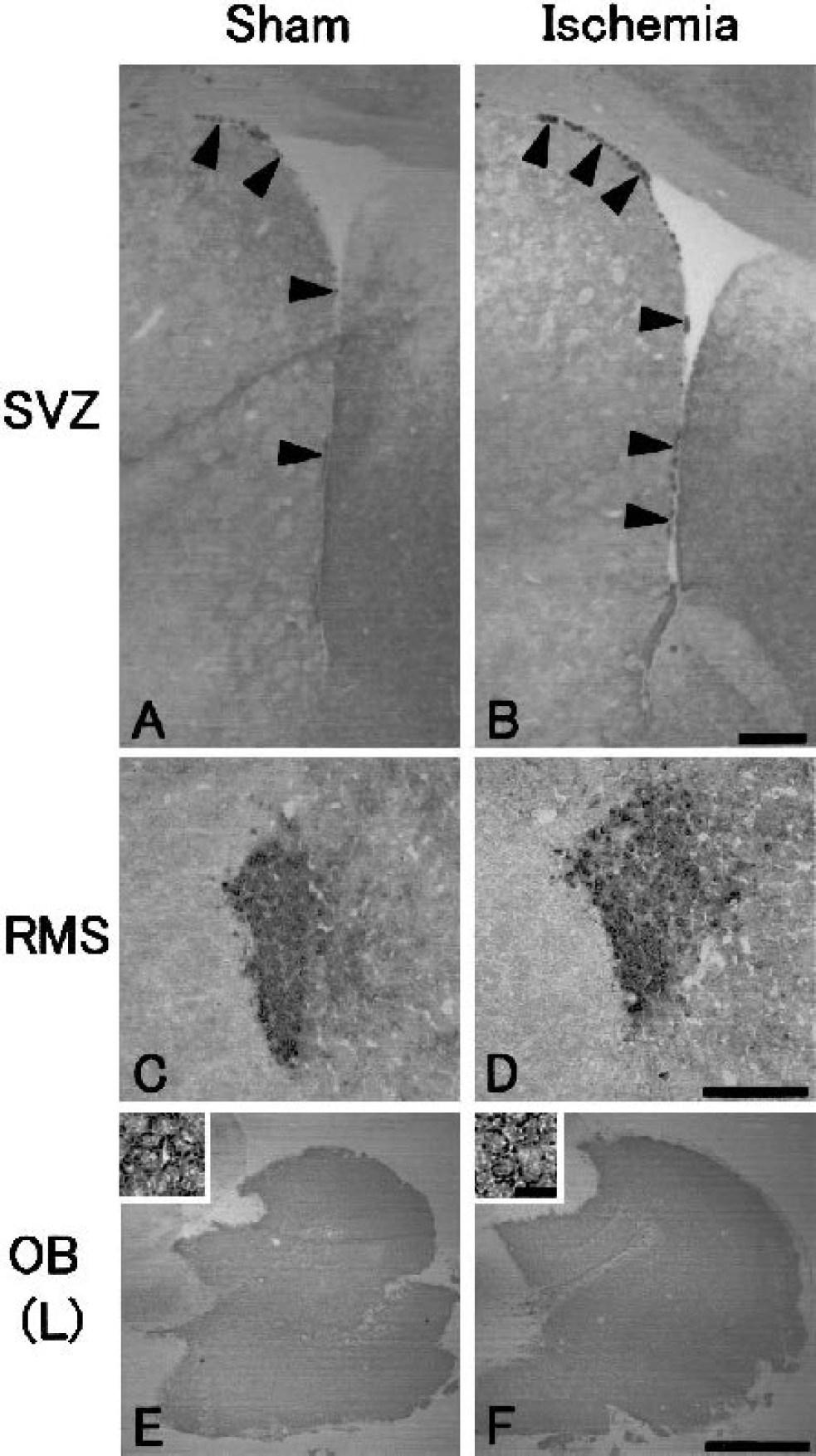
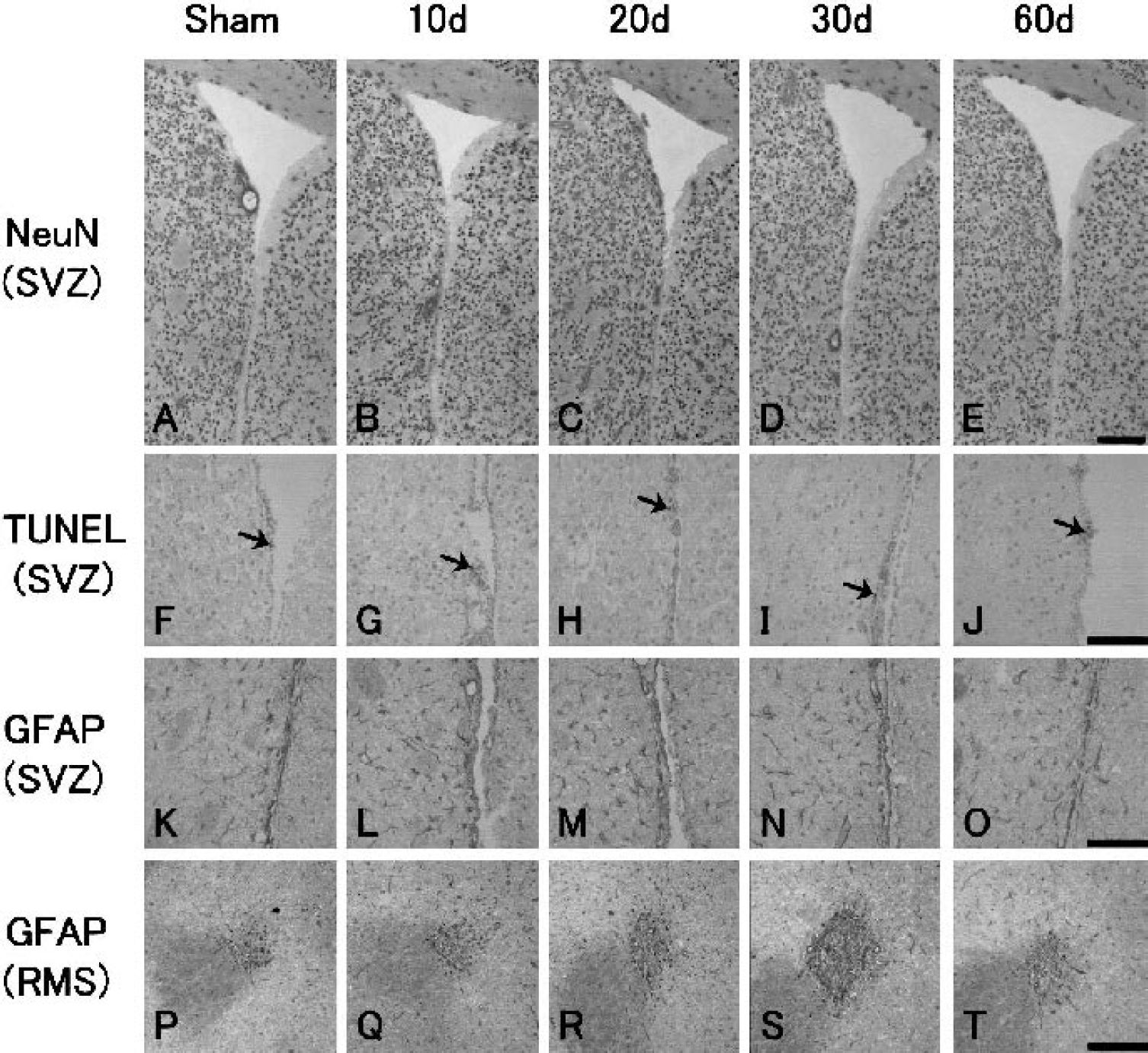
With NeuN single immunohistochemistry, no cell loss was found in the caudate putamen around the SVZ (Figs. 6A to 6E), or in the RMS and the OB (data not shown) after transient ischemia compared with sham-operated control brain. Although a few TUNEL-positive cells were present in the outside of the lateral ventricle (Fig. 6F, arrow), the RMS, and the OB (data not shown) of the sham-operated control brain, no change was found in the number of these cells after transient ischemia (Figs. 6G to 6J, arrows).
Immunohistochemistry for neuronal nuclear antigen (NeuN;
Glial cell activations around the subventricular zone and within the rostral migratory stream
In the sham-operated control brain, GFAP-positive cells were present in small numbers (231 ± 32 /mm2, Fig. 7A) in the SVZ and adjacent caudate putamen (Fig. 6K). After transient ischemia, the number of GFAP-positive cells increased in the SVZ and adjacent caudate putamen, with a peak at 10 to 30 d of 1.7- to 1.8-fold (Figs. 6L to 6N and 7A), which was still significantly elevated by 1.3-fold at 60 d (Figs. 6O and 7A).
Change of the number of glial fibrillary acidic protein–positive cells in the subventricular zone and adjacent caudate putamen
In the RMS, GFAP-positive cells were present in the sham-operated control brain (Fig. 6P). The number of GFAP-positive cells in the RMS and the area size of the RMS increased after transient ischemia at 10 to 60 d, with a peak (2.2-fold in number and 1.7-fold in area) at 30 d Figs. 6Q to 6T, 7B, and 7D).
In the OB, the number of GFAP-positive cells did not change after transient ischemia compared with that of the sham-operated control brain (Fig. 7C).
Migration and differentiation of neural stem cells
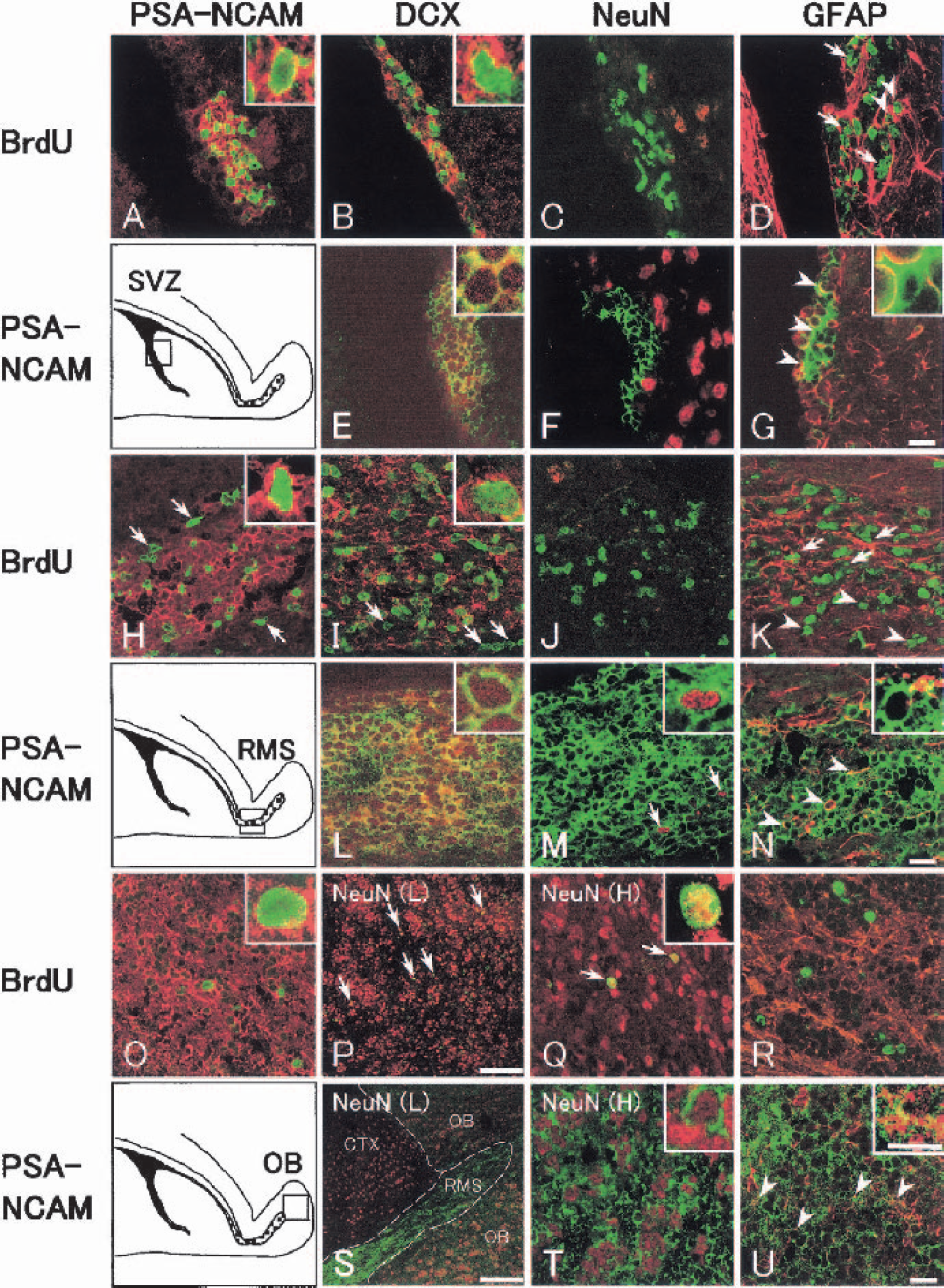
In the SVZ, some BrdU-labeled cells (green) coexpressed migration markers PSA-NCAM (red, Fig. 8A, inset) and DCX (red, Fig. 8B, inset) in both sham-operated control and postischemic brains. No double-positive cell, however, was detected with BrdU (green) + NeuN (red, Fig. 8C) in the SVZ of both sham-operated control and postischemic brains. BrdU-labeled cells (green) were usually just surrounded with some GFAP-positive cells and processes (red, Fig. 8D, arrows), and only occasionally coexpressed GFAP (red, Fig. 8D, arrowheads). In contrast, PSA-NCAM–positive cells (green) were mostly double positive (yellow, Fig. 8E, inset) with DCX (red), but again not with NeuN (red, Fig. 8F) in the SVZ of both sham-operated control and postischemic brains. A part of PSA-NCAM–positive cell bodies (green) were in contact with GFAP-positive cells and processes (red), showing a yellow color (Fig. 8G, arrowheads, inset).
Double immunofluorescence analysis in sagittal sections for bromodeoxyuridine (BrdU; green) + polysialylated neural cell adhesion molecule (PSA-NCAM; red,
In the RMS most BrdU-labeled cells (green) coexpressed migration markers PSA-NCAM (red, Fig. 8H, inset) and DCX (red, Fig. 8I, inset) in inner RMS, whereas a part of the BrdU-labeled cells did not coexpress PSA-NCAM or DCX in outer RMS (Figs. 8H and 8I, arrows) of both sham-operated control and postischemic brains. No double-positive cell was detected with BrdU (green) + NeuN (red, Fig. 8J) in the RMS of both sham-operated control and postischemic brains. BrdU-labeled cells were partly surrounded by GFAP-positive cells and processes (Fig. 8K, red) in inner (Fig. 8K, arrows) but not outer (Fig. 8K, arrowheads) RMS. Most PSA-NCAM–positive cells (green), however, were double positive (yellow, Fig. 8L, inset) with DCX (red) in the RMS of both sham-operated control and postischemic brains. A few PSA-NCAM–positive cells (green) had NeuN-positive nucleus (red) at 30 d after the ischemia (Fig. 8M, arrows, inset). A part of PSA-NCAM–positive cell bodies (green) were in contact with GFAP-positive cells and processes (red), showing a yellow color (Fig. 8N, arrowheads, inset).
In the OB, BrdU-labeled cells (green) coexpressed a migration marker PSA-NCAM (Fig. 8O, red, inset) but not another migration marker DCX (data not shown) after 30 d of transient ischemia. BrdU (green) + NeuN (red) double-positive cells (yellow) were sometimes detected in the Pl and the GrO of the OB at 30 and 60 d (Figs. 8P and 8Q, arrows, inset). BrdU-labeled cells (green) did not coexpress GFAP (red), nor were they surrounded with GFAP-positive cell bodies and processes (Fig. 8R) in the OB of both sham-operated control and postischemic brains. PSA-NCAM–positive cells (green), however, were widely located all over the OB (Fig. 8S, OB). The staining intensity of these cells, however, was much weaker in the OB than in the RMS (Fig. 8S, RMS) of both sham-operated control and postischemic brains. Almost all PSA-NCAM–positive cells had NeuN-positive nucleus in the Pl and the GrO (Fig. 8T, inset) of both sham-operated control and postischemic brains. Only a small part of PSA-NCAM–positive cell bodies (green) were in contact with GFAP-positive cell bodies and processes (red), showing a yellow color (Fig. 8U, arrows, inset).
In the cerebral cortex, BrdU-labeled cells were occasionally detected in sham-operated control brain, and often distributed as “doublets” (Fig. 9A, arrows). BrdU-labeled (green, Fig. 9B, arrow) and NeuN-positive (red) cells were located side by side, but were not double positive. These BrdU-labeled cells were not double positive with PSA-NCAM, DCX, NeuN, or GFAP (Fig. 9C) in the cerebral cortex of sham-operated control brain. BrdU-labeled cells did not change in the number, nor were they double positive for PSA-NCAM, DCX, NeuN, or GFAP in the cerebral cortex after transient ischemia (data not shown).
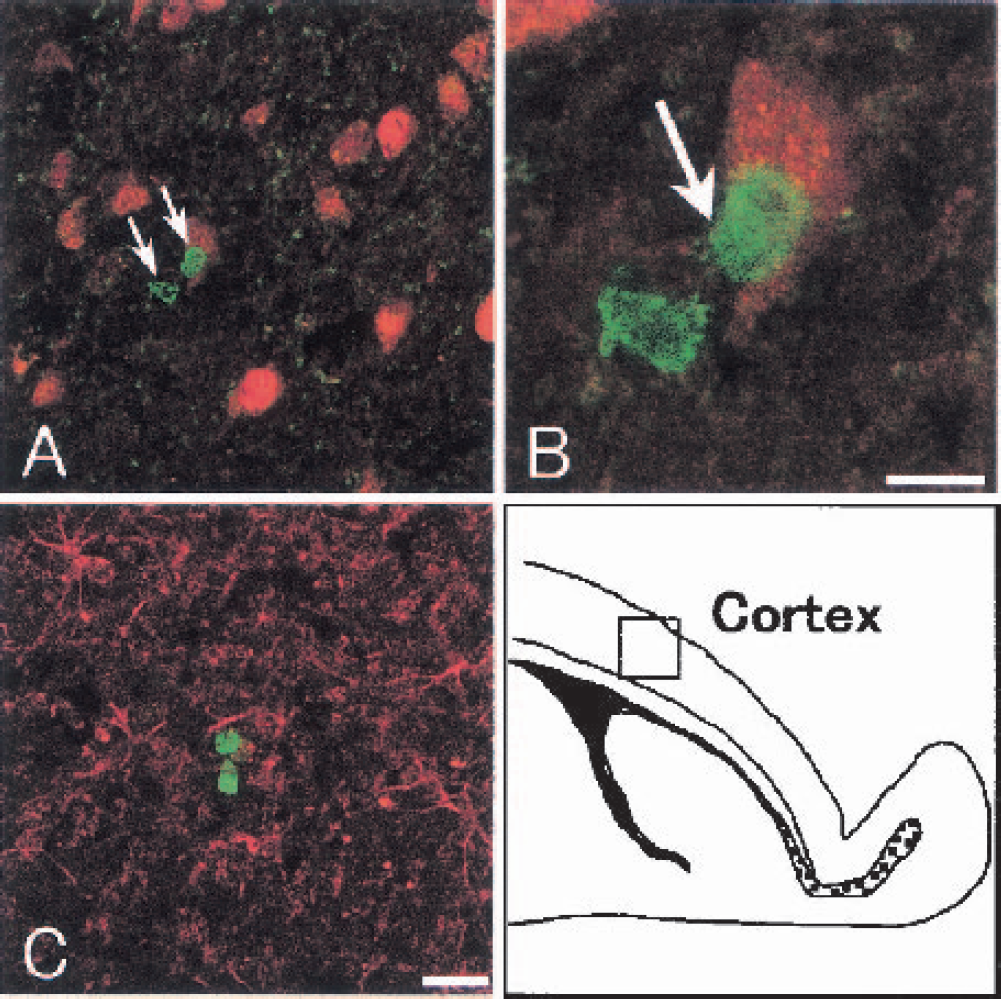
Double immunofluorescence analysis for bromodeoxyuridine (BrdU; green) + neuronal nuclear antigen (NeuN; red,
DISCUSSION
In normal adult brain, a small number of neural stem cells proliferate in the SVZ, forming a colony with positive PSA-NCAM, and the cells migrate through the RMS to the OB with differentiation into mature neural cells (Altman, 1969; Doetsch et al., 1997; Kornack and Rakic, 2001a; Pencea et al., 2001). Enhancements of neurogenesis in the SVZ after cerebral ischemia were reported in only a few studies (Jin et al., 2001; Zhang et al., 2001), but those did not clearly examine the relations between the three steps of neurogenesis such as proliferation, migration, and differentiation.
This is the first comprehensive report on such relations between the three steps of neurogenesis from the SVZ through the RMS to the OB after 5 minutes of transient forebrain ischemia in gerbils. After transient ischemia, the number of BrdU-labeled cells increased at 10 to 30 d (Figs. 2B to 2E, 3A), and the number of PSA-NCAM–positive cells increased in the SVZ, with a peak at 10 d (Figs. 3A and 4D), similar to previous reports with transient focal cerebral ischemia (Jin et al., 2001; Zhang et al., 2001). The number of BrdU-labeled cells that coexpressed PSA-NCAM (Figs. 3A and 5B, arrowheads) and the size of PSA-NCAM–positive cell colony became the largest at 10 d (Fig. 5B, open arrowhead).
New immature neurons born in the SVZ form elongate chain aggregates within the RMS and migrate through astroglial tunnels (Lois et al., 1996). In the present study, BrdU-single-labeled cells and BrdU-labeled cells that coexpressed PSA-NCAM were detected in the RMS in the sham-operated control brain. After transient ischemia, the cell numbers increased, with a delayed peak at 30 d (Figs. 2F to 2J, 3B, and 5I) compared with that of the SVZ (Figs. 2A to 2E, 3A, and 5B).
After reaching the core of the OB, young neurons finally migrate radially into the granular and the periglomerular layers to differentiate into mature neurons (Luskin, 1993; Lois and Alvarez-Buylla, 1994). After transient ischemia, the number of BrdU-labeled cells in the OB increased slightly at 20 d (Fig. 2M) and greatly with widespread distribution at 30 and 60 d (Figs. 2N, 2O, and 3C), and some of these cells were double positive with mature neuron marker NeuN (Figs. 8P and 8Q, arrows), but not astrocytes marker GFAP (Fig. 8R).
Cell proliferation was investigated as the first step of neurogenesis using BrdU in this study. After transient ischemia, about three fourths to one half of BrdU-labeled cells coexpressed PSA-NCAM for subsequent migration (Figs. 3A and 5B to 5E). Thus, the remainder was considered to be stem cells and astroglial cells that reside in the SVZ. In fact, BrdU-labeled cells that coexpressed GFAP were detected in the SVZ at 10 d (Fig. 8D, arrowheads), and some BrdU single-labeled cells showed nuclei with postmitotic speckled form at 30 d (Fig. 5D, arrows, inset) after transient ischemia.
In the present study, cell proliferation is enhanced by transient forebrain ischemia as well as transient focal cerebral ischemia (Jin et al., 2001; Zhang et al., 2001). The mechanisms of cell proliferation enhancement after ischemic insults, however, are poorly understood. One possible mechanism of this proliferation could be replacement of lost neurons. Although apoptotic mechanisms play an important role in cell replacement of the adult rodent SVZ–OB pathway under normal conditions (Biebl et al., 2000), neither change of the number of apoptotic cells nor neuronal cell loss was found in the SVZ–OB pathway (Figs. 6A to 6J) after transient ischemia. Because TUNEL-positive cells disappeared in ischemic brain by 7 d (States et al., 1966), and the clearance time of TUNEL-positive cells was reported to be 2 h and 20 minutes (Thomaidou et al., 1997), the TUNEL method could not detect change in the number of apoptotic cells accurately in the present study. Based on a study with removal of the OB in mice, however, OB is not essential for stem cells to proliferate in the SVZ or to migrate through the RMS (Kirschenbaum et al., 1999). Thus, the increase of BrdU-labeled cell number in the SVZ (Fig. 3A) did not result from ischemic cell loss, but rather from other mechanisms.
Ischemic insult is known to increase the expression of growth factors (Abe, 2000) that play important roles in stem cell proliferation (Morshead et al., 1994; Craig et al., 1996; Kuhn et al., 1997; Tropepe et al., 1997). Thus, the proliferation in the SVZ could be enhanced by stimulus of growth factors after transient ischemia. Recently, the regulation of cell proliferation in the SVZ by other molecules has been reported. Stem cells in the SVZ express ligands for EphB2, and infusion of EphB2 into the brain increases the cell proliferation in the SVZ (Conover et al., 2000). The bone morphogenetic proteins inhibit SVZ neurogenesis, whereas bone morphogenetic protein antagonist Noggin promotes SVZ neurogenesis (Lim et al., 2000). The effects of ischemia on these molecules and their role in postischemic neurogenesis remain to be determined.
Cell migration is the second step of neurogenesis that was investigated with PSA-NCAM and DCX in this study. PSA-NCAM prevents premature neurons from tight adhesion with astroglial tunnels, thus helping chain migration (Cremer et al., 1994; Hu et al., 1996). Some reports showed that PSA-NCAM could be expressed by oligodendrocytes or astrocytes as well (Ben-Hur et al., 1998; Theodosis et al., 1999; Fox et al., 2001). Similar to a previous report (Nacher et al., 2001), the present study showed that most PSA-NCAM–positive cells were double positive with DCX in the SVZ (Fig. 8E, inset) and RMS (Fig. 8L, inset) of both sham-operated control and postischemic animals. Doublecortin, a microtubule-associated protein, is also important for migration of young neurons in normal development and is expressed strongly in migrating neurons within RMS (Francis et al., 1999; Gleeson et al., 1999). Therefore, PSA-NCAM–positive cells in SVZ and RMS are most probably migrating neurons.
Corresponding to the changes with PSA-NCAM, GFAP-positive cells increased in the SVZ and adjacent caudate putamen (Fig. 7A) and the size of the RMS enlarged (Figs. 7B and 7D) after transient ischemia. Because astrocytes around the chain support the survival of migrating young neurons and enhance their migration (Mason et al., 2001), the present results indicate an enhanced support for migrating cells by the surrounding astrocytes after transient ischemia.
Cell differentiation is the third step of neurogenesis and was investigated with NeuN and GFAP as differentiated markers in this study. After transient ischemia, BrdU + NeuN double-positive cells were sometimes detected in the Pl and the GrO of the OB at 30 and 60 d (Figs. 8P and 8Q, arrows), but not in the SVZ (Fig. 8C) or the RMS (Fig. 8J). Furthermore, most NeuN-positive cells coexpressed PSA-NCAM in the Pl and the GrO in the sham-operated control and postischemic brains (Fig. 8T, inset), whereas a few such cells were also detected in the postischemic RMS (Fig. 8M, arrows). These results suggest that newborn cells finally differentiate into mature neurons predominantly after entering the OB, whereas a few cells already became matured during migration into neurons within the postischemic RMS. BrdU-labeled cells that coexpressed GFAP were detected only occasionally in the SVZ, but not in the RMS or the OB after transient ischemia. Thus, newborn cells do not differentiate into astrocytes in the RMS or the OB after transient ischemia, whereas a part of cells in the SVZ may be neural stem cells, similar to findings in a previous report (Doetsch et al., 1999).
Although BrdU + NeuN double-positive cells were observed in the cerebral cortex after a transient focal ischemia (Jiang et al., 2001), no such double-positive cell was found in the cerebral cortex of the present study (Figs. 9A and 9B). Of interest is that the number of BrdU-labeled cells did not increase, and that these cells were located as “doublets” after transient ischemia similar to daughter cells of a mitotic event under normal condition (Kornack and Rakic, 2001b). Thus, neurogenesis does not occur in cerebral cortex after transient forebrain ischemia.
In the present study, spatial and chronologic relations of neurogenesis were comprehensively shown with three steps, from the SVZ through the RMS to the OB, of gerbils after 5 minutes of transient forebrain ischemia. These observations may help clarify the mechanisms of functional recovery after the ischemia with activation of intrinsic neural stem cells.
Footnotes
Acknowledgments:
The authors thank Dr. T. Seki (Tokyo, Japan) for his generous gift of the antibody against PSA-NCAM.
