Abstract
Sixteen of 24 Sprague-Dawley rats with permanent middle cerebral artery occlusion for 24 hours were subjected to immediate or 8-hour delayed 2,3,5-triphenyltetrazolium chloride (TTC) staining (n = 8 at each time point); the other 8 animals were subjected to immediate or 8-hour delayed measurement of succinate dehydrogenase activity (n = 4 at each time point). The TTC staining was of good quality good in all animals, and the infarcted region could be distinguished easily from normal tissue. There was no significant difference in corrected infarct volume between the two groups (263.8 ± 43.1 versus 264.4 ± 54.8 mm3 [mean ± standard deviation]). The activity of succinate dehydrogenase was not significantly different when normal or infarcted tissue was measured immediately after death or with an 8 hour delay, although less activity was detected at both time points in the infarcted tissue. These results demonstrate that an 8-hour delay of TTC staining is reliable for evaluating brain infarct volume in a rat stroke model and this probably is attributable to the slow deterioration of mitochondrial enzyme activity in nonischemic brain over this time period.
Infarct volume is an important index for estimating ischemic cerebral injury. 2,3,5-triphenyltetrazolium chloride (TTC) staining is a convenient and reliable modality for evaluating infarct volume in experimental ischemic stroke that is well correlated with more traditional histologic markers of infarction (Bose et al., 1984; Bederson et al., 1986; Lundy et al., 1986; Taylor et al., 1987; Hatfield et al., 1991; Lin et al., 1993). This conclusion is based on immediate TTC staining after the elective or natural death of animals. When premature death occurs in an experiment, the animals usually are not evaluated in a fashion similar to those electively killed because sometimes it is impossible to harvest the brains immediately after death. This may lead to the exclusion from the data analysis of animals that die prematurely. Is delayed TTC staining hours after death still accurate for measuring the infarct volume? To our knowledge, this has not been evaluated carefully. The purpose of this study was to determine whether delayed TTC staining is reliable for distinguishing between normal and infarcted brain tissue and whether it accurately quantifies infarct volume. This was done by comparing TTC staining with an 8-hour delay after death to immediate staining in a rat permanent middle cerebral artery occlusion (MCAO) model.
METHODS
In a pilot experiment, eight nonischemic male Sprague-Dawley rats weighing 300 to 330 g were killed by an intraperitoneal injection of chloral hydrate (1000 mg/kg) and left at room temperature (21–23°C) for 1, 2, 4, and 8 hours (n = 2 each time point), respectively. Then the brains were removed, sectioned coronally into six slices, and incubated in a 2% solution of TTC at 37°C for 30 minutes. We found that the brains stained uniformly well even with an 8 hour delay of TTC staining. Then 24 four male Sprague-Dawley rats weighing 300 to 330 g were subjected to permanent MCAO for 24 hours and randomly divided into two group (groups A and B, n = 12 per group). Eight animals in each group underwent TTC staining and four in each group underwent succinate dehydrogenase activity quantification. Animals were allowed free access to food and water before and after MCAO. Anesthesia was given by the intraperitoneal injection of chloral hydrate (400 mg/kg) and PE-50 polyethylene tubing was inserted into the left femoral artery for continuous monitoring of mean arterial blood pressure (78901A, Hewlett-Packard, Mountain View, CA, U.S.A.) and for measuring arterial pH, arterial oxygen pressure (PaO2), and partial pressure of carbon dioxide (PaCO2) (Corning 178-pH Blood Gas Analyzer, Corning, Inc., Mansfield, MA, U.S.A.) at baseline and 15 minutes after the induction of ischemia. Rectal temperature was monitored continuously with a rectal probe inserted to a 5-cm depth from the anal ring and maintained at 37°C with a thermostatically controlled heating lamp (Model 73ATD, YSI, Inc., Yellow Springs, OH, U.S.A.) throughout the surgery.
Focal ischemic stroke was induced by the intraluminal suture occlusion model (Koizumi et al., 1986; Zea Longa et al., 1989). Briefly, the right common carotid artery and right external carotid artery were dissected, and the proximal portions of the common carotid artery and the external carotid artery were ligated. A 4-0 monofilament nylon suture (Ethilon Nylon Suture, Ethicon, Inc., Somerville, NJ, U.S.A.), with its tip rounded near a flame and coated with silicon (Bayer Inc., Leverkusen, Germany), was introduced through a cut into the common carotid artery and advanced into the internal carotid artery approximately 17 mm distal to the carotid bifurcation; mild resistance was felt, indicating that the orifice of the middle cerebral artery is occluded.
Twenty four hours after MCAO, the animals were killed by an intraperitoneal injection of chloral hydrate (1000 mg/kg). In group A, the brains of 8 animals were removed immediately, sectioned coronally into six slices with a 2-mm thickness, incubated in a 2% solution of TTC at 37°C for 30 minutes, and then fixed by immersion in a 10% of buffered formalin solution (Bederson et al., 1986). In group B, the dead animals were left at room temperature (21–23°C) for 8 hours and then subjected to the same procedure as the animals in group A. Forty-eight hours later, the brain slices were evaluated by an investigator blinded to the time of TTC staining, and the investigator determined the presence of the following staining characteristics by visual inspection: deep red, light red, pink, and white, according to the staining intensity, with red being the most intense. The borders between the stained and unstained areas were judged to be distinct, easily recognizable or unrecognizable. Then the six brain sections were photographed using a charged-coupled device camera (EDC-1000HR Computer Camera, ELECTRIM Corp., Princeton, NJ, U.S.A.). The unstained area was defined as infarcted. By using an image analysis program (Bio Scan, OPTIMAS, Edmonds, WA, U.S.A.), the uncorrected infarct area and the total areas of both hemispheres were calculated for each coronal slice. The corrected infarct area in a slice was calculated to compensate for brain edema (Swanson et al., 1990; Lin et al., 1993; Schabitz et al., 1996). Corrected infarct volumes were calculated by multiplying the corrected area by the slice thickness and summing the volume.
For the measurement of succinate dehydrogenase, the brains from four animals at each time point were cut into two hemispheres, frozen in liquid nitrogen, and stored separately at −80°C. The activity of succinate dehydrogenase in each hemisphere was assayed using a spectrophotometric method (Hochstadt et al., 1974). Briefly, the frozen brain hemispheres were thawed and homogenized (10% weight/volume) in a buffer (pH 7.5) containing 250 mmol/L sucrose and 10 mmol/L potassium phosphate. The reaction was started by adding an aliquot (50 μL) of brain homogenate to the reaction mixture (900 μl phosphate buffer [pH 7.5], 500 μg bovine serum albumin, 1 μmol k-cyanide, 10 μmol sodium-succinate, 20 μg dichlorophenol-indophenol). The reaction rate was calculated from the rate of decrease in absorbance at 600 nmol/L and expressed as μmol/mg protein/hour. The Lowry method was used to determine protein content (Lowry et al., 1951).
Values are expressed as mean ± standard deviation. A statistical analysis was performed using the Student's unpaired t test. A two-tailed P value less than 0.05 was considered significant.
RESULTS
The body weight did not differ significantly between group A and group B. No significant difference was detected in rectal temperature, mean arterial blood pressure, arterial pH, arterial oxygen pressure (PaO2), or partial pressure of carbon dioxide (PaCO2) at baseline and 15 minutes after occlusion between the two groups (data not shown).
The unstained areas in both group A and group B appeared white. The stained areas in group A were judged as deep red, and the border between stained and unstained areas was judged as distinct in all brains. The stained areas in group B were judged as pink in one brain, light red in two brains, and deep red in five brains, and the border between stained and unstained areas was easily recognizable in one brain and distinct in seven brains (Fig. 1).
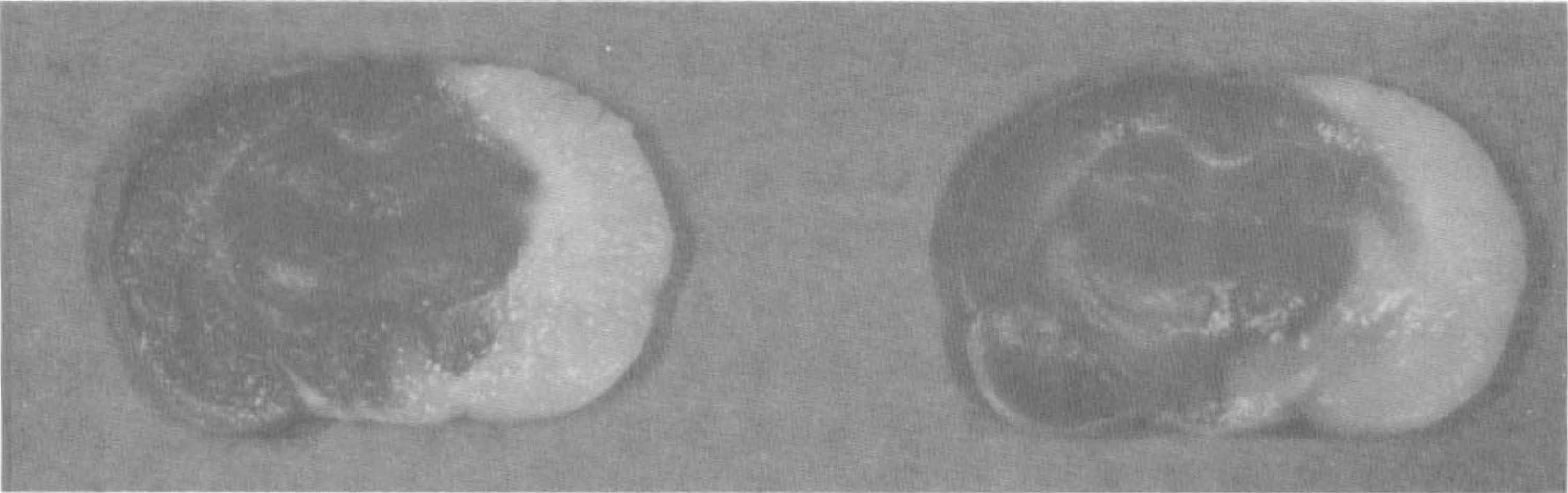
Immediate 2,3,5-triphenyltetrazolium chloride (TTC) staining after animal death shows deep red staining of normal brain tissue and white nonstaining of the infarct tissue with distinct border (left). Delay of TTC staining for 8 hours demonstrates similar findings (right).
The corrected infarct volume was 263.8 ± 43.1 mm3 in group A and 264.4 ± 54.8 mm3 in group B. There was no significant difference between the two groups (P = 0.98).
As shown in Table 1, the activity of succinate dehydrogenase in the right (infarcted) hemisphere was lower than that in the left (normal) hemisphere in both group A (P = 0.0006) and group B (p = 0.003). No significant difference of enzyme activity occurred in the normal left hemisphere between group A and group B (P = 0.31). Enzyme activity also was not significantly different in the infarcted right hemisphere between the groups (P = 0.15).
The activity of succinate dehydrogenase of the right and left hemispheres in animals studied immediately after sacrifice (group A) or with an 8 hour delay at room temperature (group B)

Unit: μmol/mg protein/h.
* P = 0.31 versus group A; †P = 0.15 versus group A; ‡P = 0.0006 versus normal; §P = 0.003 versus normal.
DISCUSSION
2,3,5-triphenyltetrazolium chloride is believed to be reduced to red formazan by mitochondrial enzymes (specifically succinate dehydrogenase) (Lippold, 1982). On immediate TTC staining after the elective or natural death of animals, the normal tissue stains deep red and infarcted tissue with loss of mitochondrial enzyme activity does not stain and appears white (Liszczak et al., 1984; Bose et al., 1984; Bederson et al., 1986; Park et al., 1988). The border between stained and unstained tissues is well demarcated and can be identified easily by visual inspection (Bederson et al., 1986; Isayama et al., 1991). Many studies demonstrate that TTC staining can rapidly and accurately detect brain infarction from 12 hours to 3 days after the onset of ischemia and is a reliable indicator of infarction extent (Bederson et al., 1986; Taylor et al., 1987; Lin et al., 1993). Therefore, TTC staining is extensively used in animal stroke experiments for quantitatively delineating the infarct volume (Memezawa et al., 1992; Hasegawa et al., 1995; Kuge et al., 1995; Reith et al., 1995; Takano et al., 1996).
Good contrast and distinct demarcation between the unstained and stained areas are crucial to define and accurately measure the infarct volume (Swanson et al., 1990). In this study, seven of the eight brains with delayed TTC staining showed distinct demarcation between infarcted and normal tissue, and the other brain had an easily recognizable border. The corrected infarct volume determined by delayed TTC staining was almost identical to that by immediate TTC staining after 24 hours of permanent MCAO, indicating that an 8-hour delay of TTC staining still is reliable for quantifying infarction.
The TTC unstained area implies infarcted tissue in which mitochondrial enzymes are denatured and dysfunctional. On electron microscopy, a considerable decrease of intact mitochondria was verified in the unstained area (Liszczak et al., 1984). The significantly lower enzyme activity of the ischemic hemisphere indicates that the TTC unstained areas are related to the reduced succinate dehydrogenase activity, but the activity in normal brain was very similar 8 hours after death to that observed immediately after death. The accuracy of TTC staining is uncertain at early stages (< 6 hours) after ischemia begins (Bederson et al., 1986; Taylor et al., 1987; Park et al., 1988; Isayama et al., 1991; Lin et al., 1993). One important reason could be the varying degrees of mitochondrial enzyme disruption over this relatively short time. Dettmers et al. (1994) found that mitochondrial enzymes retained normal activity 6 hours after MCAO in primates. Liszczak et al. (1984) demonstrated that the brains from decapitated mice kept at room temperature for 6 hours had normal TTC staining, concomitant with functioning mitochondria. One study showed that the histochemical abnormality identified by TTC is reversible and does not absolutely represent infarction within several hours (Cole et al., 1990). These observations indicate that the loss of mitochondrial enzyme activity requires many hours after blood circulation stops. Our results further confirm that the activity of succinate dehydrogenase does not significantly decrease in normal brain tissue 8 hours after death, supporting the usefulness of delayed TTC staining. Interestingly, no abnormalities of TTC staining were found in animals not subjected to focal ischemia who were left at room temperature for 8 hours and in the contralateral hemisphere of focally ischemic rats, consistent with previous finding (Liszczak et al., 1984). There is no clear evidence why the activity of succinate dehydrogenase is apparently normal in globally ischemic brains over 8 hours. We assume that the presumed slow disruption of succinate dehydrogenase activity in animals with no blood flow sitting at room temperature is related to the rapid decrease in body temperature after death. Further study is needed to clarify the disparity of TTC staining results between in vivo focal ischemia and dead animals without blood flow to the brain.
Delayed TTC staining 8 hours after death reliably measured the infarct volume in a rat stroke model. This probably is attributable to the slow degradation of succinate dehydrogenase activity, so that brains harvested within 8 hours after death still are useful for quantification of infarct volume if animals survive for more than 12 hours.
